Aluminum Nitrate: Insights and Perspectives
Historical Development
People have worked with aluminum compounds for centuries, but the intentional synthesis and use of aluminum nitrate began to catch serious attention in the industrial era. Chemists in the 1800s, seeking better oxidizers and reagents, noticed this compound held promise. Early production methods relied on treating aluminum metal or its oxide with strong acids, paving the way for modern large-scale processes. By the time industries needed strong, highly soluble oxidizing agents, aluminum nitrate had moved from lab curiosity to commercial reagent. Those early discoveries seeded many current uses, especially where precise control over oxidizing reactions mattered.
Product Overview
Aluminum nitrate delivers a reliable and potent source of aluminum ions paired with strong oxidizing properties, found both as crystalline solid and in aqueous solutions, depending on what a process demands. Industries reach for it when looking to prepare catalysts, mordants, or specialty chemicals. The substance appears as a colorless, odorless solid that dissolves freely in water. Common applications rely on these characteristics, including water purification, paper manufacturing, and laboratory synthesis. Each batch intended for commerce will have documentation that spells out its purity and recommended use, keeping both safety and performance in mind.
Physical & Chemical Properties
This compound, with the formula Al(NO3)3·9H2O, often comes as a crystalline hydrate. Nine water molecules grab hold of each aluminum nitrate unit, forming deliquescent crystals that almost seem to pull moisture right from the air. At room temperature, they keep stable, but expose them to more heat or dry air and they start dropping water. Aluminum nitrate dissolves swiftly in water, producing acidic solutions. The nitrate groups make the solution a strong source of oxidizer, eager to react with reducing agents and ready to break down under heat or in contact with organics. Most chemists and engineers remember, it doesn’t mix well with flammable materials due to its reactive nature.
Technical Specifications & Labeling
Manufacturers supply aluminum nitrate with a close eye on content and impurities. Labels detail percentage of Al(NO3)3, water of crystallization, trace metal content, acidity, and typical physical data like melting point and density. Laboratories may demand 98% or greater purity for fine work, while industry may accept certain impurities as long as overall performance holds up. Specification sheets, usually passed along with each drum or bag, outline storage recommendations—keep sealed, avoid humidity, stay clear of heat sources—plus emergency first aid and exposure limits. Anyone planning to use or store this material should carefully review these details, as the nature of nitrates means you never take them lightly.
Preparation Method
Industrial-scale aluminum nitrate usually comes from reacting aluminum hydroxide or metallic aluminum with concentrated nitric acid. The general reaction:
Al(OH)3 + 3 HNO3 → Al(NO3)3 + 3 H2O
Producers favor starting with purified aluminum hydroxide since impurities can mess with crystal formation. The reaction is monitored for temperature and acid concentration. Controlled addition prevents runaway reactions and loss of yield. The mixture then cools, letting the hydrated crystals settle out. Careful washing and drying gives the typical non-hygroscopic product. Older methods weren’t so clean, sometimes leaving behind excessive acid or side products, but modern facilities use strict monitoring to keep the output safe, pure, and reliable.
Chemical Reactions & Modifications
Aluminum nitrate acts as a decent source of aluminum ions and a powerful oxidizing agent. In water, it dissociates easily, giving up Al3+ and nitrate ions. Add it to a reducing agent, and you have a redox reaction on your hands—reason enough to label storage and prevent accidental mixing. In the lab, it reacts quickly with bases, resulting in gelatinous aluminum hydroxide precipitate and a mix of other nitrate salts. Organic chemists sometimes turn to aluminum nitrate for nitrating aromatic compounds, offering an alternative to more aggressive traditional reagents. That versatility, combined with stable storage in solid form, helps this chemical keep its status in both production and research settings.
Synonyms & Product Names
Folks in the chemical business might encounter aluminum nitrate listed under names like Nitric acid, aluminum salt, or its full systematic name, aluminum(III) nitrate nonahydrate. Many catalogs simply refer to it by its formula or Cas number. Specialty brands sometimes tweak product names depending on the degree of purity or intended end use, but the core substance remains largely the same: a reliable nitrate salt with broad industrial and scientific relevance.
