The Story of Ammonium Arsenate: Past, Present, and Future
A Walk Through Its History
Growing up in a small town with a keen interest in rocks and minerals, I often came across tales about the building blocks of chemistry. Few stories pack as much caution and intrigue as those tied to arsenic compounds. Ammonium arsenate stands out in the grand tapestry of chemical history — its origins stretching back to the 19th century when chemists first isolated and characterized countless new substances. Early researchers sought to harness every corner of the periodic table. For ammonium arsenate, its preparation arose from simple double displacement reactions, but its notoriety came quickly. Historical usage in pesticides and dyes left fierce debate, given the persistent toxicity issues. Plenty of research in the 1900s revolved around both using and regulating this compound more safely, and as the tide of safety standards rolled in, the world gradually reined in many of those original applications.
Core Features and Product Overview
Those of us who spent time with chemical catalogs or in college stockrooms remember ammonium arsenate by its white, often crystalline appearance. This salt, typically encountered as a tetrahydrate, carries a mix of curiosity and warning. It forms from the reaction of arsenic acid with aqueous ammonia, producing NH₄₃AsO₄. While you won’t find it in a typical home, it once filled bottles marked with warning labels in labs and agriculture stores. The defining aspect lies in its chemical makeup: the marriage of ammonium ions with the trivalent arsenic anion. Truthfully, it’s rarely seen outside academic or specialized research these days, reflecting how comprehension of its risks eventually overtook earlier enthusiasm for its many possible uses.
Properties and Specifications From Experience
Chemists expect a compound like ammonium arsenate to behave as a classic ionic salt—water-soluble, stable at room temperature, and sensitive to both acids and heat. Its solubility means that, once in water, it presents challenges for environmental control. From time spent in university labs, I recall how as soon as this material hit liquid, we handled it with gloves and worked under a hood—proof of how rules change when you deal with arsenic. Ammonium arsenate’s melting point sits high enough to outlast many common lab compounds, but decomposition releases toxic fumes, emphasizing why rigorous safety rules surround it. Its white crystalline form often belies the danger, which has tripped up inexperienced hands more than once.
Preparation and Chemical Reactions in Detail
In research and industry, ammonium arsenate forms by reacting arsenic acid with an aqueous ammonia solution. The balanced equation feels routine but delivers a potent reminder about respect for the lab bench. Mixing these starting materials, one gets a product that precipitates out with chilling predictability. Through years of organic and inorganic labs, I've seen colleagues treat these reactions with utter seriousness, stopping to check every label. This chemical also offers a window into broader arsenic chemistry, reacting easily with acids and alkalis. Chemists sometimes modify the compound, using it as a starting point for complex arsenate salts or catalysts. Its reactivity and utility in certain analytical methods made it a part of test kits in industry decades ago. These days, the applications get fewer as researchers search for safer alternatives.
Shifting Names and Synonyms
As with many older compounds, ammonium arsenate has traveled under various banners—ammonium orthoarsenate sometimes appears in the literature, along with designations that hark back to older chemical naming conventions. The substance also earned abbreviations in analytical texts and agricultural handbooks from a different era. Having combed through stacks of reference books, I know confusion can arise from synonym switching. These name changes reflected evolving understanding of both the compound’s structure and the risks tied to arsenic, and they drive home the point that chemistry isn’t just about test tubes, but about history and communication.
Safety and Handling—A Personal Perspective
Few substances in a chemical storeroom command the apprehension of arsenic compounds. I’ve seen experienced lab instructors go pale at the prospect of a spill. Guidelines insist on storing ammonium arsenate in tightly sealed, clearly labeled containers far from incompatible substances or busy hands. Exposure through inhalation, ingestion, or even skin contact risks acute poisoning, with symptoms that echo some of the worst nightmares of occupational medicine—nausea, cardiovascular collapse, even cancer from long-term exposure. Strict regulatory requirements now surround its use and disposal, with rules on permissible limits, spill containment, and emergency measures. Personal protective equipment stays non-negotiable. Over the past decades, advances in workplace monitoring and storage technology have cut down on accidental exposures, but the lessons taught by old accidents remain locked in every training session involving arsenic.
