4-Bromophenylacetyl Bromide: An In-Depth Commentary
Historical Development
Early work in the field of brominated aromatic compounds opened avenues for 4-Bromophenylacetyl Bromide. Chemists in the 20th century sought new reagents for the synthesis of pharmaceuticals and fine chemicals. Demand for molecules offering selectivity during acylation attention led to this compound’s wider adoption. Laboratory notebooks from those decades show frequent use of phenylacetyl derivatives in the quest for novel drug backbones. With every passing decade, labs came back to halogenated acetyl bromides, drawn by their reactivity profile. The timeline of its commercial production also reflects changes in chemical manufacturing practices, swinging towards better yields, improved purification, and stricter occupational standards.
Product Overview
4-Bromophenylacetyl Bromide occupies a unique spot in the family of aromatic acyl halides. Chemists value it for its ability to introduce the 4-bromophenylacetyl moiety into complex structures. Research and manufacturing teams leverage this property during creation of specialty chemicals. Its presence in the toolbox of medicinal synthesis signals the importance of acyl halides in both research and applied chemistry. It stands out among its analogues due to the electron-withdrawing power of both the bromine at the para position and the acyl bromide group, making it a versatile intermediate.
Physical & Chemical Properties
This compound typically appears as a colorless to light yellow liquid, with a characteristic pungent odor that experienced chemists recognize as a caution flag. It melts below room temperature and boils near 155-157°C at reduced pressure, which makes careful storage vital. Its molecular formula, C8H6Br2O, and molar mass, roughly 277.95 g/mol, show how halogenation impacts density and reactivity. Its structure includes a bromine ortho to the acetyl side chain, an arrangement that guides its behavior in organic transformations. The bromide groups at both the para phenyl and the acetyl carbon allow for diverse manipulation, contributing to its wide appeal. High reactivity toward nucleophiles and strong lachrymatory effects mean that anyone handling this must take protocol seriously.
Technical Specifications & Labeling
Quality control labs provide specifications that cover purity, common impurities, and moisture content. Purity for most bench and industrial work exceeds 98%, with gas chromatography used to confirm this. Labels carry the CAS Number 2114-00-3, hazard symbols for corrosivity and toxicity, and batch-specific data to trace each bottle back through its supply chain. Material Safety Data Sheets (MSDS) include information about safe storage, required PPE for handling, and steps to mitigate accidental exposure. Shipping containers often rely on amber glass to reduce photo-degradation and minimize risk.
Preparation Method
Laboratories rely on established synthetic pathways to obtain 4-Bromophenylacetyl Bromide. A common route begins with 4-bromophenylacetic acid, which undergoes treatment with brominating agents such as phosphorus tribromide or thionyl bromide. This reaction liberates HBr and requires a dry, inert atmosphere to prevent unwanted side reactions. Controlling temperature and stoichiometry, chemists monitor the reaction mix until GC-MS or TLC confirms complete conversion. Purification often involves distillation at reduced pressure. In some settings, additional washing steps with aqueous sodium bicarbonate and brine remove acidic or water-soluble residues. Each step demands careful exclusion of water to prevent premature hydrolysis.
Chemical Reactions & Modifications
4-Bromophenylacetyl Bromide functions as an effective acylating agent, reacting readily with amines to form amides. Medicinal chemists often exploit its reactivity to produce intermediates for APIs where a para-brominated phenyl ring improves biological activity. Nucleophilic substitution at the acyl bromide is rapid, while the bromine substituent on the ring survives many reaction conditions. Transition-metal catalysis can further modify the bromine, introducing new substituents via Suzuki or Buchwald–Hartwig coupling. This adds breadth to the synthetic transformations accessible with this building block. Acid chlorides are sometimes preferred in acylation, but the bromide offers nuanced selectivity in some protocols. Polymers and agrochemical segments both utilize its ability to form robust carbon-nitrogen and carbon-oxygen bonds.
Synonyms & Product Names
This compound carries a collection of synonyms: p-Bromophenylacetyl bromide, para-Bromophenylacetyl bromide, 2-Bromo-1-(4-bromophenyl)ethanone, or 4-(Bromophenyl)acetyl bromide. Chemical catalogs occasionally list it under trade or code names, particularly where proprietary intermediates are involved. Researchers often recognize products via their CAS Number 2114-00-3, avoiding confusion in multi-language environments.
Safety & Operational Standards
Inhalation or skin contact with 4-Bromophenylacetyl Bromide leads to acute irritation, respiratory distress, or prolonged dermal reactions. Fume hoods, gloves, splash goggles, and impervious lab coats remain mandatory for handling. Storage requires tightly sealed containers under inert gas, away from moisture and base-sensitive reagents. Spill response training features prominently in standard operating procedures. Labs and warehouses install local exhaust ventilation to trap fumes before exposure risks escalate. Emergency eyewash and drench showers, along with easy access to spill kits, minimize the impact of accidental release.
Application Area
Medicinal chemists and process engineers use this intermediate in the preparation of pharmaceutical agents, relying on its ability to generate key amides and esters. Agrochemical firms transform it further to synthesize plant protection agents. It shows up in the synthesis of dyes, specialty polymers, or as a reactive intermediate in fine chemical manufacturing. Custom synthesis companies turn to it for client-driven projects in drug development. Its reactivity profile and compatibility with palladium catalysis expand its reach in both medicinal and material science programs.
Research & Development
Innovation teams track new synthetic methods designed to cut waste and improve yields. Directed ortho-lithiation, direct bromination, and alternative halogen-exchange methods receive ongoing attention in academic literature. Startups explore greener brominating agents and solvent-free reactors to further reduce environmental impact. Patent databases reveal evolving methods to fine-tune selectivity or bypass sensitive steps. Research often correlates modifications of the phenyl ring with changes in biological activity, providing new paths in hit-to-lead optimization.
