Acetyl Thiourea: Evolution, Properties, Safety, and Future Directions
Historical Growth of Acetyl Thiourea
Chemists in the early 1900s turned their attention to thiourea derivatives as they tracked down new compounds for dyes, pharmaceuticals, and agricultural applications. Acetyl thiourea joined the scene during this period, carved out from curiosity about functional groups that merged acyl and thiourea features. Over decades, research labs across Europe and Asia documented processes that allowed acetyl thiourea to play a supporting role in synthesis work, especially as access to reagents and industrial precursors improved. These early developments came from straightforward reaction conditions—acetic anhydride or acetyl chloride meeting thiourea—laying the groundwork for easy lab and plant-scale routes that are still used today. Patents from the mid-twentieth century back up its growing portfolio, featuring acetyl thiourea as an intermediate that links up with multiple chemical families.
Product Profile and Day-to-Day Use
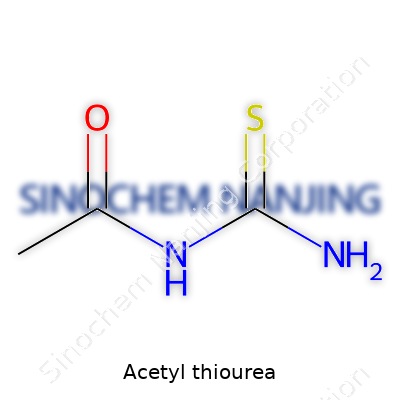
Acetyl thiourea’s formula, C3H6N2OS, captures a structural mash-up between a carbamoyl group and a thioamide. Anyone who’s handled the compound in the lab knows it appears as a white to light beige powder, bland in odor and reliable in composition. Chempurities pop up in synthesis, so product purity ranges over 99% from most large suppliers. Some applications only demand technical grade, especially during steps in making resins or curing agents. Labeling usually lists chemical names, batch numbers, handling precautions, and shelf life limitations, which comes handy during audits. Storage relies on sealed containers, placed away from heat and moisture, signaling routine care rather than anything extraordinary.
Physical and Chemical Characteristics
At room temperature, acetyl thiourea settles out as a solid that melts near 187°C. Its solubility favors organic solvents like ethanol or acetone, delivering a moderate—though not spectacular—performance in water. Chemical stability keeps it from spontaneous breakdown under regular handling, but the amide group can hydrolyze under strong acid or alkaline conditions. Lab results show the compound stays inert in air, yet develops decomposition worries above 200°C, which creates sulfur-based fumes. No strong odors or color changes show up under normal storage, making it easy to track lot integrity.
Technical Specifications and Labeling
Industrial-grade acetyl thiourea often comes with specifications such as appearance, melting point, moisture content, and assay percentages. Standard sheets outline maximum heavy metal content—often below 10 ppm—and detail impurity patterns, especially for pharmaceutical or food-related intermediates. Labels on containers must display relevant safety symbols, storage requirements, net weight, and manufacturer details to keep distribution chains transparent for regulators. Documentation leans on batch-specific data, enabling effective recalls or traceability whenever questions pop up during regulatory reviews, reinforcing trust among processors and buyers.
Preparation Route
Labs and manufacturing lines follow a few tried-and-true routes for producing acetyl thiourea. Mixing thiourea with acetic anhydride in ethanol or dilute acid works well at low to moderate temperatures, yielding a high-purity product after filtration and crystallization. The reaction sees the acyl group joining the nitrogen, followed by a quick work-up using cold water washing and vacuum drying, which strips away leftovers and boosts purity. Yield percentages tend to track above 85%, reducing chemical waste and streamlining cost profiles for bulk orders. Acetic acid forms as a byproduct, easily removed by distillation or neutralization, keeping downstream operations simple.
Chemical Reactions and Modification Profiles
Acetyl thiourea steps into both nucleophilic and electrophilic roles within organic synthesis, expanding the utility of both carbon-sulfur and carbon-nitrogen bonds. Alkylation and acylation reactions graft on functional groups, pushing the molecule toward uses in pharmaceuticals, plastic stabilizers, and vulcanization accelerators. Chemists rely on its amino and thione functionalities, driving studies in heterocyclic ring formation, for example in pyrimidine or triazole synthesis. Oxidative and reductive transformations also adapt the core for further development as herbicides or fungicide precursors. This modification flexibility lets researchers shape-morph the compound onto specialized scaffolds, a feature that keeps it relevant in today’s molecule libraries.