Safety & Operational Standards
Handling aluminum nitrate requires respect for its hazards. Its strong oxidizing property means it shouldn’t come anywhere near combustible materials or strong reducing agents. It can irritate skin, eyes, and the respiratory tract, so good practice means working with gloves, goggles, and proper ventilation. Storage generally involves cool, dry conditions, metal drums with inner liners, and clear labeling to prevent confusion. In my own work, routine double-checks of containers and clear signage in shared labs prevented accidents. Local regulations demand tracking how much is stored and where it’s kept, especially since large amounts could pose environmental or fire risks. Emergency procedures should always be posted, with neutralizing agents and spill kits close at hand.
Application Area
Aluminum nitrate finds regular use across different sectors. Water treatment plants add measured amounts to clarify and reflect contaminant removal from municipal streams. The paper industry values it as a mordant, helping dyes bond tightly to cellulose fibers. Analytical chemists use it for spotting ions in qualitative analysis. Explosives and propellant formulations often lean on its oxidizer capabilities, and the world of ceramics uses it during specialized glazing processes. In university labs, students mix it into experiment after experiment to grow skills in precipitation and redox chemistry, since its visible, reliable reactions make results easy to interpret. Each field leans on a different property—solubility, reactivity, or catalysis—meaning the market for aluminum nitrate rarely slows down.
Research & Development
Research keeps finding new ways to take advantage of aluminum nitrate’s reactivity and solubility. Over the last decade, scientists worked on tuning its release in soil amendments for agriculture, hoping to improve nutrient efficiency. Nanomaterial synthesis often uses aluminum nitrate as a clean source of aluminum during thermal decomposition. There’s a noticeable trend toward greener and safer synthetic methods. Laboratories continue to probe how nitrate ions interact with metal oxides and organics, studying catalytic pathways for waste treatment and fuel production. The work never stops; teams test alternative synthesis routes that cut down on hazardous waste, investigating solid-state reactions or using less energy-intensive conditions. Over time, safer production paired with higher purity will continue to drive new applications and expand the knowledge base.
Toxicity Research
Toxicologists studied the effects of aluminum nitrate closely, partly because both nitrate and aluminum ions come with their own baggage. Most findings point to limited acute toxicity at low exposure, but chronic or high-level exposure risks irritation, organ effects, and possibly long-term environmental harm. Studies in animals show aluminum compounds accumulate in soft tissue, so proper controls and limits in workplaces make sense. The nitrate piece can contribute to environmental eutrophication, boosting algae growth in waterways, so wastewater from plants using aluminum nitrate needs thorough treatment. Environmental guidelines set allowable discharge levels, driving both remediation and innovation in containment. Medical research stays alert to potential links between aluminum exposure and disorders such as Alzheimer’s, although definitive links remain unproven. Manufacturers and users carry responsibility for reducing risk, using personal protective equipment, and ensuring good laboratory practices stay non-negotiable.
Future Prospects
Looking forward, demand for aluminum nitrate connects directly to sectors like electronics, environmental remediation, and advanced manufacturing. As recycling of electronics ramps up, selective precipitation methods using this compound can help recover metals. Sustainable energy projects, especially hydrogen storage and battery technology, put new pressure on chemists to design materials with unique ionic properties—aluminum nitrate plays a role as precursor or catalyst. Construction materials research looks at its role in fire retardancy and high-performance concretes. My own hopes rest on smarter, safer production routes and better management of downstream waste, since the long-term health of both workers and the environment depends on these advances. As with most specialty chemicals, communication and sensible regulation shape its future as much as the underlying chemistry.
What is aluminum nitrate used for?
Hidden in Everyday Science
A lot of folks don’t realize how much of daily life connects to chemicals like aluminum nitrate. In a world obsessed with big-ticket inventions, aluminum nitrate stays mostly invisible. Still, its fingerprints show up across classrooms, labs, factories, and even our fields.
