Chloroacetyl Chloride: A Staple with a Complicated Legacy and a Demanding Future
Historical Development
Chloroacetyl chloride has earned its reputation both for its utility and dangers almost since its discovery. Chemists in the 19th century first worked with it while piecing together the puzzle of acyl halides. Textbooks barely captured just how transformative this molecule turned out to be for both industry and research. Several generations of chemical engineers have learned the hard way that an ingredient doesn't have to be produced on a grand scale to reshape production standards or influence entire markets. Chloroacetyl chloride did both, making an immediate mark in agricultural chemicals and pharmaceuticals. When regulatory agencies around the world began to understand the effects of organochlorine compounds, this chemical was already entrenched in many industries, with little room for simple substitution.
Product Overview
This clear, colorless-to-pale-yellow liquid catches anyone's eye the first time it appears in a laboratory or manufacturing operation. At room temperature, a sharp, biting odor makes its presence almost impossible to ignore. Chloroacetyl chloride carves out a niche as an acylating agent, helping introduce chloroacetyl groups into more complex molecules. Its production volume is modest by bulk standards, but the complexity of handling, storing, and transporting it always grabs more attention compared to everyday commodity chemicals.
Physical and Chemical Properties
At a glance, chloroacetyl chloride feels like a contradiction—powerful, yet easy to mishandle. It boils around 105 degrees Celsius and poses a serious inhalation hazard thanks to its corrosive vapor. Water contact brings out its true nature: hydrolysis happens so fast that choking fumes of hydrochloric acid and chloroacetic acid become an unavoidable risk. Its density and viscosity matter a lot during transfer between containers. It doesn’t take much imagination to see why so much technical effort goes into simple tasks like pumping or sampling. Compatibility with construction materials for pipes and gaskets remains a routine concern, even decades after it first entered industrial use.
Technical Specifications and Labeling
Every container, from lab bottles to bulk drums, demands clear hazard symbols—everybody knows better than to leave one unmarked. Handling instructions are also stricter compared to most other acyl chlorides. The color and purity, often above 98 percent, remain critical to meet the needs of downstream processes in pharmaceutical or pesticide synthesis. Transport documentation has to match local and international guidelines, and failure to mark a shipment correctly can halt a line or trigger a full-scale evacuation. Chemists, shippers, and safety officers share a healthy respect for the way these regulations have evolved in response to real-world incidents.
Preparation Method
Manufacturers avoid unnecessary contact between water and reactants from start to finish. Typically, the process starts with a reaction between chloroacetic acid and thionyl chloride, or sometimes phosphorus trichloride. Generating high-purity material always takes a delicate balance of temperature, agitation, and pressure. By-products—mainly sulfur dioxide and hydrogen chloride—demand careful treatment before atmospheric release. The sheer energy and waste management requirements often undercut any idea that chemistry begins and ends at the molecule itself.
Chemical Reactions and Modifications
Once produced, chloroacetyl chloride's role as a building block shows how one molecule can shape entire markets. Companies use it to convert alcohols and amines into chloroacetamides and esters, workhorse intermediates for herbicides, anesthetics, and antimalarials. In specialty polymers, its high reactivity brings about cross-linking reactions. Sometimes it even functions as a reagent in protection and deprotection steps during organic synthesis. Handling waste streams from these transformations usually requires as much creativity as the chemistry itself, since few substances challenge environmental controls the way this one does.
Synonyms and Product Names
I’ve seen it labeled as alpha-chloroacetyl chloride, monochloroacetyl chloride, and other trade names—not out of a need for ambiguity, but sometimes to align with nomenclature systems or local languages. Researchers and procurement teams stay vigilant to avoid ordering the wrong compound. Even minor differences in naming conventions have caused headaches or costly procurement mistakes, especially for teams spread across borders.
Safety and Operational Standards
Anyone who works with chloroacetyl chloride knows the line between safe operation and disaster can be razor thin. Fume hoods and full-face respirators become non-negotiable standards. Facilities incorporate multiple containment layers and neutralization stations, so that even a small leak or spill gets contained before it can trigger a wider response. Emergency showers and eyewash stations dot the workspace. In my experience, complacency never lasts long—one close call is enough to turn even the most stubborn skeptic into a champion of rigorous double-checks and buddy systems.