Aliases and Product Codes
Chemists and manufacturers circulate a list of alternative names for acetyl thiourea, such as N-Acetylthiourea or 1-Acetyl-2-thiourea. Trade catalogs sometimes show variants like Acetothiourea or Acetuoreum. The compound lands in reference systems with CAS number 591-08-4, easing cross-referencing for regulatory forms and international orders. Each code and synonym lines up with specification sheets and safety documentation, minimizing confusion across languages and labeling customs. In my experience with ordering from multinational vendors, clear identification cuts down shipment errors and workflow delays.
Safety Procedures and Operating Practices
Any chemical process worth long-term adoption builds in safety and handling measures. Acetyl thiourea doesn’t throw severe toxicity at users, but it nudges up as a skin and mucous irritant. Handling calls for gloves, eye protection, and decent ventilation, especially if dust escapes containers. Safety data sheets note the importance of minimizing inhalation or ingestion, reinforcing the habit of lab coats and routine hand-washing. In cleanup, standard spill kits with inert absorbents mop up small releases, disposing everything in sealed chemical waste. Fire hazards don’t dominate the risk list, but thermal breakdown produces pungent sulfur compounds, so fire extinguishers and exhaust hoods still sit on the standard protocol list.
Applications and Industry Demand
Acetyl thiourea carries weight in chemical manufacturing as a stepping stone for resins, pharmaceutical intermediates, and agricultural chemicals. Firms making sulfonylurea herbicides leverage this compound to bridge synthesis steps, while others tap it for rubber additives where it boosts processing reliability. Biomedical researchers test modified derivatives for antibacterials, hypoglycemics, and sometimes as anti-inflammatory leads, though not many cross the line into clinical use. Analytical labs use acetyl thiourea as a standard for sulfur content or even as a ligand in coordination chemistry. Demand rises and falls depending on agricultural cycles and new drug development efforts, but it never runs far from core industrial supply chains.
R&D and Modern Investigations
From my time in academic settings, acetyl thiourea shows up in med-chem research as a core for designing enzyme-targeting drugs. Teams use it as a building block for molecules against emerging pathogens, with thioamide groups flagged for their interaction with biological systems. Teams in polymer science explore the compound in specialty curing agents, hoping to tune mechanical properties for coatings. Environmental chemists also kick around experiments with acetyl thiourea derivatives as sensors, tracking heavy metals in water or soil samples. Industry patents draw a wide arc, spanning high-performance materials to next-generation pesticides, as synthetic chemists ramp up high-throughput screening using acetyl thiourea motifs.
Toxicity Studies and Environmental Precautions
Safety studies in rats and mice put the compound in the “low-moderate risk” bracket for acute exposure, with lethal dose values well above the thresholds typically seen in plants or workplaces. Long-term studies avoid flagging carcinogenic or genotoxic trends, but regular reviews push for limiting chronic low-dose exposure, especially in production environments with dusty conditions. Environmental breakdown runs slow unless exposed to sunlight or microbial action, suggesting companies design containment procedures at manufacturing sites. Waste effluent rules call for neutralization steps to prevent persistent build-up in streams or soil, and regulatory clamps like REACH and TSCA lay out paperwork to keep usage tracked.
Outlook and Future Trajectories
Chemists who push boundaries look at acetyl thiourea and see a foundation for specialty drugs, sustainable pesticides, and modified industrial polymers. Researchers tinker with greener, less wasteful production techniques and hunt for new ways to tweak the core for enhanced performance. Advances in analytical tools could shed new light on environmental impacts and long-range fate in ecosystems, while -omics technologies in biology might pull thiourea derivatives into the next wave of drug leads. On the industrial edge, aligning tighter with best practices in safety and fundable research promises a longer, safer, and more innovative future for acetyl thiourea across global supply networks.
What is Acetyl Thiourea used for?
What’s the Real Story Behind Acetyl Thiourea?
