An Editorial Look at Acetyl Iodide: Old Roots, Bright Prospects
Historical Development Takes the Spotlight
Chemistry circles have watched acetyl iodide for close to two centuries. The earliest narratives tie back to 19th-century synthetic chemistry when the quest for reactive acyl halides began shaping modern organic synthesis. Much of the work echoed the lab bench ingenuity of early European chemists, aiming to convert basic organics into potent intermediates. With each successful synthesis, laboratories pieced together better approaches for creating acetyl iodide, developing from rudimentary methods based on direct reaction of acetic acid with phosphorus triiodide, which often generated heat, fumes, and a sharp, pungent odor. Spirit of discovery combined with practical demand, setting this compound as a reliable building block across several domains. Years in, acetyl iodide gained a reputation not only as a reactive intermediate but as a teaching tool for dissecting reaction mechanisms. Many chemists, including myself in graduate training, first tinkered with this acyl iodide to learn about nucleophilic substitution and the peculiarities of halide reactivity. These early experiments provided a window into the fine balance between reactivity and control that defines chemical innovation and shaped lab traditions that last till now.
Product Overview in Practical Terms
Every bottle of acetyl iodide comes with a legacy of both caution and anticipation. In plain terms, it’s an acyl halide—more specifically, the iodide derivative of acetic acid. The compound serves as a bridge in synthesis, letting researchers tack on an acetyl group where it’s needed, swapping in an iodine in place of a more sluggish leaving group. Students and technicians alike know its odor signals a strong, reactive molecule. Instead of lengthy storage, most labs produce acetyl iodide on demand, recognizing the instability that defines much of the halide family. My own experience emphasizes the dance between utility and risk. Open a well-sealed vial and a wave of acetic, pungent vapor escapes—reminding everyone why tight safety routines matter. Chemists rarely see it as a destination in itself; its value plays out in the transformations it makes possible, guiding functional group changes in pharmaceuticals, dyes, and specialty chemicals.
Physical & Chemical Properties: Why They Matter
Unlike routine solvents, acetyl iodide stands out for its physical and chemical punch. This colorless to pale yellow liquid boils at around 98–100°C, though that figure can shift under lab conditions. Volatility combines with a powerful odor and tendency to hydrolyze when it meets water, breaking down to acetic acid and hydroiodic acid. The density sits higher than many organics, hovering above 2 g/cm³, and its viscosity strikes a middle ground—enough fluidity to pour, enough body to feel thicker than ether or acetone. Importantly, these properties explain the need for dry conditions. A chemist handling acetyl iodide always watches for moisture, since even a little water in air triggers fume generation and wastes precious material. The interplay of iodine’s polarizability with the carbonyl’s electrophilicity makes this compound one of the more interesting acylating agents, able to coax reluctant substrates into new reactions. Over the years, I’ve noticed that students often forget just how unstable acetyl iodide can be compared to its bromide or chloride siblings; the heavier iodine makes for a less stable molecule but an even more eager participant in exchange reactions.
Technical Specs & Labeling on the Real Bench
Technical literature tends to call acetyl iodide by its CAS number, chemical formula (CH3COI), or as ethanoyl iodide. Bottles wear hazard warnings that quickly grab your attention: corrosive, moisture-sensitive, and liable to release pungent, irritating vapors. On a shelf, it rarely stays for long—tech specs mention unstabilized forms shouldn’t be stored at room temperature for more than a few weeks, and even stabilized solutions need cool, dry storage. Labeling pushes the dangers to the front, both for regulatory reasons and everyday safety. As someone managing an academic lab, I’ve fielded questions about waste handling and spill cleanup on more than one occasion, so these warnings carry critical weight, translating technical specs into hands-on routines. Chemists keep a close eye on container integrity and clear labeling—spilled acetyl iodide means not just a chemical mess but a real health hazard, prompting full protective gear and fume hood operation.
