Aminoguanidine Bicarbonate: A Hard Look at Its Practical World
How We Got Here: Historical Development
Aminoguanidine bicarbonate didn’t appear out of nowhere. Chemists in the mid-1900s started paying attention to aminoguanidine salts due to their potential in pharmaceuticals and explosives. Tinkering with guanidine derivatives led to the first reports of aminoguanidine bicarbonate synthesis. The compound’s early applications spun out of research labs in Eastern Europe, where scientists saw its utility for selective chemical reactivity. They found that swapping out simple salts for bicarbonate made aminoguanidine less reactive to unwanted side reactions. This pushed its adaptation for more precise uses, especially when the chemical industry wanted greater control over energetic material formulations.
Getting to Know the Product
Looking at aminoguanidine bicarbonate up close, one finds a white, fine crystalline powder with a faint ammonia odor in concentrated form. Its chemical formula tends to confuse newcomers, who sometimes mix it up with the more dynamic aminoguanidine nitrate. Unlike some salts, this bicarbonate doesn’t draw too much moisture from the air, a detail that matters during storage or shipping. Molasses-like stickiness never shows up here—what you see is what you get. If someone tries to lump it together with trickier or more volatile aminoguanidine compounds, it helps to point out that the bicarbonate makes for safer and more manageable handling.
Physical and Chemical Properties
Aminoguanidine bicarbonate, packed as dense, snow-like crystals, has a melting point that holds steady until decomposition. Only at temperatures above 170°C do things start to break down, which means common lab mishaps like a forgotten flask on a hot plate won’t turn catastrophic. High water solubility means it dissolves quickly, opening doors for chemists working with aqueous reactions. Chemically, its bicarbonate group helps buffer reaction mixtures and prevents runaway acidification, something that those mixing energetic materials or crafting intermediates in drug synthesis watch closely. In solution, it stays stable under mild conditions, not prone to producing toxic vapors unless strongly heated.
The Nitty-Gritty: Technical Specs and Labeling
Technical sheets show a strong focus on purity, with commercial aminoguanidine bicarbonate topping 98 percent pure. Contaminants like chloride or nitrate sit well below set thresholds, since even a pinch of the wrong ion can ruin a batch, especially in explosives or fine chemical production. Labeling kicks up a bit more fuss thanks to evolving chemical regulations, which demand hazard signs, clear batch numbers, and strict storage warnings. Someone once told me about a batch incorrectly labeled—turns out, even the smallest paperwork slip can set back research timelines by weeks as stock gets double-checked for compliance.
How It’s Made: Preparation Method
Making aminoguanidine bicarbonate isn’t wizardry, but strict attention to detail must guide each step. Lab workers start with aminoguanidine sulfate or hydrochloride, then neutralize it with sodium bicarbonate or potassium carbonate in water. Precipitation occurs slowly, requiring patience for clean crystals to form before filtration and drying. Simple at first glance, but scale changes everything. Plants running multi-ton synthesis make continuous tweaks for crystal quality and yield. Any shortcut on water quality or filtration shows up immediately in both the final product and downstream applications, which is why reputable producers go overboard on purity checks.
Getting Reactive: Chemical Behavior and Modifications
Aminoguanidine bicarbonate’s reactivity has earned it a reputation in organic synthesis. The molecule acts as a solid intermediate for building more complex heterocycles—of special note are tetrazoles and triazines, both heavy hitters in medicine and agrochemicals. It can donate nitrogen atoms under certain conditions, fill out multi-component reactions, or serve as a buffering agent in cases where reaction pH gets unruly. Chemists lean on its capacity to transform when heated with other reagents, churning out vital intermediates for dyes or drug frameworks. Some labs use it as a stabilizer or a precursor for nitro derivatives, tuning reaction conditions for specificity.
What’s in a Name: Synonyms and Product Titles
Aminoguanidine bicarbonate goes by more than one handle. Some call it guanyl hydrazine bicarbonate, while suppliers often abbreviate it as AGBC or just “aminoguanidine salt.” Regional naming preferences pop up too, adding to confusion for buyers sifting through global catalogs. Unlike more famous chemicals, aminoguanidine doesn’t have a well-marketed brand name, so research teams must look for chemical identifiers on technical sheets to confirm they’ve got the real deal.
