Adenosine Triphosphate Disodium Salt: An Evolving Foundation in Life Sciences
Historical Development
Stories about adenosine triphosphate disodium salt stretch back to the early 20th century, a time when scientists hunted for the fuel that keeps living cells going. The tale starts with Karl Lohmann, who identified ATP in 1929 while working on mechanisms of muscle contraction. The early years saw ATP as a curiosity, but after Fritz Lipmann outlined its starring role in cellular energy transfer, the floodgates opened. Labs across continents recognized ATP as more than a molecule; it offered a tangible key to understanding muscle physiology, cellular respiration, and biosynthesis. By bridging chemistry and biology, ATP’s significance expanded rapidly. During my own biology studies, I saw how historic debates about chemical energy storage—once confined to classrooms—were grounded in ATP’s chemical dynamics, showing how laboratory investigation shapes classroom insight.
Product Overview
Adenosine triphosphate disodium salt appears as a finely granulated white to off-white powder. Purified ATP, in this sodium salt version, offers greater stability in both storage and preparation—important for research teams seeking consistency across experiments. Scientists rely on this form for delivering precise concentrations without hassle. It dissolves in water, creating clear solutions that drive cellular experiments, enzyme assays, and energy metabolism studies. My own hands-on work with ATP focused on enzyme kinetics, and this product eliminated concerns around weigh-outs and pH swings, giving more repeatable results than less stable forms.
Physical & Chemical Properties
Structurally, this salt form brings together the adenine base, a ribose sugar, and three phosphate groups. Its molecular formula is C10H14N5Na2O13P3, and it packs a molar mass just under 551.2 g/mol. Under standard lab humidity, ATP disodium salt stays robust, holding form when stored below -20°C in sealed containers. Its solubility tops 50 mg/mL in water, with pH values near neutral, which fits most biochemical workflows. The phosphate backbone signals strong negative charge, so it behaves predictably in electrophoresis and chromatography, making purification more manageable in both research and industrial scale-up. From my experience, ATP’s swift hydrolysis under heat or alkaline conditions keeps everyone mindful—one missed ice bath, and you’ll be restocking.
Technical Specifications & Labeling
The ATP disodium salt involves strict quality parameters: purity exceeding 98%, measured by HPLC; moisture and impurity thresholds; and certificates for endotoxin-free status, especially for clinical or injectable applications. Labels highlight batch number, manufacturing date, expiry, storage temperature, and, crucially, the absence of harmful heavy metals. For scientists, seeing “Pyrogen-Free” and “Non-GMO” tags builds confidence before trials. My own experiments ran into issues with impure ATP, which threw off kinetic measurements—a clear label with third-party validation made a big difference in experimental trust.
Preparation Method
Production starts with fermentation processes, where select microbial strains churn out adenosine monophosphate (AMP) or adenosine diphosphate (ADP). Inorganic phosphate donors, such as phosphoryl chloride or polyphosphate, aid in assembling that third phosphate. Chemists then use sodium salts to buffer and stabilize the product. Filtration, crystallization, and lyophilization round out the process, with final sterilization for sensitive tasks. Scaled synthesis prioritizes eco-friendly solvents and recycling, on both cost and regulation fronts. Teams must monitor each batch for contamination—those lessons came home for me during internships, where one deviation in pH at crystallization ruined weeks of work.
Chemical Reactions & Modifications
ATP’s high-energy phosphate bonds enable it to transfer a phosphoryl group to target molecules in kinase assays. In research, scientists use chemical labeling (such as attaching fluorescent or radioactive tags) to probe pathways or track nucleotide hydrolysis. Enzymes can transform ATP to its diphosphate and monophosphate siblings, allowing researchers to study branching pathways from one basic starting material. I once ran side-by-side ATP labeling and hydrolysis checks, and seeing the differences gave direct feedback about batch quality and assay conditions—no need for guesswork, just solid chemistry.
Synonyms & Product Names
In journals and catalogs, this compound appears as ATP-Na2, adenosine 5'-triphosphate disodium salt hydrate, or simply sodium ATP. Brands might pitch it as their “UltraPure ATP Disodium Salt” or “Bioreagent-Grade ATP Na2.” Across research teams, folks settle on “ATP disodium” for simplicity. Consistency in naming streamlines procurement, compliance, and cross-lab reputation—the lack of ambiguous codes makes coordination much easier for technicians and procurement officers.