Teaching and Discovering in the Lab
Teachers and students use aluminum nitrate in real experiments. High school chemistry labs often pull out jars of the stuff to make lessons visible. Mixing solutions, making colored flames, or learning about reactions—it all gets clearer with simple, reliable chemicals. In college and research spaces, people dig further. Aluminum nitrate can help break down other compounds or reveal how metals and acids behave. In my student days, our group tested water samples with aluminum salts and learned plenty about acids, bases, and hidden contaminants. Experiments like that stick in the mind better than any textbook chapter.
Industry: Factories and Fields
Factories use aluminum nitrate to help make paints, pickles, and treated leather. Tanners rinse hides with solutions to create stronger, more flexible leather goods. Dye makers use it to set colors inside cloth, keeping blue jeans bright through cycles in the wash. In ceramics, the chemical prepares clays and helps glazes show richer color. Some manufacturers turn to aluminum nitrate when they need to clean metal or to get special surface qualities without harsh acids.
Agriculture isn’t left out. Farmers and soil scientists sometimes use aluminum nitrate to test soil acidity. With modern environmental concerns, testing for heavy metals and pH changes has grown into a huge challenge, especially for those near cities or older industrial sites. Results from these tests guide planting patterns and fertilizer use. Better data can help keep toxins out of crops and drinking water, giving peace of mind to anyone who eats food from that ground.
Public Safety and the Environment
Like any strong chemical, there’s a trade-off. Aluminum nitrate can burn skin or eyes; it needs careful handling. I’ve seen workplaces improve when teams set up clear training and stockpile gloves, goggles, and proper ventilation. Disposal matters, too, since it dissolves quickly and would otherwise add nitrogen to water sources. Extra nitrogen in local streams can spark algae blooms, which kill fish and threaten nearby farms.
Pushing for Solutions
Some old habits still pop up, especially in smaller shops that reuse containers or wash runoff into storm drains. Honest, hands-on education can help. Posters in break rooms, onsite demos, and real-world stories come through louder than rulebooks. Regulators and managers who ask workers for their feedback often spot problems faster than any outside inspector. Changes in packaging and clearer safety labels also make a difference.
At every stage—from mixing in a test tube to shipping drums cross-country—real improvements start with workers, teachers, and neighbors who understand what chemicals can do. Aluminum nitrate won’t grab headlines, but respect for it pays off with safer schools, cleaner rivers, and stronger community trust.
Is aluminum nitrate hazardous to health?
Daily Life and Chemical Risks
Working in a lab for a few years teaches a person how certain chemicals demand more respect than others. Aluminum nitrate tends to show up on lists that warn users to pay attention. In a world where chemical safety often gets reduced to nameless numbers, dealing with real compounds makes everything personal.
Direct Exposure and How it Affects People
Most folks never encounter pure aluminum nitrate at home, but plenty of workers handle it in research, water treatment, or making specialty products. Inhalation of its dust can catch even experienced professionals off guard. Eyes itch, throats burn, headaches linger. These aren’t stories from a century ago—these are common complaints from those moving or measuring the powder today.
Documented Health Effects
Peer-reviewed articles and safety data sheets spell out the concerns. If someone gets the solid or solution on their skin, irritation follows. Splashed in the eyes, the burning and redness build fast. Breathing the dust brings on coughs and tightness in the chest. Swallowing even moderate amounts by accident causes stomach pain and nausea. Firefighters and emergency medical workers recognize these symptoms and train for chemical incidents where aluminum nitrate gets released.
Environmental Harm Signals Human Risk
The stuff washes away easily and interacts with soil and water. In my area, the wastewater treatment workers always keep an eye on nitrate compounds, since too much causes trouble for fish and can pollute the water supply. What harms the ecosystem often finds a path to humans, increasing risks bit by bit even when each step seems minor.
Why Proper Safety Measures Matter
Stories of poor chemical storage echo in every lab. One person forgets gloves, another skips the goggles, and the consequences add up. Local hospital records confirm the uptick in eye injuries and accidental poisonings linked to industrial-grade nitrates. At my old university, a single spill closed off an entire floor for half a day. That incident forced the whole department to rethink training and spill preparedness.
