Allyl Isothiocyanate: Deep Dive into Its Past, Present, and Prospects
Historical Development
Allyl isothiocyanate (AITC) entered the scene through horseradish roots and mustard seeds long before folks understood chemistry the way we do now. People in past centuries used pungent mustards and herbs both as food and as primitive medicine. The story turns a corner with chemists like Auguste Cahours in the 1800s, who managed to tease apart its structure and draw a map for others to follow. Not just a food component, AITC became interesting for its sharp bite and for early observations that it kept bacteria at bay. So it didn’t just land in the spice rack. By the middle of the twentieth century, its value extended into labs and factories, where industry extracted or synthesized it for all sorts of specialized uses, from flavoring to fighting fungus.
Product Overview
You don’t have to glance far to spot AITC in daily life. Think wasabi, mustard, and radish heat—the kind of zing that can clear sinuses and bring tears to your eyes. Purified allyl isothiocyanate won’t linger in shadows, either. Shift away from natural sources and you find a clear, colorless to pale yellow liquid produced on large scales, packaged for producers of condiments, fumigants, and plenty in between. European and American flavor houses pour it into mustards, pickles, and sauces. Agriculture puts it to work protecting crops and seeds. Heck, it even features in some pest control products designed for stored grain or root vegetables.
Physical and Chemical Properties
AITC easily volatilizes at room temperature, filling space with that unmistakable aroma. It weighs in at 99.15 g/mol and carries the formula C4H5NS. Most people encounter it dissolved in vinegar or oil, but straight from the source, its boiling point hovers around 152°C, and it lights up flammable warnings at 41°C for flash point. Solubility shows a split personality—mixes neat with most organic solvents, but acts stubborn in water unless you coax it. Its reactive N=C=S group gives it teeth, snapping up nucleophiles in reactions and breaking down under alkaline or acid conditions.
Technical Specifications and Labeling
Suppliers who know their audience provide purity grades upward of 95%, flagged with tight specs for refractive index and specific gravity. Labels must show CAS Number 57-06-7, hazard warnings for skin and eyes, and safe storage advice—more to protect the user than the bottle. Rules in the EU, the US, and Japan demand that food-grade material comes with batch traceability, precise ingredient volumes, and revised allergies statements. You won’t find it in retail spice jars in pure form, but bulk containers show regulators’ influence on designated uses and handling.
Preparation Method
Traditional extraction still plays a part: grind up mustard seeds, add water, let the enzyme myrosinase split sinigrin, and AITC pops out. For larger volumes, chemists aim for direct synthesis. The most common approach routes allyl chloride through potassium thiocyanate, using solvents and controlled temperatures to corral the product and filter out byproducts. Refinement depends on fractional distillation or solvent extraction. This method’s reliability—now decades old—keeps factory output steady, even when seed supplies run thin.
Chemical Reactions and Modifications
AITC’s electronegative elements make it a key player in organic chemistry. Pharma researchers and flavor scientists count on its isothiocyanate group to bond with amines, alcohols, and thiols, spinning out derivatives that change flavor, stability, or biological activity. Add acids or bases, and the backbone can break down, losing its bite to form allyl amines or secondary amides. Chemists keep probing fresh reactions—forming heterocycles for drug discovery, or blending new analogues that last longer in prepared foods or agro products.
Synonyms and Product Names
Allyl isothiocyanate won’t always show up under its scientific name. You see “mustard oil” or “synthetic mustard oil” on food ingredients, “3-isothiocyanato-1-propene” in lab catalogs, or even “oil of mustard” in agricultural bulletins. Depending on market, regulatory filings, or country, these synonyms crop up in patents, product datasheets, and food labels—the mix sometimes adding to the confusion, especially for importers keeping tabs on customs declarations or safety sheets.
Safety and Operational Standards
Several industry watchdogs call for careful handling. AITC can hit the skin, eyes, and lungs hard. Acute exposure stings and irritates, and higher concentrations, especially during spills or in manufacturing, spark alarms for chemical burns or respiratory distress. OSHA sets airborne limits to keep it below damaging levels. Protective gloves, goggles, and fume hoods fall under standard requirements, and everyone from production line operators to laboratory scientists gets periodic training in spill and exposure response. European regulations classify it as hazardous, driving both packaging requirements and transport limits. Risk communication, PPE, and regular audits factor into operations—from shipping docks to bottling lines.