Walk into any chemical lab and you’ll find bottles with complicated names. Acetyl thiourea sits quietly on a shelf, but don’t let its low profile fool you. It pulls its weight in many industries. I remember seeing it for the first time in a university lab, where one of the senior researchers explained how it ended up in everything from dye-making to medicine. Turns out, Acetyl thiourea steps in just about anywhere reactions need a little nudge.
Why This Compound Matters in Pharmaceuticals
Pharmaceuticals rely on molecules that build, block, or balance chemical structures in the human body. Acetyl thiourea contributes its sulfur and nitrogen to form the backbone of many drug molecules. These days, drug discovery moves fast, but chemists are careful about what they put in a synthesis route. Reliable chemical building blocks like acetyl thiourea help researchers build new medicines that tackle infections and even cancers. I recall reading about one group that used it to create more potent versions of antiviral agents because it fits so well into the assembly line of organic synthesis.
Dyes and Pigments: Bringing Color to Industry
Color matters in textiles, plastics, and inks. Making a dye or pigment that stays put on fabric—even after many washes—gets tricky. Acetyl thiourea stabilizes the color molecules, stopping them from breaking down too quickly. In my own small experiments with fabric dyes, introducing sulfur-containing agents often made the difference between a bright, long-lasting color and one that washed out after a cycle or two.
Agrochemicals: Protecting Crops and Boosting Yields
Feeding billions means crops need protection from pests and disease. Farmers want chemicals that work but break down safely over time. Acetyl thiourea shows up in the recipes of herbicides and fungicides. It offers a structure that gets absorbed by plants efficiently while targeting harmful invaders. Plenty of field studies mention products made from it that show strong disease control without lingering too long in the environment, a major win for safety and sustainability. We need more chemicals that do their job and then step out of the way.
Material Science: Unexpected Roles
Materials making, especially with polymers and specialty papers, tends to be a world of trial and error. Polymer chemists mix and match small molecules to invent new properties. Acetyl thiourea sometimes takes on the role of a cross-linker—helping make things like tough coatings, or heat-resistant plastics. I watched a demonstration where scientists used it in fire-retardant treatments for textiles, showing its benefit isn’t just theoretical—real-world applications keep coming as our needs change.
Challenges and Solutions Connected to Acetyl Thiourea
No chemical works in isolation. One concern that research journals point out is the possible environmental impact if acetyl thiourea waste isn’t treated carefully after use. Traces in water systems could build up, so waste treatment facilities need to stay alert. Manufacturers should keep investing in cleaner synthesis methods and better disposal practices. Building partnerships between industry and research—testing greener routes and designing smarter products—goes a long way. Companies already experimenting with biodegradable versions show that progress is possible when there’s a push for change.
Moving Forward
Everyday life rarely brings attention to the chemicals that keep industries moving, but acetyl thiourea proves that small compounds play supporting roles with big effects. Practical action and careful handling keep its benefits high and its risks low. That’s what really keeps innovation safe and steady.
What is the chemical formula of Acetyl Thiourea?
The Chemistry and Identity of Acetyl Thiourea
Acetyl thiourea often makes its way into research labs and chemical supply shelves, but many folks forget its roots and role. The formula for acetyl thiourea is C3H6N2OS. What that means in simple terms is that it carries three carbons, six hydrogens, two nitrogens, one oxygen, and one sulfur atom per molecule. With a backbone like that, it can easily slot itself into bigger, more complicated chemical reactions. In my own college lab days, getting this formula right meant could avoid making simple mix-ups and focus on the real work of experimentation and observation.
Applications Reach Further Than Textbooks Show
This compound isn’t one of those celebrity molecules people read about in every pharmacy magazine or pop-science blog, but it sits at the crossroads of medicine and materials science. Acetyl thiourea shows up as a building block for new drugs, dyes, pesticides, and even some vulcanizing agents. In a lab book from the early 1980s, researchers pointed out how its derivatives fought bacteria and hinted at possible cancer research connections. Rolling back over the research, it’s not hard to find chemists using this formula to shape molecules that can slip into new organic frameworks.
Safety and Environmental Footprints
Getting to know a chemical means also grasping the risks. Acetyl thiourea carries a moderate toxic profile. Spills and exposure bring problems like skin irritation or headaches. Proper handling practices come from years working with it in tight lab quarters — gloves, eye protection, and a reliable fume hood are a must. Many experienced techs learn through hard lessons that documentation and labeling go a long way in reducing surprise exposures. Safety data sheets warn about storage away from heat and open flames, as this compound breaks down and releases unpleasant, sometimes unsafe fumes.