Application Areas—Lessons From Use and Misuse
All arsenic compounds share a haunted legacy in agriculture. In my own reading of local environmental history, I found old farm newspapers advertising arsenic-based chemicals as must-have solutions for pests. Ammonium arsenate once had a niche in these mixes, sought for its ability to take down insects and fungal threats. Shifts in regulatory priorities—prompted by mounting evidence of health and ecosystem harm—forced a sharp retreat from such uses beginning in the late 20th century. Its other roles in ceramics, glass coloring, and certain laboratory assays became rare as voluntary and legal restrictions tightened. Current interest in ammonium arsenate often centers on teaching, demonstration experiments (conducted under careful controls), or controlled syntheses for analytical research. The long shadow of environmental persistence and bioaccumulation keeps interest at arm’s length; chemists and engineers routinely substitute less hazardous materials.
Research, Development, and Ongoing Curiosity
Curiosity still runs deep among scientists who focus on the chemistry of heavy elements. Research teams continue to explore modification techniques for arsenate compounds—each innovation aiming to lock away arsenic in less mobile or less bioavailable forms. In niche areas of inorganic chemistry, researchers use ammonium arsenate as a tool for studying arsenic remediation strategies. Water treatment experts analyze arsenate behavior in environmental systems, drawing on the documented properties and reaction patterns as templates for new filtration or binding technologies. While the commercial outlook for ammonium arsenate shrank, the legacy of research into its reactivity and stability provided blueprints for safer chemical development. This cycle of use, restriction, and targeted study defines plenty of elements from chemistry’s darker chapters.
The Challenge of Toxicity and Lessons Learned
The dangers behind ammonium arsenate rarely need overstating. Chronicling toxicity studies, it’s easy to see why safety cultures around this material matured so quickly. Arsenic targets multiple organs, turns up in pathology reports tied to skin lesions, cancers, and cardiovascular failures. Toxicity spans acute and chronic exposures, as seen in both accidental and occupational records. Regulations and best-practice guides reflect hard-earned wisdom from the front lines of chemistry and public health. Growing awareness of ground and water contamination forced changes in disposal and cleanup, pushing laboratories and factories to overhaul their arsenic use. Reflecting on these lessons, I’m reminded that the story of ammonium arsenate is as much about caution and stewardship as it is about technical progress.
Future Prospects—A Glimpse Ahead
Peering into the future, ammonium arsenate sits as a textbook case of technological progress checked by safety responsibility. Most prospects for the compound stand tied to niche scientific inquiry. Safer substitute compounds and stricter laws have shifted both markets and research agendas. Any remaining work with ammonium arsenate happens alongside new detection and containment technologies, shaped by the legacies of past use and by community standards that now demand much more rigorous controls. Innovations in environmental science prompt continued monitoring for arsenic species, ensuring that future generations deal with fewer consequences of prior misuse. The cautionary story of ammonium arsenate guides both research and regulation, acting as a reference point in the journey toward safer chemistry and more responsible development.
What is Ammonium Arsenate used for?
Why Ammonium Arsenate Matters
Ammonium arsenate lands in that small and uneasy group of chemicals that carry a reputation based on risk as much as usefulness. Its name alone is enough to make most folks tighten up. The reality remains: chemists discovered its utility years ago, and its presence still lingers in certain corners of science and industry. During some summer days in a research lab, I saw firsthand why such substances keep cropping up in controlled environments. They aren’t everyday tools, but their impact is undeniable.
Historic and Present-Day Uses
The story of ammonium arsenate runs back well over a century. In earlier decades, manufacturers turned to it when producing insecticides. It helped tackle pests that would decimate crops. Farmers had few options then. Arsenic-based compounds could wipe out invaders that otherwise destroyed entire fields. Momentum for these uses waned, though. Research underscored the chemical’s danger to more than just insects—it threatened animals and people alike. Across the world, as stories of poisonings piled up, countries shut down the bulk of agricultural and household use.
