Allyl Glycidyl Ether: A Deep Dive into Its Origins, Chemistry, and Impact
Historical Development
Allyl glycidyl ether first gained commercial traction after World War II, at a time when the chemical industry pushed boundaries with new polymers and specialty resins. The insatiable demand for innovative materials drove researchers to explore small molecules with more than one reactive group. The molecule’s dual nature appealed to chemists who saw the value in quick, reliable crosslinking. Over the decades, its production method shifted away from batch processes using sodium hydroxide and epichlorohydrin, evolving toward safer routes with better yields and fewer by-products.
It’s clear that the interest in allyl glycidyl ether grew as epoxy technology took off. Epoxy resins meant for protective coatings, electronics, and adhesives used this compound for improved performance and reliability. After tough environmental scrutiny in the 1970s, producers shifted away from wasteful syntheses. Process improvements shaped current standards for purity and safety— not just better for manufacturing but reassuring for anyone handling the substance.
Product Overview
Allyl glycidyl ether sits among the unsung heroes in many industrial formulas. Chemists see this liquid as a versatile building block, thanks to its reactive epoxy and allyl functionalities. Colorless and with a mild, ether-like aroma, this compound pours easily and dissolves in alcohol, ethers, and aromatic hydrocarbons. Each barrel promises chemical engineers plenty of options when seeking to modify resins or boost adhesion promoters. Guarantors of quality aim for minimal water content, as water triggers premature curing.
Requiring proper labeling and documentation, suppliers provide this molecule with clear CAS number identification and thorough certification. Besides confidence about purity, end-users often want batch-specific data, especially in pharmaceuticals and high-end electronics manufacturing.
Physical & Chemical Properties
Allyl glycidyl ether registers a boiling point around 156°C, staying liquid across typical laboratory and plant conditions. The molecular structure—combining an allyl group and an epoxide ring—hands it a unique reactivity profile. Its density hovers near 0.968 g/cm³ at room temperature, with modest viscosity and a flash point close to 37°C, so it demands careful storage. This compound’s volatility is enough to merit solid ventilation design, even before considering its low explosive limits.
The molecule doesn’t come with high water solubility, yet it mixes well with a variety of organic solvents. Chemists rely on this miscibility to craft specialized polymer networks and reactive diluents. In personal experience, even slight impurities in feedstock can alter end properties dramatically, making quality control essential—not just a paperwork task, but a process that shapes final product integrity.
Technical Specifications & Labeling
Suppliers label drums or bottles with the name “Allyl Glycidyl Ether” alongside synonyms—AGE, 3-allyloxy-1,2-epoxypropane, Glycidyl allyl ether—ensuring clarity in global trade. Key numbers take center stage: boiling point, molecular weight (114.14 g/mol), refractive index, and purity—usually >99%. Transport and GHS hazard symbols stay prominent on containers, signaling potential health risks and regulatory rules for moving chemicals between countries.
Material safety data includes everything from storage guidance to first-aid response. Any user, from researchers to plant technicians, expects hazard ratings and personal protective measures spelled out. A robust product information sheet separates responsible vendors from the rest.
Preparation Method
Large-scale preparation of allyl glycidyl ether typically starts with epichlorohydrin and allyl alcohol. Most manufacturers select a base-catalyzed reaction tailored for efficiency, suppressing side-reactions and maximizing selectivity. Chloride ion removal, drying, and fractional distillation complete the process. Working in industry for a decade sharpened my appreciation for how subtle tweaks—different catalysts, temperature shifts—can impact yield and by-product formation. Safe venting and scrubbing equipment prevent fugitive emissions, now a standard for any modern plant.
Continuous flow reactors add another layer of control, addressing previous bottlenecks in scale-up and reducing batch-to-batch variability. Even in laboratory work, keeping a close eye on the reaction exotherms plays just as vital a role as scale, especially since uncontrolled polymerization can lead to dangerous runaways.
Chemical Reactions & Modifications
Chemists prize allyl glycidyl ether for its dual reactivity—thanks to the strained epoxide ring and flexible allyl group. In epoxide chemistry, it acts as a reactive diluent or as a monomer in copolymer chains. Ring-opening reactions with amines, acids, or thiols build countless derivatives used in adhesives and tough coatings. The allyl group brings additional modification options, like crosslinking via free radical polymerization—opening doors to tailor materials for specific flexibility or durability demands.
