Aluminum Dichromate: From Laboratory Curiosity to Industrial Chemical
Historical Development
Aluminum dichromate has a story shaped by discoveries in both basic chemistry and real-world necessity. Chemists first observed the strong oxidizing properties of dichromate compounds two centuries ago, as the chemistry of chromium gained attention after French chemist Louis Nicolas Vauquelin isolated the element at the tail end of the 18th century. The union of aluminum and dichromate likely emerged out of efforts to expand the catalog of metal dichromates for pigment, tanning, and oxidation reactions throughout the 19th and early 20th century. In research circles, chemists started blending chromium(VI) oxoanions with lightweight metals to explore new possibilities in catalyst design and analytical reagents, often using simple double displacement reactions between dichromate salts and aluminum salts in aqueous solution. The search for efficient oxidizing agents and pigment materials kept interest in this compound alive, even as its hazards became more widely recognized.
Product Overview
Today, laboratories and some niche industrial users still produce aluminum dichromate on demand, though it never reached the commercial scale of potassium dichromate or sodium dichromate. Its limited solubility in water and less predictable behavior compared to other dichromates have kept it on the fringes, appreciated mainly by chemists needing a source of chromium(VI) in a less common form. Most orders flow through chemical supply houses under tight regulation. Users expect a solid, orange-to-red crystal or powder, always handled in sealed, labeled containers due to strict safety mandates. Some specialty glassmakers and research synthesis chemists rely on its oxidizing punch or its ability to provide chromium in certain reactions.
Physical & Chemical Properties
Aluminum dichromate stands out thanks to its striking red-orange hue, a telltale sign of chromium(VI) at work. It forms crystalline solids, though one quickly notices poor solubility in both water and organic solvents compared to its alkali counterparts. It breaks down in water with slow but steady hydrolysis, gradually shifting toward barely soluble chromium(III) hydroxide and a range of oxyanions in solution. Its thermal stability falters at moderate temperatures: heat breaks it down to chromium oxides and leaves behind brittle metal oxides. As an oxidizer, it ranks among the strongest, grabbing electrons away from organic matter or metals with little provocation. The powder often clumps, takes on moisture, and stains everything it touches a persistent orange—signals to any careful chemist that both gloves and an exhaust hood are essential in the lab.
Technical Specifications & Labeling
Chemical suppliers must spell out technical details with precision. Most labels carry the formula Al2(Cr2O7)3, a purity rating (often 98% or greater), batch number, and a stark warning about toxicity and reactivity. Particle size may matter for certain applications, though the market rarely demands specialized grades. Materials Safety Data Sheets (MSDS) accompany every purchase, formatted to international standards like the Globally Harmonized System (GHS). Labels display hazard pictograms—a flame over a circle to warn of oxidizer risk, a dead tree and fish for environmental hazard, an exclamation mark for health risk. Storage containers must seal tightly, remain in cool, ventilated cabinets away from fuels, acids, or organic material. Labs keep logs of who handles the compound, how much gets used, and how they dispose of any residue.
Preparation Method
Lab technologists usually prepare aluminum dichromate by reacting a sodium dichromate solution with a water-soluble aluminum salt such as aluminum sulfate. The reaction releases sodium ions and precipitates an orange-red solid that can be filtered, washed, and dried. Few modern chemists scale up this process, mainly because the product forms in low yield, tends to lose chromium(VI) through hydrolysis, and challenges any cost-effective filtration. The classical double displacement relies on excess reagent and precise stoichiometry to control contamination from leftover sodium or sulfate. Some researchers skip intermediates and add dichromic acid slowly to aluminum hydroxide, trying to coax out bigger crystals. Every method calls for careful waste management: anything containing extra chromium needs deactivation before disposal.
Chemical Reactions & Modifications
Aluminum dichromate stands as a textbook oxidizing agent. Organic chemists sometimes exploit its relentless appetite for electrons, using it to convert alcohols to aldehydes or ketones, or to break up stubborn carbon bonds in complex molecules. Its reactions can turn dangerous outside tightly controlled systems. As it reacts, the orange or red color fades to dull green or blue-green—evidence of chromium(VI) morphing into safer chromium(III). Strong acid boosts its reactivity, though this drives the release of corrosive fumes and heightens the risk of toxic exposure. Attempts to coat surfaces or pigment glass involve melting or fusing aluminum dichromate with silicates or clay, which releases more stable chromium oxides while the aluminum supports a finished product. Modifications remain limited, since almost every tweak leads to a product less stable than the starting material, or less useful than the far safer potassium and sodium dichromates.