Application Area
Agricultural chemicals took up chloroacetyl chloride early in its commercial history, particularly for herbicide synthesis—cases like the widespread use in dichloroacetate production come to mind. It appears in pharmaceutical factories just as often. Analgesics, sedatives, and muscle relaxants all emerge from intermediates that start with a chloroacetyl chloride transformation. Textile processors, dye manufacturers, and even some flavor and fragrance producers occasionally draw on its unique chemical reactivity. Its influence stretches beyond basic chemistry: some sectors depend on it so deeply that supply disruptions can halt entire product lines.
Research and Development
Lab teams everywhere see chloroacetyl chloride as both challenge and opportunity. It enables the development of novel compounds that might one day fight disease or save crops, but it also casts a long shadow over many research paths. Each year, specialty journals report tweaks to improve selectivity, yield, or safety during its use. Recently, focus has shifted toward green chemistry approaches, including solvent minimization, improved by-product handling, and finding less hazardous acylating agents. Computational chemistry now aids process optimization, aiming for steps with lower heat output and less gas release—real-world proof that innovation often springs from necessity rather than convenience.
Toxicity Research
Occupational health studies have documented the acute and chronic dangers for decades. Exposure brings rapid, severe respiratory and tissue damage. Animal studies and worker incident reports underscore the non-negotiable need for full personal protective equipment and fail-safe engineering controls. Efforts continue to penetrate the mechanisms behind its toxicity, hoping to find antidotes or rapid-response treatments, but at present, risk reduction through avoidance and containment remains the only tried-and-true answer.
Future Prospects
Every indication suggests chloroacetyl chloride will stick around for years, driven by global growth in agriculture and pharmaceuticals. More stringent environmental and occupational safety regulations will shape production and handling standards. Emerging chemical processes with less hazardous alternatives may eventually replace it in select applications, but the gap between research promise and large-scale adoption remains wide. Teams dedicated to green chemistry might one day write its epitaph, but until then, safe use and rigorous oversight represent the best approach for workers and the chemical industry.
What is chloroacetyl chloride used for?
Tough Stuff, Real World Impact
Walking through the aisles of any pharmacy or hardware store, you’re likely to cross paths with the end result of some tough chemistry. Chloroacetyl chloride is one of those unassuming players that helps make a surprising range of things work better or simply work at all. Its roots start deep in industrial chemistry. As somebody who’s spent time in both college labs and the real world dealing with chemical safety, I can vouch for both its value and its risks.
Big Role in Pharmaceuticals and Crop Protection
Big pharma keeps chloroacetyl chloride busy. Drug makers lean on it for a reason: it’s a must-have for making broad-spectrum antibiotics like chloramphenicol. It’s also in the background during the creation of pain medications and even heart treatments. Without it, the supply chain in medicine would face a crunch.
The world’s crops gain ground from it too. Herbicides based on chloroacetyl chloride give farmers a fighting chance against weeds. While many may criticize chemical farming, without effective weed control, fields can lose yields. That means higher prices at the grocery store and less food on the table for families. Countries that rely on grain imports particularly value any tool keeping fields productive.
Useful in Plastics and Dyes
Life needs color and durability. Factories transforming raw materials into plastics and dyes have quietly put chloroacetyl chloride to work for decades. It reacts predictably and efficiently, forming special bonds in plastics, coatings, and specialty dyes. You’ll find its influence in everything from colorful textiles to strong synthetic fibers. These items show up in car seats, sports jerseys, and rain jackets. The chemical’s role rarely gets a headline, but without it, modern materials would be tougher and costlier to make.
Safety Isn’t Optional
Not many people get close to chloroacetyl chloride without gloves, goggles, and lots of respect. It’s corrosive and releases toxic fumes if it touches water or air moisture. The people handling it daily know all too well what can go wrong. About a decade ago, I saw a minor spill at an industrial site. The crew reacted quickly: full hazmat suits, rapid exhaust fans, and a shelter-in-place order for the plant until the air cleared.