Production, Handling, and Quality
Synthesizing acetyl thiourea looks straightforward enough on paper. Mixing acetic anhydride with thiourea in the right solvent — often water or ethanol — produces the target compound if the temperature sits in a safe range. Sloppy measurement or heating too quickly ruins the batch. From personal experience, the sharp odor lingers long after cleanup unless ventilation gets addressed early. In industry, automated equipment does much of the mixing and separation, but small-batch chemists still rely on glassware, patience, and time-tested procedures to keep yield and purity high.
Troubleshooting and Best Practice
Labs run best on trust, clear communication, and a shared respect for the power of chemicals like acetyl thiourea. Storage containers need clear labeling, tight seals, and separation from acids or strong oxidizers. Waste disposal stays straightforward: seal and ship to qualified handlers, never send waste down the drain. Accidents happen, but teams that drill response routines stand the best chance of moving past a spill or exposure without harm. Every time, I see chemistry students gain confidence as they connect the dots — from formula memorization to responsible use — and realize each detail protects both people and projects.
How should Acetyl Thiourea be stored?
Understanding What We’re Dealing With
Acetyl thiourea looks harmless sitting in the lab, but this isn’t something anyone should shrug off. It plays a role in all kinds of chemical syntheses, but basic mishandling can lead to ruined stock or safety problems. The white crystals may not smell menacing, yet long-term exposure can hit the skin or lungs. Keeping it in good shape is only half the reason proper storage remains so important.
The Real Risks if You Ignore Best Practices
Take it from those who’ve spent time around ill-kept storerooms: chemicals exposed to heat or moisture rarely behave as the safety data sheets describe. Acetyl thiourea breaks down if it sits too long in a damp spot. Humidity isn’t just a minor detail—it opens the door for clumping, changes in reactivity, or even the growth of mold and bacteria that thrive on anything with free nitrogen or sulfur. Not only does this jeopardize experiments, but residue or vapors can build up, quietly hurting those who work nearby.
Once, I returned to a container of thiourea left near a window. Sunshine streaming in had warmed the stash just enough to let condensation sneak inside. When I cracked that lid two weeks later, the small, stubborn clumps made any weighing pointless. Money and time, down the drain. Why risk it, knowing the routine for safer handling never gets complicated?
Simple Steps People Overlook
Most good storage starts with a strong, airtight container. Glass or high-density plastic work best. Leave paper cartons to dry foods—the acids and moisture in the air have a way of working through the edges over time. Toss in a silica gel pack, especially if you’re in a humid or coastal lab. Too many folks skip this, thinking the lid will handle everything. It’s better to play it safe.
Next, avoid any temptation to stuff Acetyl thiourea up on a high, sunny shelf. Direct light kicks off unwanted reactions, sometimes speeding up decomposition. Every proper chemistry storeroom keeps its sensitive chemicals in a cool, shaded spot. Aim for temperatures below 25°C. Even brief heat spikes in the middle of summer can shorten shelf life.
Labeling shouldn’t need explaining, yet bottles without clear dates or hazard warnings still pop up in crowded cabinets. If two jars somehow mix—maybe by a gloved hand brushing against both in a rush—you open new chances for side reactions. A clear label is a simple fix that keeps everyone on the same page.
Acetyl thiourea doesn’t enjoy neighbors like strong acids or oxidizers. Separate storage means accidents can’t snowball. If something spills or breaks, there’s less risk of toxic gases forming or runaway reactions starting. Keep incompatible chemicals apart, even if it means reorganizing a shelf.
Solutions for Real-World Labs
Busy labs cut corners during hectic stretches, but building storage habits into the layout solves more trouble than strict policies alone. That might mean placing a sealed bin close to the workspace and setting lab reminders to check for cracks or leaks monthly. Share tips at safety meetings and use photos to show storage mistakes. These simple fixes cost little but shield everyone from big headaches or ruined supplies.
Digital inventory can alert teams when older chemicals approach expiry, nudging you to use up or replace material before storage turns into waste. Records don’t always seem urgent, but knowing what sits on a shelf wards off confusion years down the line.