One memorable project involved using this compound to adjust the flexibility of a high-gloss, solvent-free epoxy. It only took a small fraction to transform the resin’s feel and durability—no textbook can substitute the insight earned from such hands-on experimentation. Research journals detail catalytic systems that fine-tune molecular attachments, letting companies remain competitive by offering custom-aged, functionalized materials.
Synonyms & Product Names
Throughout the world’s laboratories and manufacturing plants, you’ll hear “AGE” tossed around, but the same substance answers to 3-allyloxy-1,2-epoxypropane, glycidyl allyl ether, or allyl-2,3-epoxypropyl ether. Each global region carries its own registry numbers or shorthand, so staying vigilant about product codes and shipping documents prevents mix-ups, especially when importing specialty chemicals.
Safety & Operational Standards
Any professional who’s handled allyl glycidyl ether knows that safety goes beyond just gloves and goggles. Inhalation of its vapors irritates mucous membranes, and skin contact causes sensitization. Consistent air monitoring in enclosed spaces brings peace of mind. Companies must post clear signage, maintain spill kits, and train workers on rapid response. Cross-contamination with food or drink gets ruled out by strict area segregation and robust procedural barriers.
I recall a factory audit that emphasized regular leak checks on seals and valves. Even one overlooked gasket invited headaches—flaring off volatile organic compounds and triggering regulatory fines. Reputable manufacturers invest in well-drained, explosion-proof storage rooms, treating worker safety as a smart business practice rather than a costly burden.
Application Area
Allyl glycidyl ether quietly shapes daily life, moving from specialty coatings that shield electronics to adhesives critical in aircraft and cars. Composite manufacturers employ it to modify resins, because it boosts toughness without making curing unmanageable. Optical fiber coatings depend on its high purity and tailored curing profile. Waterborne coating chemists pull it in to achieve precise balances of hardness and flexibility.
In prototyping, I’ve mixed AGE into epoxy blends for prototypes that withstood high-impact knocks and aggressive solvents. Professional painters, construction crews, and electronics assemblers count on the results, often without ever seeing the chemical itself.
Research & Development
R&D teams explore new catalysts and environmentally friendlier routes to allyl glycidyl ether, as industry goals shift towards greener production. Innovative surface coatings demand oligomers crafted from this building block, fine-tuned for UV curing and reduced volatile emissions. At several chemistry conferences, I’ve watched researchers debate how to reduce residual monomer content to ever-lower levels. Analytical chemists invest in new GC-MS methods to spot and minimize impurities that would otherwise cripple sensitive electronics or medical devices.
The search for novel derivatives never stops. Collaboration between universities and material companies leads to the development of new networks—epoxy-allyl systems that promise unmatched solvents or corrosion resistance for offshore infrastructure.
Toxicity Research
Extensive animal and cell studies map the risks of allyl glycidyl ether. The compound exerts cytotoxicity, with evidence of respiratory, dermal, and reproductive hazards at high exposures. Researchers have revealed the importance of chronic exposure tracking; data shows inhalation and skin contact can cause systemic effects well beyond immediate irritation. Regulatory agencies scrutinize every new study, as updated findings potentially reset permitted workplace exposure limits.
As someone who has worked alongside toxicologists, I’ve seen the effort invested in tracking exposure incidents and developing protocols for emergency decontamination. At the same time, epidemiological data proves sparse for low-level, chronic exposure, driving interest in long-term biomonitoring for exposed workers.
Future Prospects
New regulations raise the bar for purity, emissions, and traceability in every batch of allyl glycidyl ether. Manufacturers funnel resources into process updates, answering consumer demands for safer, more sustainable products. Applications in advanced photopolymers, flexible electronics, and low-VOC building materials drive the hunt for next-generation derivatives.
Looking ahead, the blend of regulatory tightening, technical innovation, and changing market needs keeps allyl glycidyl ether relevant. Industrial and academic researchers find themselves constantly revisiting this building block—a testament to its adaptability, yet a reminder that safe handling and ongoing toxicity review will remain non-negotiable priorities for years to come.
What is Allyl Glycidyl Ether used for?
Why People Use Allyl Glycidyl Ether
Ask anyone who has spent time in a polymer lab or factory, allyl glycidyl ether (AGE) isn’t a household name but it sure shows up in plenty of products around you. I remember wiping down a new desk and seeing the resin shine, that tough and glossy feel under my hand—that moment’s brought to you by compounds like AGE.
