Allyl Propionate: Chemistry's Quiet Contributor
A Brief Look Back
Years ago, chemists searching for new flavors and fragrances kept stumbling upon interesting esters. Allyl propionate didn’t stir headlines but quietly emerged in the early twentieth century, first in research as part of a group of small molecules changing the flavor industry. Synthetic processes became increasingly refined in the decades that followed. Today, many perfumers and food technologists know allyl propionate fairly well, far from its early obscurity. It became a staple among synthetic esters after scientists understood its chemistry and recognized the value of its fruity, pineapple-like scent—one that hints at summer for many people, though most have never heard its name.
The Skinny on the Molecule
Allyl propionate grabs attention in food labs because it’s an ester. Its formula looks like C6H10O2—three simple elements combined to give both a sweet, fruity flavor and a sharp, crisp aroma. Left at room temperature, it forms a colorless liquid with a distinct scent you’d recognize from certain candy. Allergies rarely follow it, but regulators still keep tabs to ensure levels don’t exceed what’s safe. Its boiling point is close to 125 degrees Celsius, so it’s stable under most kitchen or processing conditions. Solubility in water is low, yet it blends well with alcohol, making it useful in various chemical mixes. None of these physical traits are surprising for an ester, yet they define why manufacturers keep ordering it.
Technical Details Without the Jargon
Labeling and regulation remain straightforward—most jurisdictions rely on international chemical safety norms. Denotation as “allyl propionate” covers its essential identity, sometimes popping up as “propionic acid, allyl ester.” Regulatory agencies in Europe and the US both recognized it as safe when used properly: grasping this comes from decades of safety reports and practical use without incidents. The clear liquid may be shipped under basic hazardous material rules, but those who work with it, whether in a food factory or fragrance plant, seldom run into trouble if standard procedures are followed.
How Chemists Piece It Together
Synthesis isn’t rocket science—usually it’s about reacting allyl alcohol with propionic acid. In practice, someone combines propionic acid with allyl alcohol in the presence of an acid catalyst. This process, called esterification, isn’t just textbook learning; it’s done at scale every week. Controls over temperature and acid concentration keep things smooth and efficient. If you’ve worked in a lab, there’s a good chance you’ve handled reactions like this, with water splitting away as a byproduct. The method stands as a model of efficient organic synthesis, one that universities teach their undergraduates and big manufacturers apply with industrial flair.
Chemical Tweaks and What They Mean
Reactions with allyl propionate don’t end after the first esterification. Chemists value the molecule’s allyl group for further modifications. That double bond attached to the propionate structure allows for additions or substitutions, leading to derivatives prized in industry. Hydrogenation can remove some double bond character, or halogenation creates more reactive compounds. There’s plenty of room for improvisation, and these reactions feed right into the broader world of chemical manufacturing, not just flavors and scents.
Other Names You’ll Hear
In addition to “allyl propionate,” the substance might turn up as “propionic acid, allyl ester” or sometimes in ingredient labels as “artificial flavor.” Chemical databases use identifiers like CAS numbers, but in most everyday settings, workers call it by its core name or as a component of an additive blend. Those who spend their days blending flavors, mixing perfumes, or cataloging chemicals see these alternate names pop up on manifests and safety reports, but the core identity remains familiar in labs and factories alike.
Staying Safe: Real-World Handling
Safety in the lab or in manufacturing lines matters far more than a pamphlet can express. Allyl propionate has a moderate vapor pressure, meaning it can give off enough vapor to irritate eyes or noses if ventilation isn’t right. Gloves, goggles, and a good fume hood solve most exposure problems, and chemical spill clean-up mirrors what you’d expect from a mild irritant. Everyone I’ve worked beside knows the MSDS protocols and treats it with the same respect as any other volatile ester. Thankfully, incidents remain rare due to these established habits, but complacency always threatens upsets. Following operational standards is as much about paying attention in the moment as it is about ticking boxes on a form.