Acetyl thiourea’s long-term value depends on storage done right—tight lids, low light, secure shelves, and a little attention each week. That’s how you keep the lab safe, the results reliable, and expenses under control.
Is Acetyl Thiourea hazardous to health?
What Acetyl Thiourea Is and Where You’ll Encounter It
I first came across acetyl thiourea in a chemistry lab, buried in an old bottle on a crowded shelf. Mainly used as a building block for other chemicals, it pops up in research, pharmaceuticals, and sometimes in specialty industrial processes. Most folks won’t come across it at home, but its hazards are a real-world concern for anyone working near it, and for the companies that manufacture or ship it by the ton.
Health Risks and Real-Life Exposure
Acetyl thiourea looks harmless—white powder, slight odor, boring at first glance. That dull exterior doesn’t reflect its risk profile. The main dangers come if it touches skin, gets in your eyes, or if you breathe dust or vapors. Skin exposure can trigger redness and sometimes a nasty rash; direct splashes in the eye burn and may even injure the cornea. Nobody wants the stuff in their mouth, but swallowing it often leads to nausea, abdominal pain, and sometimes headaches or worse. I’ve seen fellow researchers slip up and need quick treatment. For anyone with asthma or breathing conditions, inhaling even small amounts of powder can tighten the chest or start a coughing fit. Toxicological reports flag its potential to irritate mucous membranes, and regular handling without proper gloves boosts the odds of dermatitis.
Cancer Risk and Chronic Health Effects
Thioureas, including the acetyl variant, put up some red flags in animal tests. Studies in rodents linked chronic exposure to changes in thyroid glands and, in some cases, tumors. Scientists don’t call acetyl thiourea a human carcinogen right now, but those findings tug at old worries. There’s not enough human data to draw hard lines, but a lack of evidence doesn’t prove it’s safe. The longer someone deals with these chemicals, the more careful they have to be. Job sites with poor ventilation drive up risk sharply.
Why This Matters and Steps to Minimize Harm
Ignoring chemical hygiene never pays off, and that’s especially true for materials like acetyl thiourea. Accidents don’t happen daily in most labs and plants, but long-term safety depends on strong habits and clear procedures. Gloves made of nitrile or PVC—never plain latex—keep it off your skin. Goggles and a face shield stop splashes cold. Most labs insist on solid lab coats and chemical hoods because fume inhalation creates problems fast. People who work directly with this compound should get training and regular reminders to avoid shortcuts.
Cleanup matters. If acetyl thiourea spills, sweeping dry powder always sends dust into the air, so wet methods and proper waste bins matter. Facilities with local exhaust ventilation cut down on airborne exposure. For me, keeping a spill kit nearby always felt smarter than trusting good luck. Medical staff should know symptom lists in case anyone feels unwell at work. Strict labeling and storage policies ensure nobody grabs a dangerous chemical by mistake.
What the Experts Say and How Companies Should Adapt
Guidance from occupational health bodies points to the need for limits on allowed exposure, especially in enclosed spaces. National Institute for Occupational Safety and Health (NIOSH) and European regulators place this compound on their watchlist, though rules sometimes lag behind science. Companies serious about worker safety must draw up exposure control plans, substitute less hazardous chemicals if they can, and adopt closed systems to limit human contact. Fast, honest reporting of incidents puts everyone on guard. My best advice? If you don’t need to use acetyl thiourea, choose something milder.
Just like other chemicals, the risk all comes down to respect, knowledge, and routine. Chemical hazards never sleep, and neither do the people who have seen the costs of ignoring them.
What are the main applications of Acetyl Thiourea in industry?
A Useful Tool in Chemical Synthesis
Acetyl thiourea finds its way into chemical labs and production plants more often than people outside the industry might guess. Its appeal comes from a simple structure that reacts with a range of molecules. Chemists use it to build more complex organic compounds, taking advantage of its ability to donate or accept functional groups. Over the last decade, the chemical sector leaned into these applications, because compounds made from acetyl thiourea eventually show up in pharmaceuticals, plant protection, and specialty chemicals.