Synonyms & Product Names
The chemical registry lists this compound as aluminum dichromate, but a buyer might bump into names like aluminum chromate(VI), dialuminum dichromate, or the more formal aluminium dichromate (British English spelling). Trade catalogs refer to it under its CAS number, 7789-23-3. Mislabeling can occur in less regulated settings, but international law sets a clear expectation for accurate identity—no supplier that values its reputation risks cutting corners on correct phrasing.
Safety & Operational Standards
Safety sets the rhythm for anyone working with aluminum dichromate. Chromium(VI) compounds can trigger severe health effects: inhalation of dust or vapors attacks the lungs, while skin contact risks painful ulcers or allergic reactions. Regulatory bodies insist on fume hoods, nitrile gloves, goggles, and disposable gowns as bare minimum. Any spill receives immediate cleanup with special neutralizing agents; the company health officer trains workers to avoid dry brushing and never uses vacuum cleaners. Disposal takes top priority, since chromium waste falls under hazardous waste law in the US, Europe, and beyond. Transport regulations demand secondary containment, emergency plans, and—often—notification to local authorities. Medical monitoring for regular users picks up signs of chromium exposure before health problems set in. Anyone with personal experience working in a chemistry lab knows the tension that comes with handling a bottle of orange powder, and the relief after it has been safely neutralized and removed.
Application Area
This compound’s reach stays fairly specialized. Industrial glassworkers value it for certain decorative effects impossible to achieve with other chromium sources. It plays a role in sensors and test kits developed for detecting organic molecules, since its strong oxidizing power can trigger clear, visible changes. Researchers might use it as a model compound or a standard in calibration routines. Alkali dichromates outpace it in bulk metal treatment, photography, and wood preservation, meaning aluminum dichromate fills in gaps or covers applications calling for a particular balance of aluminum and chromium in a process. Rarely, a catalyst designer or synthesis chemist finds just the right use for its oxidizing punch.
Research & Development
Academic work on aluminum dichromate slowed as knowledge of chromium toxicity deepened and safer substitutes appeared. Research articles from the 1970s and 1980s described kinetics, crystallography, and redox chemistry—today’s interest sprinkles into studies on remediation or new detection methods for chromium(VI) in waste streams. A handful of researchers explore surface catalysis or the structure-directing effects of aluminum in mixed oxides. The emphasis clearly tilts toward safer chemistry, more sensitive environmental detection, and engineering routes to limit chromium’s health risk. My own time in the laboratory backed up this trend: as techniques advanced, aluminum dichromate shifted from a go-to reagent to a chemical curiosity, pulled off the shelf only for specific, high-value work or to investigate chromium’s stubborn environmental persistence.
Toxicity Research
The deep orange-red hue signals a danger hidden at the molecular level. Toxicologists long ago showed chromium(VI) reaches deep into the lungs, damages DNA, and threatens both worker and community health. Studies on rats and mice link exposure to everything from skin burns to cancer. Soil and water contaminated with dichromates show lasting effects, disrupting microorganism activity and harming aquatic life. Regulators pay close attention: occupational limits demand air sampling, urine tests for exposure, and stringent clean-up targets for contaminated sites. Chemical companies fund research on less hazardous oxidants, and enforcement of strict exposure limits on the factory floor helped to drive much of the dichromate chemistry out of daily use.
Future Prospects
Aluminum dichromate’s future depends on the push for cleaner and safer alternatives, as society pulls back from hazardous chromium(VI) compounds. Advanced catalysts, oxidizing agents based on benign elements, and green synthesis technology keep inching forward. Some researchers focus on new spinel materials or hybrid metal-oxide particles that capture the positive attributes of dichromate without the legacy of toxicity. Work continues into more effective remediation techniques, using reduction with iron or sulfur compounds to sweep up and detoxify waste streams—a necessity in places where old stocks or contaminated soils persist. At the same time, the legacy of aluminum dichromate underscores the need for strong oversight, clear safety information, and honest dialogue between manufacturers, scientists, and regulators.
What are the common uses of Aluminum Dichromate?