Preparation Method: Context from the Lab Floor
The classic synthesis routes for acetyl iodide echo organic chemistry’s inventive spirit. Most routes start with acetic anhydride or acetic acid and react with either iodine and red phosphorus or, more commonly, phosphorus triiodide (PI3). The basic procedure often mixes these reactants under controlled cooling, then distills the product away as the reaction proceeds. In my years of lab experience, I’ve seen the search for alternatives: using other iodination agents, tweaking stoichiometry, and seeking greener, more sustainable choices. For example, some researchers push for milder reagents or look into catalytic alternatives that minimize waste. Whether in a teaching lab or during scale-up for a pilot plant, the challenges stay the same—controlling exothermic reactions, managing hazardous gases, and aiming for high yield without sacrificing safety. Each generation of chemists fine-tunes technique, learning from past incidents and improving efficiency—a real testament to the hands-on perseverance behind every lab-bench bottle.
Chemical Reactions & Modifications: The Big Picture
Acetyl iodide steps into many organic synthesis stories as both a reagent and a supporting character. Its major draw lies in its ability to acetylate alcohols, amines, and other nucleophiles more rapidly than its chloride or bromide cousins, largely due to the excellent leaving ability of iodide. That speed and vigor come with tradeoffs, especially the need for careful moisture exclusion and vigilant temperature control. I’ve watched new researchers misjudge its power—quick substitutions can mean tough-to-predict side reactions or decomposition. Still, in the hands of a practiced chemist, acetyl iodide opens doors to making esters, amides, and acylated compounds that are otherwise stubborn targets. Beyond straightforward acetylation, it plays a part in halogen exchange, feeding into cross-coupling reactions where an iodine makes a more reactive partner for metal catalysts. Swap the conditions, and you get creative routes to organoiodine compounds, vital in radio-labeling studies and new drug scaffolds.
Synonyms & Product Names: Cutting Through the Jargon
Alongside the chemical name acetyl iodide, the industry and textbooks reference this molecule as ethanoyl iodide. In research literature, abbreviations like AcI sometimes pop up, especially in reaction schemes. Despite these different labels, the substance remains the same—one of the less common acyl iodides in routine chemical inventory, but crucial wherever its unique reactivity fits the bill. In practice, clear labeling trumps clever nomenclature; most labs I’ve worked with stick to full names to avoid mix-ups, especially when dealing with classes of similarly reactive acyl halides.
Safety & Operational Standards Grounded in Experience
Every time acetyl iodide appears on the lab schedule, safety prep jumps to the top of the agenda. As a corrosive, irritant, and potential environmental hazard, this substance demands respect. Gloves, splash goggles, and fully functioning fume hoods stay non-negotiable. Any spill or accidental exposure needs rapid response: chemical spill kits for neutralizing acids, full lock-down of affected zones, and clear reporting. I remember early-morning drills where new lab members practiced mock spill responses, reinforcing that complacency around acetyl iodide can end with chemical burns or worse. Regulatory bodies insist on proper training and PPE use, a lesson reinforced by any lab that’s seen what happens when protocols slip. Waste handling also draws attention—not just because of possible toxicity but due to regulations governing halogenated organic disposal. In my own settings, close review and repeated training keep incidents rare, but any sign of lax standards raises immediate concern. Regular audits and reporting, not just paperwork, help keep places safe and compliant.
Application Area Drives Continued Demand
Although acetyl iodide rarely features in flashy press releases, its real value shows up across research, specialty synthesis, and drug discovery. Medicinal chemists embrace it for crafting acetylated intermediates that act as prodrugs or metabolic probes. Material scientists turn to it for tailored surface modifications, thanks to its swift acylating power. As someone invested in teaching, I find it’s a great case study for demonstrating the reactivity ladder among acyl halides, letting students see why leaving group ability matters. Pharma labs use acetyl iodide both as a strategic building block and as a test case for optimizing reaction conditions—shaving time off development cycles when fast acetylation makes all the difference. Its role also stretches into chemical manufacturing at small scales, where efficiency and selectivity trump raw throughput.