Lab experience showed that ammonium arsenate still finds a foothold in analytical chemistry. Well-trained hands weigh, dilute, and react this compound in minuscule amounts, especially in tests that call for standard arsenic solutions. Quality control labs sometimes lean on such precise measurements to calibrate equipment. Rarely, it serves as a reagent in specific chemical syntheses or in separating materials in mineral analysis. These situations rarely spill into public view, because the risks demand tight controls—complete with proper gear, fume hoods, and strict procedures.
Health and Environmental Risks
Few things create as much anxiety in the world of chemicals as arsenic. Ammonium arsenate reflects that concern. The science is clear: contact or inhalation brings high toxicity. Chronic exposure causes cancer, damages skin, and impacts the nervous system. Water contamination stands out as a serious threat, and soil pollution isn't far behind. Stories from contaminated communities push many to demand answers and action. The chemical’s hazard profile basically defines its limitations. Personal experience in labs confirms the obsessive checks, special gloves, and emergency protocols that keep the smallest spill from turning into major trouble. Recalling those days, one never forgets the invisible threat of a misplaced drop or careless breath.
Addressing the Challenges
Few chemicals spur stricter rules than those with arsenic. Regulation tightened everywhere. The Environmental Protection Agency keeps watch in the United States, just as European and Asian counterparts do. Disposal now comes with mandatory reporting and permits. Special containers, trained collectors, and locked storage help prevent leaks. Research centers substitute less toxic compounds where they can—teaching new scientists that safety trumps tradition. Industry continues to root out old applications, moving away from arsenic wherever modern science offers an alternative.
An honest look at ammonium arsenate pushes the need for education. It’s easy to ban a substance but much harder to clean up its legacy or protect workers still exposed. Open disclosure, government oversight, and real-world training matter as much as technical innovation. The lessons learned from this compound shape how science and society manage all hazardous materials today.
Is Ammonium Arsenate toxic or hazardous?
What Makes Ammonium Arsenate Dangerous?
Ammonium arsenate might not sound familiar at the grocery store, but it grabs attention in a lab or factory. This chemical mixes ammonia with arsenic, something most people know from history books as a dangerous poison. Its structure puts it in the same family as other chemicals that have impacted communities around the world. Arsenic itself has a long-standing reputation for health risks, and adding ammonia doesn’t soften the blow.
Direct Health Risks
Touching or inhaling ammonium arsenate introduces risks fast. I remember reading medical case studies where workers unaware of proper safety measures paid a high price—headaches, stomach pain, skin problems, and long-term organ damage shaped their daily realities. This chemical gets absorbed through the skin or lungs, making even a short exposure risky. Chronic contact piles up in the body, often hitting the liver, kidneys, and nervous system hardest. Signs aren’t always obvious until things get serious.
A few decades ago, some communities ended up with arsenic in their water. Rates of cancer and heart disease shot up. Arsenic in any form doesn’t respect industry boundaries—it finds ways to move through air and water. The Environmental Protection Agency classifies arsenic compounds as known carcinogens, and that label brings weight. Workplace regulations require air quality checks and protective gear, not out of caution, but hard-learned necessity.
Environmental Impact
Ammonium arsenate isn’t just tough on people; it’s rough on the environment too. Runoff or accidental spills can poison soil and water. I’ve watched neighbors worry as fish in their local lakes disappeared following exposure to arsenic-based compounds decades back. Plants, animals, and whole ecosystems still carry scars. The chemical doesn’t break down easily; it lingers, bioaccumulates, and travels up through the food web.
Handling and Storage: No Room for Error
Every parent teaches their kids to keep cleaning chemicals out of reach. Labs and factories take that logic further with locked cabinets, special signage, and rigorous training—not because they’re afraid of inspectors, but because the risk is real. Ammonium arsenate reacts with acids and heat, sometimes releasing toxic gases. Emergency rooms have seen patients who never knew what hit them, just from being too close without protection.