Safety & Operational Standards
ATP disodium salt remains safe when handled with gloves and goggles in standard laboratory settings. It offers minimal inhalation or dermal risk at normal concentrations, though ingestion and dust exposure should be avoided. Safety datasheets warn about mild irritation, but ATP itself poses little acute risk compared to solvents or acids. Dedicated workspaces with clean benches help, since contamination from ATP can affect downstream tests and even attract mold or microbial growth on open plates. During my own bench work, cleanup and fresh gloves proved essential, especially with large batches or if solution handling grew sloppy. Clear instructions on spillage, storage, and waste cuts down risks and supports good science.
Application Area
ATP disodium salt matters most in cell metabolism studies, where it serves as a go-to marker for energy dynamics. Medical research leans on ATP measurement to assess tissue viability, muscle fatigue, cancer metabolism, and ischemic injury. Molecular biology teams use ATP to drive ligase reactions (connecting DNA), guide luciferase assays (bioluminescence), and test kinase drug candidates. Pharmaceutical manufacturers scale up ATP as part of diagnostic kits, toxicity screens, and therapy validation. My classmates used ATP for rapid detection of microbial contamination in food safety labs, where a quick “glow” confirmed if surfaces carried bacteria—simple, sensitive, and convincing results.
Research & Development
Today’s R&D labs focus on fine-tuning ATP’s stability and delivery, particularly for injectable drugs in cardiovascular or nervous system conditions. Projects look at ATP analogs that resist enzymatic breakdown or penetrate tissues more readily. Synthetic biology teams design pathways where ATP acts as a programmable signal, unlocking new biomanufacturing and biosensing tools. Testing batches for impurities, enzymatic inhibitors, or breakdown products sharpens the product’s reliability, especially for clinical-grade supplies. I’ve watched teams modify ATP analogs for use in designer kinase screens, showing how small tweaks on an old molecule open wide new avenues.
Toxicity Research
Oral, dermal, or inhalational ATP exposure at normal laboratory concentrations brings negligible acute toxicity, but high systemic doses—particularly by injection—lower blood pressure or affect heart rhythms. Animal studies at excessive concentrations underscore the balance between ATP’s therapeutic promise and physiological disruption: too much can trigger vascular collapse, convulsions, or sudden fatigue. Regulatory agencies set maximum thresholds for residual ATP in injectable solutions. While handling ATP never posed a threat during my training, safety teams kept documentation up-to-date for regulatory and audit purposes, reinforcing a culture of respect for both chemical and biological hazards.
Future Prospects
The future for ATP disodium salt looks lively. Diagnostic companies explore assays for detecting pathogens and environmental contaminants within minutes. Pharmaceutical firms pursue new formulations to assist in organ preservation, transplant medicine, or targeted cell therapies. Research into ATP analogs could lead to tailored cancer drugs and metabolic boosters. Green chemistry approaches may lower process waste and enable massive, low-cost synthesis for underserved regions. The interplay between synthetic chemistry, biotechnology, and medicine will keep ATP in the spotlight for years ahead. My experience in both the classroom and the lab tells me that as our understanding of energy management grows sharper, demand for clean, reliable ATP produces a ripple effect, improving research, clinical care, and even environmental safety.
What is Adenosine Triphosphate Disodium Salt used for?
How the Body Draws on ATP
Energy shapes everything I do, right down to the cellular level. That energy flows through a tiny molecule called adenosine triphosphate, better known as ATP. The disodium salt version makes it easier to dissolve in water, which matters both in labs and in the body. ATP gives muscles fuel to contract, powers the nerves that carry thoughts and feelings, and helps healing. Without it, nothing gets done.
ATP Disodium Salt in Medicine
People might not think of ATP as medicine, but it sometimes becomes exactly that. Doctors and researchers use adenosine triphosphate disodium salt during heart stress tests or to study how heart rhythms change. It pushes the heart into action so medical staff can record responses and spot problems.
Some folks facing cancer or muscle weakness try ATP supplements, hoping to regain strength. Clinical research has explored those uses. For example, trials tracked if ATP infusions helped patients recover from surgery or chemotherapy. Results remain mixed, but some patients do say they feel stronger or less tired. The FDA has not approved ATP for all uses or for over-the-counter sale, and the long-term effects of supplementation need more research. Doctors monitor any ATP use closely.