Where It Ends Up
Allyl propionate dominates in flavor formulation. Those fruity notes don’t come from magic—they’re engineered using this ester, layered into pineapple, apricot, and tropical flavors that pop in both cheap candy and gourmet foods. Perfumers grab it when they want to brighten a citrus or floral base, balancing cheapness with impact. In industrial contexts, it sometimes shows up as a solvent because of its compatibility with other organics, but its value always circles back to taste and smell. Over the years, I’ve talked with food scientists who rely on it for new recipes, admitting that its impact far outweighs its cost compared to natural extracts.
Digging Into the Science
Research continues to study not just how best to synthesize allyl propionate but also to minimize unwanted by-products, improve yields, and cut costs for flavor houses. Food safety teams at research universities focus on metabolic fate in living bodies, testing how esters like allyl propionate break down during digestion. Newer developments push for “greener” production: biocatalysts are edging into pilot plants to avoid strong mineral acids and lower environmental footprints. These moves matter for both efficiency and public acceptance, as sustainability now sits high on many companies’ lists in ways never seen even a decade ago.
What We Know About Toxicity
Most toxicity data comes from animal studies and years of routine use. Allyl propionate stays well below the danger threshold in typical applications. Poisoning is essentially unheard of in food use because regulatory bodies lock down limits and track exposures. Acute exposures at much higher levels—like if a drum leaked in a warehouse—could trigger headaches, irritation, or nausea, but warnings and standards keep these events rare. Chronic exposure studies do not suggest severe long-term risks, yet ongoing review keeps regulators satisfied. Personal workplace anecdotes back up the lack of real-world incidents beyond the minor complaints found with many strong-smelling esters.
Looking Ahead
Although not an eye-catching topic at parties, allyl propionate affects everyday life far more than most realize. Changes in regulations, advances in green chemistry, and shifts toward “natural” flavors continue to shape how companies use and produce it. Expansion into new markets comes as more regions build up packaged food and fragrance industries. Researchers push for new biocatalytic methods, pursuing cleaner and safer production lines. Technological advances may someday replace traditional synthesis altogether, especially if public pressure leads to wider adoption of renewable feedstocks. In the end, the future stretches with possibilities: innovation, safer workplaces, and ever-newer flavors for the next generation of candies and perfumes.
What is Allyl Propionate used for?
A Sweet and Fruity Note: Where You’ll Smell Allyl Propionate
Walk into a bakery and catch the aroma of pastries, or take a swig of fruit-flavored soda—odds are some of those rich, inviting notes come from allyl propionate. This colorless liquid pops up in the world of flavors and fragrances, thanks to its naturally sweet, pineapple-tinged scent. Just a whiff brings to mind juicy fruit, and manufacturers lean on that big time. Artificial flavors for candy, drinks, and even ice cream use it to add depth or replicate flavors impossible to deliver with fruit alone. Perfumers like a dash of this stuff for fruity blends, giving high-end and drugstore brands a consistent finish batch after batch.
Food Industry Trusts, But Regulates
I once worked in a bakery that made its own syrups for flavoring cakes. Reading the labels of the raw syrup ingredients showed how often chemicals like allyl propionate cropped up. Food scientists pay close attention to the amounts used. Too much, and foods taste artificial; too little, the flavor falls flat. The Food and Drug Administration recognizes this ingredient as generally safe in small doses. That fact matters—for everyone from candy makers to soda giants—since nobody wants a product that could harm consumers or spark a recall. The European Food Safety Authority did its own assessment, reaching a similar safe use determination. They both encourage keeping things within reason, advocating proper handling and strict adherence to recommended limits.
From Air Fresheners to Cosmetics
Step beyond the kitchen, and allyl propionate finds a new life. Air fresheners and household cleaners bring it into play for masking strong odors with sweetness. Anyone who has tested scented lotions or soaps might recognize part of that tropical or fruity peak. In my own home, a few drops of an off-the-shelf fragrance oil made cleaning less of a chore, simply by making everything smell nicer. Cosmetic manufacturers seek out stable and pleasant-smelling compounds, which keep products appealing and easy to market. People want their shampoo to evoke something fresh or exotic, and this compound fits the bill.