Working Toward Safer Alternatives
Chemistry doesn’t stand still. Researchers keep searching for alternatives that do the same job without risking so much. Many companies swapped out arsenic-based products in agriculture and manufacturing. Public reports show less contamination when industries switched to safer compounds for processes like wood preservation or glassmaking. Giving professionals information and access to substitutes helps reduce exposure and long-term health costs, and it keeps communities safer as well.
Supporting Safe Practices
One solution starts with better awareness and stricter regulation. Looking back at disasters, most took root with ignored warnings or a lack of transparency. Local governments now require strict labeling and clear instructions for hazardous substances. Having seen family and friends affected by environmental pollution, I value community transparency and workplace training above all. Keeping dangerous chemicals under lock and key, and investing in real safety education pays back in healthy lives and clean neighborhoods.
What are the safety precautions when handling Ammonium Arsenate?
Understanding the Danger
Ammonium arsenate doesn’t play around. My chemistry professor always started safety talks with real stories—one about a forgotten flask, another about a hurried mistake. It stuck with me. This compound, with its arsenic component, can wreck your health. Inhalation, ingestion, or even simple skin contact can cause severe poisoning, and there’s a real cancer risk if you let your guard down. Government agencies, from OSHA to the CDC, highlight the same message: treat every moment of exposure as a moment of risk.
Practical Safety Precautions
I remember the old metal cabinet in our lab, painted red just for the nastier chemicals like ammonium arsenate. There’s a reason for storage rules. Keep ammonium arsenate in tightly sealed containers, away from acids and dampness, in a spot that only trained staff can access. The goal is to limit any chance of accidental mixing or inappropriate grabs in a rush.
Gloves aren’t enough on their own—choose thick, chemical-resistant ones. Splash goggles, a lab coat (preferably one that stays in the lab), and a recommended mask or, even better, a full respirator fit for arsenic dust prevent dangerous particles from getting a foothold. If you get careless, a noseful or a trace on your lunch can change your day—or worse, your life. Taking five seconds to gear up beats years of treatment for arsenic’s side effects.
Work in a fume hood every time. You can’t rely on wide-open windows or room fans; they don’t cut it with this kind of poison. Fume hoods draw contaminated air away before it even gets close to your lungs. Always check that airflow works before opening your chemical bottle.
Cleanliness, Disposal, and Spills
My lab supervisor drilled us on spill drills. With ammonium arsenate, speed and accuracy matter. If you spill, restrict the area fast. Lay absorbent, scoop up solids with a disposable scoop, and use wipes soaked in water, not something acidic, or you risk making things worse. Bag every cleanup cloth and tool, then tag it for hazardous waste disposal—no shortcuts, no trash cans. Follow EPA guidelines: only certified waste handlers get to touch those bags after you’re done.
Clean hands before leaving the lab—even if you wore gloves. One tiny trace can stick to your phone, your keys, or your sandwich and come along for the ride. Every surface needs a regular scrub with a soft bleach solution, and every break means soap, warm water, and a two-minute scrub.
Training and Emergency Plans
Lab signs only do half the job. Anyone using, transporting, or even walking near ammonium arsenate works better after real, hands-on training. Know emergency numbers by heart, and review the map to the nearest eyewash station or shower every week. Didn’t do a practice run? Don’t be surprised if panic throws you off.
Ammonium arsenate isn’t something you forget about or take lightly. It’s about staying alive, staying strong, and showing respect—not just for the rules, but for your own future and anyone around you.
What is the chemical formula of Ammonium Arsenate?