Application Area
Food manufacturers count on its natural antimicrobial strength—think mustard pickles that don’t go slimy—and its pungency for recreating familiar tastes. Grain warehouses fog it through bins to knock back insects and molds, taking advantage of its volatility. Over in healthcare labs, AITC joins the arsenal for probing enzyme function and cancer biology, thanks to its ability to modify cysteine groups in proteins. Surface cleaners sometimes borrow its germ-knocking properties. Even fragrance stocks mix it in for the “fresh-cut radish” note.
Research and Development
Labs focusing on cancer therapy look at AITC derivatives hoping to block cell growth or tweak immune signaling. A cluster of studies dig into its effects against E. coli, Salmonella, and Listeria in food handling and hospital sanitation. Researchers test formulations to keep its heat locked for processed foods, while others try encapsulation tricks to mask odor and control volatility. Agro-chemists are tweaking its structure to curb toxic residues and lengthen its protective reach in soil and grain storage. Patents on new synthesis routes and ingredient blends keep flowing, hinting at strong commercial curiosity.
Toxicity Research
AITC doesn’t let its spicy reputation go unchecked. Animal tests show that single doses above 151 mg/kg can cause obvious health problems. Extended exposure sketches out worries for workers or populations around manufacturing plants. Regulatory experts comb through data for skin irritation, inhalation toxicity, and risks in food overconsumption. Recent studies watch for impacts on gut microbes and metabolic enzymes, since even low concentrations can hit sensitive groups. Animal tests provide numbers for safe ingestion, but human data for chronic exposure remain under discussion. Governments and safety panels post limits—like 1–5 ppm in foods—as a buffer zone.
Future Prospects
Global interest keeps rising, not just for food but for natural preservatives and pharmaceutical platforms. Demand for “clean label” foods, less reliant on synthetic antimicrobial agents, has food technologists looking at AITC with renewed excitement. Regulatory reviews shape innovation—every new application, from biodegradable packaging to pest-resistant crops, faces close scrutiny. Advances in gene editing and green chemistry may one day tailor AITC-producing plants for agriculture or therapeutics. As synthetic biology matures, more folks will likely find ways to produce and modify it sustainably. In my own experience, the steady march of both industrial and academic research means AITC will ride along at the crossroad of food safety, pharmacology, and agritech for the foreseeable future.
What is Allyl Isothiocyanate used for?
Allyl Isothiocyanate in Your Everyday Life
The sharp sting you feel biting into wasabi or horseradish has a name. That’s not just a culinary trick; it comes straight from a chemical called allyl isothiocyanate. I’ve made plenty of homemade pickles, and I’ll admit, the tang and nose-tingling sensation gives pickled veggies their memorable kick. Allyl isothiocyanate gives that distinctive heat and aroma you find in mustards, wasabi, horseradish, and some root vegetables. Researchers believe plants produce it as a natural defense, driving away herbivores and pests.
Why Food Producers Count on It
Food safety isn’t just a buzzword. Food spoilage drains manufacturers, leads to waste, and leaves customers annoyed. Allyl isothiocyanate shows real promise as a way to extend shelf life. Think of it as a sort of edible bouncer. Studies in food science journals, including those by the Journal of Food Protection, show that this compound puts the brakes on bacteria—especially in packaged vegetables and meats. In my own kitchen, adding horseradish to homemade preserves didn’t just boost flavor—it seemed to help them last longer too.
Getting Down to Health and Nutrition
Some folks look at spicy foods as nothing more than a thrill. Yet, allyl isothiocyanate is catching attention among nutritionists. Early research highlights its antioxidant potential and shows it can help kickstart certain detox enzymes in the liver. Scientists at Johns Hopkins found it could play a part in reducing inflammation, though you’d have to eat a lot more mustard greens than most people do to get those benefits. Of course, nobody should start chugging wasabi from the tube, since too much can cause gastrointestinal upset.
Industrial Uses—Beyond the Kitchen
The story doesn’t stop in the pantry. Manufacturers use this compound in pest control products and to add extra punch to pepper sprays, making use of its powerful, irritant properties. Its strong scent means it gets a seat at the table in some perfume labs, too. I once worked a stint in a packaging warehouse, and learned that this compound sometimes lines food packaging to keep mold and microbes under control. It’s found its way into the toolbox of chemical engineers, thanks to its pungency and antimicrobial kick.