Why Regulation Still Matters
Although allyl propionate makes food and fragrances more exciting, health agencies keep a watchful eye. Accidental overexposure in factories or labs can cause irritation, especially to eyes and skin. Ethical manufacturers invest in protective equipment and ventilation, not just to keep up with audits, but because workers’ health matters in practice. I have met people managing fragrance production lines who talk about the importance of airtight storage and regular safety drills. In the hands of trained staff, problems stay rare. The push for clearer labels and ingredient transparency only grows, answering consumer calls for greater control over what enters their lives and bodies.
Balancing Pleasure and Protection
Allyl propionate’s biggest appeal comes from its ability to add punch to products that people use every day. Its use continues, thanks to relentless testing, oversight, and a willingness by manufacturers to put consumer safety at the center. Instead of seeing these ingredients as villains, I’ve learned through direct experience and research that with clear oversight and open communication, chemistry can serve both flavor and peace of mind. Relying on safe storage, regular training, and sensible regulation allows industries to put out products people enjoy, without losing sleep over unintended harm.
Is Allyl Propionate safe for use in food or cosmetics?
Understanding Allyl Propionate
Allyl propionate brings a sweet, fruity scent perfect for products designed to please the nose or the palate. You come across its flavor in foods labeled as “fruit punch,” “pineapple,” or “butterscotch.” The perfume industry leans on it for that crisp, inviting top note in certain colognes and skin-care products. Even household products benefit, adding a subtle fragrance often mistaken for the real thing.
Safety in Food Applications
Safety takes center stage for food additives, and allyl propionate sits under close watch by regulatory authorities. The U.S. Food and Drug Administration (FDA) lists it as “Generally Recognized As Safe” (GRAS) for limited use in foods. Scientists and regulatory panels recognized its absence of toxicity when used in the incredibly tiny amounts you’d find in food—think parts per million. Imagine putting a teaspoon of sugar into a whole Olympic swimming pool; that gives a good sense of scale for how little gets used. The Joint FAO/WHO Expert Committee on Food Additives also cleared allyl propionate as safe when used at low concentrations common in flavoring applications.
Still, safeguards remain. The concentrations allowed in food aren’t random—they’re based on studies involving rats and other lab animals, set far below any dose connected to negative effects. If the food industry suddenly flooded products with large doses far above the GRAS limit, you’d likely see irritation or toxicity. That’s true of almost anything considered safe: too much salt, caffeine, or vitamin A also causes trouble. Responsible manufacturers keep allyl propionate use well beneath those upper thresholds, so consumers run into no real risk.
Cosmetics and Skin Contact
Allyl propionate’s fruity scent brings it into all kinds of beauty products—body sprays, facial cleansers, hand lotions—often in even smaller amounts than in foods. Cosmetic chemists pay close attention to ingredients that touch skin; no one wants a nasty rash. The Cosmetic Ingredient Review (CIR) panel published a safety assessment finding allyl propionate safe as used, again citing its low concentrations, rapid evaporation, and checks on its potential to cause irritation. Allergy cases appear extremely rare, according to patch test data. For many, this means a product smells like real fruit without much risk at all, although those with very sensitive skin should always patch-test new products.
Why Trust Matters
Controversy sometimes flares up around obscure ingredients or anything with a chemical-sounding name. Yet a substance isn’t dangerous simply because it’s hard to pronounce. As someone who likes to read ingredient lists, I’ve learned it makes sense to look up what regulatory bodies and peer-reviewed research say. The experts examining allyl propionate back its safety in the minimal amounts found in products you use or eat today. Food scientists, toxicologists, and health agencies stay alert for any new evidence, adjusting rules if something shifts, which builds trust in the process.
Paths for Improvement and Transparency
No one benefits from secrecy in product formulation. Clearer labeling empowers consumers who want to make their own choices, especially those with allergies. Regular review of safety studies keeps ingredient lists up to date, giving confidence to both shoppers and regulators. Manufacturers can do their part by publicizing data and giving people more tools to research ingredients for themselves. With honest communication and responsible science, safe use remains within reach, and trust continues to grow.