Staying Safe: Standards and Practices
Anyone who’s ever spent time in a chemical plant or university stockroom knows that safety with aminoguanidine bicarbonate goes beyond mere labeling. It isn’t an explosive by itself, but its close relationship to other aminoguanidines means safety protocols overlap. Proper ventilation and protective equipment help prevent potential respiratory or skin contact hazards. While its environmental profile looks better than that of nitrates or perchlorates, waste procedures still require controlled disposal to keep local waterways clean. In the U.S., storage must follow OSHA’s general industry guidelines, with secondary containment for bulk shipments and spill-proof containers for lab use.
The Real World: Application Area
Applications for aminoguanidine bicarbonate keep growing. The compound found its first fame stabilizing energetic mixes in propellant and pyrotechnic industries, giving safer shelf-life without detonating unpredictably. Drug researchers picked up on its ability to form frameworks used in blood pressure and diabetes medications. I recall one pharmaceutical tech describing how a simple batch of aminoguanidine bicarbonate helped her team fine-tune new antihypertensive agents. Further out, researchers use it to manufacture specialty foams, textile processing chemicals, and even corrosion inhibitors in pipeline maintenance. This versatility relies on the molecule’s acute reactivity under tightly controlled conditions.
R&D and Toxicity Research
Pushes in research and development have centered around new derivatives that improve therapeutic index or reduce side effects. Toxicity studies dating back to the 1960s flagged concerns about liver and kidney impact at very high doses in rodents, which led to cautious optimism during early diabetes drug trials. Over time, understanding grew that well-controlled, low-level exposure posed minimal risk, but gloves and fume hoods remain standard wherever the powder is in play. Teams working in environmental chemistry continue tracking breakdown products to make sure secondary metabolites from larger-scale use don’t trickle into sensitive watersheds. Part of the ongoing appeal, though, is that its toxicity profile hasn’t hampered continued investigation into new uses in agricultural or medical settings.
Looking Forward: Future Prospects
Aminoguanidine bicarbonate stands to gain ground as new regulations push the chemical industry toward less hazardous intermediates. Researchers explore how its reactivity serves as a springboard for “green” synthesis, reducing waste streams and dangerous byproducts. There’s growing interest in using this compound to address challenges in sustainable manufacturing for dyes, consumer goods, and advanced polymers. I’ve met teams who look at aminoguanidine as their best shot at crafting specialty polymers for electronics and energy storage, particularly where custom nitrogen-doping serves niche performance needs. Staying on top of safety updates, improving recyclability, and designing more targeted applications will let the compound keep pace with stricter rules and expectations, both in the lab and on the factory floor.
What is Aminoguanidine Bicarbonate used for?
Why This Chemical Matters
Aminoguanidine bicarbonate doesn’t pop up much in everyday conversation. Most folks probably haven’t even seen the name outside a lab or a factory. The truth is, it underpins more than most people realize. In my years working with scientific and industrial topics, I’ve learned how substances like this create ripples far beyond the obvious.
Pharmaceutical research drives much of the demand. This chemical often enters the spotlight in studies tackling diabetes and its complications. Scientists look at aminoguanidine because it interrupts those pesky advanced glycation end-products (AGEs) that accumulate when blood sugar stays high over time. These AGEs can speed up damage to blood vessels and nerves—two big problems in diabetes. Trials in animals showed the compound could slow or prevent some of this damage, especially nerve-related issues or heart problems linked to long-term high blood sugar. Although it didn’t make the jump to a widely approved drug, the work wasn’t wasted. Researchers still revisit it as a reference point, especially when developing new treatments for chronic complications.
Moving away from medicine, explosives and propellants often rely on compounds far from household items—and aminoguanidine bicarbonate lands squarely in their world. Manufacturers use it as a building block for triazoles and tetrazoles, chemicals with the power to push rockets skyward or clear a path through tough terrain. These aren’t areas most people see close up, but anyone watching a fireworks display or thinking about satellite launches bumps into these technologies at some level. Handling and producing such energetic compounds brings real safety and environmental concerns. I’ve visited factories where strict protocols guard against accidents—it’s clear that any misstep carries serious risks. This highlights the need for strong training and safety measures, not just for workers but for the companies that rely on their expertise.