It’s not just about workers. If leaks reach rivers or the air outside, local folks could face coughs, burns, or worse. Chemical makers, regulators, and emergency crews carry heavy responsibilities. In countries with fewer safety laws or outdated equipment, the risks multiply. Too often, news reports still tell stories of accidental releases that result in hospitalizations or even deaths.
Getting Better at Doing Better
Because of its power and hazard, tighter controls, better training, and modern equipment are essential. Regular safety drills and up-to-date containment systems save lives. Innovations in plant design and extra sensors that spot vapor leaks early keep risk down. Making sure every worker—no matter the country—gets solid safety education and has what they need means fewer lives upended by avoidable mistakes.
At the same time, research teams keep working on friendlier alternatives for the dirtiest jobs chloroacetyl chloride handles. Some new molecules or changes to process design can mean less danger for workers and neighbors without giving up the quality or cost savings that industries need. Big progress may not come overnight, but each step forward brings fewer headlines about accidents and more about smart, safe chemistry in action.
What are the safety precautions when handling chloroacetyl chloride?
Why Extra Caution Matters
Chloroacetyl chloride usually turns up in the stories of chemical manufacturing or academic research, and every chemist or lab worker I’ve known treats it with a kind of respect usually reserved for explosives. There’s good reason for this. Contact with skin, accidental inhalation, or even quick splashes can bring on toxic effects that put people in the emergency room. I can’t count the warnings posted in every lab or production area that stores this chemical, but the atmosphere always feels more strict around it.
The Biggest Dangers
The scent alone—pungent and acrid—tells you moving quickly matters. This compound reacts quickly with water, releasing hydrochloric acid. That means it can turn the humidity in the air (or the moisture on your skin or eyes) into something immediately dangerous. Most stories about accidents start with a small spill that wasn’t contained, or someone whose gloves had a tiny tear. Effects start fast: breathing trouble, deep skin burns, and permanent eye damage. People who experience exposure often don’t want to talk about it after—the pain and panic stay with them.
Personal Protective Gear Isn’t Optional
If you work anywhere near chloroacetyl chloride, you suit up. Not just lab coats and regular gloves, but chemical splash goggles, face shields, thick nitrile or Viton gloves, and full-coverage lab jackets. Double-gloving helps because one layer can fail fast, especially if sweaty hands and chemical drips get involved. In most labs I’ve worked in, anyone handling this compound works inside a fume hood, no questions asked. These hoods pull the fumes away before they get into the breathing zone. It’s not paranoia. Most people learn the hard way that even diluted fumes burn the inside of your nose and throat.
Engineering Controls and Safety Protocols
Equipment plays a part in safety. Splash guards, secondary containment trays, and labeled chemical storage cabinets offer real protection. I always check that the emergency shower and eyewash stations work before starting any job. In one place I worked, management required us to do a quarterly dry run, practicing how fast we could reach the eyewash station with both eyes shut. It felt silly until someone spilled a few drops during an experiment and lost all sense of direction—muscle memory takes over when panic hits.
What Good Training Looks Like
Book learning only goes so far. Solid safety culture comes from seeing people do it right. New staff shadow experienced colleagues before handling anything dangerous on their own. Safety data sheets stay posted, but coworkers and supervisors remind each other all the time which containers need special attention. In some places, workers keep a binder showing step-by-step clean-up procedures for different chemicals—a lifesaver during an accident. I’ve seen supervisors toss out defective gloves or expired reagent bottles on the spot, with no hesitation or negotiation.
Beyond the Obvious: Mindset and Practical Solutions
Confidence grows out of good habits. Rushing or skipping a step costs too much. I once had a colleague who saved hours by prepping all protective gear ahead of time and running through a mental checklist every day, from checking ventilation fans to confirming label accuracy. Simple steps like pairing up with a coworker and verbalizing every action prevent slips. In feedback meetings, people talk honestly about near-misses—the mistakes nearly made. Real solutions come out of these talks, like installing mirrors near hoods so coworkers can spot problems, or standardizing glove quality across the department.