What is the chemical formula of Allyl Propionate?
What Sets Allyl Propionate Apart
Fragrances and flavors come alive with the help of a handful of big players. Allyl propionate, with a sweet, pineapple-like aroma, often sits near the top of the list in both perfumery and food additives. Its impact reaches everyday life, but every good story has a backbone—this one has a chemical formula: C6H10O2.
Chemistry Behind the Blend
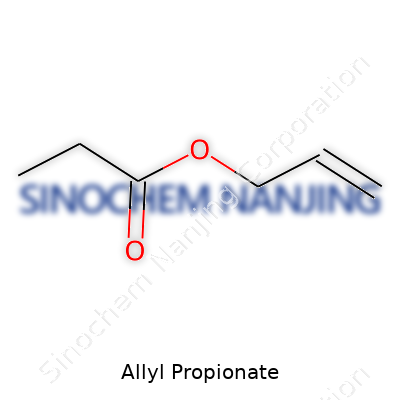
Allyl propionate stands as an ester. In plain terms, esters pop up anytime you mix an acid with an alcohol and toss out a water molecule. With this one, the building blocks are propionic acid and allyl alcohol. The word “allyl” points to a three-carbon chain with a double bond; “propionate” adds another three-carbon chunk. Put these together, and you shape a molecule that brings out strong, fruity notes. Its structure looks like this: CH2=CH-CH2-O-CO-CH2-CH3. Here’s how you get C6H10O2 out of that: six carbons, ten hydrogens, and two oxygens, all stuck together just right.
Everyday Encounters
Eating pineapple-flavored candy or even smelling some floral perfumes, you're probably picking up on esters like allyl propionate. The food industry leans on this compound because it gives a refreshing, uplifting scent that mimics fruits without needing to crush real pineapples by the truckload. Synthetic options knock down both cost and spoilage. Perfume makers like it because it lingers and layers well with other notes.
Impact on Health and Safety
Like most strong-smelling chemicals, allyl propionate deserves a careful approach. Research points out that high concentrations might irritate the skin, eyes, or respiratory system. The food industry keeps quantities low, well below levels that could cause harm. For workplaces that make or blend this chemical, safety teams stick to protective gear and solid ventilation to manage the risks.
Environmental Notes
Nature can break down allyl propionate, but large-scale spills or careless disposal push problems into soil and water. Laws in most countries stick to strict rules about handling and discarding synthetic esters to keep communities and local wildlife safe. Investing in clean production methods and closed systems goes a long way toward shrinking the chemical’s environmental footprint.
Staying Informed
With new flavors, fragrances, and synthetic chemicals arriving all the time, folks in industries that use allyl propionate keep an eye on evolving safety studies and ingredient trends. Health and food regulatory agencies worldwide—like the US Food and Drug Administration (FDA) and the European Food Safety Authority (EFSA)—review scientific evidence before allowing flavor and fragrance use in products for sale. These agencies check for toxic effects, long-term exposure risks, and environmental impact before stamping “safe” next to flavor compounds.
The Takeaway
Behind a bright flavor or fresh fragrance hides a story best told through the lens of science. C6H10O2 offers a great example of turning raw chemistry knowledge into something people taste or smell every day. Choosing to learn about the molecules behind familiar experiences helps bridge gaps between science, manufacturing, and daily life. Responsible handling and steady research make sure compounds like allyl propionate stay useful and safe in the years to come.
What does Allyl Propionate smell like?
Aroma Straight From the Lab
Allyl propionate leaves a mark on the senses. Pull the stopper off a small bottle in the lab, and a strong fruity scent fills the air. Think of pineapples and pears sitting on a sun-warmed kitchen table, ripening until they get that sharpness just shy of being too much. There’s a clarity to the smell that makes your nose perk up—bright, sparkly but not sugary. It lacks the heaviness found in some fruit esters.
