Ammonium Bisulfite: Exploring Its Story, Science, and Impact
Historical Development
Ammonium bisulfite’s story began inside the rise of industrial chemistry in the 19th century. Large textile mills and paper manufacturers needed new ways to treat fibers and wood pulp. Early chemists learned that sulfur dioxide, dissolved in water and reacted with ammonia, gave a liquid that could break tough plant bonds. Ammonium bisulfite did the job, helping the sulfite process outpace earlier, less efficient methods. Today, the chemistry looks simple, but the social shift was huge: forests turned to paper quicker, textile makers improved dyeing, and water treatment plants found a partner for dechlorination. This single chemical rode the industrial revolution’s energy while shaping manufacturing’s path for over a century.
Product Overview
Ammonium bisulfite, most often found as a 40-65% aqueous solution, shows up in everyday industry with a slightly yellow to greenish tint. It’s never as glamorous as the headline ingredients, but white liquor or pulp cooking baths depend on it. Water treatment barely moves without ammonium bisulfite to remove excess chlorine. In oilfields, it prevents oxygen corrosion inside pipelines and injection systems. Food processors depend on it to slow browning in vegetables and shrimp. Cleaners, chemical labs, and even some electronics outfits order drums or totes as part of their monthly supplies. The stuff blends into both mega-scale and small lab operations, sticking around wherever chemistry needs reliable reduction reactions.
Physical & Chemical Properties
Pour ammonium bisulfite solution and the faint scent of sulfur greets you, like damp wool or weak match heads. Its chemical formula, NH4HSO3, describes a mix of inorganic ions that dissolve in water right away. Compared with similar agents, it's not explosive and doesn't build up pressure unless you mix it with acids or oxidizers. If you leave a container open, ammonia vapor floats off, making nearby air unpleasant and irritating to the nose and eyes. In routine lab settings, the solution feels slippery to touch and can sting if left on skin for long. Its density runs just above plain water, and with heating or neutralization, the chemical breaks down, freeing sulfur dioxide and ammonia gas. Such properties make rigorous sealing and ventilation common sense when stocking or handling it.
Technical Specifications & Labeling
Labels on ammonium bisulfite containers list concentration by percent weight, usually 40% or 60%, and show the batch date, recommended storage range (most settle on 10-25°C), and pH (tending toward the mildly acidic). Manufacturers add details like net mass, gross mass, and a hazardous material warning, since the chemical needs careful transport and vented storage. Common packaging includes drums, IBC totes, or even bulk tanker loads for industrial customers. Standard specs track heavy metals, clarity, and sulfur dioxide content; buyers ask about iron content or purity to match their applications. On the MSDS sheet, you'll see hazard codes for skin, eyes, and respiratory irritation, along with first-aid guidance for spills or brief exposure.
Preparation Method
Making ammonium bisulfite involves bubbling sulfur dioxide gas through a chilled aqueous ammonia solution. The gas dissolves in water, forming sulfurous acid, which then reacts with ammonia, giving ammonium bisulfite in solution. Operators must keep pH and temperature under control; unchecked, excess ammonia or SO2 could waste reagents or push the reaction toward other byproducts. Plants often recirculate unused gases to minimize emissions. Proper mixing, ventilation, and gas monitoring keep the process both safe and efficient. Scale-up from lab to full plant happens with little fuss because the underlying reaction is so reliable. Wastewater cleaning becomes important, since spent process liquids can still contain ammonia or sulfite residues.
Chemical Reactions & Modifications
Add acid to ammonium bisulfite and sulfur dioxide bubbles back out, ready for capture or venting. Toss in an oxidizer like hydrogen peroxide and watch the sulfite convert to sulfate, ending up as a neutral salt. Some labs tweak the formula with ammonium thiosulfate or by controlling temperature, steering the product’s reactivity or shelf life. Water treatment uses these reactions on the fly: the solution strips chlorine or chloramine out of city water, forming harmless ammonium chloride or nitrogen gas. In pulp and paper, adjustments in pH steer the balance between delignification and fiber strength. Tweaking ammonium bisulfite’s chemistry unlocks new performance, whether balancing an industrial process or treating delicate produce for extended storage.