Understanding What Aluminum Dichromate Brings to the Table
Aluminum dichromate isn’t a name people hear every day, but it plays a role in several industries, usually out of sight from daily life. With a deep orange color and strong oxidizing power, this chemical gets put to work in places where reliability counts. Over the years, I’ve run into it most in industrial labs and manufacturing, so I’ll share what I’ve learned in those settings.
Industrial Applications in Oxidation and Organic Synthesis
Chemists know aluminum dichromate as a workhorse oxidizing agent. Instead of wasting time with slow changes, this compound speeds up reactions and drives tough processes home. Think about turning alcohols into acids or aldehydes—this substance takes on the job cleanly and without much fuss. Organic labs and research teams rely on it when they can’t use gentler options, since it doesn’t back down from thick, stubborn chemical structures. In practice, it helps shave hours or even days from reaction times, which can make a difference between meeting a deadline or not.
Role in the Production of Dyes and Pigments
The world wouldn’t be as colorful without chemicals like aluminum dichromate. In the process of making strong dyes and pigments for plastics, textiles, and paints, manufacturers often use this compound to ensure vivid, lasting colors. Having consulted for pigment makers, I saw how certain reds and oranges don’t come out quite right without heavy-traffic oxidizers like this one. It works by opening up chemical rings or rearranging molecules, which can make a simple dye ten times more resistant to fading in sunlight.
Surface Treatment and Metal Finishing
Metal parts rust if left alone, and not every coating sticks to bare metal. Industrial workers use aluminum dichromate in surface preparation, especially for aluminum and steel. By cleaning, etching, and passivating the surface, it helps primers and other coatings grab on for years instead of months. This kind of surface work isn’t flashy, but it matters every time you walk under a bridge, drive a car, or use a metal tool. It’s part of that invisible workforce preventing accidents and saving repairs. From my hands-on time in automotive plants, I can say corrosion creeps in fast—any tool that holds it back earns respect.
Lesser-Known Uses Worth Noting
Some industries put aluminum dichromate to work in chemical analysis and testing. Metallurgists sometimes use it as a reagent for identifying elements during quality control checks. Labs also use it for cleaning glassware that regular soap won’t touch. The stuff will even strip away stubborn organic stains, but safety isn’t negotiable. It’s toxic, so no one should handle it without real protection and training. That’s a lesson I got early—one careless moment can mean burns or worse.
Balancing Benefits with Responsibility
Aluminum dichromate delivers real results, but safety stays front and center. Its toxic nature puts responsibility on the handlers. Strict safety laws, right disposal methods, and continuous training all keep accidents down. Some companies are searching for greener alternatives, but not every process has a ready substitute. Until that changes, workers, suppliers, and researchers all have to stay sharp, not just for productivity, but for everyone’s safety. No corners can get cut when dealing with strong chemicals like this one.
Is Aluminum Dichromate hazardous to health?
Looking at Hazardous Chemicals—Straight Talk
Aluminum dichromate isn’t a chemical you stumble across in the kitchen or hardware store. It’s uncommon in daily life, but it shows up in some labs and certain manufacturing. The conversation about this compound always swings back to health risks, and honestly, the concerns aren’t overblown.
So, What’s the Worry?
The main thing to understand is that this stuff isn’t just an ordinary aluminum salt. Chromates, the family it belongs to, contain hexavalent chromium (Cr(VI)). If you’ve heard stories about dangerous chrome compounds, they weren’t fiction. The science on hexavalent chromium is as established as it comes. It’s a strong oxidizer—meaning it can react harshly with living tissue. Even at very low levels, it can spark trouble for the human body.
Once it lands on skin, chromate can trigger allergic reactions—rashes, ulcers, or worse. Dust or fumes drifting in the air are even more dangerous. Breathing that in means giving those tiny particles a direct path to the lungs, and the links to cancer are solid. Studies among workers exposed to hexavalent chromium show lung cancer cropping up much more often. Risk doesn’t stop at cancer—people develop nasal ulcers, chronic nosebleeds, asthma-like symptoms, and damage to the kidneys and liver. I’ve seen plenty of veteran chemists treat chromate spills as code-red situations, and they do that for good reason.
PPE Isn’t Optional for a Reason
The right equipment separates safe work from a trip to the ER. Gloves, goggles, and fitted respirators don’t just look good for lab safety posters—they actually block the routes chromate uses to invade the body. Clothes that fully cover the skin cut down the chance for contact, and good ventilation stops workers from breathing it in. Labs with poor airflow or workers who brush off their gear don’t get away with it forever. Eventually the exposure adds up.