Understanding the Substance
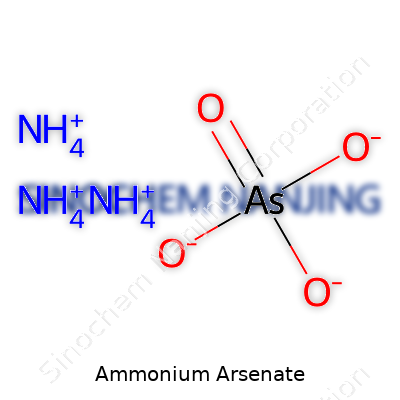
Ammonium arsenate, with its chemical formula (NH4)3AsO4, lands on the radar of anyone with an interest in chemistry, environmental health, or industrial safety. This crystal-like compound contains three ammonium ions and one arsenate ion. Its makeup is straightforward on paper, but the effects and uses tell a deeper story. Sometimes, the simplest formulas remind us how dangerous the world of molecules can get.
Why This Chemical Matters
In the field of chemistry, recognizing formulas like ammonium arsenate is not just a trivia exercise. It’s all about vigilance. Arsenic often gets a lot of attention for being both toxic and historically misused. Knowing the formula isn’t only about passing an exam. It’s about understanding the risks and safeguards—especially for those who come across these substances in labs, factories, or even old pesticides.
Where It's Found
Though it’s rarely seen in everyday products now, ammonium arsenate once had its heyday in agriculture. People used it to fight off bugs and weeds. Over time, the facts caught up: both arsenic and many of its compounds pose serious health threats. Despite its decline in popularity, traces can still turn up in soil and groundwater in places where arsenic-based chemicals linger from past use. This legacy brings concerns for environmental health professionals working to protect communities from accidental contamination.
Health and Safety Risks
The combination of ammonium and arsenate is not something to take lightly. Breathing in dust or accidentally ingesting even tiny amounts carries the threat of arsenic poisoning—a risk with long-term symptoms ranging from skin lesions to cancer. The formula tells us there are three ammonia derivatives holding hands with a single arsenate, painting a clear picture of how the molecule interacts in the body. Because of arsenic’s notorious toxicity, laws in many countries now restrict the sale and transport of compounds like ammonium arsenate.
Building Safer Practices
Learning the formula is one thing. Respecting it is another. Safety starts with education. Every chemistry student, industrial worker, or environmental technician should get comfortable reading chemical formulas and understanding the practical consequences on human health and nature. That was drilled into me during my first toxicology seminar, when professors drove home the dangers linked to unfamiliar white powders. Established regulations, like wearing gloves and keeping good ventilation, work best when people actually know why they matter.
Looking for Solutions
Prevention stands as the best approach. Labs and workspaces use rigorous labeling and documentation for all compounds, but complacency still creeps in. Regular training, surprise inspections, and real stories about chemical mishaps help drive the point home more than any poster ever does. For older sites where arsenic compounds might still hide in the soil, systematic testing and cleanup programs save lives and restore trust. These steps rely on everyone knowing exactly what is at stake—starting with a seemingly basic formula.
Final Thoughts
The heart of this issue lies in a simple formula, but the consequences reach into safety, public health, and environmental protection. Recognizing (NH4)3AsO4 isn’t just for exam season. In my experience, respect for chemistry starts with small details—and those details can make all the difference.
How should Ammonium Arsenate be stored and disposed of?
Hidden Danger in the Supply Room
Ammonium arsenate rarely grabs headlines, but its risks don’t drop just because it stays out of the news. Even a small lab bottle on a shelf tells a bigger story about chemical safety and human health. I remember walking into an old school storeroom and seeing containers covered in thick dust, some marked “arsenate” with faded labels. Those containers hardly felt dangerous, but looks fool a lot of people. Ammonium arsenate contains arsenic; a known carcinogen, ranked high among substances that cause cancer and nerve damage. Mishandling brings real risk, both for those working directly and for their neighbors and the surrounding community.
The Smart Way to Store
Nobody wants to discover a leaking bottle or spilled powder in the back of a closet. Storage should reflect just how serious the risk is. Strong containers work best—chemical-grade glass or plastic with airtight seals. Clear, current labels make all the difference; a mix-up could end in tragedy.