Synonyms & Product Names
Order sheets rotate through names like “ammonium hydrogen sulfite,” “ammonium acid sulfite,” or simply “ABS solution.” CAS registry number 10192-30-0 tags almost every technical document. Labels from different makers might show “Yellow Bisulfite” or “Ammonium Sulfite, Acid Salt.” In commercial catalogs, all these names mean the same blend: a water-based sulfite salt ready to reduce oxidants or treat wood. Switching suppliers means checking synonym lists to avoid confusion, because the field still uses all these terms interchangeably.
Safety & Operational Standards
Everyone who works with ammonium bisulfite faces a few non-negotiable rules. Gloves and goggles sit near every drum. Store containers in well-ventilated rooms, away from acids or oxidizers. Always close lids; open containers let out not just ammonia but sulfur dioxide, stinging eyes and throats. Workers who handle spills flush the site with lots of water, then neutralize residues with mild alkali before sending waste to treatment. Most companies run regular training on chemical safety, reading MSDS forms and accident reporting. Federal and state agencies regulate handling, with rules for air emissions, wastewater limits, and emergency response drills. Once, I handled a small leak and learned quickly how pungent and stubborn the vapors can be, impressing on me why the rules exist and why workers value those thick gloves and tight goggles.
Application Area
Pulp and paper plants use ammonium bisulfite in cooking liquors, softening wood so fibers release smoothly. In municipal water plants, as soon as finished water leaves chlorine tanks, pipes carry it through ammonium bisulfite to strip extra chlorine. Oil refineries and drilling systems treat water with it to keep rust from sabotaging thousand-dollar pipes. Shrimp and fruit processors soak produce in a mild solution, fighting browning as food waits for package or delivery. Cleaners blend it into de-scaling solutions for boilers and heat exchangers, dissolving stubborn deposits. In labs, researchers lean on ammonium bisulfite to cut oxidants or stabilize short-lived reagents. The chemical bridges the gap between raw materials and finished goods in quiet, behind-the-scenes fashion, shaping everything from cleaner tap water to brighter copy paper.
Research & Development
Recent years have brought energetic research aimed at making ammonium bisulfite greener and safer. Chemists run experiments to reduce fumes during manufacturing and look for ways to recover spent ammonia or sulfur dioxide from waste. Papers from China and Europe show new blending methods that cut heavy metal contamination, especially for food and water-grade product. In the lab, researchers study how ammonium bisulfite’s reducing ability might control novel pollutants or serve niche syntheses. A few teams in environmental science investigate advanced application techniques, like microencapsulation, aiming to boost effectiveness and limit worker exposure. Oil and gas engineers check how the chemical holds up in new shale formations, while food scientists test ways to combine it with natural preservatives, stretching shelf life with fewer added ingredients. The research energy springs from two needs — keeping the old jobs safe and opening up new uses wherever reduction chemistry matters.
Toxicity Research
Extensive toxicology reports shape real-world rules for ammonium bisulfite handling. Acute exposure causes stinging eyes and lungs, much like sulfur dioxide or ammonia. Most workplace limits come from these short-term respiratory effects; at levels above 5 ppm in the air, workers report headaches and throat pain. Long-term animal studies show no mutagenic or cancer-causing risk at routine doses. In food and water, trace residues get strict monitoring, since excessive sulfur compounds can trigger allergic reactions, especially in people with asthma. Environmental labs report quick breakdown after release, provided the chemical isn’t washed into streams in bulk. Safety teams in factories use this research to build proper ventilation and spill controls, aware that even small leaks can make an enclosed room quickly uncomfortable or unsafe.
Future Prospects
Ammonium bisulfite faces both challenge and opportunity in coming decades. Tighter regulation on ammonia emissions pushes manufacturers to recycle process gases or develop closed-loop systems, which keep efficiency high and cut accidental releases. In water treatment, as chlorine alternatives (like ozone or UV) get popular, demand shifts but does not vanish; operators will keep ammonium bisulfite on standby for emergencies or maintenance cycles. In oil and gas, deoxygenation with ammonium bisulfite anchors corrosion control—not flashy, but cost-effective compared to pricier chemicals. Green chemistry research probes plant-based or lower-impact alternatives, but for now, this bisulfite stands as a mainstay. Ongoing studies into micro-dosing and safer formulations show promise in shrinking risk in food and workplace settings. Enduring demand and active research point to ammonium bisulfite holding onto its under-the-radar but essential role across many sectors.
What is Ammonium Bisulfite used for?
What is Ammonium Bisulfite Used For?