Most use comes down to its handy structure: an epoxide group on one end and an allyl group on the other. Chemists love raw materials with these two reactive spots. The epoxide end opens doors to building tough plastics that resist cracks and heat, while the allyl end bonds on to other things seamlessly. Plugging AGE into epoxy resins gives manufacturers a way to control flexibility or add extra resistance against chemicals, ultraviolet rays, and moisture—all from a single additive.
Applications on the Factory Floor and Beyond
AGE pops up in building materials, paints, coatings, and adhesives. The paints on bridges or industrial machinery owe much of their weatherproof toughness to how this ether helps build a sturdy, cross-linked network. Furniture coatings and automotive parts use it for the same reason—nobody wants a chipped or faded bumper after a year on the road.
You find AGE in electronics, too. The circuit board inside your phone, TV, or computer likely carries resins modified with this ingredient. These boards deal with heat and electrical stress every day. AGE gives manufacturers more control over how resins cure and allows the boards to survive repeated heating-cooling cycles. That means fewer breakdowns and longer gadget lifespans.
Health and Safety: Real Concerns
Working with AGE isn’t as simple as pouring milk. AGE can irritate the skin and eyes, and if inhaled in higher concentrations, it’s no joke for your lungs either. I’ve seen technicians in full gear handling the stuff—and there’s good reason for that. Regulatory agencies, including OSHA and the EPA, have published strict guidelines for storage, handling, and disposal. Manufacturers using AGE spend time and money to ensure vapors don’t escape into the air, and workers have barriers and protective gear.
Moving Toward Safer Alternatives and Practices
No chemical provides every benefit with zero risk. Over the years, companies have looked for alternative substances with lower toxicity, but replacing AGE isn’t always easy due to its unique structure and performance. Research continues in this direction, especially in places where frequent exposure could harm workers over decades.
I’ve seen success come from companies investing in better engineering controls—like closed systems and advanced ventilation—so workers barely come near the raw material. Training also helps: if folks know how to protect themselves, injuries and spills drop way down. For end-users, the good news is that most products containing AGE get processed so thoroughly the leftover chemical content is almost undetectable.
Keeping Innovations Safe
AGE delivers strengths modern products demand, with manufacturers weighing its benefits against what’s safest for workers and the environment. The path forward looks like it will pair careful handling, new safety tech, and steady research for ever-safer upgrades—and that’s the sort of balance the chemical industry chases every day.
What are the safety precautions when handling Allyl Glycidyl Ether?
Why This Chemical Deserves Respect
Allyl Glycidyl Ether, with its sharp odor and high reactivity, has earned a spot on my personal list of chemicals that demand constant respect. Years back, I watched an experienced lab hand mishandle just a small amount of this stuff. He got careless, knocked over a bottle, and ended up with a rash on his wrist where the glove had slipped. That pain made a lasting impression on all of us in the room. I realized then how quickly things can turn sideways if you don't approach chemicals like this one the right way.
One of the biggest risks here comes from its volatility. This ether isn’t just a bit irritating—skin contact can cause burns or allergies, and its fumes are rough on the lungs. Getting your nose too close spells trouble fast. It only takes a tiny splash or careless breath to invite a big problem.
Smart Handling Starts Before You Open the Bottle
Before getting started, gear up with the right stuff. I reach for chemical-resistant gloves, not the cheap latex kind, and a well-fitted apron. Goggles are non-negotiable. In my first months of serious lab work, I saw a colleague brush an eye with a contaminated glove. We lost the rest of our shift to an emergency flush and doctor visit. Even a splash from the cap or residue on the outside of the bottle can get you.
Good airflow matters as much as anything. Don’t trust a regular bench or windows. I stick to the fume hood every time. This chemical’s vapors creep, and the hood’s constant suction really is essential. At home or in workspaces without industrial ventilation, there’s no safe way to use it. This isn’t about being overly cautious; it’s about protecting your lungs and everyone else’s in the building.
Storage and Spills: Lessons Learned the Hard Way
A leaky cap or a cracked glass bottle taught me that storage can't be an afterthought. Allyl Glycidyl Ether reacts with moisture and can catch fire near heat sources. Keep it in a tight-sealed container—lined metal or certain plastics work—and stash it away from direct sunlight, strong acids, or oxidizers. Many experienced techs still make the mistake of storing incompatible chemicals together, leading to dangerous mixes or even fires.