What About Safety and Environmental Concerns?
Regulators, including the FDA and the European Food Safety Authority, review compounds like allyl isothiocyanate for food safety. Most assessments call it generally safe at the typical levels used in food. Exposure in large amounts, though, can irritate the skin, eyes, and respiratory tract. In my experience making large batches of mustard, even a whiff can set eyes watering. Food workers and packaging employees need to manage exposure, and responsible handling protects both people and the planet. Research teams continue to monitor how this substance behaves in the environment and whether production leaves any mark.
Room for Smarter Solutions
Brighter food labels can help folks with sensitivities know what they’re getting. Producers who try out allyl isothiocyanate as a natural preservative might be able to cut back on artificial additives. Calling on more sustainable extraction methods—rather than chemical synthesis—could shrink the environmental footprint, too.
Allyl isothiocyanate adds heat, preserves food, and finds its way into bug sprays and even fragrances. Using it wisely means paying attention not just to taste, but to health, safety, and the world around us.
Is Allyl Isothiocyanate safe for consumption?
The Everyday Chemical Hidden in Your Mustard
Allyl isothiocyanate sounds like something you'd find in a chemistry textbook. In reality, it gives that sharp, pungent kick to mustard, horseradish, and wasabi. I’ll never forget spooning too much wasabi on my sushi as a kid — the burning sensation in my nose earned an instant lesson in moderation. Even with that fiery reminder, I always came back for more, probably because this chemical is what makes these condiments stand out at summer barbecues or on hot dogs at the ballpark.
Why Allyl Isothiocyanate Gets Scrutinized
Food safety draws lots of attention, and with good reason. Chemicals that bring flavor or preservation to the table walk a thin line between tasty and risky. Allyl isothiocyanate gets tested because it’s strong; its natural defense role in plants makes it an irritant. The U.S. Food and Drug Administration (FDA) lets food manufacturers use it, but only up to certain limits. Europe also keeps it under a tight watch. Too much of anything, even something natural, can cause problems, so regulators don’t let companies use it carelessly.
What Scientists Found
Animal studies show high doses can cause stomach or liver problems. Give lab rats huge amounts of allyl isothiocyanate, and their organs struggle. These doses go way above what anyone would get from a deli sandwich. In reasonable quantities, evidence doesn't support it as a threat. Studies in people, plus decades of mustard and horseradish eating, point to general safety. A few folks get skin or nose irritation from the fumes — something I’ve felt myself. Breathing in that spicy aroma too close to the jar is enough to make your eyes water, but it passes quickly. The body can handle small amounts just fine, breaking it down and getting rid of it within hours.
Potential Health Perks
This intense chemical is not just a flavor booster. Research out of Japan and the US hints at some health perks. Some scientists link the compounds in mustard, wasabi, and related plants to lower risks of certain cancers. This ties back to how plants defend themselves: the same stuff that deters bugs or grazing animals can harm cancer cells. These benefits still need more human trials, and nobody recommends eating piles of horseradish just for cancer prevention. Eating a range of veggies remains the smarter choice. Still, people curious about food science find it fascinating how plant chemicals protect us and themselves.
Sensible Eating: The Middle Road
Folks often want yes-or-no answers: is something safe, or not? Reality lives in the middle. Allyl isothiocyanate keeps flavor interesting and food safer, but as with hot sauce or spicy peppers, too much will let you know. Sticking to normal food use keeps things low-risk. Pregnant women and young kids usually handle small amounts in meals with no trouble, but high concentrations or supplements haven’t been proven safe. Moderation rules in my own kitchen. If I make a potato salad and add mustard, nobody blinks. If I ate a spoonful of the pure stuff, the experience would be memorable for the wrong reasons.
Smart Choices at the Table
Every time we pick up a bottle of mustard, horseradish, or wasabi, we take in a pinch of allyl isothiocyanate. National health authorities keep tabs on the research. If they spot trouble, ingredients can be pulled or regulated further. For now, the real story looks reassuring. Mustard on a sandwich or the kick in your sushi roll is part of a diverse diet. Listen to your body. Enjoy the flavor, and keep it balanced with the rest of your meal. That sharp taste is more than safe — in the right amount, it’s part of what makes food satisfying.