Aminoguanidine bicarbonate also supports some pretty niche production processes. Dye and photographic chemical synthesis use it to tweak color or stabilize images. Unlike smartphone pictures that vanish with a swipe, chemical photos hang onto their color and composition for decades, and small changes in formulation can affect lasting power. Chemical manufacturers tell me that tweaking inputs like aminoguanidine can tip the balance toward deeper, longer-lasting colors or sharper image contrast.
Weighing Opportunities and Risks
It’s tempting to see every specialty chemical as a double-edged sword. Opportunities can’t come without risks. In pharmaceutical settings, aminoguanidine showed real promise, then ran into safety limitations as researchers dug deeper. Side effects appeared in some test subjects. With explosives, the benefits are obvious for those who need the power, but so is the worry over accidents or improper use. Waste from dye or photo chemical production can raise environmental red flags if not handled properly. Responsible stewardship matters. Whenever I talk to chemists in these industries, safe handling and thorough risk assessment form the backbone of their approach.
There’s room for growth if researchers can iron out safety and stability issues. Better regulation, smarter handling systems, and early investment in alternatives or less hazardous ways to achieve similar results make a difference. Ongoing progress in chemical engineering could make manufacturing cleaner and safer, reducing the environmental footprint while protecting workers.
Aminoguanidine bicarbonate won’t steal headlines, but its imprint runs through modern science, tech, and industry. Getting the most out of it without letting downsides take over—that’s where attention should stay.
What is the chemical formula of Aminoguanidine Bicarbonate?
Understanding the Formula
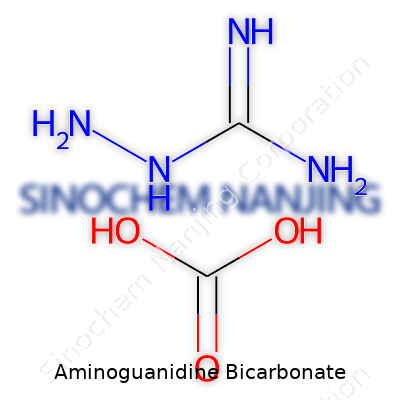
Chemistry gets pretty personal once a person starts digging into specific compounds. Aminoguanidine bicarbonate, for example, carries more weight than its name suggests. The chemical formula here is CH6N4·H2CO3. Most scientists break that down to clearly state one aminoguanidine molecule paired with a single molecule of bicarbonate. Sometimes industry calls it aminoguanidine hydrogen carbonate, but the composition does not change—only the language.
Why the Formula Matters
My own path through chemistry taught me early on—understanding a formula gives more than trivia answers. Each part links directly to the behavior of the compound and explains how it reacts or stays stable in various environments. Aminoguanidine bicarbonate comes up mostly in sectors like pharmaceuticals, textile processing, and sometimes water treatment. Chemists pay close attention to formulas since the tiniest change can throw off purity, safety, or intended results.
Aminoguanidine’s makeup—four nitrogens, six hydrogens, one carbon—means it brings reactive nitrogen to any synthesis. Add the bicarbonate (one extra carbon, two hydrogens, three oxygens), and it changes the solubility and stability compared to free aminoguanidine. Laboratories rely on knowing this, especially since aminoguanidine on its own can be dangerous, unstable, or too reactive for storage. Bicarbonate tames it, making shipping and handling easier and safer.
How Formula Drives Decisions in Real World Tasks
There’s always debate about what makes a formula truly “matter.” In practice, mistakes with chemical formulas can mean record-setting costs or harm. For instance, pharmaceutical companies face strict audits; if someone swaps aminoguanidine sulfate for aminoguanidine bicarbonate, the effect on drug quality could be serious. The carbon dioxide produced once the bicarbonate decomposes in a reaction needs to be watched closely as well.