If a spill happens, grabbing a handful of paper towels won't cut it. I've seen people try that, then panic as the chemical chewed through gloves or splashed up. Use absorbent materials meant for solvents, and control airflow to draw away vapors as you clean. Getting trained on how to use these kits beats Googling emergency instructions in panic mode.
Why Routine Matters
I recognize strict routines can seem annoying, especially to folks just starting out. But there are reasons every chemical safety sheet gets drilled into new hires. Wear the gear, use the hood, read the safety sheet every single time, even if you know it backward. I’ve learned through mishaps—none of them mine, thankfully—that short cuts end up costing more time and health in the end.
Eyes, nose, and skin all deserve serious guarding. If exposure happens, emergency showers and eyewash stations need to be close by and checked monthly. Quick action can make the difference between a scare and a hospital visit. Take training seriously and never work alone with Allyl Glycidyl Ether. Having backup nearby has saved more than one person from real harm.
Chemical safety is a living practice. People bring their lived experience, and over time, these habits become second nature. Being careful isn’t about fear—it’s about respect for yourself and others sharing the workspace.
What is the chemical formula and structure of Allyl Glycidyl Ether?
Basic Facts and Chemical Identity
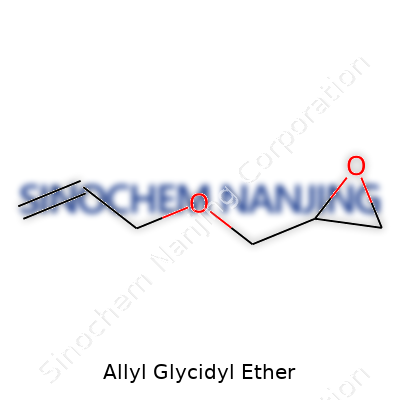
Walk into any polymer lab, and you’ll likely run into Allyl Glycidyl Ether (AGE) — a colorless liquid with a sharp odor. Chemical diagrams might seem mysterious for newcomers, but this compound has a clear formula: C6H10O2. Its molecular structure includes an allyl group and a glycidyl (epoxide) group, which gives it a dual personality. The molecule can be written as CH2=CH–CH2–O–CH2–CHOCH2, joining the vinyl group, ether linkage, and reactive epoxide ring.
Where Structure Drives Use
The way AGE binds these two groups lets it stand out. The allyl moiety tends toward reactions with free radicals, while the epoxide opens doors for ring-opening reactions with acids and bases. Many chemists, drawn by its reactivity, use it as a bridge-builder in polymers and resins. Acrylic and epoxy manufacturers often reach for AGE to tweak their products’ flexibility and stickiness.
Years ago, while working with adhesives, I learned that AGE’s structure allowed us to tailor properties that standard epoxies couldn’t deliver alone. The ether oxygen provides some added stability. The epoxide ring breaks open under the mildest conditions, making further reactions quick and clean. Its structure also means it dissolves well in many organic solvents, an advantage when you’re mixing complex resin blends.
Health, Safety, and Responsible Handling
Despite its popularity in industry, AGE isn’t gentle on people. It irritates eyes and skin, and can trigger coughing or headaches if vapors fill the lab. Long-term overexposure can sensitize some users, making protective gear (goggles, gloves, working vents) a baseline in labs using this compound. Reading the safety data sheet, you’ll see warnings about accidental spills or skin contact.
Environmental Perspective and Alternatives
AGE’s reactive groups provide big value, but they present waste challenges. Wastewater or emissions require real caution. Facilities working with AGE must keep emissions low and prevent its leaking into streams or soil. I once witnessed a spill in a small research lab; even minor accidents forced a scramble for containment and cleanup. These experiences pushed my team to evaluate greener or less hazardous alternatives where possible.
Solutions for Risk and Sustainability
Safer practices cut risks significantly. Use closed mixers and local exhaust systems, especially in factories or larger operations. Online resources, like those hosted by the American Chemical Society, detail step-by-step cleanup guidelines and long-term monitoring. Solvent alternatives or new formulations without AGE remain a developing frontier, but some researchers experiment with less volatile epoxides or bio-based monomers to limit exposure and environmental damage. Anytime supply-demand shifts threaten the production chain, companies who have already diversified their chemistries breathe easier.