What are the health effects of Allyl Isothiocyanate exposure?
What Is Allyl Isothiocyanate?
Allyl isothiocyanate, a chemical known for its strong smell, often jumps out of foods like mustard, horseradish, and wasabi. This compound gives those condiments their fiery kick. Manufacturers use it for flavoring, but it also ends up in products meant to repel insects or fight bacteria. Everyday life puts people in contact with it more often than they might realize.
Impact on the Body: Short-Term Exposure
Breathing in a little allyl isothiocyanate sets off instincts most people recognize — eyes water, throat stings, and coughing kicks in. Even a whiff of too much wasabi at sushi restaurants can surprise someone and prove that the body recognizes it as a threat. Inhaling larger amounts or exposing skin can bring on blisters, burns, and breathing problems. The compound’s strong smell acts as a warning sign, and the body reacts quickly to protect itself.
Beyond irritation, high doses can cloud thinking and slow reaction times. Emergency rooms sometimes see cases after industrial accidents, especially where flavor factories or cleaning companies handle the chemical in large batches. Quick removal from exposure and medical attention usually leads to recovery, but severe burns or breathing problems may take longer to heal.
Long-Term Health Risks
Continued contact with allyl isothiocyanate, especially at workplaces, raises deeper concerns. Repeated breathing in vapors or touching liquid form increases the chance of chronic breathing issues, similar to what smokers or people exposed to air pollution experience. Some workers report asthma-like symptoms, nosebleeds, and rashes that won’t clear up.
Research from government agencies, like the US National Institute for Occupational Safety and Health, shows that chronic overexposure could damage the lungs and weaken immune defenses. Animal studies raise questions about possible liver and kidney impacts, though results in humans stay less clear. While cancer hasn’t been directly linked, the warning flags are up for people who work around these chemicals day after day.
Exposure in Foods
Spicy foods using natural allyl isothiocyanate rarely raise health risks. Most people’s bodies handle small food-borne amounts easily, even if they cause a bit of sinus drama. Those with sensitive digestive systems sometimes feel more discomfort — heartburn or stomach pain after a wasabi-heavy meal isn’t rare — but dangerous effects from eating such foods stay uncommon.
Problems grow when people eat products using artificially high concentrations. Some imported or industrially flavored condiments contain stronger doses than homemade recipes, so reading ingredient lists matters. If a sharp, unfamiliar heat hits the mouth or throat, it’s time to pause and check what went into the food.
Keeping Exposure in Check
The easiest way to dodge trouble is to stay alert and read product information. At work, protective gear — gloves, goggles, face masks — earns its place in the toolbox when handling allyl isothiocyanate. Factories can use ventilation systems and train workers about safe storage. At home, keeping strong-smelling oils out of reach from kids and pets makes sense.
Public health experts push for tighter regulation, clear labeling, and better education about this and similar chemicals. Everyday folks play a part by asking questions, learning what’s in their food and cleaning products, and not brushing off sensations of irritation or pain. Our senses usually do a good job alerting us when something in the air or on the plate doesn’t belong.
How should Allyl Isothiocyanate be stored?
The Real Deal with Allyl Isothiocyanate
Walk into a lab that handles Allyl Isothiocyanate, and the sharp, pungent odor lets you know right away: this material doesn’t mess around. It’s used to give mustard its kick, but in its pure form, it demands careful handling. People working with it deserve safe environments, and that starts with smart storage. Lab disasters rarely begin with splashy mistakes—they sneak in through carelessness, corner-cutting, or simple forgetfulness like leaving bottles unsealed.
Health Risks Put Safety First
This is one of those chemicals where human experience truly matters. Years of chemistry work taught me that a whiff too many in an unventilated storeroom leaves your nose tingling and your eyes watering. According to the CDC, Allyl Isothiocyanate irritates skin, eyes, and lungs; chronic exposure can lead to respiratory problems and even harm your digestive tract. OSHA guidelines flag it as a hazardous material for good reason: one spill can escalate quickly, especially if cleanup gear isn’t nearby or if containers aren’t kept in good condition.