Facts Matter: What the Agencies Say
The World Health Organization, OSHA, and EPA all agree about the risk. Hexavalent chromium isn’t a “maybe” carcinogen—it’s marked as a proven cause of cancer. Workplace limits exist for a reason. OSHA sets the exposure cap at just 5 micrograms per cubic meter of air over an eight-hour workday. Even that small amount can’t be dismissed. If you check recent fines and citations, some companies still miss the mark, and workers pay the price in their health.
What Companies—and Workers—Can Do
No chemical can be handled on trust. Anyone in a facility using aluminum dichromate needs training that actually sticks. Not a quick slideshow—a real walk-through of worst-case scenarios and emergency response. Routine air monitoring shows if the place is safe, not just the paperwork. Spill kits near work stations, medical checks for team members, and swapping out aluminum dichromate for a less risky chemical all rank higher on the “to do” list than cutting corners. Every company that uses hazardous stuff has already learned the cost of doing less—the lawsuits and lifelong health impacts aren’t worth it.
A Final Word: Don’t Underestimate the Risks
Many chemicals deserve respect, but few are as unforgiving as chromates like aluminum dichromate. If you see it in a lab, personal protection comes before productivity. If you work with it, demand the right gear and proper procedures. Lives really do depend on how seriously risks like this get taken.
What is the chemical formula of Aluminum Dichromate?
The Formula and How It Comes Together
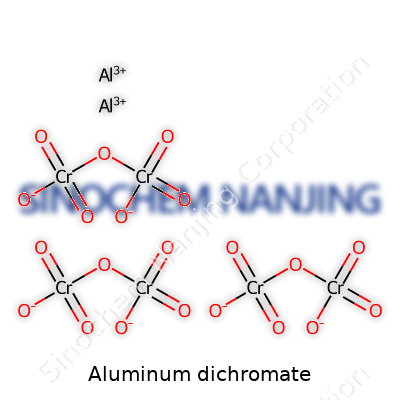
Aluminum dichromate has the chemical formula Al2(Cr2O7)3. This formula comes from simple ionic rules: aluminum brings a 3+ charge, dichromate brings a 2- charge, and balancing these requires two aluminum ions for every three dichromate ions. It’s not just a collection of symbols — that formula tells you exactly how many atoms come together, and what kind of interactions they’ll have. I remember my early chemistry lab, face-to-face with a yellow-orange beaker, startled by how quickly a small scoop could stain hands or desktops. The color of the dichromate ion has stuck with me as much as the formulas ever did.
Why Does This Matter?
Many folks think this compound sits quietly in a laboratory, but it looms large in discussions about both industry and safety. Crystals like these have a role in pigments, oxidation reactions, and sometimes even prepping metal surfaces. The dichromate inside brings a strong oxidation hand. This property helps strip electrons in reactions, making it valuable but also potentially hazardous. I once saw a messy spill at a plating shop. Cleanup wasn’t just about sweeping—it needed real caution. The hexavalent chromium inside dichromate seizes headlines for all the wrong reasons. Breathing in dust or getting it on your skin isn’t a minor problem. Research from the National Institute for Occupational Safety and Health (NIOSH) flags hexavalent chromium as a carcinogen, so working with aluminum dichromate takes careful handling.
The Bigger Chemical Story
If you break down the formula, every piece carries weight. Aluminum’s lightweight, strong nature lifts entire industries — think airplanes, construction, and sporting goods. Dichromate’s job as an oxidizer, though, draws heavy regulation. For all its clever chemistry, the environmental footprint stands out. Where chromium travels, contamination risks track along. A study led by researchers at Johns Hopkins showed groundwater near chromium sites sometimes carries levels that concern scientists and homeowners alike. This isn’t theory for communities next to old chromate works. They’ve seen health impacts in neighbors, not just labs.
Safer Handling and Future Outlook
Regulatory agencies like OSHA don’t just wave their hands at these issues. They set exposure limits and require gear and ventilation. Even high school labs have moved away from free-wheeling use of strong dichromate salts. In industries where these chemicals linger, stricter reporting and restricted waste disposal keep dangers in check. I’ve visited plants swapping older, riskier chemicals for safer modern alternatives. Some have turned to less harmful oxidizers or sealants for metal finishing, showing that innovation follows real concern.