Research & Development Hooks and Hurdles
No research program involving acetyl iodide stands still. The ongoing pursuit of greener processes means teams keep testing alternative iodination techniques, aiming to cut down on phosphorus waste or reduce solvent hazards. There’s increasing curiosity about deploying flow chemistry setups to tame the volatility and scale-up risks. My own contacts in process development tell stories of moving away from classic batch reactors, instead using continuous systems that allow for safer, more controlled handling. Still, hurdles remain—especially around storage, long-term stability, and minimizing byproduct formation. Innovations digging into catalyst design, alternative precursors, and waste valorization keep fueling academic publications as well as industry R&D. Investment in better monitoring tools—like inline IR or NMR—means researchers catch decomposition early, preventing bigger losses and accidents. Every new insight gets folded back into protocols, slowly raising the bar for what counts as best practice.
Toxicity Research Seeks Reliable Answers
Acetyl iodide’s reactive nature sets off alarm bells among toxicologists. The irritant action—eyes, skin, lungs—reflects both the acidity of its hydrolysis products and the underlying volatility. Animal toxicology data remain limited, but by analogy to acyl chlorides and bromides, it’s clear this compound needs robust controls in any setting. Human data point mostly to short-term exposure impacts, like respiratory irritation and burns, underscoring why fume hoods and direct handling bans make sense. Lab incident reports—collected over my years of teaching—demonstrate that even trained staff occasionally let their guard down; burns and mild poisoning symptoms surface most when rushed protocols replace careful preparation. Most regulations advise against chronic exposure, and waste handlers echo these risks. Better, more transparent research into metabolic fate, breakdown products, and chronic exposure is overdue; advances here will help refine safe handling guidelines and inform environmental management.
Future Prospects Shape the Conversation
Looking ahead, acetyl iodide stands at an interesting intersection of tradition and emerging need. As synthesis turns greener and more modular, new routes and handling methods could break longstanding bottlenecks. Innovation around iodination chemistry keeps opening fresh opportunities—radio-labeling, targeted organic frameworks, and plug-in intermediates for custom molecules. At the same time, sustainability pressures steer everyone toward minimizing hazardous waste and boosting atom efficiency. That challenge brings practical shifts: more recyclable reagents, real-time monitoring, and tailored containment to make traditional reagents like acetyl iodide less risky. From my side of the bench, the push to use these classic chemicals in smarter, safer, and more responsible ways feels just as crucial as their performance in the latest synthesis. By connecting better science with sharper safety and sharper awareness of their environmental footprint, compounds like acetyl iodide will keep earning their place in every chemist’s toolkit.
What is acetyl iodide used for?
Unlocking Acetyl Iodide's Role in the Lab
Stepping into a chemistry lab, folks handling organic synthesis have come across acetyl iodide sooner or later. This clear, sometimes strong-smelling liquid pulls its weight as a reagent. Chemists reach for it to introduce acetyl groups into molecules—think of it as a tool for rearranging molecular furniture, especially when they want to convert alcohols into esters. I remember working in a university lab, and everybody respected how reactive this stuff could get. You add acetyl iodide to the mix, and suddenly reactions that stalled with other reagents seem to move forward.
Why the World of Pharmaceuticals Pays Attention
Medicines often start out in glass flasks before making it onto pharmacy shelves. Drug makers need to tweak chemical structures delicately. There’s a constant search for ways to make synthesis steps more efficient without introducing unnecessary byproducts. Acetyl iodide steps in as a tidy source of acetyl groups, ensuring certain intermediates form with fewer impurities. This translates to better drug purity and easier clean-up down the line.
Special Spot in Organic Chemistry
Acetyl iodide doesn’t top the list for everyday chemical supply rooms, partly because you can’t just leave it sitting around—it breaks down when exposed to water or even the air. For those who do need it, it gets made fresh from sodium iodide and acetyl chloride. The real appeal comes in reactions that ask for a more direct or assertive acetylation. Sometimes, traditional sources like acetic anhydride don’t cut it, especially when faced with more stubborn compounds. In those cases, acetyl iodide’s punch gets the job done.
