Allyl Acetate: A Deep Dive into an Unsung Chemical Player
Historical Development
Looking back, chemists in the early twentieth century chased better methods to create complex molecules. Allyl acetate first came together through the work of scientists exploring unsaturated esters. The process of manufacturing plastics, resins, and coatings benefited from this work. The real breakthrough came when new catalysts like palladium and newer reactor technologies allowed for commercial synthesis on a big scale, helping industrial chemistry take massive steps forward. Refineries often searched for efficient ways to harness byproducts of petroleum—allyl alcohol and acetic acid both came from this line of thinking, finding a route right to allyl acetate. Chemical producers recognized its versatility and started scaling up during the mid-century boom, giving industry a dependable building block for everything from adhesives to specialty polymers.
Product Overview
Allyl acetate is an ester created by combining allyl alcohol with acetic acid. Its structure includes an allyl group bonded to an acetate, giving it a reactive double bond while also letting it dissolve into other liquids with ease. In practice, containers of allyl acetate line shelves in research labs, paint factories, and ingredient rooms in perfume facilities. Its subtle, almost fruity smell belies its industrial backbone. Market demand rises and falls in step with construction, automotive, and consumer goods sectors, so producers closely track end-user habits and material cycles before investing in plant expansions.
Physical & Chemical Properties
Transparency and liquid form mark allyl acetate in a pure state. Its boiling point hits around 140 degrees Celsius, while the flash point—vital for safety protocols—lies near 23 degrees Celsius. Density clocks in just below water, so spills don’t sink to the bottom of a containment basin. Chemically, it resists water mixing but dissolves in most organic solvents, giving paint chemists and resin producers real flexibility. The double bond in its allyl group drives plenty of reactions, letting it become the backbone of new polymers or act as a stepping-stone to other organic molecules.
Technical Specifications & Labeling
Industrial buyers specify impurities down to parts per million for allyl acetate. A common technical grade falls above 98% purity, with moisture and acid contents tightly controlled. Barrel and drum labels show chemical formula C5H8O2, United Nations number 1301, and warnings about flammability in big red letters. Most producers follow rules laid down by organizations such as the Global Harmonized System and local hazard communication standards, with Material Safety Data Sheets explaining every risk in direct language. Storage vessels keep out sunlight, and temperature is monitored—regulatory audits sometimes show that a lot can go wrong if details slip through the cracks.
Preparation Method
To prepare allyl acetate, chemical plants usually use the catalytic reaction of allyl alcohol with acetic acid. Palladium-based catalysts work best under moderate pressure and heat, producing high yields without much waste. This process offers a clever way for companies to turn two relatively cheap starting materials—both often derived from petroleum—into a higher-value product. Engineers focus on controlling reactor temperature and catalyst quality for each batch. Any deviation can mean off-spec product or dangerous pressure buildups, so the whole process needs tight controls and quick response when parameters drift.
Chemical Reactions & Modifications
Allyl acetate jumps into a wide range of synthetic pathways. Most notably, chemists hydrolyze it to make allyl alcohol, which then builds up everything from flame-retardant plastics to herbicides. Its double bond kicks off further reactions such as epoxidation, giving manufacturers a route to glycidol and glycidyl ethers—materials used in specialty adhesives and epoxy resins. Researchers also explore grafting reactions, using the allyl group’s reactivity to attach functional arms onto polymers, leading to better performance in coatings and binders. Its chemical flexibility means new downstream products keep showing up in the market as designers push boundaries.
Synonyms & Product Names
On shipping papers and regulatory forms, “allyl acetate” might show up under several names. Some call it 2-propenyl acetate or simply acetic acid allyl ester. Certain companies market it under proprietary names for use in fragrances or custom plastics, but usually, the IUPAC and CAS conventions dominate. In trade, knowing all possible synonyms protects against costly mix-ups—especially in multilingual trading hubs, where labeling errors can shut down production lines or trigger regulatory headaches.