Laboratory Uses and Scientific Research
Every biology lab I’ve seen keeps some ATP on hand. Many experiments start with ATP solutions, especially when studying how living things use energy. Researchers use ATP disodium salt to:
- Trigger muscle fibers in a dish, letting them watch contractions in real-time.
- Measure enzyme reactions, because ATP helps signal when things start or stop.
- Study nerve cells, testing how they fire off impulses or release neurotransmitters.
Bacteria and viruses need energy just like we do. Scientists use ATP testing to check for contamination. Hospitals and food plants swab surfaces and read ATP levels with a bioluminescence test, then clean harder if numbers run high.
Industrial and Food Quality Testing
Food safety experts rely on ATP for helpful shortcuts. Quick tests spot bacteria or fungi in water, on surfaces, or on tools. Swabs can check inside food factories or healthcare settings. Anything that glows in the presence of ATP suggests living cells, and more cleaning gets done. Clean environments protect real people.
Thinking Twice about Supplements
Ads often promise ATP supplements will turn workouts into record-breakers or erase fatigue overnight. There’s no magic bullet. The gut breaks down most ATP before it can get into the bloodstream, so pills rarely deliver as promised. Athletes and patients deserve straight talk. Focusing on balanced diets and managed exercise works better than chasing after shortcuts.
Where Research Can Go Next
Scientists still learn new things about how ATP helps the body cope with disease or extreme strain. Researchers at hospitals and universities want to know if ATP injections can protect the brain after a stroke or support weak muscles in the elderly. Careful studies matter most, because mistakes hurt real patients. Early results show promise but always call for patience and caution.
Responsible Handling Matters
If someone brings ATP disodium salt into a classroom, hospital, or factory, training and clear rules become essential. The chemical itself doesn’t pose grave danger, but people should still wear gloves or goggles and keep the powder dry. ATP only helps when used with wisdom and care.
How should Adenosine Triphosphate Disodium Salt be stored?
Care Matters for Lab Reagents
Anyone who has spent long afternoons in a lab knows chemicals rarely forgive carelessness. Adenosine Triphosphate Disodium Salt (ATP) offers an excellent example. This molecule drives energy transfer in living cells. Scientists depend on it for everything from enzymatic reactions to cell viability assays. Some chemicals handle a little rough housing, but ATP quickly lets you know if it isn't happy.
Heat and Light: The Usual Suspects
ATP, plain and simple, decays with heat. High temperatures accelerate its breakdown, so a cool, stable environment makes a world of difference. Store ATP at -20°C or below. Most freezers in research labs work fine for this job. Always shield the container from direct light. Even room light acts slowly but surely to break down ATP. Keep the bottle in its original, amber vial or wrap it in foil to add another line of defense.
Moisture: The Hidden Threat
A curious fact about ATP’s disodium salt: it loves water, to its own disadvantage. Let it meet humid air and it pulls in water, turning clumpy and sometimes unusable. Leave the cap off for a few minutes, and suddenly the powder doesn’t dissolve the way you remember. I’ve learned the hard way to work quickly with the vial and reseal it right away. A glove box filled with dry nitrogen or argon works well in big labs, but a desiccator cabinet with a fresh packet of silica gel helps too, even on a tight budget.
Dealing with Solutions
ATP’s dry powder form resists breakdown better than its dissolved form. In solution, ATP falls apart faster than you might expect. I make fresh solutions before each project. Frozen aliquots help save time—just avoid thawing and refreezing over and over. Every freeze-thaw cycle chips away at the ATP you count on. Prepare single-use aliquots, using small tubes labeled with the date, and toss anything that looks off.
Chemical Compatibility
One detail worth mentioning: ATP can react with trace metals or contaminants in your water. Ultrapure water from a good filtration system works much better than tap or distilled water. Avoid storing solutions in metal containers or using metal tools. plastics or glass cause fewer headaches. I rinse my glassware thoroughly before touching ATP to avoid any hidden contaminants.
Safety Always Comes First
Even if ATP doesn’t rank high on the hazardous list, good lab practice always applies. Wear gloves, eye protection, and a lab coat. Have a spill kit ready and know where the nearest eyewash station stands. Close attention to chemical hygiene means fewer disasters and more reliable experiments, based on decades of findings in safety studies.