The storage space must keep moisture away because ammonium arsenate can break down, releasing toxic arsenic compounds. People sometimes underestimate the need for real ventilation, but on a warm summer day, vapors can build fast. Lock the storage door—this is not a material for open shelving or anywhere kids or untrained visitors might wander through. I once talked to a facility manager who kept their chemicals in a locked, fire-resistant cabinet with only trained employees allowed in; that’s the right mindset.
Disposal—No Shortcuts Here
Safe disposal is not about tossing containers in the trash or washing residues down the drain. Authorities don’t just suggest proper disposal—they demand it, for good reason. Waste services see ammonium arsenate as hazardous waste. If it ends up in the ground, it taints water supplies and harms plants, animals, and people over many years.
The easiest thing I’ve found? Call certified hazardous waste disposal companies. Trying to neutralize it yourself, without advanced training and the right safety gear, risks spills and chemical burns. These professionals collect, handle, and transport the material according to strict EPA rules that protect workers and everyone further down the line. I saw an old expired bottle removed by a professional crew wearing full-face protection, double gloves, and chemical suits—the scene made me realize just how serious this process gets.
Responsibility and Reputation
Nobody wants a fine or a scandal. Environmental agencies track hazardous substances closely, and records of poor handling follow companies and managers for years. Doing things the right way—secure storage, clear labeling, controlled access, authorized disposal—is more than following laws; it shows respect for coworkers and neighbors.
In my experience, good safety training makes mistakes a lot less likely. Teaching people about what’s really in those old bottles, making sure the right gear is available, and keeping emergency lines open mean small labs and big companies alike can be confident that ammonium arsenate stays where it belongs—locked away and out of the news.
| Names | |
| Preferred IUPAC name | Ammonium arsenate |
| Other names |
Diammonium arsenate Ammonium orthoarsenate |
| Pronunciation | /əˌmoʊniəm ɑːrˈsiːneɪt/ |
| Identifiers | |
| CAS Number | 7784-41-0 |
| Beilstein Reference | 82292 |
| ChEBI | CHEBI:22243 |
| ChEMBL | CHEMBL1201188 |
| ChemSpider | 55729 |
| DrugBank | DB14633 |
| ECHA InfoCard | 03e1ad35-711a-4e64-8ea4-2cf07e6a4b7f |
| EC Number | 232-221-1 |
| Gmelin Reference | 837 |
| KEGG | C01250 |
| MeSH | D000648 |
| PubChem CID | 24808 |
| RTECS number | CA2450000 |
| UNII | 7X4F2OHL6T |
| UN number | UN1557 |
| Properties | |
| Chemical formula | (NH4)3AsO4 |
| Molar mass | Molar mass: 398.29 g/mol |
| Appearance | White crystalline powder |
| Odor | odorless |
| Density | 2.525 g/cm³ |
| Solubility in water | soluble |
| log P | -2.25 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 11.6 |
| Basicity (pKb) | 3.19 |
| Magnetic susceptibility (χ) | -62.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.500 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1488 kJ/mol |
| Pharmacology | |
| ATC code | V03AB13 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled or in contact with skin; may cause cancer; causes damage to organs through prolonged or repeated exposure. |
| GHS labelling | GHS05, GHS06, GHS08, GHS09 |
| Pictograms | GHS06,GHS08,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. H373: May cause damage to organs through prolonged or repeated exposure. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P201, P202, P260, P264, P270, P272, P273, P280, P301+P310, P302+P352, P304+P340, P308+P313, P314, P330, P391, P405, P501 |
| NFPA 704 (fire diamond) | 3-1-2-OX |
| Lethal dose or concentration | LD50 oral rat 6 mg/kg |
| LD50 (median dose) | LD50: 6 mg/kg (oral, rat) |
| NIOSH | AS8225000 |
| PEL (Permissible) | PEL: 0.01 mg/m3 |
| REL (Recommended) | 0.2 mg As/m³ |
| IDLH (Immediate danger) | 100 mg As/m3 |
| Related compounds | |
| Related compounds |
Sodium arsenate Potassium arsenate Ammonium arsenite |