Over the years, experts have flagged real-world incidents where the wrong chemical grade ended up in production lines, causing recalls. Problems like that could be avoided just by double-checking formulas like CH6N4·H2CO3 before buying or using chemicals.
Toward Safer, Smarter Chemistry
Careful use of aminoguanidine bicarbonate links back to broader safety. Training, access to accurate chemical databases, real-time digital tracking in warehouses—these steps root out confusion. Digital solutions can highlight the formula and make side-by-side comparisons with similar compounds, giving lab techs, buyers, and process engineers a fighting chance to spot differences before problems spread.
The old way of copying formulas from a notebook has led to too many near-misses. Now, open-access chemical reference tools and chemistry education make it much harder to justify mistakes around formulas like this one. The future offers even more—improvements around digital labeling, supply chain transparency, and hands-on safety training so people respect the science, not just its paperwork trail.
Is Aminoguanidine Bicarbonate safe to handle?
Facing Chemistry in Real Life
Anyone handling chemicals in the lab or on the production floor knows there’s no coasting by on guesswork. Aminoguanidine bicarbonate turns up in research and industry, usually as a reagent for the synthesis of pharmaceuticals, dyes, and agricultural compounds. Looking at safety, I always hold chemicals to a tough standard—because years of working with everything from solvents to acids have shown me what can go wrong with even small mistakes.
Diving Into Aminoguanidine Bicarbonate
Aminoguanidine bicarbonate has some chops, especially in experimental work, but safety deserves a closer look. This compound is a white crystalline powder. It doesn’t look out of place in a chemical cupboard next to things like urea or ammonium salts, but looks can fool you. The substance is known to release toxic nitrogen oxides and ammonia when heated or mixed with strong acids. That kind of decomposition shows up as an obvious risk hazard, especially in confined spaces.
Health risk is not only about toxicity. Inhaling dust or letting it contact your skin should absolutely be avoided. It causes irritation, and prolonged exposure may trigger more severe effects. The European Chemicals Agency includes aminoguanidine bicarbonate as a compound that can be hazardous if not managed well. The main problems stem from its potential role as an irritant, along with risks tied to improper storage and mishandling.
Lessons Learned the Hard Way
I’ve watched people get complacent with lab chemicals. No one wakes up thinking, “Today I’ll damage my lungs,” but it happens too often. When I worked on corrosion inhibitors and research on enzyme reactions, I saw plenty of talented techs pull gloves straight on after lunch and dive in, only to start sneezing from unexpected powder exposure. Aminoguanidine bicarbonate fits into the same rulebook. If you don’t use gloves, goggles, and sometimes even a dust mask, you take an unnecessary risk—especially when weighing out powder or dumping waste.
Not everyone has access to a fume hood, especially in smaller facilities or teaching labs, so extra caution is needed. I always kept spills to a minimum and cleaned up right away. Trying to shortcut cleanup or pretending dust won’t become airborne doesn’t work out. Even small exposure can leave you coughing or fighting eye irritation.
What Actually Works: Lowering Risk
A sensible safety protocol addresses the realities of the chemical, not just theoretical dangers. Store aminoguanidine bicarbonate in tightly sealed containers, away from acids and sources of heat. Don’t lean on memory or instinct, even if you’ve handled “worse” before. Read through the manufacturer’s Safety Data Sheet, and update your process if new health info comes out.
Ventilation always ends up as one of the biggest difference makers. Even a basic bench-top extractor draws away dust, trimming down the chances of inhalation risks. Gloves and glasses should come standard, not as an afterthought. If a spill happens, scoop it up carefully—don’t sweep or vacuum dry powder since it can spread dust.
In training others, I stress straightforward habits: always label your containers, don’t assume any exposure is harmless, and never work alone with hazardous substances. Accidents need a second set of hands and a clear head. You can’t “tough out” chemical exposure—it doesn’t care if you’ve been around the block or just started out.
Why All This Matters
Chemicals like aminoguanidine bicarbonate might not be front-page material, but the stakes are high every day they get opened and weighed in the lab. Good habits don’t just protect you—they keep co-workers safe and help labs avoid the painful costs of accidents. It’s not just about regulatory compliance. A safety-first attitude belongs right down at the bench, mixed in with every scoop and every reaction started.