Labeling and Inventory
Neglected, expired reagents cause disasters ranging from wasted money to ruined results. Track the arrival date and the first day opened. Mark bottles clearly and keep an up-to-date inventory. It sounds simple, but I’ve seen too many labs with piles of unmarked vials and faded labels. Digital inventory systems, or even a simple spreadsheet, help reduce confusion and waste. Organized storage offers benefits that quickly add up in both safety and budget.
Responsible Disposal
Don’t pour old or unused ATP down the drain. Read your local waste disposal guidelines. Many research facilities offer chemical take-back programs or special disposal bins for nucleotides and similar compounds. Protecting lab co-workers and the environment depends on careful disposal, and taking the extra step now pays off later.
What is the chemical formula of Adenosine Triphosphate Disodium Salt?
ATP in Science and Daily Life
Every biology class points to ATP as “energy currency,” but the details behind this phrase rarely make it past a quiz. For me, seeing adenine, ribose, and three phosphates hooked together in ATP’s structure clicked much later—college chemistry pulled back the curtain. In pure form, ATP carries the chemical formula C10H16N5O13P3. In lab settings, things get a little more practical. The compound shows up mostly as the disodium salt to improve water solubility and stability. Here’s the formula that turns up on reagent labels: C10H14N5Na2O13P3. The shift from C10H16 to C10H14 and the addition of Na2 marks the replacement of the two acidic protons with sodium ions. This configuration holds up on a shelf and can mix right into aqueous solutions.
Why This Formula Makes a Difference
In practical biology and medicine, ATP disodium salt plays a role exactly because of these sodium partners. Once, during a summer research gig, I watched how painstakingly fresh solutions got prepped from the dry disodium salt. Plain ATP breaks down in water and acid in a blink. The disodium salt resists that, holding together long enough for cell biology experiments, enzyme tests, and even clinical investigations.
Tasks such as “luciferase testing” in bioluminescence labs or enzyme activity assays in pharma research depend on that reliable, shelf-stable form. Labs trust what’s written on suppliers’ labels because mixing the wrong salt or hydrating the wrong base formula leads to wasted batches or skewed test results. Drug developers, for example, lean on ATP assays when screening compounds for energy metabolism impacts. The presence of sodium balances pH and ensures solubility in blood or cellular fluids, even if most people don’t see the difference between ATP and ATP disodium salt on a data sheet.
Safety, Storage, and Lab Use
Pure ATP doesn’t just degrade—it attracts water and turns to sludge, and I’ve lost count of the number of ruined, sticky ampoules in busy research shops. The disodium salt version packs tighter in containers and survives shipment better. Simple mistakes, like storing ATP disodium salt in a humid environment, still ruin powders, but best-case storage practices keep it around for months. Always, handling involves gloves and quick transfer because no one wants to breathe dried nucleotide dust or sprinkle it on a bench.
Big industrial labs and biotech shops train staff on how to tell their salts apart. Specific formulas end up on inventory reports, including precise molecular weights and sodium content. Consider how gene editing, cell culture, or pharmaceutical compound testing stop cold without the exact compound in hand. Inconsistent use messes up downstream results, costing money, time, and sometimes causing regulatory headaches if audits find the wrong reagent listed in a method.
Room for Getting It Right
After seeing how error-prone chemical prep can get, I started triple-checking CAS numbers and chemical formulas. One good policy comes from keeping hard copies of supplier certificates and cross-referencing with inventory. Most errors in life science research trace back to missing details in how a salt gets listed or misunderstood during ordering. Anyone new to lab work benefits from seeing common salts in person, measuring them out, and reading up on why each atom and ion counts. This hands-on familiarity prevents problems and gives the kind of confidence needed in a field where details matter at every level—even something as seemingly simple as ATP disodium salt’s formula.
Is Adenosine Triphosphate Disodium Salt safe for human use?
What We Know About ATP Disodium Salt
Adenosine triphosphate (ATP) powers nearly every cell in the human body. Within the body, ATP helps muscles contract, nerves send signals, and cells produce energy. Plenty of folks look at ATP supplements—often in the form of the disodium salt—and wonder if popping a pill can deliver the same benefits they saw in textbooks. The short answer is complicated. ATP in food and ATP in a lab aren’t the same thing when it comes to safety or results.