Safety & Operational Standards
Allyl acetate acts as a significant fire risk due to its low flash point. Handling it demands attention to spark-proof equipment, constant ventilation, and static discharge controls. Workers suit up in chemical-resistant gloves and splash shields to avoid skin and eye contact—its vapors can cause irritation and more serious problems if inhaled for too long. Emergency drills play a key role in safe handling, especially where large drums get moved in and out of storage. Regulatory bodies such as OSHA and the European Chemicals Agency set limits on workplace exposure and enforce proper emergency response plans, pushing companies to constantly update their training and hazard controls.
Application Area
Allyl acetate serves as a backbone for many consumer and industrial products. Paint manufacturers blend it into resins for strong, weather-resistant coatings. Plastics makers use it as a feedstock for manufacturing flexible material found in hoses, wire coatings, and packaging films. Scent formulators add small quantities to fragrances and personal care items, giving them a crisp, slightly fruity undertone. Some pharmaceutical labs look at allyl acetate for building more complex molecules, tapping its reactive allyl group for custom synthesis work. This range of uses keeps demand steady across sectors, fueling investments into both traditional and novel applications.
Research & Development
Innovation often starts in the lab, where chemists screen new catalysts that can lower reaction temperatures or boost yields in allyl acetate production. Recent years saw breakthroughs in recycling palladium, making large-scale processes more sustainable and cost-effective. Other researchers dig into sustainable feedstock sources, like bio-based allyl alcohols, to cut the industry’s carbon footprint. Performance additives based on allyl acetate lead to more durable paints and adhesives. Scientists also tailor its reactivity for making eco-friendly plasticizers, aiming for regulatory approval on materials with lower toxicity. This pipeline of R&D helps keep the compound in the spotlight among specialty chemical suppliers and applied research institutions.
Toxicity Research
Concerns around allyl acetate’s impact on health push researchers to study its breakdown products and behavior in living systems. Animal tests indicate irritation potential to airways and skin. At high concentrations, more severe symptoms may show up, including systemic toxicity. Regulatory agencies collect this data and set occupational exposure limits to prevent problems among workers who handle the chemical every day. Environmental scientists monitor how quickly allyl acetate and its derivatives break down if spilled, checking for bioaccumulation or groundwater impacts. This type of research serves public safety, keeping both the community and the environment out of harm’s way.
Future Prospects
Interest in greener chemistry promises fresh directions for allyl acetate manufacturing. Startups and research labs push for renewable raw materials, cutting fossil fuel reliance by developing ways to produce allyl alcohol from plant-based sources. Process optimization tends to lower waste and energy consumption, addressing both cost and environmental demands. Application areas keep growing as industries hunt for adaptable intermediates that can enhance product durability and performance without new safety headaches. The story of allyl acetate turns on clever synthesis and thoughtful safety—core values that shape an industry always looking for the next smart solution.
What is allyl acetate used for?
Understanding Where It's Used
Most folks probably don’t recognize the name allyl acetate, but manufacturers have been working with it for decades. It’s a colorless liquid with a strong, fruity smell. That smell isn’t just for show—a lot of the world’s biggest consumer brands use it to create flavors and scents. I’ve spent time in labs and plants where just a bit of this stuff will knock the socks off anyone not prepared for its punch.
The main reason people care about allyl acetate is because it acts as a shortcut. Chemists make things with it that they’d struggle to create otherwise. Take allyl alcohol, for example. This alcohol forms the backbone of many everyday plastics, resins, and coatings. Instead of taking the hard road, companies react allyl acetate with a base, and, just like flipping a switch, they have their allyl alcohol. This chemical ends up in water purification membranes, clear plastics, and even specialty adhesives.
In the World of Flavors and Fragrances
Here’s where things get interesting. That fruity aroma means flavor and fragrance companies put allyl acetate to work. They don’t bottle it straight, though. They use it to make compounds that mimic the smell of pears, apples, and tropical fruit. Folks in the flavor industry use these derivatives to give chewing gum, candies, and sometimes even toothpaste a certain zing. I once heard a flavorist say, “A few drops will make a pound of product bloom.”