How should Aminoguanidine Bicarbonate be stored?
Understanding the Substance
Aminoguanidine Bicarbonate turns up in labs and production sites that work with specialty chemicals. Handling any chemical with complex properties calls for safe storage practices, and this compound is no exception. My time in research facilities made it clear: proper storage isn’t just about regulatory compliance, it’s about keeping people and products safe. Let’s dig into some concrete advice rooted in what works, not just what’s on paper.
Storage Environment Matters
This substance prefers a cool, dry location away from sunlight, heat, and any source of ignition. I once watched a team have to toss weeks of work because a sample drew in moisture from humid air—the compound had caked and started degrading. Humidity spells trouble, so controlling room moisture helps keep the material stable longer. No one likes tossing away valuable material due to storage mistakes.
Choose a section of your storage area designed to prevent temperature swings. Direct sunlight or heat can start chemical changes, so a climate-controlled shelf away from windows or furnaces has always struck me as the smart call. At one university, a dedicated cabinet just for chemical stabilizers made the difference between smooth workflow and repeat accidents.
Avoid Mixing with Other Chemicals
This isn’t a compound that gets along with everything. Strong acids, oxidizers, and reducing agents could trigger reactions. Contamination raises risks, so storing the material in its tightly closed original container helps. Whenever we mixed up storage by accident, it meant lost time and greater danger. In my experience, having a clear label and assigned spot on the shelf often does more than expensive warning systems ever could.
Container Choice and Maintenance
Sealed plastic or glass containers serve best. Metal often reacts with chemicals over time, sometimes releasing gas or changing the material’s properties. Chemical suppliers I know use thick, screw-top plastic jars and check lids for cracks or fit issues every time they take out a sample. It isn’t complicated—just one simple check keeps storage issues off your worry list.
Documenting Storage Practices
Anyone who’s worked in regulated environments knows the real value of documentation. Keep a log of storage locations and status checks. After a spill incident in one workplace, management tracked storage changes for every reactive chemical. It turned up small lapses that no one caught until they kept honest notes—lessons learned sometimes the hard way.
Training and Communication
Not every member of a team comes with years of chemical handling experience. Providing clear instructions about safe storage, including what not to store together and how to spot container problems, pays off quickly. Every person should know the emergency protocol if a spill or exposure occurs, and real-world drills teach better than lectures ever will.
A Safer Approach Protects Everyone
Careful storage of Aminoguanidine Bicarbonate protects staff, property, and research investments. Most mishaps I’ve seen in labs started with shortcuts or unclear instructions. Paying daily attention to storage temperature, moisture, compatibility, and container condition is not just good practice—it keeps things safer and helps work progress as planned.
What are the potential hazards or side effects of Aminoguanidine Bicarbonate?
What Sets Aminoguanidine Bicarbonate Apart
Aminoguanidine Bicarbonate doesn’t often draw attention from the general public, but anyone working in labs, chemical plants, or research centers has probably crossed paths with it. Its use stretches into the pharmaceutical and chemical synthesis spaces, and even pops up in materials science. That reach makes it important to ask hard questions about what risks this powder brings to the table. My experience handling chemical agents in a university lab taught me one thing: if you haven’t considered the human toll and environmental load of a compound, you’re skimming the surface.
Direct Impact on Human Health
Anyone who has opened a fresh container of Aminoguanidine Bicarbonate knows you can’t ignore its potential for irritation. Skin, eyes, and the respiratory tract are all fair game. The dust can sting the eyes and dry out your hands fast, and breathing in fine particles over time raises the odds of coughing, throat pain, or worse. Sometimes I’d catch myself rubbing my eyes before washing them, and that’s how you end up red-eyed and tearing up for hours.
The data on serious toxicity in humans can feel thin, but rodent studies showed large exposures result in headaches, dizziness, and nausea. One peer-reviewed article from Toxicology Letters highlights that aminoguanidine compounds alter blood pressure and kidney function in mammals at higher doses. This isn’t surprising, given their intended use as advanced glycation end-product inhibitors — they interfere with biochemical pathways, and those don’t always stop at their targets.