Safety and Production Challenge
Nobody grabs a bottle of acetyl iodide without a game plan. The substance reacts fiercely with moisture, giving off hydrogen iodide, which can corrode metals and hurt lungs if released as a vapor. Even in busy labs, most researchers only work with acetyl iodide in tiny batches inside well-ventilated fume hoods. From my own experience, getting through a full synthesis run without someone telling stories about a leaky flask feels rare. Mistakes stick with you; handling such chemicals calls for sharp training and equipment checks every time.
Greener Paths for Modern Chemistry
There’s growing interest in steering clear of harsh or dangerous reagents wherever possible. Chemists have started swapping acetyl iodide with milder acetylation agents or catalysts, cutting down on risks for workers and the planet. Some companies push for continuous-flow setups, producing acetyl iodide right before use so there’s less leftover to stash or spill. Peer-reviewed studies back this shift, showing lower accident rates and cleaner waste streams as a result.
Respecting Complexity and Progress
Acetyl iodide isn’t on every shopping list, yet its value in niche reactions keeps it relevant. It delivers reliable performance in skilled hands, as long as safety stays front and center. As green chemistry grows, the hope is safer alternatives will continue to spread—based on real lab evidence, not just marketing promises. Folks in research and industry know that trust in a reagent builds over years, one reaction at a time.
How should acetyl iodide be stored?
Understanding the Risks of Acetyl Iodide
Acetyl iodide attracts quick attention from anyone who’s ever opened a bottle in a well-ventilated lab and immediately picked up a sharp, onion-like smell. It doesn't just assault the senses; it carries real health hazards. One drop on skin or splash near the eyes can trigger serious injury. Breathing in too much can damage lungs and mucous membranes. Anyone who's handled this chemical remembers the caution in the air—literally and figuratively.
The main reason for all this worry comes down to the way it reacts with water and moisture around us. Acetyl iodide hydrolyzes quickly, breaking down to acetic acid and hydrogen iodide—both dangerous, corrosive substances. Even a small leak in storage lets moisture sneak in and starts a chain reaction of decomposition, releasing gases that build up pressure or creep into the work area. One careless moment can lead to a mess that's not just inconvenient but actively harmful.
Storing Acetyl Iodide Safely
Rule number one: keep it bone-dry. If you have a freshly delivered container, check the seal. Glass bottles with airtight stoppers—the kind with ground glass and a tight fit—work best. Most labs worth their salt stash acetyl iodide in desiccators or dry boxes. Even if you're using it in a busy space, resist the urge to store it near any water source, including sinks, wet benches, or wash bottles.
Some labs prefer amber glass, and there’s a reason. Acetyl iodide doesn’t handle light well. Sunlight or UV will push it to break down faster, meaning extra risk and wasted money. So, dark glass tucked away in a shaded, stable cabinet offers real benefits. Flimsy plastic just doesn’t cut it here; this chemical eats through soft containers over time, turning storage into a leaky disaster.
Temperature matters. A cool room, not a freezer, slows down those reactions. Too cold, and bottles may crack during handling; too warm, and decomposition ramps up. For years, chemists have kept acetyl iodide in refrigerator chambers dedicated to flammable and reactive reagents, set right above freezing. Not everyone has the budget for specialized fridges, but every chemical storeroom deserves a corner with extra protection: secondary containment trays, away from acids, bases, and metals that spark violent reactions.
Personal Experience and Protective Measures
Anyone who’s transferred acetyl iodide in the middle of a summer afternoon knows what a splash feels like running up against your gloves. Good PPE goes beyond standard nitrile: lab coats, goggles, and face shields. Use gloves with serious chemical resistance—laminated or heavy-duty neoprene. Even short tasks call for a fume hood; don’t believe the myth that small amounts in open air are harmless. I once worked with a researcher who thought “just a quick pour” was safe outside the hood. Minutes later, we had a whole section of the lab clearing out with headaches and burning eyes.
Labeling matters, too. “Corrosive. Decomposes with moisture. Handle only under dry conditions.” Every bottle should say all that, right on the front. No guessing if you’re reaching for the right chemical under time pressure. And every log book should track how long a container has been open. Old stock turns unpredictable. If the bottle’s sticky on the outside or the color’s changed, it’s safer to dispose of it than risk fighting a runaway reaction.