Ammonium bisulfite holds a quiet but crucial place in the world of chemicals. If you ever catch a whiff in paper mills or hang around a water treatment plant, you might notice its sulfur-like scent. In my time working near industrial areas and environmental testing labs, I never saw ammonium bisulfite make headlines, but it kept showing up behind the scenes. It’s not the type of compound folks brag about, but there’s no denying its impact when it comes to keeping things running cleanly and safely.
Water Treatment and Beyond
Water treatment companies rely on ammonium bisulfite as a reducing agent. One of its big jobs is to get rid of excess chlorine in wastewater. Chlorine serves a key role most of the time, disinfecting water so we’re not swimming with microbes. After it does its job, though, leftover chlorine can damage the very rivers and fish those plants aim to protect. A dose of ammonium bisulfite grabs that chlorine and neutralizes it, stopping the harm before it flows downstream. I’ve watched this process in water labs, where folks use simple color changes to spot if there’s chlorine left. That small drop of ammonium bisulfite takes care of the rest, making water safe to release back into the wild.
Power in the Paper Industry
The papermaking business also finds a good friend in ammonium bisulfite. For those who haven’t seen a pulp mill up close, the air around one fills with the odor of rotten eggs and mystery chemicals. One of the steps in turning wood into white paper relies on cooking chips with chemicals to break down tough fibers. Here, ammonium bisulfite comes into play. Its ability to break down lignin—the glue that holds wood together—lets the pure fibers separate more easily. I’ve read through technical logs and talked with plant workers who see this process chop down raw timber into a soft, almost fluffy stock. Manufacturers can shape it into everything from newsprint to packaging. By using ammonium bisulfite, mills can run more efficiently and cut back on harsh byproducts other chemicals would leave behind.
Cleaning and Preservation
In oil and gas pipelines, another battle wages underground. Iron and steel pipes risk attacking sulfide gases, leading to corrosion and heavy repairs. Ammonium bisulfite works as a scavenger here, snatching up oxygen and sulfur compounds that corrode metal. In my own experience doing fieldwork near an aging refinery, maintenance teams always kept close track of their oxygen levels. Even a brief drop in scavenging chemicals could lead to rust and leaks, costing thousands in repairs. Using ammonium bisulfite means fewer pipe failures, safer deliveries, and fewer environmental worries.
The Balance Between Utility and Risk
Chemicals earn their keep by solving problems. Ammonium bisulfite achieves a lot with a little, but it doesn’t work without respect. High concentrations pose health risks. In hands-on settings, I’ve seen crews suit up with gloves and masks just to avoid splashes and fumes. Breathing it in or getting it on skin leads to headaches, rashes, or worse. Proper handling isn’t fancy—just practical habits, good training, and solid storage. It’s hard not to think of the countless hours spent checking safety records and fine-tuning chemical feed rates. The world runs better when folks look out for one another, and in this case, making sure ammonium bisulfite does its job without stepping out of line makes all the difference.
Is Ammonium Bisulfite hazardous to health?
Digging Deeper Into a Common Compound
Most people won’t come across ammonium bisulfite outside of a lab, a power plant, or an industrial site. It pops up in places where folks treat water, make paper, and even in some food processing. At first glance, it just looks like another chemical helping people get jobs done quietly in the background. Still, talk to anyone who’s handled the stuff, and you’ll hear stories that can raise some eyebrows.
The Real Risks In the Air and On the Skin
Step into a room where ammonium bisulfite is in the air, and a sharp, biting smell hits pretty quickly. That’s sulfur dioxide being released—nobody forgets that stale-match stink. Inhaling vapors can trigger coughing, throat irritation, or worse for those with asthma or other lung troubles. OSHA and NIOSH list exposure limits for a good reason. Folks working with this compound, especially in larger operations, often suit up with masks and gloves.
Spill a bit on bare skin and it can cause itching or a mild burn, especially if left unwashed. Eyes get the worst of it, as a splash could mean stinging, watering, and serious irritation. Most safety sheets call for rinsing with water for at least fifteen minutes if that happens. That’s not scare tactics—it’s based on real lab incidents and years of industrial experience.
What Science Says About Long-Term Exposure
Researchers have looked into the effects of chronic, low-level contact. They don’t pull any punches. Long-term exposure can lead to sore throats, recurring cough, or even chronic bronchitis. Some animal studies suggest higher risks of respiratory stress with enough frequent contact. There’s no solid link to cancer or persistent organ damage, but folks who work with it daily seem more likely to report ongoing irritation, which lines up with what safety experts have found across several industries.