It pays to understand what a chemical like aluminum dichromate really represents — not just its formula, but the real-world implications packed inside. This turns simple memorization into a call for respect and responsibility, especially in schools, shops, and factories where it might show up. Seeing both the utility and danger, and pushing for alternative solutions where possible, keeps chemistry moving in the right direction.
How should Aluminum Dichromate be stored safely?
Understanding Aluminum Dichromate’s Risks
Anyone who’s worked in a lab or an industrial setting knows that certain chemicals demand respect. Aluminum dichromate belongs in that group for a good reason. Bright orange, highly toxic, and an oxidizer that doesn’t forgive mistakes, this compound presents health and safety challenges that go beyond what's printed on its label.
Why Proper Storage Matters
Aluminum dichromate earns its reputation for being hazardous because it releases toxic chromium(VI) compounds. Breathing in its dust can damage lungs and poison the body. Skin contact causes severe irritation or burns, and even small spills make a mess that’s tough to clean safely. From what I’ve seen, the biggest drama happens in areas that undervalue preventive thinking.
Real-World Advice for Storage
Many chemical incidents happen not during wild experiments, but while moving, storing, or opening compounds. In my time at a university research facility, I saw how casual storage turns into real danger. A bottle perched on a top shelf, the wrong kind of container, or a dusty old label — just a few steps from disaster.
Aluminum dichromate belongs in a dedicated chemical storage cabinet. Flammable storage rules do not fit this material. Instead, focus on a well-ventilated, cool, and dry location. Keep it far from organic materials, paper, wood, or anything flammable, because this oxidizer can start fires without warning. Forget cardboard boxes, plastic bags, or any container that doesn’t close tightly and resist corrosion. Use glass or high-grade polyethylene. Label containers clearly and make sure the hazard symbol sticks out.
Handling and Personal Protection
Experience teaches that personal protective equipment is never optional with aluminum dichromate. Nitrile gloves, lab coats, and chemical splash goggles should become habits. Some folks think a dust mask is enough, but that can’t block chromium(VI) particles. A properly fitted respirator makes more sense for anyone opening or transferring the powder.
I’ve watched even seasoned chemists get careless under time pressure. Accidents usually follow. Wash hands thoroughly after handling, and never leave residue on benches or containers. Change gloves and discard them through hazardous waste channels, not regular trash.
Minimizing Exposure and Accidents
Remote storage locations seem inconvenient, but distance really helps reduce unintentional exposure. Keep inventory small — nobody needs to keep more of this stuff than a project requires. Record every transfer and disposal, and never store aluminum dichromate near acids or reducing agents, which can react violently if they come together.
Spill kits designed for oxidizers are worth the investment. I’ve seen that quick access to neutralizing agents stops problems from spreading. Staff should know how to use these kits, not just see them gathering dust under a bench.
Safe Disposal
Never pour leftover aluminum dichromate down a drain. Every community’s disposal rules look a little different, but most require hazardous waste pickup. Follow local, state, and federal guidelines without shortcuts. Handing this work off to professionals costs less than dealing with fines, lawsuits, or the health effects that follow sloppy disposal.
Building a Safety-First Culture
No label or storage cabinet can replace the value of training. Regular safety meetings, refresher courses, and real discussion about what can go wrong turn rules from paperwork into something people trust and follow. Safe storage habits do not come from fear — they grow from respect for what these materials can do, and a commitment to keeping everyone safe.
What precautions should be taken when handling Aluminum Dichromate?
Why Aluminum Dichromate Demands Respect
If you’ve worked in a chemistry lab, you probably recognize that orange-red color from across the room. Aluminum Dichromate isn’t some routine cleaning powder or seasoning you can toss around without worry. This stuff is toxic, both for the folks handling it and for the environment. Breathing in the dust or getting it on your skin can set off allergic reactions, damage organs, and even cause cancer if you're exposed long enough. I couldn’t believe the headaches that lingered after a sloppy experiment back in college; many people brush off the warnings, but personal experience brings the message home fast.
Real Precautions—Not Just Lab Posters
Most safety signs look nice and neat, yet dealing with Aluminum Dichromate takes more than reading some laminated rules on the wall. It starts with gear—nitrile gloves, safety goggles, and a thick lab coat are non-negotiable. In grad school, I used to cut corners with gloves, thinking quick rinsing would protect my hands. One careless day, an itchy rash told me otherwise. Even a little dust can start problems. The best labs run fume hoods continuously, drawing away any airborne bits before they land in your lungs. If you’re working outside a fume hood, you’re gambling with your health.