Reducing the Risks for Everyone
Building a solid storage routine saves people, not just property. The science behind acetyl iodide’s hazards is well-documented, and routine safety checks aren’t about red tape; they catch leaks and prevent accidents. Training new lab members, sharing real stories of what can go wrong, and taking the time to check each bottle sets a standard that gets noticed. These aren’t just rules—they’re habits that protect livelihoods and keep a lab running smoothly, year after year.
What are the hazards or safety precautions of handling acetyl iodide?
Getting to Know the Risks
Acetyl iodide stands out with its sharp, irritating odor and volatile personality in the laboratory. Anyone who has opened a bottle can tell you: this chemical wastes no time announcing its presence. It reacts quickly with water and releases fumes that hit the eyes and throat hard, and if you’re working in a cramped lab, the discomfort builds up fast. The risks aren’t just in making your eyes water. Acetyl iodide can corrode skin, eyes, and airways, and once contact gets past a certain point, recovery turns from a rinse to a full medical emergency.
Health Dangers on the Table
Back in my university days, a researcher in the next lab wound up coughing and gasping after an acetyl iodide spill. It only took a few drops and a few seconds of exposure. That incident made one thing clear: this stuff grabs onto moisture in the air—and in your body. Vapors irritate your lungs and eyes right away. If it lands on your hands, the sensation stings and the burn lingers. Chronic inhalation isn’t common in most settings, but even short-term exposure makes the case for respect and caution.
Fire and Reaction: More Than Just a Splash
A bottle of acetyl iodide brings fire risk to the bench. It catches fire if a hot plate or open flame gets too close, and once ignited, it gives off fumes of hydrogen iodide and acetic acid. As someone who’s seen small accidents spiral quickly, I know spills can combine with even trace amounts of water or alcohols, causing heat and hazardous releases. This is no place for shortcuts—protective habits help stop a small error from turning into lab evacuation chaos.
Precautions Every Handler Learns, Sometimes the Hard Way
Goggles and gloves aren’t optional. Nitrile keeps the chemical off your skin better than latex. A lab coat never feels quite thick enough, but it adds a layer. Acetyl iodide needs to be opened and measured in a chemical fume hood—plain ventilation or a cracked window won’t draw fumes away fast enough. Working alone isn’t smart; a partner can catch slips and react fast to problems.
Chemical storage matters more than folks sometimes realize. Acetyl iodide stays stable in tightly sealed glass containers, kept cool and away from water, bases, and oxidizing agents. In practice, even a drop of condensation inside the bottle changes its shelf life and safety. Emergency eyewash and showers must sit within a short sprint from your workbench.
What Actually Makes a Difference
So much of the risk drops if people slow down and follow basic routines: label every container, double-check equipment, and keep no more on hand than necessary. Teams talk through mishap plans—who fetches water, who calls security, who manages the cleanup. Spills need more than just paper towels; neutralizing agents and proper disposal containers make sure fumes and residues disappear safely. Regular training drills keep steps fresh in everyone’s mind.
Flirting with acetyl iodide’s hazards for efficiency never pays off. Safety starts with respect for the chemical and care for the people working with it. Over time, small habits stack into real protection.
What is the chemical formula of acetyl iodide?
Understanding Acetyl Iodide
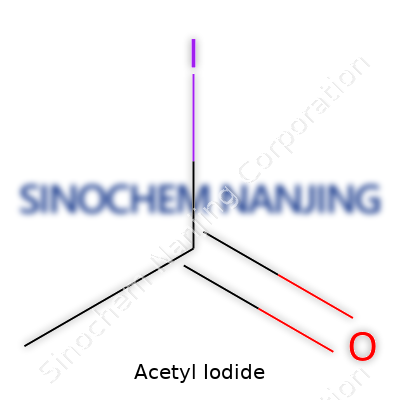
Acetyl iodide doesn’t make the headlines every day, but ask any synthetic chemist about it and you might hear a story or two. Chemistry classrooms skim over it, but once you step into real labs, this compound is more than a trivia answer. The chemical formula for acetyl iodide is CH3COI. Simple on paper, it represents a blend of an acetyl group and an iodine atom.