Human Use and Safety Data
Most research on ATP disodium salt comes from sports nutrition studies and clinical trials. Athletes chase better endurance, and doctors sometimes use ATP disodium salt in an IV to test for certain heart or lung conditions. Injections under a doctor’s care show a fair safety record. Some people feel flushed, short of breath, or might notice a racing heart right after a dose—especially at higher concentrations. These symptoms tend to fade in a few minutes. Oral ATP supplements don’t always clear digestive enzymes, so only a tiny fraction actually gets into the bloodstream. That limits both the benefits and the risks when taken by mouth.
The U.S. Food and Drug Administration hasn't approved ATP disodium as a drug or over-the-counter medicine. ATP supplements fall under dietary supplement rules, which means less strict testing. Producers are supposed to follow safe manufacturing, but products may not always contain the stated amount or be completely free of contaminants. That’s a gamble for anyone expecting consistent safety or purity.
Possible Benefits, Lingering Risks
For most healthy people, the body’s system for making ATP runs smoothly. Extra ATP won’t override normal limits, because digestive tissues break it down before much crosses into the blood. There’s little strong evidence that swallowing ATP disodium salt will improve workouts, brain function, or energy levels in routine use. A small number of medical trials tried high-dose ATP with certain tumors and muscular disorders, always under medical supervision. So far, some people with chronic fatigue or fibrosis felt less exhausted, but side effects included mild stomach upset, heart fluttering, or headaches.
Nobody knows if long-term ATP disodium salt use leads to trouble, since very few studies lasted longer than a couple of months. Researchers keep a close watch for allergic reactions, spikes in uric acid (which can fuel gout attacks), and changes in blood pressure. If someone already has heart disease or kidney issues, extra ATP could raise risks.
How to Stay Safe
Consumers thinking about ATP disodium salt should choose products tested for purity and published by reputable labs. The best bet for safety is talking with a health care provider, especially for those on prescription drugs or dealing with medical conditions. Doctors know how different supplements interact with heart rhythm medicines, blood pressure medications, and anticoagulants. The body usually breaks down ATP quickly, but even a small risk isn’t worth facing alone if there’s a medical history in play. At-home use won’t match the monitoring in a clinical trial.
Research still hasn’t proven a strong benefit for the average person using ATP disodium salt. Most folks will get better returns from regular exercise, balanced nutrition, and plenty of sleep. For now, safety questions stick around simply because there’s little oversight and not enough well-controlled studies on long-term human use.
What is the recommended dosage for Adenosine Triphosphate Disodium Salt in laboratory applications?
Understanding the Right Dose Matters
I’ve watched many lab protocols trip up just because of ambiguous instructions about concentrations. When preparing Adenosine Triphosphate (ATP) Disodium Salt for cell culture, enzyme assays, or bioluminescence work, accuracy counts more than almost anything else. Too little ATP and the reaction falls flat; too much and you overwhelm the system or introduce unintended effects, like cell toxicity or buffer imbalance.
Industry Standards and Common Practices
In enzyme activity testing, ATP levels hang around 1–5 mM. Research published in Journal of Biological Chemistry and cell biology protocol books from teams at major universities often list 1 mM as a starting point, fine-tuning from there based on the enzyme and substrate specifics. Sometimes you need to push the concentration higher when dealing with more stubborn targets, but going much past that generally doesn’t help and, in my experience, makes troubleshooting headaches worse.
For cell culture—especially with mammalian or bacterial cells—we see ATP dosages in the range of 0.1–2 mM. A study from the University of Tokyo back in 2017 highlighted how higher ATP levels above 2 mM started to stress out kidney cell cultures. In ATP-based bioluminescent assays, like ATP quantification kits, instructions usually recommend between 1–10 μM, depending on detection sensitivity. Trying to match textbook figures without thinking critically about the readout can waste both time and precious reagent.
Mind the Solution Prep
ATP disodium salt, pure as it might look, often clumps or degrades when exposed to moisture or strong acid. Always measure ATP salt in dry conditions. Quickly dissolve in water or lightly buffered solutions, adjusting pH to about 7.0–7.4. This keeps the ATP stable and ready for whatever reaction you’re setting up. I learned my lesson the hard way once: If pH drifts below 6.5, breakdown starts almost immediately. For experiments running over several hours, keep tubes cold and prepare fresh stock every week or two.