Rules and Solutions Make a Difference
OSHA and NIOSH, respected safety authorities in the U.S., set clear exposure limits for soluble aluminum compounds. Many industrial sites use fume hoods and dedicated safety showers because those simple tools save time and health. Workers train every new team member until routines stick. My own practice always revolved around triple-checking labels and closing containers tight. Good habits start with education, but real culture change follows when supervisors and employees watch out for each other.
Handling and Prevention Over Guesswork
Anyone handling aluminum nitrate benefits from learning the emergency measures—flush with plenty of water, seek fresh air, seek medical attention for major concerns. Clear labels, available personal protective gear, and practical safety drills transform confusion into confidence at every level. Tools and rules only go so far if people cut corners. Trust in coworkers helps, but systems catch mistakes before they turn into emergencies.
Creating a Safer Place for Everyone
One tech at my facility set out plain-language safety posters, and in a year, our incident rate dropped. Sharing information openly—without jargon—keeps people grounded. Whether it’s a hands-on demonstration or a reminder to wash up after a shift, building these habits protects everyone who steps into a room with aluminum nitrate.
What is the chemical formula for aluminum nitrate?
Why Chemical Formulas Matter in Everyday Science
Looking up the formula for a compound like aluminum nitrate takes me back to crowded high school labs, marker stains on hands, and that anxious feeling before a chemistry quiz. We use science every day—sometimes without even realizing it. Kitchen recipes, gardening tricks, cleaning supplies, DIY repairs, all involve some trial, error, and chemistry. Aluminum nitrate, though not something you’ll find lined up in your spice cupboard, has a real presence across fields, from factories and classrooms to environmental cleanup.
The Formula at the Center: Al(NO3)3
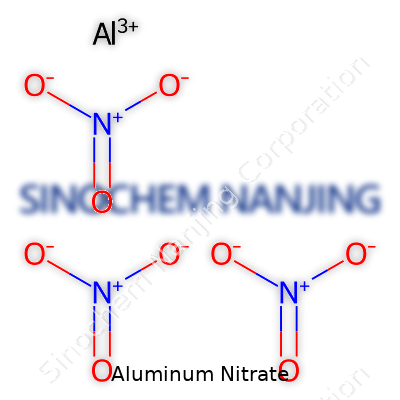
The formula for aluminum nitrate is Al(NO3)3. One aluminum atom, three nitrate groups. The nitrate has its own structure—a nitrogen atom surrounded by three oxygens, carrying a -1 charge as a unit. Aluminum, with its +3 charge, pairs handily with three nitrates to balance everything out. Call it chemical harmony or just a rule of nature, but everything adds up cleanly—no leftover charges.
Why This Compound Matters
Years ago, a family friend started a business treating municipal water in our town. I watched as massive bags with chemical names I could barely pronounce rolled into the plant. Among them: aluminum nitrate. They used it to help get rid of phosphates and pesky impurities that turned the town’s tap water murky every spring. It sounds almost industrial, but communities across the world rely on this compound for cleaner, safer water. It’s less about chemistry class and more about public health.
Lab techs mix aluminum nitrate with sodium carbonate or sodium hydroxide to create alumina gels, useful in everything from water purification to making tiny refining catalysts. In my college years, I met researchers using it to prepare special ceramics. Some even apply it when they want to produce a controlled release of nitrogen, for fertilizers or certain lab applications.
Staying Safe Around Aluminum Nitrate
Everyday folks don’t usually handle aluminum nitrate powder, but it’s good to know what you’re dealing with. This compound dissolves easily in water and acts as a strong oxidizer, which means it plays nicely with water but not so much with organic materials or heat. There’s real risk if someone stores it near flammable stuff. That’s not just a rule, it’s a recommended practice after learning how quickly certain oxidizers can fuel a fire. Stories from local fire departments show small accidents can spiral fast if chemicals and caution aren’t properly paired.
Challenges of Chemical Access and Transparency
A lot of schools and small labs struggle to safely manage chemicals like this. Funding shortfalls sometimes push basic safety updates down administrators’ lists. In my volunteer work, I’ve seen chemical storage closets with faded labels and open bags. It feels risky. Wider access to practical chemical safety training would go a long way. If schools, municipalities, and small labs could easily find up-to-date safety guides and affordable personal protective gear, accidental exposures and fires would drop.