Looking Forward
Chemistry’s not just about mixing and measuring — it’s about the choices that steer safety and progress. AGE’s formula provides the power to build reliable materials, but its risks require responsibility. Respecting the molecule in both the lab and the world beyond it keeps innovation moving forward, without putting health or habitats in jeopardy.
How should Allyl Glycidyl Ether be stored?
Why Storage Matters with Allyl Glycidyl Ether
Allyl Glycidyl Ether brings up some old memories from my time working in a small lab, where the whiff of certain chemicals always signaled a job that demanded more respect and care. This compound has a punch when it comes to sensitivity—it reacts with air and moisture, and a sharp sense of responsibility keeps accidents away. Ignoring routine can have real consequences.
Fire departments and industrial safety reports often show how ignoring even one step in chemical storage can set off a chain of problems. Take it from seasoned plant operators: even a small spill can become an emergency, so storing Allyl Glycidyl Ether isn’t something folks just guess their way through. Sealed containers, out of sunlight, and far from anything that even hints at heat. It’s not about ticking off checklists, but about keeping the workplace, and everyone in it, upright and safe.
Daily Habits Make a Difference
Working with volatile chemicals taught me that clear routines save the day. Labeling every container the moment it’s opened, keeping logs, marking use-by dates—these steps shrink the risks. Reaching for a flask and knowing exactly what’s in it, or seeing at a glance whether a batch’s shelf life has passed, saves a world of trouble. One time, I caught a cracked lid just in time, and it drove home that inspection routines can’t be taken lightly.
Guidelines set by agencies like OSHA and the National Fire Protection Association draw on thousands of incidents. They call for Allyl Glycidyl Ether to rest in a tightly sealed drum, kept in a cool, well-ventilated room, where accidental heating or static buildup can't catch anyone by surprise. Ventilation does more than just freshen the air—it chases off vapors, knocking down the risk of inhalation or fires. The right storage temperature—right around what’s considered room temperature—slows down chemical changes and keeps the compound stable. Pushing past those conditions gives moisture a way into the container, and corrosion or dangerous breakdowns can follow.
Simple Choices Boost Safety
Sometimes it’s easy to cut corners—maybe a container seems “good enough” or a storage room looks safe with the naked eye. But Allyl Glycidyl Ether requires a non-reactive lining in its container—stainless steel or high-quality glass. Forgetting that detail causes leaks and contamination, costing time and sometimes a whole batch of product. Storing the ether away from acids, bases, and oxidizers keeps reactive mishaps at bay.
Spill kits and eye wash stations must sit close at hand. The best labs don’t just have them—they train every worker to use them, and keep clear walkways so everyone knows exactly which route leads to safety. Regular drills help people react to spills or inhalation hazards without hesitation.
Responsible Disposal and Handling
Part of safe storage is planning for what happens after use. Wastes and residues need closed, marked containers, kept in a separate area until pickup. The safest chemical facilities keep cleaning materials and protective gear near the storage zone, and encourage workers to report any irregularities right away.
In the end, storing Allyl Glycidyl Ether is less about strict rules on paper, and more about building a culture of care. If someone new asks why storage steps matter, stories from the field work better than any manual. Good habits, real attention, and simple respect for the hazards go a long way.
What are the physical and chemical properties of Allyl Glycidyl Ether?
Physical Traits You Notice Right Away
Allyl glycidyl ether grabs attention with its clear, colorless liquid form. This stuff gives off a strong, sharp scent—not unlike some glues or solvents found in workshops. You’ll find it carries a relatively low boiling point of around 156°C, and a specific gravity just a touch below water at about 0.97, meaning it tends to float. It slides easily across surfaces with its low viscosity, and easily dissolves in most common organic solvents such as ether or alcohol. Water won’t make it vanish—allyl glycidyl ether fights with moisture and only mixes sparingly.
This liquid’s volatility matters most in daily handling. Small spills can fill a room with its pungent odor quickly. Some people develop headaches or nose irritation from lingering in those vapors. You learn fast to work with it in a well-ventilated spot or under a fume hood.
Chemical Features That Matter
The backbone of allyl glycidyl ether includes two reactive sites: the epoxy ring and the allyl group. Both spots show a willingness to jump into new reactions. The epoxy group brings plenty of tension—like a coiled spring—ready to open up and link to other parts. This behavior lies behind its use in making specialty polymers, adhesives, and as a reactive diluent for epoxy resins in everything from electronics to automotive parts.