Safety and Good Laboratory Habits
Safety isn’t just about gloves and goggles. High levels of ATP in some biochemical reactions can mess with cellular ion balances or feed unexpected side reactions. The Occupational Safety and Health Administration doesn’t list ATP as especially hazardous, so acute risk is minimal. But using common sense counts—a small spill won’t hurt, but sloppy technique always comes back to bite. Keep ATP at -20°C, label it well, and write down how much you used. That notebook entry saves you from repeating a failed experiment.
Why Precision Makes a Difference
Fudging concentrations, especially in projects where every dollar comes out of limited grant budgets, is a luxury most teams can’t afford. Reliable results demand standardized stock solutions, regular calibration of pipettes, and checking the math every time. If your published methods are followed elsewhere, or your team relies on repeatable experiments, it’s these fundamental practices that stand between chaos and clear data.
Adjustments and Troubleshooting
Protocols can set you on the right path, but troubleshooting requires close attention to the system in front of you. If reactions sputter out or plates stain oddly, walk back through each step—starting with ATP concentration. I’ve seen whole projects turn around just by realizing the stock was off by half a log unit, traced to misreading the bottle’s label.
Reaching Out for Expert Support
Stumped on a dosage? Technical bulletins from suppliers like Sigma-Aldrich or Thermo Fisher offer practical guidelines. If questions remain, peer-reviewed publications often break down concentrations by experiment type and organism. Don’t hesitate to email the author or a technical sales rep—chances are, they’ve faced the same stumbling blocks as everyone else.
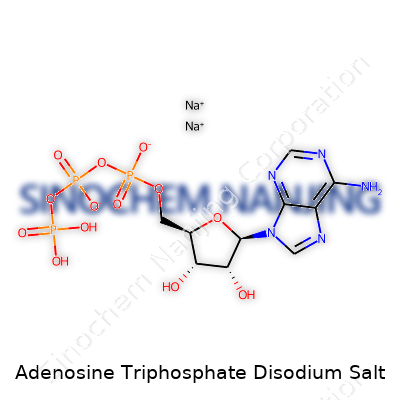
| Names | |
| Preferred IUPAC name | disodium [(2R,3S,4R,5R)-5-(6-aminopurin-9-yl)-3,4-dihydroxyoxolan-2-yl]methyl triphosphate |
| Other names |
ATP disodium salt Disodium adenosine 5′-triphosphate Disodium ATP Adenosine 5′-triphosphate disodium salt |
| Pronunciation | /əˌdiː.nəˌsiːn traɪˈfɒs.feɪt daɪˈsoʊ.di.əm sɒlt/ |
| Identifiers | |
| CAS Number | 987-65-5 |
| Beilstein Reference | 4125914 |
| ChEBI | CHEBI:30616 |
| ChEMBL | CHEMBL1429 |
| ChemSpider | 94732 |
| DrugBank | DB00171 |
| ECHA InfoCard | 03a78c1819d2-3e34-4a64-bfd6-8ec95b6edc73 |
| EC Number | EC 232-146-4 |
| Gmelin Reference | 8905 |
| KEGG | C00002 |
| MeSH | D000255 |
| PubChem CID | 190249 |
| RTECS number | AU7350000 |
| UNII | 8L70Q75FXE |
| UN number | 2811 |
| Properties | |
| Chemical formula | C10H14N5Na2O13P3 |
| Molar mass | 605.18 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.118 g/cm³ |
| Solubility in water | soluble in water |
| log P | -4.1 |
| Acidity (pKa) | 2.28 |
| Basicity (pKb) | 1.95 |
| Magnetic susceptibility (χ) | -55.0e-6 cm³/mol |
| Refractive index (nD) | 1.570 |
| Viscosity | Viscous oil |
| Dipole moment | 5.9 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 531.6 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | B06AB04 |
| Hazards | |
| Main hazards | Irritant to eyes, skin, and respiratory tract. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P272, P273, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P333+P313, P337+P313, P362+P364, P403+P233, P501 |
| Lethal dose or concentration | LD50 (mouse, intraperitoneal): 245 mg/kg |
| LD50 (median dose) | 830 mg/kg (Intravenous, Mouse) |
| NIOSH | Not Listed |
| PEL (Permissible) | Not Established |
| REL (Recommended) | REL: 0.06 mg/m^3 |
| IDLH (Immediate danger) | Not listed |
| Related compounds | |
| Related compounds |
Adenosine diphosphate Adenosine monophosphate Guanosine triphosphate Cytidine triphosphate Uridine triphosphate |