Big perfume houses rely on related chemicals, too. Some of the light, sweet notes in high-end perfumes start out from allyl acetate. A little goes a long way, which matters in an industry where every drop gets measured and costs can spiral. These uses often never make it onto a product label, but they shape the smells and tastes most people recognize.
A Critical Tool for Making Plastics and Pesticides
Now, turn to the industrial world. Engineers often need compounds that can keep up with tough conditions, like high heat and moisture. Allyl acetate helps produce polymers that stand up to stress and weather well outdoors. It’s handy for making electrical laminates and even bulletproof glass. For anyone working in manufacturing, reliability comes down to choices like this—by picking the right building blocks, companies make sturdier, longer-lasting products.
Agriculture also owes a tip of the hat to this chemical. Some pesticides begin their life in a reactor filled with allyl acetate. It helps chemists tweak how plant-protection agents perform. Many modern pesticides break down faster in the environment, which gives farmers more options while protecting water and wildlife.
Risks, Safety, and What Could Change
With these big benefits also comes risk. I’ve seen lab guides list allyl acetate as a potential irritant, something that causes pain if you breathe too much or get it on your skin. Good ventilation and solid safety training matter—workers shouldn’t take shortcuts here. Both the U.S. Environmental Protection Agency and OSHA track these risks, and smart companies adapt by training staff, keeping workspaces clean, and always labeling containers correctly.
Green chemistry startups have begun building safer, bio-based substitutes. Finding new ingredients reduces hazards and keeps costs down over time. More companies are eyeing renewable plant-based routes because supply chain issues and environmental regulations keep piling up. That could tilt the balance over the next decade, bringing safer products to shelves and lessening environmental footprints.
What is the chemical formula of allyl acetate?
Understanding Allyl Acetate: The Formula at a Glance
Allyl acetate carries the chemical formula C5H8O2. Those five carbon atoms, eight hydrogens, and two oxygens don’t just form a string of letters and numbers on paper—they represent a crucial building block in manufacturing and daily life. Growing up in an industrial town, the smell of solvents and chemicals like allyl acetate lingered near factories. We rarely thought about what powered these plants, but understanding the backbone of such compounds matters, especially as society grows more aware of chemical footprints.
Why This Simple Formula Drives Industry
Allyl acetate isn’t just a shelf item at a chemical supply company. Its formula, C5H8O2, gives manufacturers a shortcut to important products. The backbone of perfumes, plastics, and even some pharmaceuticals can trace roots to this molecule. Take synthetic glycerol as an example. Most folks recognize glycerol from skincare products, but industry needs it in bulk for all kinds of uses. By reacting allyl acetate with water, plants can churn out glycerol without relying on animal fats, helping to meet both demand and ethical standards.
The typical production of allyl acetate happens by reacting propene with acetic acid and oxygen. Factories have used this process since the early twentieth century. As global demand for plastics and flexible coatings increased, the C5H8O2 formula became a mainstay in chemical plants. Old photos from industry trade magazines show massive steel reactors and monitoring panels, all devoted in part to shepherding this conversion. The numbers sound dry, but the scale is massive: millions of tons roam across supply chains each year.
Public Health: Less Toxins, More Transparency
Living near production zones brings up another side of the allyl acetate story. Its sharp odor signals the presence of a reactive chemical, and breathing too much of it can irritate the eyes and lungs. Chemical plants follow strict guidelines to limit emissions, but communities have a right to clear answers about what’s in the air. After pushback from local families, several plants ramped up free air quality monitoring and shared data monthly. Real-time updates helped spark more trust between workers, residents, and companies.
Navigating the Road Ahead
Sustainability grows in importance every year. Biotech firms now hunt for greener ways to make allyl acetate using enzymes and plant-based feedstocks. These efforts aim to cut greenhouse gas output and offer biodegradable pathways where possible. Such research holds promise; a student-led startup near my hometown even won funding to scale up a plant-based propene alternative. Reducing fossil input ties into climate goals and can reshape chemical economics over the next decade.