Temperatures, strong acids, or bases easily crack open the epoxy ring. Watch out for chain reactions that can spiral into runaway polymerizations if someone gets careless with process controls. I’ve seen a mishap or two in university labs—the jar heats up, contents foam and overflow, sometimes in a spectacular (and smelly) mess.
Health and Environmental Impact
Handling allyl glycidyl ether with bare hands isn’t wise. It irritates skin and eyes almost instantly, and inhaling the fumes brings on coughing and burning sensations. Chronic skin contact can trigger allergies for some unlucky folks, so those blue chemical gloves and goggles become your best friends.
Every chemical brings risks if spilled or dumped. This liquid makes its way into stormwater if ignored, and aquatic life suffers—allyl glycidyl ether poisons fish and insects even in low concentrations. Clean-up rarely works with a mop and bucket; chemical spill kits and trained staff make a difference in keeping damage contained. Local fire departments and hazmat teams usually know how to neutralize it but staying cautious with storage and disposal works better than any heroic clean-up.
Solutions for Safer Handling
Companies and labs building a culture of safety prove the best defense. Good airflow, proper gloves, and dedicated chemical storage shelves set the foundation. Emergency showers and eye-wash stations never get ignored in well-run spaces. Regular training prevents accidents—people know how to recognize symptoms, handle spills, dispose of waste, and treat affected coworkers instantly.
Replacing allyl glycidyl ether sometimes makes sense where health risks outweigh the benefits. Some manufacturers swap in less aggressive alternatives—though trade-offs in durability or performance sometimes follow. Green chemistry research pushes to design safer substitutes. Until those options hit the mainstream, informed respect and careful protocols keep users safe and the environment cleaner.
| Names | |
| Preferred IUPAC name | 2-(Prop-2-en-1-yloxy)oxirane |
| Other names |
AGE 1-Allyloxy-2,3-epoxypropane Glycidyl allyl ether 2,3-Epoxypropyl allyl ether Allyl glycidol ether |
| Pronunciation | /ˈæl.ɪl ɡlɪkˈsɪd.ɪl ˈiːθər/ |
| Identifiers | |
| CAS Number | 106-92-3 |
| Beilstein Reference | 1220712 |
| ChEBI | CHEBI:42358 |
| ChEMBL | CHEMBL135724 |
| ChemSpider | 12980 |
| DrugBank | DB14006 |
| ECHA InfoCard | 03abea4a-8cac-4928-adb4-69c47bb2e7c1 |
| EC Number | 202-936-7 |
| Gmelin Reference | 7396 |
| KEGG | C19533 |
| MeSH | D004055 |
| PubChem CID | 7890 |
| RTECS number | RD0350000 |
| UNII | G5A4I80Y7F |
| UN number | UN2341 |
| Properties | |
| Chemical formula | C6H10O2 |
| Molar mass | 100.13 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Strong, unpleasant |
| Density | 0.963 g/mL at 25 °C (lit.) |
| Solubility in water | miscible |
| log P | 0.27 |
| Vapor pressure | 3.7 mmHg (20°C) |
| Acidity (pKa) | 15.9 |
| Basicity (pKb) | 13.97 |
| Magnetic susceptibility (χ) | -8.11×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.423 |
| Viscosity | 2.5 mPa·s (25 °C) |
| Dipole moment | 2.45 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 340.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -4063.7 kJ/mol |
| Pharmacology | |
| ATC code | |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H302, H312, H315, H319, H332, H341, H351, H411 |
| Precautionary statements | P210, P261, P280, P301+P310, P305+P351+P338, P331 |
| NFPA 704 (fire diamond) | 3-2-2 |
| Flash point | 43 °C (109 °F) (closed cup) |
| Autoignition temperature | 215 °C (419 °F; 488 K) |
| Explosive limits | 1.8% - 18.3% |
| Lethal dose or concentration | LD50 (oral, rat): 640 mg/kg |
| LD50 (median dose) | 238 mg/kg (rat, oral) |
| NIOSH | AS3325000 |
| PEL (Permissible) | PEL: 1 ppm (3.6 mg/m³) |
| REL (Recommended) | 1 ppm |
| IDLH (Immediate danger) | 500 ppm |
| Related compounds | |
| Related compounds |
Epoxy compounds Allyl compounds Glycidol |