Why Worry: Everyday Chemicals and Secret Dangers
Chemicals like ammonium bisulfite fly under the radar because they have industrial uses most folks don’t see firsthand. Out of sight, out of mind. Still, it’s easy to downplay the risk. Plenty of companies have tried to cut corners, skipping out on recommended ventilators or relying on open-air dilution “to take care of” fumes. The truth comes from the emergency room visits by workers who develop sudden respiratory symptoms during cleanup or large-scale processing.
Having worked in places where mixing chemicals was part of the daily grind, I’ve seen safety ignored more often than big corporations want to admit. Small lapses—like a missing glove or a forgotten mask—turn into big incidents quickly. Ammonium bisulfite doesn’t have the drama of gasoline or bleach, but it can quietly cause harm if used without care.
Keeping Health Risks Low
Clear instructions, strict labeling, and reliable ventilation save lives. People who run water treatment plants or manage paper mills should press for better training and regular safety drills. Workers on the front lines—those handling open vats, hoses, and sprayers—need the right gear and encouragement to speak up about conditions that don’t seem safe. A big part of that means backing up any safety policies with real action and a no-excuses approach to replacing worn-out equipment.
Instead of quietly accepting a little throat burning as normal, workers ought to have ways to report and respond quickly. Companies benefit, too—lower accident rates mean less downtime, fewer lawsuits, and happier, healthier teams. Everyone wins with a culture that treats chemical hazards, including ammonium bisulfite, with open eyes and respect informed by science, not just gut feelings or cost-saving temptations.
How should Ammonium Bisulfite be stored?
Why Handling Ammonium Bisulfite Matters
You won’t have to look far to spot ammonium bisulfite on the labels of water treatment agents or industrial chemicals. This clear yellowish solution helps keep pipes corrosion-free and controls oxygen in systems like boilers. Its usefulness often hides its hazards—expect a sharp smell, strong reactivity, and some unpleasant risks if it’s not handled right. Mishandling leaks, fumes, or spills lands more often on workers than on abstract lists. My own experience, having watched warehouses handle the stuff, reminds me no one wants the stress of a splash or whiff.
Storage Conditions: Not Just a Suggestion
Controlling temperature comes first. Warm weather and sunlight turn ammonium bisulfite into a recipe for pressure buildup. Store drums and containers indoors, away from direct sun or radiant heat. Think of a stable room temperature, generally below 30°C, with a preference for a cool, shaded spot. Fans or air conditioning help if the building gets warm in the afternoon.
Humidity matters, too. Excess water exposure sets off chemical reactions that generate toxic gases. Always keep containers tightly closed and dry, making sure the lids seat properly after every use. Warehouses need to keep floors clean and dry underneath containers, since pooled water can corrode storage drums from the outside, and that leads to trouble.
Container Choice: Not All Plastics Are Equal
Don’t repurpose any old barrel just to save money. Only use high-density polyethylene drums or glass bottles certified for chemical storage. Steel, especially unlined steel, doesn’t get along with ammonium bisulfite. Leaked product finds every nick or scratch in cheap containers, speeding up leaks and creating a bigger clean-up job.
Look for containers with strong seals and labels showing handling instructions. Storage shelves or pallets handle the weight, but small chemical cabinets are a smart backup for jobs with limited space. If the storage area ever gets wet (think leaks from above or a spill), mop everything right away. Chemical safety seems dull until one small slip leads to a mess.
Mixing and Cross-Contamination Hazards
Never keep ammonium bisulfite near acids, oxidizers, or strong bases. Accidental mixing starts fast reactions, sometimes with choking fumes or even small fires. Use a separate section or at least a dedicated rack in the storeroom. Color-coded tape worked well for our team: blue for bisulfite, red for acids, green for oxidizers. Even a sticker can make a difference during a busy shift.
Ventilation and Spill Planning
Poorly ventilated storage brings risks up a notch. Fumes linger and, caught unprepared, even experienced staff might get a nasty headache or worse. Good exhaust fans draw away invisible hazards. Keep absorbent pads and neutralizer nearby in case of small spills. For bigger releases, stick to approved containment gear and call your supervisor—don’t try to handle a pool of chemical solo.