Why This Formula Matters
Anyone who’s tried to make complicated molecules knows acetyl iodide has a character of its own. Its formula tells you so much: two carbon atoms, three hydrogens, one oxygen, and a single iodine. That iodine gives it some punch — a good leaving group in countless organic transformations. In pharmaceutical labs, where new drugs are made bench-scale before mass production, acetyl iodide offers a straightforward way to introduce an acetyl group into molecular frameworks.
Chemistry shapes the world around us, and small compounds like acetyl iodide play unsung roles in this daily transformation. Acetyl iodide can help turn alcohols into esters or acids, swap out functional groups, or speed up industrial reactions. I remember having a bottle of the stuff during an undergrad project — the sharp, vinegary scent clued me in to its volatility. Safety goggles were not optional.
What We Learn from Its Formula
Chemists rely on clear rules, and a formula like CH3COI gives structure. It tells you not only which atoms you’re dealing with, but also how they might interact. In a practical sense, having iodine stitched into the molecule changes its reactivity compared to other acyl halides like acetyl chloride (CH3COCl). The larger, heavier iodine atom can alter reaction rates and mechanisms.
Take the pharmaceutical world as an example. Introducing an acetyl group in the right spot can transform a molecule’s behavior in the human body. With the heavy iodine atom in acetyl iodide, reactions sometimes become more selective and predictable, bringing hope that a useful intermediate won’t get sidetracked by unwanted byproducts.
Health, Safety, and Environmental Concerns
Handling acetyl iodide means respecting both its power and danger. The iodine in the structure makes this substance less common in big industry, mainly because of cost and disposal challenges. It fits the bill for specialized syntheses, but only folks who understand the risks reach for it. Exposure can burn skin, corrode metals, and react violently with water.
Thinking about sustainability in chemistry, compounds built with scarce elements like iodine push researchers to consider greener alternatives. Incorporating more benign reagents or recycling iodine waste is not just a lab topic; it’s a matter of responsible stewardship in industries that feed, clothe, and heal us.
Moving Forward
Knowledge of chemical formulas like CH3COI gives a foundation for innovation, safety, and responsibility. Building better processes — with an eye on minimizing hazards and waste — means chemistry evolves. Students learn to respect the flask and its contents. Industries find ways to do more with less. Even a ‘simple’ formula carries weight that goes well beyond the page.
How is acetyl iodide synthesized or produced?
The Chemistry Behind Acetyl Iodide
Acetyl iodide isn’t something you find on store shelves. Its sharp smell and strong reactivity command respect in any lab, and anyone working with it knows safety gear is non-negotiable. The method most folks use for making this chemical boils down to a reaction between acetic anhydride and hydrogen iodide. Scientists prefer this route because it’s straightforward and gives a good yield. Hydrogen iodide can be hard to handle; it fumes, it bites at your nose, and it reacts right away with moisture. Mixing it into acetic anhydride forms acetyl iodide—sometimes there's a bit of heating involved, often under controlled conditions to keep things steady and avoid breakouts of dangerous fumes. The result is a colorless liquid that’s eager to react.
Why This Synthesis Matters
Acetyl iodide doesn't get big headlines, but it makes itself valuable in organic chemistry circles. Its uses might look niche from the outside, but for chemists chasing new compounds or streamlining a synthesis, this chemical can open doors no other reagent can. For instance, acetyl iodide helps researchers pack in iodine atoms into organic molecules efficiently. This is a big deal in the world of pharmaceuticals, where making small tweaks to a molecule’s structure can spell the difference between a powerful antibiotic and a dud.
Plenty of synthetic protocols in drug research lean on reliable reagents that just work. Acetyl iodide, by swapping in an acetyl group quickly, gets the job done with less waste and fewer steps. That translates to lower costs and less environmental impact compared to roundabout chemical routes. In my own graduate lab days, we eyed every reagent for reliability and yield. Acetyl iodide earned its place in our fridge—taped up in a fume hood, of course—because it delivered, even if we grumbled about the smell.