Tracking chemical formulas such as C5H8O2 may seem like a detail designed for chemistry textbooks, but awareness pays off in larger ways. Choices made at the factory level can send ripples through local environments, everyday products, and global sustainability targets. A simple chemical formula stands for more than ingredients—it fuels the engines of change.
Is allyl acetate hazardous or toxic?
What Exactly Is Allyl Acetate?
Allyl acetate shows up in more industrial settings than the average person might expect. It’s a colorless liquid with a sweet, fruity odor, commonly used in making flavors, fragrances, and some plastics. Most people don’t realize how much time goes into keeping chemicals like this contained and managed—having worked in a food manufacturing facility, I learned firsthand that every barrel and drum requires strict tracking. Even trace exposure raised concerns on the factory floor.
Health Risks and Toxicity
People have good reason to question the safety of industrial chemicals. Research from the National Institute for Occupational Safety and Health shows skin contact with allyl acetate may lead to redness, irritation, and blisters. Breathing in its vapors could cause a sore throat, coughing, headaches, or dizziness. Longer exposures—especially without proper safety gear—risk more severe effects, including damage to lungs and nervous system.
The chemical doesn’t rank near the worst offenders like benzene or formaldehyde, but its hazards pile up when workplace controls slip. Swallowing even small amounts causes nausea, vomiting, and intestinal pain. I remember a coworker experiencing a minor splash; the site nurse responded within minutes, washed the area extensively, and sent him for a quick check-up. That incident left a lasting impression: protocols exist for good reason.
Environmental Impact
Anytime a factory handles allyl acetate, accidental spills threaten water and soil. This chemical breaks down eventually in the environment, but aquatic life faces the brunt before that happens. Even moderate releases harm fish and disrupt small aquatic organisms. Facilities near water sources must act quickly to prevent runoff from entering drains and streams.
Regulations and Worker Protection
Government agencies keep a close eye on allyl acetate. The Occupational Safety and Health Administration (OSHA) has set exposure limits for workplaces, and the Environmental Protection Agency monitors its use to avoid spills. These rules are not just boxes to check off; I saw employees lose their tolerance for shortcuts after learning about chemical burns the hard way. Every weekly briefing drove home that skipping gloves or masks wasn’t an option.
In my experience, companies that train workers and supply protective gear see fewer accidents. Simple fixes—like good ventilation, PPE, and fast emergency response—make a world of difference. Spills are rare with proper storage. For those who handle chemicals for a living, clear labels, up-to-date safety data sheets, and regular training stand as non-negotiable.
Possible Solutions and Safer Practices
The goal goes beyond just avoiding accidents. Substituting less hazardous chemicals in those flavor and fragrance labs helps reduce risk. Engineers designing storage areas now focus on spill containment right from the blueprints rather than retrofitting later. Encouraging a “safety-first” attitude isn’t abstract theory—it’s something everyone from line workers to senior managers must live daily.
Consumers play a part, too, by supporting manufacturers with transparent safety records and robust third-party oversight. After all, safeguarding health and protecting the planet works best as a shared effort.
How is allyl acetate produced?
Everyday Chemistry Shaping Industry
I remember watching a glass chemist at work, the way steam swirled above beakers and the sharp smells that crept through the lab. That different, sweet scent comes up again with allyl acetate, a compound not many people outside the chemical industry know. But companies use it everywhere—paints, adhesives, and even in the flavors sprinkled into candy or chewing gum. So, how do you actually produce something so useful, and why does it matter?
The Nuts and Bolts of Production
The main recipe for allyl acetate uses propene (a gas made during oil refining), acetic acid, and oxygen. The process goes by a technical name: catalytic acetoxylation. It relies on palladium as the catalyst. That means mixing up propene and acetic acid with a dash of air, then pushing them over palladium supported on silica or alumina at around 100°C to 150°C. Out of that reactor comes allyl acetate, water, and carbon dioxide.