Building a Responsible Chemistry Community
The chemical formula for aluminum nitrate might look simple: Al(NO3)3. The real impact comes with education, transparency, and responsibility. Whether you’re purifying water, making ceramics, or just curious about what’s in your school’s storeroom, accurate chemical knowledge and up-to-date safety practices mean more trust and less risk.
How should aluminum nitrate be stored?
Understanding the Nature of Aluminum Nitrate
Aluminum nitrate brings its own set of challenges to any lab or storeroom shelf. This chemical attracts moisture from the air with a remarkable hunger, leaving clumps or even watery pools if the cap isn’t tight or the room keeps a dampness to it. More than once, I’ve opened a bottle that someone left a bit loose, only to find a sticky mess and glare at the label that should’ve warned me. On top of that, the compound mixes with water to make acidic solutions—nothing fit for careless handling. I’ve watched careless storage eat through shelves and leave surprise stains that never quite wash away.
Getting Storage Conditions Right
Keeping aluminum nitrate in top shape calls for a sealed container—one that holds tight, preferably made of glass or high-grade plastic. Even a small leak lets air sneak in and start the slow melt. I keep these chemicals off the bottom shelf, far away from splash-prone sinks and never near anything flammable. This isn’t just habit; the stuff reacts with reducing agents. Sitting aluminum nitrate next to a simple bottle of metal powder or even fuel oil creates a risk nobody wants to face during a long shift.
Coolness and dryness give the right protection. A dark, climate-controlled room often works best. Humidity invites clumping, while temperature swings encourage condensation; both can change a clear set of crystals into sludge or unexpected liquid. Lab managers always talk about using desiccators (those airtight boxes with moisture-absorbing beads at the bottom), and for once, this step really does cut down on headaches over the long haul. Between the proper jar and a dusting of silica gel, bottles from last year can look almost new if checked regularly and handled carefully.
Personal Responsibility and Overall Safety
Common sense mixes with training here. Every dusty shelf, faded label, and jar half-forgotten in the corner stands as a silent warning. I’ve seen people shortcut steps because they felt too busy or too sure in their knowledge; a rusty crust on the lid or a warped plastic lid told the story later. Cleaning up spills isn’t just a chore—it means wearing real protection: gloves that resist acids, safety glasses, and quick access to plenty of fresh water.
Alarm bells ring in good labs for a reason. Storage rooms often post clear instructions and update them after every close call. Keeping a spill kit nearby, along with neutralizing agents, gives everyone breathing room to make corrections on the fly. I remember a time a student dropped a bottle, the glass shattered, vapors starting to rise—without the right storage procedures and an alert team, it would have burned through both bench and reputation.
Looking for Better Solutions
Some companies invest in digital humidity and temperature trackers. These offer precision and keep a record of every blip that might spell trouble. Older labs benefit from simple fixes—rotating stock to avoid forgotten containers, tightening labeling rules, and running regular checks where everyone on the team gets involved. The simplest habits go furthest: closing lids, cleaning drips, locking up after every use.
People ask why this matters. Imagine the risks to staff, to property, and to future experiments if chemicals break down or spill. The costs of one mistake can far outweigh a lifetime of careful habits, and fixing these problems at their root—through training, equipment, and real accountability—pays off every single day.
Is aluminum nitrate soluble in water?
Solubility Matters
I spent a lot of years with my hands in science projects and pool supplies, so it’s hard to ignore how some chemicals seem to disappear almost instantly when they touch water. Aluminum nitrate catches the eye because it doesn’t need much persuading to dissolve—just pour it in and watch it vanish. That property sets it apart from a lot of other compounds out there, including other aluminum salts that tend to be stubborn and hang around longer in water or soil.
Everyday Relevance
Aluminum nitrate isn’t something most folks keep under the kitchen sink, but its solubility shapes actual industry practices. Water treatment plants, for example, often rely on chemicals that dissolve fully and quickly. The quick-dissolving nature of aluminum nitrate means it doesn’t leave behind any gritty residue. It acts fast, mixes easily, and doesn’t clog pipes, which cuts down on headaches for workers keeping municipal water clear and safe. Speaking as someone who’s scraped stubborn stuff out of filters, fast solubility saves both time and repair costs.