Training and Documentation
Everyone on site needs a briefing on ammonium bisulfite before storage duties. Share the safety data sheet with the team and keep it accessible. Even seasoned operators value reminders. Inventory logs and regular checks also point out leaks or swelling drums early, catching problems before they reach outside the store room.
Practical storage is less about fancy systems and more about staying alert, using the right container, respecting boundaries, and working as a team. Ammonium bisulfite earns respect for its chemical bite—a lesson learned best from others’ stories, not personal accidents.
What are the safety precautions when handling Ammonium Bisulfite?
Understanding What You’re Working With
Ammonium bisulfite keeps popping up in industries like water treatment and paper manufacturing. Most people who work with it come across problems from unexpected spills, skin contact, or breathing in its vapors. Having spent years on shop floors and in labs, I’ve seen good routines make the difference between routine work and rushed trips to the clinic.
Protecting Your Health Starts with Basic Gear
The best protection doesn’t come from any single piece of equipment. Doing the simple things matters. Gloves made from nitrile or neoprene offer a strong barrier and last through long shifts. Goggles with tight seals keep splashes out, and it only takes one careless moment for eyes to burn for hours. Lab coats or coveralls keep the chemical off your clothing and skin. It’s tempting to skip respiratory gear, but a mask with acid gas cartridges keeps fumes from sneaking in, especially where ventilation struggles to keep up. The Centers for Disease Control (CDC) and OSHA have both tracked hundreds of workplace accidents just because small steps got ignored or gear felt uncomfortable for an afternoon.
Good Ventilation Means Fewer Accidents
Poor airflow lets ammonium bisulfite build up in the air, and there’s no shortcut around that. Many old buildings still rely on cracked windows or basic fans. Investing in extraction systems and checking that air filters work properly goes a long way. I remember a workplace that didn’t upgrade its ventilation until people started getting dizzy halfway into every shift. Only after proper hoods and exhausts went in did issues drop to zero. Regular air monitoring should become habit — testing for sulfur dioxide and other gases means you catch leaks before they become ER visits.
Storing Chemicals Safely
Ammonium bisulfite reacts with air and oxidizes into sulfur dioxide, a toxic gas. Keeping containers tight and away from sunlight slows down this reaction. Storage areas stay safer when kept cool and dry, with clear signage warning of potential leaks. Housekeeping is not just about avoiding fines; spills left overnight can start eating into concrete floors or corroding nearby metals. Using secondary containers, like a tray under a jug, contains accidents without a mad scramble for sand or absorbent pads.
Spill Response Requires Speed and Preparation
Every shop or plant benefits from routine drills. People panic less if they know their roles and where to find spill kits. Neutralizing agents can turn a dangerous chemical pool into something safer to sweep up. Training everyone — not just supervisors — means the first person at the scene acts immediately. I’ve watched situations go from manageable to dangerous in under five minutes, just because a couple of workers waited for a manager.
Training Makes All the Difference
It’s easy to point to gear and rules, but real safety grows from practice. New employees learn more from walking through possible accident scenarios than reading binders. Retelling stories about near misses keeps safety personal. Posting clear signage about risks — in English, Spanish, or any language workers use — stops accidents before they begin. Companies with the lowest injury rates usually have the most open conversations about safety problems, and it shows up in everything from lower insurance costs to happier, healthier teams.
What is the chemical formula of Ammonium Bisulfite?
Understanding What’s in Ammonium Bisulfite
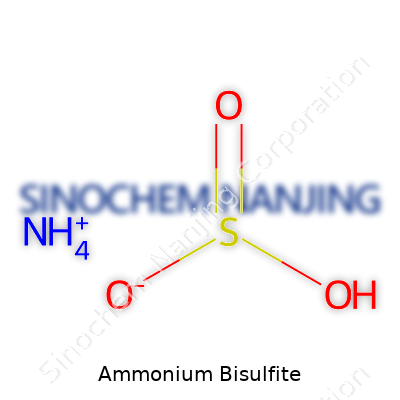
Ammonium bisulfite comes with the chemical formula NH4HSO3. This formula means the compound forms from one ammonium ion (NH4+) and one hydrogen sulfite ion (HSO3-). You find it in some unexpected household products as well as big industrial operations. Think of it as a behind-the-scenes helper in a lot of standard processes.