What stands out about this method? For starters, it skips chlorine. Producers used to use allyl chloride as a starter ingredient, but that pathway leads to harsh byproducts and wastes. Catalytic acetoxylation steps around some tough safety and environmental problems, which makes sense given how much fuss regulators have stepped up in the past twenty years. I can recall walking through older plants and seeing hazmat signage everywhere because of chlorine. That shift to cleaner methods has real consequences for workers and for the air around a busy chemical town.
Environmental Reality
Producing chemicals always brings up bigger questions: where does the feedstock come from, what waste do you dump, and how much energy does the process guzzle? Using propene, a byproduct of fossil fuel cracking, means allyl acetate production still leans on oil and gas. It isn’t a green dream yet. Modern plants recover unused propene and recycle it, cutting down losses. And compared to older pathways, newer production setups send less noxious stuff down the drain.
Energy demand still nags at the conscience. Running these reactors at high enough temperature means burning fuel somewhere in the chain. If companies shift toward renewable-powered plants, the carbon footprint: shrinks, but so far, petrochemicals still drive the train.
Why Getting It Right Matters
Making big batches of allyl acetate safely and efficiently matters for both the paycheck and the planet. Poor handling of acetic acid or oxygen risks high-pressure accidents—combustion in the works. Plant shutdowns over safety violations cost serious money and rile up the neighborhood. I’ve seen first-hand how communities push back if a plant fouls the air or nearby river, leading to tighter rules and more expensive upgrades.
With cleaner chemistry, better reactor designs, and tools for handling waste, the chemical industry saves on fines and legal headaches. Newer processes even let some plants use less precious metals, slashing catalyst cost. Every corner shaved off production cost gives industries an edge—but skimping on standards just swings back as lawsuits and bad press.
Looking Ahead
Green chemistry has promise here. Research teams try out new catalysts, using less palladium or looking for ways to pull oxygen from the air more cleanly. Bio-derived routes for propene pop up in journals, though they’re not mainstream yet. It reminds me of how fast old rules vanish once the technology gets good enough to catch.
At the end of the day, making allyl acetate mirrors bigger choices about efficiency, safety, and impact. Each advance means less waste, safer jobs, and maybe—sooner than later—paints and adhesives built from more renewable roots.
What are the storage and handling requirements for allyl acetate?
A Closer Look at Allyl Acetate in Everyday Industry
Allyl acetate pops up in factories more often than most people would guess. It shows up in the production of plastics, coatings, and some adhesives. This creates an obligation: treat the substance with care, both for worker safety and for the sake of the environment. Just one whiff will remind anyone working in a lab or plant that this chemical packs a volatile punch. Having spent years around chemical warehouses, I’ve seen how even seasoned teams slip up on routine checks if protocols grow stale. So, how this compound is tucked away and moved around is not a classroom exercise—it’s critical.
Controlling Temperature and Airflow
Let allyl acetate sit at the wrong temperature and the risk of fire or explosion rises quickly. Its flash point sits at around 13°C (55°F), which means leaving it by a window in July or next to a heater in winter could spell disaster. Storage calls for a cool, well-ventilated space. Flammable liquid storage cabinets provide one solution. These cabinets protect against accidental heat sources and shield the material from direct sunlight. Underground, rooms dedicated to flammables—fitted with fire suppression and proper airflow—offer another safeguard. Poor ventilation ramps up not only the danger of ignition but also exposes workers to fumes that can irritate lungs and mucous membranes. Any warehouse manager who’s had to clear a building due to a chemical cloud knows those numbers on the safety sheets are there for a reason.
Choosing Safe Containers and Storage Practices
The type of container matters as much as the room it’s kept in. Allyl acetate tends to chew through plastics and rubber, so stainless steel or glass offers a better choice. Neglecting this detail often leads to slow leaks, which don’t just waste money — they add headache to any clean-up operation. Tight-fitting lids, careful labeling, and shipping only the amount needed also cut down on the risk. In facilities that take safety seriously, supervisors check seals and markings by hand, and conduct regular inventory sweeps. Automating some of the checks with barcodes and digital logs cuts down on human error. Still, nothing beats a trained eye for spotting trouble before it grows.