Keeping Perspective
Chloroacetyl chloride invites attention and caution. The consequences of getting careless go beyond minor injuries, so every extra step—whether rehearsing safety drills, choosing the right gloves, or checking the ventilation fan twice—keeps people whole and work running smoothly. Learning from experienced colleagues, paying attention to every detail, and speaking up before things go sideways matter most of all.
What is the chemical formula and structure of chloroacetyl chloride?
Understanding Chloroacetyl Chloride
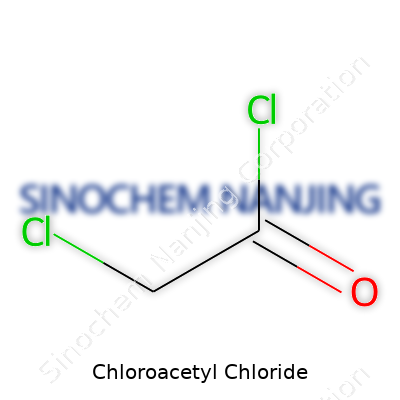
Chloroacetyl chloride, found in labs and factories, carries the chemical formula C2H2Cl2O. With its sharp, acrid smell and fuming behavior, this compound shows just how strong its reactive nature is. The structure tells a lot about why it acts the way it does. You get a two-carbon backbone—one of them binds to both a chlorine atom and a carbonyl group (C=O), while the other just carries two hydrogens. Then, on that same carbon, you find a chlorine atom replacing the usual hydrogen. Staring down its skeletal formula, the arrangement looks like ClCH2COCl. In simple terms, you’ve got one chlorinated methyl group attached to a carbonyl carbon, which is also tied to another chlorine.
The form it takes in space matters for chemists. That carbonyl carbon stands as an inviting target in many chemical reactions. Its two “handles”—the chlorine atom and the carbonyl—make it especially useful for building up more elaborate molecules. I’ve worked with this stuff in an organic lab, and even a drop can fill a room with serious fumes. The safety sheet sits right next to the flask, and protective gear is a must: gloves, goggles, and a hood. Chloroacetyl chloride does not mess around with water, either. It reacts and gives off corrosive, toxic gases like hydrogen chloride. Spills have people scrambling, not just for the mop but for emergency ventilation.
Why It Matters in Chemistry
Chloroacetyl chloride earns its spot in modern industry because it jump-starts so many important reactions. In pharmaceuticals, it acts as a key step in making antibiotics and pain medications. Agrochemical companies use it in the synthesis of herbicides and insecticides that shape world agriculture. This material links simple building blocks into structures that fight infection or protect crops from damage.
The business of handling chloroacetyl chloride cuts close to the cores of safety and stewardship. Accidents with this chemical don’t just mean ruined experiments; they mean evacuation alarms and trips to the hospital. Back at my old university, a fume hood malfunction mid-synthesis shut down the whole lab. That wake-up call reminded everyone of the real-world weight chemicals carry. This compound teaches respect, and every bottle on the shelf stands as a reason for checklists and backup plans.
Tackling the Challenges
Making safer workplaces starts with training and the right equipment. Clear labeling, airtight storage containers, and regular safety audits cut down on risk. Modern labs often use scrubbers to trap escaping fumes before they reach workers. Research continues into how to make these reactions cleaner and less hazardous. Some startups look for alternative reagents that still deliver useful chemical reactivity without the intense risks. In the meantime, those who work with chloroacetyl chloride rely on up-to-date safety data, engineering controls, and constant vigilance.
Chloroacetyl chloride remains a cornerstone in chemical manufacturing. Its structure and reactivity drive innovation in medicine and agriculture, but demand the highest standards of care and respect. Direct experience underscores just how much goes into managing these substances—and why every precaution matters in the quest for progress.
How should chloroacetyl chloride be stored and transported?