Don’t Go Solo
At my first industrial internship, they drilled into us: never work alone with chromates. Every time someone needed to weigh the powder, there was a buddy system. Simple—if something goes wrong and you start coughing or freeze up, there’s someone right there to pull you out or call for help. This isn’t busy work or bureaucracy. It’s basic self-preservation.
Cleanup Isn’t Optional
Even the smallest spill deserves action. I remember once in a teaching lab, a classmate ignored a tiny sprinkle on the bench. By the next morning, someone leaned into the same spot and ended up with a chemical burn. Small amounts can do big damage; the material doesn’t magically disappear. The only approach is to soak up spills with damp paper towels and bag the waste for hazardous disposal. Wiping with a dry cloth just stirs up more dust. Local environmental rules matter here—this isn’t stuff you put down a sink or in the trash. I’ve gathered container after container of used gloves, towels, and pipette tips. It adds up, but it keeps the rest of the building safe.
Training Makes the Difference
Rules mean nothing if people don’t understand why they exist. Too many folks end up with respiratory issues or skin conditions that could have been avoided with a real conversation about the risks. Learning from those who’ve had a close call beats reading an instruction manual every time. At every lab I’ve worked in, we held regular drills and walked through the steps for safe handling and emergency response. This practice makes people confident, not careless.
Smart Steps Forward
No one wakes up in the morning hoping to go home covered in rashes or facing a trip to the emergency room. Taking care with Aluminum Dichromate isn’t about paranoia—it’s about respect for something powerful and dangerous. With solid gear, honest teamwork, and never shortcutting cleanup, anyone can get the job done and leave the lab in better shape than they found it.
| Names | |
| Preferred IUPAC name | dialuminium tris(oxidochromate(VI)) |
| Other names |
Aluminium dichromate Aluminum bichromate |
| Pronunciation | /əˈluːmɪnəm daɪˈkroʊmeɪt/ |
| Identifiers | |
| CAS Number | 7789-23-3 |
| 3D model (JSmol) | Al2(Cr2O7)3 3D model (JSmol) string: `Al2(Cr2O7)3` |
| Beilstein Reference | 3972333 |
| ChEBI | CHEBI:91256 |
| ChEMBL | CHEMBL1201561 |
| ChemSpider | 19550806 |
| DrugBank | DB14506 |
| ECHA InfoCard | ECHA InfoCard: 100.028.888 |
| EC Number | 236-750-7 |
| Gmelin Reference | 1865 |
| KEGG | C18797 |
| MeSH | D000579 |
| PubChem CID | 24543 |
| RTECS number | BD9275000 |
| UNII | VK8T24F7L3 |
| UN number | UN1439 |
| Properties | |
| Chemical formula | Al2(Cr2O7)3 |
| Molar mass | 451.996 g/mol |
| Appearance | Orange red crystals |
| Odor | Odorless |
| Density | 2.61 g/cm³ |
| Solubility in water | Soluble |
| log P | -2.97 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 6.1 |
| Magnetic susceptibility (χ) | +1200.0e-6 cm³/mol |
| Refractive index (nD) | 1.760 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 237.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1827 kJ/mol |
| Pharmacology | |
| ATC code | V09AA03 |
| Hazards | |
| Main hazards | Toxic if swallowed, causes severe skin burns and eye damage, may cause cancer, may damage fertility or the unborn child, may cause respiratory irritation, very toxic to aquatic life. |
| GHS labelling | GHS02, GHS05, GHS06, GHS08, GHS09 |
| Pictograms | GHS05,GHS06,GHS08,GHS09 |
| Signal word | Danger |
| Hazard statements | H272, H301, H314, H317, H334, H341, H350, H360Fd, H372, H410 |
| Precautionary statements | P260, P264, P270, P271, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P308+P313, P310, P314, P321, P330, P362+P364, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Lethal dose or concentration | LD₅₀ (oral, rat): 62 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 26 mg/kg |
| NIOSH | DT5725000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Aluminum Dichromate: "0.05 mg/m³ (as Cr VI), 8-hour TWA (OSHA) |
| REL (Recommended) | 200 μg/m³ |
| IDLH (Immediate danger) | 15 mg Cr(VI)/m3 |
| Related compounds | |
| Related compounds |
Chromyl chloride Chromium trioxide Ammonium dichromate Potassium dichromate Sodium dichromate |