Ventilation and Cooling: Two Storage Cornerstones
Keep it cool, and keep air moving. That’s the golden rule here. Don’t tuck Allyl Isothiocyanate on a regular shelf or leave it out on a bench. Ideal storage calls for a dedicated flammable chemical cabinet, well-ventilated and marked with hazard symbols. Temperature matters—most sources agree it stays stable below 25°C. Rising mercury nudges up vapor pressure, letting fumes escape, so a standard office storeroom won’t cut it. I’ve seen labs try to stash it away on top shelves; this is both unsafe and disrespectful to anyone sharing the space.
Keep It Dry and Out of Sunlight
Sunlight does more than just bathe a room in warmth—it breaks down sensitive compounds, especially those with double bonds like isothiocyanates. Ultraviolet kicks off unwanted reactions, which can lead to degraded material or, worse, pressure build-ups in sealed vials. Moisture is just as unwelcome. Allyl Isothiocyanate reacts with water, sometimes producing corrosive byproducts and fumes. A dry, shaded storage area with controlled humidity protects both the compound and those storing it.
Container Choice Can’t Be Ignored
Think twice before pouring this compound into a random jar. Use tightly sealed glass containers with PTFE-lined caps. Plastic won’t last; it softens or cracks, especially if exposed to fumes for too long. Label these bottles with the date, hazard symbols, and emergency contact info. No one wants to play guessing games with mystery bottles, least of all in an emergency.
Training and Emergency Prep: Everyone’s Job
Throwing rules at people doesn’t always work. Back when I started out, the best labs paired written protocols with hands-on drills, reminding everyone that storing a chemical safely is just as important as using it correctly. Every person involved should know how to grab a spill kit or find the nearest eyewash. Spill protocols, fire plans, and ventilation checks make a huge difference when the unexpected happens.
Staying Ready, Staying Safe
People who treat storage as a living system—checking logs, running drills, fixing fridges—send a message. By respecting a compound like Allyl Isothiocyanate, they respect each other. And as someone with a few too many stories of close calls, I’m convinced: small daily habits in storage often mark the line between routine days and disaster.
What are the main applications of Allyl Isothiocyanate in industry?
Spicing Up Food Safety and Shelf Life
Anyone who enjoys wasabi or horseradish recognizes the pungent kick that shoots through the nose. That sharp bite comes from allyl isothiocyanate—a compound drawn from mustard, horseradish, and related plants. The food industry leans on this ingredient not just for its flavor. Its strong antimicrobial properties help preserve foods naturally, which reduces the need for synthetic preservatives. In my years working with food scientists, I've seen how companies searching for cleaner labels turn to natural solutions like this compound. Studies back this up, showing that small doses slow the growth of common spoilage bacteria and fungi. This helps extend the shelf life of bread, cheese, meats, and salads, keeping waste down and safety up.
In Pest Control, Nature Lends a Hand
On farms and in storage facilities, pests and molds pose huge threats to harvests. Synthetic pesticides, once standard, now face stricter regulation and pushback from people worried about chemical residues in their food. Some farm consultants, noticing this shift, encourage clients to try biofumigation. In this method, crushed mustard plants or pure allyl isothiocyanate turn into a vapor that suppresses insects and nematodes without leaving behind toxic traces. Farmers using this method see fewer crop losses and reduce dependence on harsh chemicals. This matters because safer pest control strategies protect not just crops but also the workers who apply these treatments and the soil ecosystems at risk from overuse of synthetics.
Personal Care Products with a Tangy Edge
Unexpectedly, this same chemical pops up in personal care aisles. Toothpastes and mouthwashes take advantage of its antibacterial power to fight odor-causing germs. Small concentrations get the job done without numbing the tongue. In the world of cosmetics product development, formulators look for ingredients that deliver on both performance and safety. Regulation around these ingredients remains tight—manufacturers must follow thresholds set by agencies like the FDA or EFSA to make sure people avoid irritation.
Boosting Pharma’s Arsenal
The medical field generally approaches new chemicals with a lot of caution, but the potential for plant-derived compounds remains strong. Researchers in labs across the globe explore whether allyl isothiocyanate fights inflammation and helps clear out certain bacteria that shrug off mainstream antibiotics. Imagine hospital surfaces treated with a plant-based cleaner that stands up to resistant germs. That kind of technology would make a difference in keeping vulnerable patients safer. Still, moving from promising data in a petri dish to a product on the market takes time, careful clinical trials, and clear communication about both the benefits and risks.