Risks and Realities in the Lab
Easy to make doesn’t mean risk-free. If water sneaks into the reaction mix, acetyl iodide breaks down, giving off hydrogen iodide, which can eat through your gloves and leave you coughing. Labs working with it need good ventilation and glassware with airtight seals. Spills and mishaps aren’t just annoying—they can put health and safety at risk for everyone nearby.
From an environmental angle, the mainstream production process creates acidic byproducts that need proper disposal. A few companies have developed sealed system synthesis to cut down airborne hazards and waste, but these set-ups need good training and strict oversight. There’s a real push for “greener” chemistry these days. Swapping out harsh reagents for more eco-friendly ones, engineers and scientists can lighten the load on landfills and wastewater streams.
Room for Improvement
It’s easy to overlook small chemicals like acetyl iodide. Their direct uses might stay out of the public eye, but the steps taken in the lab ripple out—shaping how medicines, polymers, and specialty materials come to life. Investing in safer, cleaner production methods could make a real difference. Modern chemists need reagents that aren’t finicky, don’t pollute, and aren’t hiding big risks in a glass bottle. New techniques for making acetyl iodide, whether by electrochemical processes or using recyclable catalysts, could cut down on waste and dial up safety.
In the end, every bottle of acetyl iodide started with a deliberate reaction and a lab worker who respected its power. It’s another reminder that good science depends on both know-how and careful judgment—plus a healthy respect for what happens when you mix the right chemicals in just the right way.
| Names | |
| Preferred IUPAC name | Ethanoyl iodide |
| Other names |
Ethanoyl iodide Iodoacetic acid Acetyliodide |
| Pronunciation | /əˈsiːtɪl ˈaɪədaɪd/ |
| Identifiers | |
| CAS Number | 507-02-8 |
| Beilstein Reference | 1209221 |
| ChEBI | CHEBI:39174 |
| ChEMBL | CHEMBL1681751 |
| ChemSpider | 11754 |
| DrugBank | DB13845 |
| ECHA InfoCard | 100.007.010 |
| EC Number | 209-704-8 |
| Gmelin Reference | 1149 |
| KEGG | C00835 |
| MeSH | D000080 |
| PubChem CID | 67665 |
| RTECS number | AK4550000 |
| UNII | JJ5M9J76GI |
| UN number | UN2189 |
| CompTox Dashboard (EPA) | DTXSID0034262 |
| Properties | |
| Chemical formula | CH3COI |
| Molar mass | 168.97 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | pungent |
| Density | 2.798 g/mL at 25 °C |
| Solubility in water | Reacts |
| log P | 1.33 |
| Vapor pressure | 17 mmHg (20 °C) |
| Acidity (pKa) | -2.5 |
| Basicity (pKb) | -6.7 |
| Magnetic susceptibility (χ) | -57.5 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.577 |
| Viscosity | 0.83 cP (20°C) |
| Dipole moment | 2.45 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -59.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | '-199.6 kJ/mol' |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS02, GHS05 |
| Signal word | Danger |
| Hazard statements | H302 + H314 + H332: Harmful if swallowed. Causes severe skin burns and eye damage. Harmful if inhaled. |
| Precautionary statements | P210, P261, P280, P301+P330+P331, P305+P351+P338, P309+P310 |
| NFPA 704 (fire diamond) | 3-2-2-W |
| Flash point | 10 °C (50 °F; 283 K) |
| Autoignition temperature | 215 °C |
| Lethal dose or concentration | LD50 (oral, rat): 820 mg/kg |
| NIOSH | NA0450000 |
| PEL (Permissible) | PEL: 0.01 ppm (as Iodine) |
| REL (Recommended) | 'Acetyl iodide (CH3COI), 99%' |
| Related compounds | |
| Related compounds |
Acetyl chloride Acetyl bromide Acetic anhydride Acetic acid Iodoform |