Fertilizers, Labs, and the Classroom
Schools and science labs stock this compound because students need to learn about reactions that require completely dissolved ions. In fertilizer production, turning raw ingredients into solutions that plants can take up makes a difference. Plants can’t use aluminum directly, but aluminum nitrate supplies nitrogen—key fuel for growth. Farmers and gardeners look for products guaranteed to dissolve in irrigation water since unpredictable solubility invites a patchwork result, wasting both money and effort.
Safety and Environmental Impact
On the flip side, fast solubility means runoff can carry aluminum nitrate far away from its starting point. Excessive nitrate in water poses risks—streams get choked with algae, aquatic life suffers, and drinking water quality takes a hit. That’s not just textbook concern; plenty of communities have flagged contaminated wells in recent years. The Environmental Protection Agency (EPA) monitors nitrates because of links to health problems, especially for infants.
Responsible Use and Smarter Choices
Anyone using aluminum nitrate in large amounts should remember, what dissolves goes somewhere. Simple steps like carefully measuring doses, monitoring for leakage, and storing bags indoors out of the weather cut back on waste. For big operators, using on-site treatment and runoff containment stops problems before they get to the groundwater table. If the job allows, switching to less soluble alternatives reduces risk even more.
Expert Voices and Proven Facts
Chemists have checked and rechecked solubility. Aluminum nitrate ranks high, dissolving at more than 70 grams per 100 milliliters of water at room temperature. That’s not just my word—peer-reviewed handbooks and safety data sheets share this number. Its place on lists of highly soluble inorganic salts isn’t up for debate.
Key Takeaway
Understanding how aluminum nitrate plays with water keeps mistakes small, whether you’re fine-tuning a high-school experiment, running a treatment plant, or farming on a large scale. The science is settled: if you mix aluminum nitrate with water, it goes right in. Respect that chemistry, and you’ll sidestep a lot of real problems down the line.
| Names | |
| Preferred IUPAC name | aluminium trinitrate |
| Other names |
Nitric acid, aluminum salt Aluminum(III) nitrate Aluminum trinitrate Aluminium nitrate Aluminium trinitrate Aluminum nitrate nonahydrate |
| Pronunciation | /əˈluː.mɪ.nəm ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | 13473-90-0 |
| Beilstein Reference | 80387 |
| ChEBI | CHEBI:32599 |
| ChEMBL | CHEMBL1201563 |
| ChemSpider | 15616 |
| DrugBank | DB14638 |
| ECHA InfoCard | ECHA InfoCard: 100.028.763 |
| EC Number | 221-854-6 |
| Gmelin Reference | 778 |
| KEGG | C09971 |
| MeSH | D008545 |
| PubChem CID | 101912 |
| RTECS number | BD0825000 |
| UNII | 9L6Q14F4UW |
| UN number | UN1438 |
| Properties | |
| Chemical formula | Al(NO3)3 |
| Molar mass | 213.00 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 2.72 g/cm³ |
| Solubility in water | 52 g/100 mL (20 °C) |
| log P | -0.48 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 3.11 |
| Basicity (pKb) | 9.12 |
| Magnetic susceptibility (χ) | +59.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.47 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 146.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1334 kJ/mol |
| Pharmacology | |
| ATC code | V07AY05 |
| Hazards | |
| Main hazards | Oxidizing agent, causes severe skin burns and eye damage, harmful if swallowed, may intensify fire. |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS03,GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H272, H315, H319, H335 |
| Precautionary statements | P210, P220, P221, P264, P280, P301+P330+P331, P305+P351+P338, P308+P313, P370+P378, P501 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Autoignition temperature | 400 °C (752 °F) |
| Lethal dose or concentration | LD50 (oral, rat): 3671 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 3,670 mg/kg |
| NIOSH | UN1438 |
| PEL (Permissible) | PEL: 2 mg/m³ |
| REL (Recommended) | 2 mg/m³ |
| Related compounds | |
| Related compounds |
Aluminum sulfate Aluminum chloride Sodium nitrate Potassium nitrate |