Looking Beyond the Present
One challenge with any bioactive compound, including allyl isothiocyanate, comes from handling and safety. Strong flavors or fumes can irritate skin, eyes, and lungs if people ignore proper training or protective equipment. The chemical’s volatility means storage hurdles and potential environmental releases need careful management. Companies in food, agriculture, and health are working together with regulators to set clearer standards and monitoring systems. New technology—like encapsulating the ingredient in powders or gels—helps control release and boosts safety.
Drawing from experience and industry data, it’s clear that allyl isothiocyanate does more than deliver a spicy jolt on your plate. It sits at the intersection of tradition and innovation, giving businesses more options for safe preservation, pest control, and even personal and health care. Smart development and careful oversight unlock the broad benefits while keeping people and the planet top of mind.
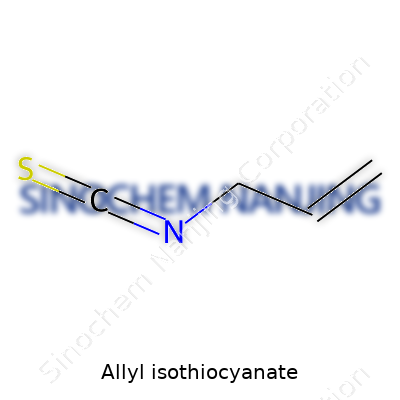
| Names | |
| Preferred IUPAC name | 3-isothiocyanatoprop-1-ene |
| Other names |
3-Isothiocyanatoprop-1-ene Allylsenföl AITC 3-allyl isothiocyanate Mustard oil 3-Isothiocyanatopropene |
| Pronunciation | /ˈæl.ɪl aɪ.soʊˌθaɪ.oʊ.saɪˈəˌneɪt/ |
| Identifiers | |
| CAS Number | **57-06-7** |
| Beilstein Reference | 1209223 |
| ChEBI | CHEBI:2756 |
| ChEMBL | CHEMBL47453 |
| ChemSpider | 5344 |
| DrugBank | DB06710 |
| ECHA InfoCard | 100.004.200 |
| EC Number | 200-309-2 |
| Gmelin Reference | 8223 |
| KEGG | C06572 |
| MeSH | D000579 |
| PubChem CID | 5975 |
| RTECS number | WL4375000 |
| UNII | F862O2R043 |
| UN number | UN1548 |
| CompTox Dashboard (EPA) | DTXSID7020287 |
| Properties | |
| Chemical formula | C4H5NS |
| Molar mass | 99.15 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Pungent, horseradish-like |
| Density | 1.018 g/mL at 25 °C |
| Solubility in water | 0.6 g/100 mL (20 °C) |
| log P | 1.77 |
| Vapor pressure | 3 mmHg (20°C) |
| Acidity (pKa) | 10.8 |
| Basicity (pKb) | 3.52 |
| Magnetic susceptibility (χ) | -12.47×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.520 |
| Viscosity | 2.19 mPa·s (20 °C) |
| Dipole moment | 1.73 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 295.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 126.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2066 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | A01AD09 |
| Hazards | |
| GHS labelling | **GHS02, GHS05, GHS06, GHS07** |
| Pictograms | GHS02,GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H315: Causes skin irritation. H319: Causes serious eye irritation. H335: May cause respiratory irritation. |
| Precautionary statements | P210, P233, P261, P264, P270, P271, P273, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P311, P312, P330, P337+P313, P362+P364, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2 |
| Flash point | 28 °C (82 °F; closed cup) |
| Autoignition temperature | 435 °C |
| Explosive limits | 1.6–10.2% |
| Lethal dose or concentration | LD50 oral rat 151 mg/kg |
| LD50 (median dose) | LD50 (median dose): 151 mg/kg (rat, oral) |
| NIOSH | SA9125000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Allyl Isothiocyanate: 0.02 ppm (0.08 mg/m³) |
| REL (Recommended) | 0.025 mg/m³ |
| IDLH (Immediate danger) | 40 ppm |
| Related compounds | |
| Related compounds |
Isothiocyanate Methyl isothiocyanate Phenyl isothiocyanate Sulforaphane |