Why Handling This Chemical Brings Real Risks
Chloroacetyl chloride doesn’t mess around. One broken seal or a careless transfer, and you’ve got fumes, burns, and a big safety incident brewing. People who’ve worked with it know the harsh odor and the sting on bare skin, so trying to cut corners on containers or transport never pays off. In a busy lab, I once watched a small leak fill a space with a choking cloud in seconds. It’s easy to underestimate just how aggressive this stuff gets, especially toward moisture and organic materials.
Safe Storage Starts With Good Containers
Steel drums line many chemical storerooms, but chloroacetyl chloride chews through regular metals and moisture like acid. Stainless steel with special linings or heavy-duty glass containers handle it much better. Don’t trust any old container; it will eat right through and leave disaster behind. Rusty seals and aging valves lead to regret and big clean-ups. Keeping stocks in a dry, cool room with no sunlight stops the chemical from breaking down or causing pressure buildup.
Personal experience has taught many in the chemical trades: stray drops on shelves raise a warning smell, and humidity inside storerooms spreads the risk. So, working dehumidifiers and good airflow matter a lot. If you can’t trust the air or sealing, you shouldn’t store this chemical there.
Transport Calls for Sharp Focus
Nobody wants to drive a truck with this stuff through a summer traffic jam. Chloroacetyl chloride releases hydrochloric acid gas on contact with water, so even a small leak on a rainy day causes panic. Hazmat rules in most countries demand leakproof, labeled drums or bulk tanks, all tightly strapped down. Drivers need full training and a spill kit close by. A rookie mistake—like missing a cracked gasket—can trigger a highway lockdown or a hospital visit.
Regulations put the spotlight on route planning. Take the path away from schools and homes, avoid bridges or tunnels when possible, and check the weather for storms or flooding. I’ve seen shipments get rerouted on wet days because the risk isn’t worth it.
Why People and Procedures Make the Difference
Accidents don’t come just from bad containers; they come from rushing, distraction, or forgetting the little steps. Wearing the right gloves and goggles, following the checklist, double-sealing every cap—these aren’t just annoyances. They’re why people go home safe and doors stay open for business the next day.
Regular drills, updated emergency plans, and tight coordination with local fire departments help keep small mistakes from turning into headline disasters. From my time in medium-sized chemical plants, the teams that met weekly, checked up on their own work, and raised concerns without fear, always had fewer scares.
Practical Solutions for a Tough Chemical
Tamper-evident sealing and digital record-keeping keep storage honest. Routine inspections pick up hidden leaks or corroded spots. Good insurance makes sense, but prevention works better. Sometimes older containers hold surprises—a swap now saves money and headaches later. Staff deserve clear training and time to do things right, not rushed handovers or half-told instructions. Investing in better storage and safer truck fleets does more than tick boxes; it saves lives and reputations.
At the end of the day, chloroacetyl chloride rewards attention and good habits. Trying to take shortcuts with such a stubborn chemical rarely ends well, and the stories of what went wrong stick around a long time. Getting storage and transport right means everyone sleeps a little better.
What are the hazards and potential health effects of chloroacetyl chloride exposure?
The Nature of the Risk
Many workers in chemical plants or labs walk past drums of chemicals without looking twice at the hazard labels. Chloroacetyl chloride stands out among these as one of the nastier substances. Even small leaks or spills create a dangerous situation; this chemical reacts violently with water, kicking out harsh fumes of hydrogen chloride and phosgene. A damp floor, a little rain on an open container, and suddenly the air feels like tear gas has filled it. This stuff has landed workers in emergency rooms with blistered airway linings and corroded skin.
Sneaky Routes of Exposure
It only takes a bit to start causing trouble. If chloroacetyl chloride lands on the skin, its corrosive bite burns fast. Eyes take even more damage — one splash can ruin sight in seconds. Inhaling the fumes stings the throat and lungs, making it hard to breathe. Folks donning protective suits and gloves know that a torn glove means their workday can end at the hospital. I’ve seen supervisors insist on face shields and respirators because even hardened old-timers won’t cut corners around this chemical.