Expert Handling in Perfumery
Perfumers have reached for allyl propionate for years. Just a touch can brighten up a “fruit salad” note, adding crispness without overwhelming other ingredients. Pear, pineapple, strawberry—these fruit notes get a genuine lift. It doesn’t scream artificial; it balances sweetness with a whiff of green, almost like sniffing freshly sliced fruit rather than a candy wrapper. Some say it even adds a glimmer of marzipan or a faint winey backdrop, which gives more depth.
Why It Stands Out
Fruits feel more inviting with a bit of allyl propionate. Natural fruit flavors in foods often fall flat once processed. Scientists in the flavor industry know about this challenge. Pop open a bottle of factory-made pear juice and you’ll notice something missing. Allyl propionate serves as an enhancer, filling in those missing aromatics so the end product tastes closer to an actual fruit. It’s no gimmick—regulated and used in small amounts, it stays safe and approachable for consumers. The average palate recognizes “something fruity” before picking up on specifics.
Potential Issues in Use
Big flavor often triggers suspicion. “Synthetic” carries baggage for consumers tuned into food labels. Some people shy away from additives with names they can’t pronounce. Maybe you remember walking down the grocery aisle, flipping a bottle of flavored water, and puzzling over the ingredients. An unfamiliar name might signal “fake” or “unhealthy,” even though allyl propionate occurs naturally in some fruits. Regulators keep a close watch over its use because high levels could irritate, but used right, it stays well below risky thresholds.
Building Trust with Transparency
One solution for the stigma lies in open communication. Food companies could share simple breakdowns of why allyl propionate, and similar molecules, show up in foods and fragrances. Pointing to its natural occurrence in pineapple or cheese bridges the gap. Education—through social media, packaging, or even documentaries—can change perceptions. Showing the actual safety data, and not just relying on buzzwords like “natural,” makes a stronger case than hiding behind technicalities.
Safer Handling, Better Outcomes
Companies make the workplace safer by training staff to handle concentrated allyl propionate with gloves and good ventilation. Inside factories and flavor houses, no one wants lasting irritation. Strong smells may trigger headaches at full strength, so using it at the lowest effective dose matters. Experienced workers learn the value of respect for even pleasant-smelling molecules. Some in the business believe that spending extra on better extraction and blending equipment more than pays off by keeping both products and people safe.
In Everyday Life
Snack on a pineapple-flavored candy or sip on a pear drink, and you’ve likely experienced allyl propionate—even if you never heard of it. The aroma may not get headline attention like vanilla or cinnamon, but it plays its part in the background, helping everyday foods and fragrances taste and smell the way you remember from childhood.
Where can I buy Allyl Propionate?
Why Buying Allyl Propionate Isn’t Like Picking Up Groceries
Allyl propionate isn’t something you stumble across on the shelf at a local store. Anyone searching for this compound probably works in fragrance production, flavor synthesis, or another technical field. So you can’t rely on big box retailers or everyday e-commerce sites. Choices narrow quickly to chemical suppliers and specialized distributors. From personal experience in scent chemistry, a buyer needs to know more than just where to click “buy” — safety, legitimacy, and compliance all come into play.
Understanding Who Sells Allyl Propionate
Large distributors such as Sigma-Aldrich, TCI Chemicals, and Alfa Aesar carry a range of specialty reagents. They supply research labs and industrial facilities. These companies present purity standards, offer analysis certificates, and follow regulations most countries expect. Online ordering is possible, but certain hurdles come into play. These include account setup, verification, and agreement to terms around chemical handling.
Smaller specialty suppliers often fill gaps when larger distributors charge too much or limit smaller quantities. Perfume supply houses and flavor houses sometimes offer allyl propionate as a raw ingredient for formulation labs. Here’s where authenticity matters. Reputable suppliers show transparent batch records, clear labeling, and professional communication. Cheap deals found on anonymous websites usually ring alarm bells. Fakes, dilutions, or mislabeling can turn a straightforward purchase into a safety hazard. Scams aren’t rare — I’ve seen hobbyists burn money and face setbacks after buying from the wrong place.