Training and Spill Response
No matter how strong the container or how cool the room, people make all the difference. Clear protocols for moving, loading, and emergency response keep small mistakes from ballooning into disasters. Proper gloves, chemical splash goggles, and flame-resistant uniforms serve as everyday armor. Companies that hold monthly drills not only meet regulatory requirements but also create habits that stand up in an emergency. I once watched a rookie mist over the threat of static electricity. Training pointed out that something as simple as the right footwear and grounded equipment stops ignition before it starts.
Keeping Eyes on the Big Picture
Effective storage and handling of allyl acetate stand as a checkpoint for a company’s wider safety culture. Regulators like OSHA and EPA lay out clear lines for fire suppressants, ventilation specs, and accident logs. Still, it takes a hands-on approach from everyone involved to keep risks in check. Digital tracking, mechanical upgrades, and thorough staff education each bring real value. As demand for industrial chemicals rises, stakes only move higher. Solutions start with basic diligence—then grow with each lesson learned on the shop floor.
| Names | |
| Preferred IUPAC name | Prop-2-en-1-yl ethanoate |
| Other names |
Allyl ethanoate Acetic acid, allyl ester 2-Propenyl acetate |
| Pronunciation | /ˈælɪl əˈsiːteɪt/ |
| Identifiers | |
| CAS Number | 591-87-7 |
| Beilstein Reference | 1209074 |
| ChEBI | CHEBI:140453 |
| ChEMBL | CHEMBL1200271 |
| ChemSpider | 6762 |
| DrugBank | DB14019 |
| ECHA InfoCard | 100.003.194 |
| EC Number | 205-538-0 |
| Gmelin Reference | Gm. 5914 |
| KEGG | C06730 |
| MeSH | D000573 |
| PubChem CID | 7842 |
| RTECS number | AT2450000 |
| UNII | LHM6999UI7 |
| UN number | 1098 |
| CompTox Dashboard (EPA) | DTXSID1020296 |
| Properties | |
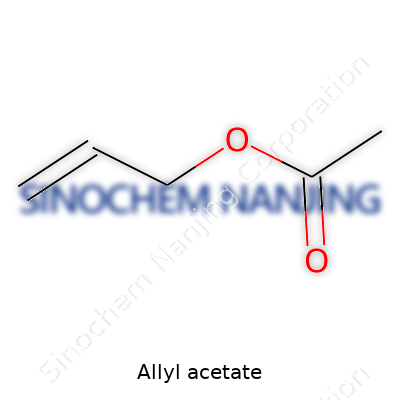
| Chemical formula | C5H8O2 |
| Molar mass | 100.12 g/mol |
| Appearance | Colorless liquid |
| Odor | fruity, pungent |
| Density | 0.936 g/mL at 25 °C (lit.) |
| Solubility in water | 1.00 g/100 mL (20 °C) |
| log P | 0.76 |
| Vapor pressure | 24 mmHg (20°C) |
| Acidity (pKa) | 4.75 |
| Basicity (pKb) | 13.48 |
| Magnetic susceptibility (χ) | -12.36×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.400 |
| Viscosity | 2.39 mPa·s (25 °C) |
| Dipole moment | 1.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 322.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -370.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1948 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V03AB36 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07, GHS08 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H301, H311, H331, H314 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P301+P310, P303+P361+P353, P305+P351+P338, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-2 |
| Flash point | -17 °C |
| Autoignition temperature | 225°C (437°F) |
| Explosive limits | 2.4–11.6% |
| Lethal dose or concentration | LD50 oral rat 830 mg/kg |
| LD50 (median dose) | 930 mg/kg (rat, oral) |
| NIOSH | AM2875000 |
| REL (Recommended) | 0.1 ppm |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Allyl alcohol Acetic acid Vinyl acetate Ethyl acetate Isopropenyl acetate |