Long-Term Health Effects
Pain and blistering happen in real-time, but injuries can linger for months. Scar tissue replaces soft tissue after deep burns. Breathing in those acidic clouds? That can leave lifelong asthma or bronchial scars. A Harvard survey on chemical industry workers found long-term lung problems became much more frequent among those handling chloroacetyl chloride compared to workers not exposed to it. Treating these health issues drains public health resources, and it’s tough to put a price tag on someone’s ruined lung function.
The Workplace Factor
Hazards like this never stay contained by accident. One study from the American Chemical Society recorded over a dozen accidental releases at manufacturing sites in the last ten years. Most came down to lapses in protective equipment use or improper storage. Storing it near water sources creates a recipe for disaster. Old pipes and poorly kept containers raise the chance of an unwanted reaction happening. Short staffing or rushed shifts only make it more likely that someone forgets extra eye protection or mislabels a drum.
Prevention: Real Steps, Real Results
Clear training helps, and so does strict supervision on the shop floor. Teams need to practice drills, not just read wall posters. I’ve witnessed management transform accident rates by rewarding the right safety habits — not punishing mistakes, but handing out bonuses for thorough double-checks. Workers told me that real investment in new gloves, newer acid-resistant clothing, and good face shields stopped injuries. Even simple things like shower stations close to the mixing area can mean the difference between saving a limb or losing it. Regular air quality checks pick up small leaks before they turn into clouds of toxic gas.
Community Awareness
People living near chemical plants rarely get a say in what’s stored next door. Community groups push for public disclosure of chemical hazards and better emergency plans. Fencing off danger does little if residents don’t know what to do if sirens go off. Some towns now distribute emergency kits and teach people how to seal windows and shelter if a leak drifts their way. Public reporting of near-misses pushes management to stay sharp. My advice: ask questions about what chemicals are handled nearby; don’t wait for an accident to learn the hard way.
| Names | |
| Preferred IUPAC name | 2-chloroacetyl chloride |
| Other names |
CAC Chloroacetylchloride Acetyl chloride, chloro- Monochloroacetyl chloride |
| Pronunciation | /ˌklɔːr.oʊ.əˈsiː.tɪl ˈklɔːr.aɪd/ |
| Identifiers | |
| CAS Number | 79-04-9 |
| Beilstein Reference | 1209226 |
| ChEBI | CHEBI:35214 |
| ChEMBL | CHEMBL14220 |
| ChemSpider | 13031 |
| DrugBank | DB14051 |
| ECHA InfoCard | 100.004.672 |
| EC Number | 200-900-5 |
| Gmelin Reference | 8229 |
| KEGG | C01747 |
| MeSH | D002682 |
| PubChem CID | 6579 |
| RTECS number | AF9625000 |
| UNII | Y7C3G9D895 |
| UN number | UN1752 |
| Properties | |
| Chemical formula | C2H2Cl2O |
| Molar mass | 112.94 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Pungent odor |
| Density | 1.42 g/cm³ |
| Solubility in water | Reacts |
| log P | 1.09 |
| Vapor pressure | 13 mmHg (20°C) |
| Acidity (pKa) | 1.0 |
| Magnetic susceptibility (χ) | -7.23 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.468 |
| Viscosity | 1.388 cP (25°C) |
| Dipole moment | 1.90 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -217.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -507.2 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D14AB09 |
| Hazards | |
| Main hazards | Causes severe skin burns and eye damage; may cause respiratory irritation; toxic if inhaled, swallowed or in contact with skin; reacts violently with water. |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H301, H311, H314, H331, H335, H410 |
| Precautionary statements | P210, P260, P262, P264, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P330, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-3-2-W |
| Flash point | 66 °C |
| Autoignition temperature | 633°F (334°C) |
| Lethal dose or concentration | LD₅₀ oral rat 414 mg/kg |
| LD50 (median dose) | LD50 (median dose): 160 mg/kg (rat, oral) |
| NIOSH | CAS No. 79-04-9 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Chloroacetyl Chloride: 0.05 ppm (Ceiling) |
| REL (Recommended) | 0.001 ppm |
| IDLH (Immediate danger) | 2 ppm |
| Related compounds | |
| Related compounds |
Acetyl chloride Chloroacetic acid Chloroacetyl-CoA Chloroacetamide |