Long-term Hazards and the Unknowns
Long-haul exposure carries risks beyond simple irritation. If the powder soaks into gloves or sits on surfaces, repeated skin contact ups the chance of dermatitis, eczema, or even allergic skin reactions. Scientists haven’t settled whether long-term inhalation leads to chronic lung conditions, but the possibility should keep anyone spraying it around on their toes.
More worrisome, experiments in animals flagged certain aminoguanidine salts as potential mutagens — meaning they might mess with DNA. The evidence from a handful of cell culture and animal tests never landed on the kind of certainty you’d want for a blanket ban, but it lays a foundation for caution. Each step in a research pipeline that skips over hazard analysis just hands off risk to the next person.
Environmental Fallout
Leaving chemical powders in the wild has never ended well for rivers, soil, or wild creatures. Aminoguanidine Bicarbonate breaks down when exposed to moisture and air, but in the meantime, run-off could contaminate waterways. Aquatic toxicity data are scarce, but related compounds harm invertebrates. Keeping waste away from drains and using approved disposal channels has to stay a top priority.
Staying Ahead With Smart Habits
Steady habits matter more than lab safety posters. Simple rules like gloves that fit, up-to-date eye protection, and decent ventilation save headaches — or hospital trips. I learned to check expiration dates and store dry, reactive powders in airtight containers after a minor spill cooked an awful smell into our storeroom one summer during my post-grad years.
Companies and labs should update safety data sheets each year and train new folks on potential hazards. Routine air monitoring and regular reviews of disposal practices help keep everyone in one piece. These habits mean people spend more days healthy and go home with all their fingers, lungs, and eyesight intact.
| Names | |
| Preferred IUPAC name | aminomethanidinhydrogencarbonate |
| Other names |
Hydrazinecarboxamidine bicarbonate Guanidine, amino-, bicarbonate Aminoguanidine carbonate Aminoguanidine hydrogen carbonate |
| Pronunciation | /əˌmiːnoʊˈɡwænɪˌdiːn baɪˈkɑːrbəˌneɪt/ |
| Identifiers | |
| CAS Number | 2582-30-1 |
| Beilstein Reference | 3854940 |
| ChEBI | CHEBI:135749 |
| ChEMBL | CHEMBL127464 |
| ChemSpider | 161127 |
| DrugBank | DB01414 |
| ECHA InfoCard | 100.086.293 |
| EC Number | 207-894-1 |
| Gmelin Reference | 87123 |
| KEGG | C06547 |
| MeSH | D03AX Aminoguanidine Bicarbonate |
| PubChem CID | 139605 |
| RTECS number | BP8450000 |
| UNII | Y44VTJ1H73 |
| UN number | UN3249 |
| CompTox Dashboard (EPA) | DTXSID80993260 |
| Properties | |
| Chemical formula | CH6N4·CH2O3 |
| Molar mass | 136.11 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.54 g/cm3 |
| Solubility in water | Soluble in water |
| log P | -2.9 |
| Acidity (pKa) | 8.45 |
| Basicity (pKb) | pKb = 4.55 |
| Magnetic susceptibility (χ) | -65.0×10⁻⁶ cm³/mol |
| Dipole moment | 2.62 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 189.5 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | V03AX03 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. H315: Causes skin irritation. H319: Causes serious eye irritation. H335: May cause respiratory irritation. |
| Precautionary statements | P264, P270, P280, P301+P312, P305+P351+P338, P337+P313, P330 |
| NFPA 704 (fire diamond) | 2-0-0 |
| Autoignition temperature | 185 °C |
| Lethal dose or concentration | LD50 Oral Rat 1560 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 >2000 mg/kg |
| PEL (Permissible) | Not established |
| REL (Recommended) | 300 mg/m3 |
| Related compounds | |
| Related compounds |
Aminoguanidine Aminoguanidine hydrochloride Aminoguanidine sulfate Hydrazine Guanidine Biguanide |