Pharmaceutical and Drug Manufacturing
Drug makers draw from a limited pool of safe and cost-effective starting materials. Acetyl thiourea fits in this mix thanks to its sulfur and nitrogen-rich design. Scientists use it to create drugs that target infections and inflammation. Research articles published over the last few years highlight work on antiviral and antifungal agents produced using this compound. Compared to earlier options, acetyl thiourea often streamlines synthesis, helping drug makers save raw materials and limit toxic byproducts.
Agrochemicals and Pesticides
In farming, the story changes. Pest control companies work with acetyl thiourea to tweak how their products interact with plant pests and soil fungi. Some successful modern fungicides trace back to this starting point. By integrating acetyl thiourea into the molecular backbone, agrochemical innovators create new modes of action—important, since pests keep building resistance to old formulas. Consider the statistics: nearly 40% of crop losses connect to diseases and pathogens. Tools like acetyl thiourea help seed companies and farmers keep up with these threats while managing cost and regulatory hurdles.
Metal Extraction and Recovery
Mining industries also spend time with acetyl thiourea. Hydrometallurgy uses it for selective extraction of precious metals like gold and silver. Labs tune process conditions so this compound binds specifically to these metals. Compared to historical extraction methods, which dumped lots of mercury or cyanide into waterways, use of acetyl thiourea gives a cleaner, more manageable reaction. The costs of waste treatment drop, safety goes up, and companies avoid the worst fines and lawsuits—a big motivation as rules tighten worldwide.
Dye and Pigment Production
Color and pigment makers turn to acetyl thiourea for more than just its chemistry. Its ability to participate in building complex ring systems means new colorants become possible without relying on old, less environmentally friendly processes. Data from recent years shows that pigments derived from acetyl thiourea offer higher stability in outdoor applications. Paint producers and textile dyers, who face rising demand for weather-resistant and fade-proof products, push for these innovations with increasing urgency.
Path Forward for Safer and Smarter Use
Industries constantly face questions about resource efficiency and environmental impact. Acetyl thiourea solves problems in several areas, but safe management depends on skilled technicians and responsive regulation. Shortcuts in disposal or handling threaten more than just profit—they risk health, groundwater, and reputations. Newer research points toward greener synthesis, recycling, and smarter waste treatment for acetyl thiourea. Professionals invested in chemical manufacturing should keep pushing for these updates, drawing from both academic studies and what works at scale in real factories.
| Names | |
| Preferred IUPAC name | 1-Acetamidothiourea |
| Other names |
Acetylthiourea Thioacetanilide N-Acetylthiourea 1-Acetyl-2-thiourea |
| Pronunciation | /əˈsiː.tɪl θaɪ.oʊˈjuː.rɪə/ |
| Identifiers | |
| CAS Number | [591-08-2] |
| Beilstein Reference | Beilstein 413972 |
| ChEBI | CHEBI:21540 |
| ChEMBL | CHEMBL187675 |
| ChemSpider | 11118 |
| DrugBank | DB14163 |
| ECHA InfoCard | 100.032.341 |
| EC Number | 220-984-1 |
| Gmelin Reference | 82442 |
| KEGG | C19152 |
| MeSH | D000080 |
| PubChem CID | 6616 |
| RTECS number | YN8225000 |
| UNII | K5H49F030P |
| UN number | 2811 |
| Properties | |
| Chemical formula | C3H6N2OS |
| Molar mass | 119.16 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.42 g/cm3 |
| Solubility in water | soluble |
| log P | -0.77 |
| Vapor pressure | 0.00017 mmHg at 25°C |
| Acidity (pKa) | 11.12 |
| Basicity (pKb) | 12.08 |
| Magnetic susceptibility (χ) | -55.1×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.605 |
| Dipole moment | 4.07 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 155.8 J mol⁻¹ K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -111.6 kJ/mol |
| Pharmacology | |
| ATC code | N02BG06 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | Precautionary statements of Acetyl Thiourea: "P261, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-1-1 |
| Flash point | 113 °C |
| Lethal dose or concentration | LD₅₀ (oral, rat): 1250 mg/kg |
| LD50 (median dose) | LD50 (median dose): 125 mg/kg (Oral, Rat) |
| NIOSH | SY8925000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Acetyl Thiourea: Not established |
| REL (Recommended) | Relatively Non-Hazardous |
| Related compounds | |
| Related compounds |
Thioacetamide Thiourea Acetylurea N-Methylthiourea Butyryl thiourea |