Safety, Legality, and Documentation
Countries treat potentially hazardous chemicals with strict guidelines. U.S. and EU buyers will find allyl propionate listed under chemical inventories like TSCA and REACH. Legit suppliers ask for end-use information and verify business credentials. Medical or food applications raise the bar — food-grade certifications and origin traceability can slow the purchase, but also keep products safe. If a vendor ships without questions or paperwork, that’s a red flag. Proper companies include Safety Data Sheets (SDS), and sometimes demand proof of training on safe handling.
Beyond Price: Real Value Comes from Accountability
High price tags scare some buyers away, leading them to chase gray-market deals. Reliable suppliers justify their prices with safety measures, regulatory compliance, storage infrastructure, and insurance. Anyone who cuts corners risks accidents, unwanted legal trouble, or contamination. Staying away from cut-rate offers is my personal rule. When a supplier can answer technical questions and share documentation without delay, it signals reliability.
Choosing the Right Path
Reach out to established chemical distributors that serve your country or field. Check for a visible address, working phone number, and traceable order process. Industry forums and professional networks sometimes highlight lesser-known but trustworthy sources—word of mouth from seasoned buyers saves headaches. If you’re not sure where to begin, universities with chemistry or perfumery departments sometimes share leads on reputable sellers. Purchasing through a registered business or institution further streamlines approvals and compliance steps.
The urge to save time or money sometimes leads buyers to take shortcuts. In my experience, staying patient, double-checking credentials, and buying from sources that are used to serving laboratories or manufacturers pays off. Allyl propionate plays an invisible role in countless finished products, but buying it shouldn’t feel like a shot in the dark.
| Names | |
| Preferred IUPAC name | Prop-2-enyl propanoate |
| Other names |
Propanoic acid, allyl ester Propionic acid, allyl ester Allyl propanoate |
| Pronunciation | /ˈæl.ɪl proʊˈpiː.ə.neɪt/ |
| Identifiers | |
| CAS Number | 142-19-8 |
| Beilstein Reference | 1041582 |
| ChEBI | CHEBI:88571 |
| ChEMBL | CHEMBL3185952 |
| ChemSpider | 62416 |
| DrugBank | DB14134 |
| ECHA InfoCard | 100_100_887 |
| EC Number | 211-926-9 |
| Gmelin Reference | 8197 |
| KEGG | C14418 |
| MeSH | D017760 |
| PubChem CID | 8954 |
| RTECS number | UF8225000 |
| UNII | 71EC8F9205 |
| UN number | UN2342 |
| Properties | |
| Chemical formula | C6H10O2 |
| Molar mass | 114.15 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | pineapple, sweet, fruity, cheese |
| Density | 0.889 g/mL at 25 °C(lit.) |
| Solubility in water | Insoluble |
| log P | 1.94 |
| Vapor pressure | 0.9 mmHg (20 °C) |
| Acidity (pKa) | pKa ≈ 25 |
| Basicity (pKb) | pKb ≈ 15.88 |
| Magnetic susceptibility (χ) | -7.44×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.414 |
| Viscosity | 1.174 cP (25 °C) |
| Dipole moment | 2.06 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 383.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -322.05 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3106.5 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H315, H317, H319, H335 |
| Precautionary statements | H302: Harmful if swallowed. H315: Causes skin irritation. H319: Causes serious eye irritation. H335: May cause respiratory irritation. |
| NFPA 704 (fire diamond) | 2-3-2 |
| Flash point | 66 °C |
| Autoignition temperature | 455°C |
| Explosive limits | Explosive limits: 2.6% to 10.4% |
| Lethal dose or concentration | LD50 (oral, rat): 4300 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 6,440 mg/kg |
| NIOSH | SNH |
| PEL (Permissible) | 'PEL (Permissible) of Allyl Propionate: 2 ppm (TWA)' |
| REL (Recommended) | 0.01 |
| IDLH (Immediate danger) | 150 ppm |
| Related compounds | |
| Related compounds |
Allyl acetate Propionic acid Allyl alcohol Propionitrile |