Why Knowing the Formula Matters
People hardly ever read the fine print on product labels unless they have to. Anyone with asthma or allergies learns quickly that chemical names and ingredients affect their health. With ammonium bisulfite, understanding its chemical formula isn’t just for scientists. The formula tells us how the compound reacts when mixed with water, air, or other chemicals. In the pulp and paper industry, folks use it for removing oxygen from water. This step protects expensive metal equipment from rust and corrosion. One wrong ratio in the formula, and suddenly safety tanks turn hazardous or paper quality drops.
Lessons From Everyday Life
Growing up in a rural area, I watched my dad maintain old farm machinery with industrial chemicals. Some were harsh, others safe if handled carefully. Ammonium bisulfite came up once when treating boiler water. My father understood the dangers of mixing the wrong chemicals. He’d always run through the labels and double-check formulas before pouring anything into a big tank. These small actions protect property from damage and people from burns or respiratory trouble.
Behind the Scenes in Different Industries
The food industry uses ammonium bisulfite under very controlled conditions, acting as a preservative agent or process aid. It helps keep food color and extends shelf life, but strict rules say how much you can add. Anyone working with it needs to observe the established safety guidelines. The textile sector turns to this chemical during the dyeing process, where its reducing properties make colors last longer and look richer. Not every chemical labeled “ammonium” or “sulfite” works the same way, which shows why paying attention to the formula is so important.
Putting Safety First
Getting careless with chemicals usually leads to trouble. Documented cases have shown that ammonium bisulfite can cause breathing problems if stored in a poorly ventilated room. Workers sometimes underline the importance of the right ratio: a bit too much, the solution eats through metal pipes; too little, and bacteria start to grow. Safety organizations like OSHA and the CDC provide detailed instructions—the kind of practical knowledge you hope everyone follows. Anyone can look those up, and it pays off to read and follow them.
Moving Toward Better Solutions
Many companies track and update their processes every year. They look for smarter ways to use chemicals—by using less dangerous substitutes or switching to cleaner additives. That sometimes starts with a close look at chemical formulas and how they interact. Proper staff training and easier-to-read labels also make a big difference. Regular audits and better handling routines keep both workers and the public safe, while cutting costs down the line.
| Names | |
| Preferred IUPAC name | ammonium hydrogen sulfite |
| Other names |
Ammonium hydrogen sulfite Ammonium acid sulfite Diammonium dithionite Ammonium bisulphite |
| Pronunciation | /əˈmoʊniəm baɪˈsʌlfaɪt/ |
| Identifiers | |
| CAS Number | 10192-30-0 |
| Beilstein Reference | 3596807 |
| ChEBI | CHEBI:38158 |
| ChEMBL | CHEMBL1231877 |
| ChemSpider | 25956 |
| DrugBank | DB11110 |
| ECHA InfoCard | 100.028.229 |
| EC Number | 232-260-9 |
| Gmelin Reference | 120152 |
| KEGG | C06199 |
| MeSH | D000648 |
| PubChem CID | 23238 |
| RTECS number | BO0875000 |
| UNII | V3R8C78A4L |
| UN number | UN2693 |
| CompTox Dashboard (EPA) | Ammonium Bisulfite: "DTXSID3020003 |
| Properties | |
| Chemical formula | NH4HSO3 |
| Molar mass | 101.11 g/mol |
| Appearance | Clear yellowish liquid |
| Odor | pungent odor |
| Density | 1.3 g/cm³ |
| Solubility in water | Very soluble |
| log P | -4.3 |
| Vapor pressure | < 0.01 mmHg (20°C) |
| Acidity (pKa) | pKa1 = 5.3 |
| Basicity (pKb) | 6.94 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.415 |
| Viscosity | 10 – 15 cP |
| Dipole moment | 4.12 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 199.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -623.03 kJ/mol |
| Pharmacology | |
| ATC code | V03AB37 |
| Hazards | |
| GHS labelling | Warning, H302, H315, H319, H335 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. |
| Precautionary statements | P210, P221, P261, P271, P273, P280, P301+P312, P304+P340, P305+P351+P338, P312, P330, P337+P313, P403+P233, P501 |
| NFPA 704 (fire diamond) | NFPA 704: 2-0-1 |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 (oral, rat): 2,400 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 640 mg/kg |
| NIOSH | SCG |
| PEL (Permissible) | 5 mg/m³ |
| REL (Recommended) | 25-35% |
| Related compounds | |
| Related compounds |
Ammonium sulfite Ammonium sulfate Sodium bisulfite Sodium sulfite |