Toxicity Research
Toxicologists classify 4-Bromophenylacetyl Bromide as a severe irritant, with acute toxicity following inhalation, skin contact, or ingestion. Laboratory animal studies suggest moderate systemic toxicity, leading teams to emphasize risk minimization over substitution. There is no compelling evidence of chronic toxicity or carcinogenicity, but repeated exposure leads to cumulative irritation or sensitization. Occupational safety teams use real-time air monitoring and personal exposure records to safeguard workers. Any spill calls for full evacuation, decontamination, and incident reporting.
Future Prospects
As industries demand higher purity and better safety, manufacturers look for cleaner, more efficient routes to 4-Bromophenylacetyl Bromide. Regulatory agencies push for greener reagents, improved PPE, and process automation to further cut risk. Researchers in pharmaceuticals and agrochemicals continue to find new uses for brominated aromatic building blocks, driving steady demand. Advances in catalyst technology could lead to selective transformations that make the most of both bromide groups. Improved analytical methods give labs tighter control over impurity profiles, supporting stricter standards. The compound stands at the intersection of risk and reward, underscoring the need for technical expertise, robust documentation, and a steady adaptation to best practices.
What is the chemical formula of 4-Bromophenylacetyl Bromide?
Digging Into the Structure
4-Bromophenylacetyl bromide brings its own story to the table when looking at its chemical makeup. It carries the formula C8H6Br2O. On paper, it looks straightforward: a phenyl ring with a bromine atom at the fourth position, joined to an acetyl bromide group. Molecules like this aren't just a neat arrangement of carbon and bromine. Behind that formula, I see years of organic chemistry packed into a handful of letters and numbers, each piece chosen for a purpose.
Why Chemical Precision Remains Essential
In the laboratory, missing a single digit or forgetting a bromine atom can shift a reaction in the wrong direction. I remember a college research project, chasing down a reaction that just wasn’t yielding results. Turned out the chemical catalog had swapped a methyl for a bromo on a phenyl ring. It cost us a month. For teams synthesizing advanced pharmaceuticals or specialty materials, that error could mean millions of dollars lost, or worse, the wrong compound entering testing.
Accurate formulas also let chemists talk to one another in a common language. Researchers in Mumbai or Boston glance at C8H6Br2O and know what bonds connect and which functional groups lie on the molecule. This global shorthand reduces confusion and speeds up discovery. Checking for purity, running NMR or IR spectra, making sure no stray bromine or carbon has snuck into a reaction—all rely on that initial, exact formula.
Current Use and Where It Leads
4-Bromophenylacetyl bromide is no stranger in organic synthesis labs. It often acts as a building block for more complicated molecules, especially for those working on pharmaceuticals, fragrances, or advanced plastics. The extra bromine atom makes it a handy candidate for further substitution, giving chemists flexibility in shaping the final product. Every year, dozens of research papers explore what new compounds can be achieved with this specific blueprint.
Precision isn't just some academic exercise; it echoes out to real-life applications. Imagine a medication that’s off by just a single group of atoms. Medicines, polymers, or specialty coatings all trace their reliability back to knowing exactly what’s coming out of each reaction flask.
Addressing the Risks and Finding Solutions
Mislabeling or misidentification of chemicals causes more than research setbacks. Safety incidents happen when formulas are off. Workers need to handle chemicals like 4-Bromophenylacetyl bromide cautiously: this compound can release corrosive hydrogen bromide fumes if not stored correctly. Clarity in documentation and labeling avoids dangerous surprises in the lab and supports the kind of traceability that regulators and safety officers expect. I’ve seen safety audits pick up on mislabeled bottles from suppliers, and the fines, as well as trust lost, linger much longer than the cleanup.
Handling these risks starts with robust databases, double-checking entries in electronic lab notebooks, and relying on trusted suppliers with a reputation for precision. Training plays a big part, too—making sure everyone from students to senior researchers understands why those formulas matter past the surface level.
Direct Paths to Stronger Practices
Early career chemists and students need exposure to the fundamentals of chemical nomenclature and structure drawing. Digital tools now offer 3D visualizations, minimizing the chance for error. Open-access databases and peer-reviewed publications help professionals verify chemical data before moving forward with new syntheses.
In my experience, the persistent drive for accuracy not only strengthens science but keeps everyone along the supply chain safe—from manufacturer to researcher to end user. Reading a formula like C8H6Br2O properly is more than a technical detail; it shows a respect for the discipline and the real-world impacts every experiment brings with it.
What are the common uses or applications of 4-Bromophenylacetyl Bromide?
Behind the Science: What Drives Its Use
Ask any organic chemist who spends long hours in the lab about core reagents, and 4-Bromophenylacetyl Bromide usually comes up. This compound plays a key role in the world of pharmaceutical and pesticide synthesis. It’s not a household name, yet plenty of researchers depend on it to form more complex molecules.
Building Blocks in Drug Design
Pharmaceutical companies lean on this compound for making a wide range of active ingredients. The bromine atom gives the molecule high reactivity, helping researchers join different molecular groups together. Imagine you’re tasked with creating a targeted cancer drug. Chemists need a solid way to introduce acetyl groups to their base structure or attach a bromine-bearing fragment. This is where 4-Bromophenylacetyl Bromide comes in. It acts like a construction tool, fitting new widgets onto an existing scaffold during a sequence of chemical reactions.
Many new painkillers, antibiotics, and anti-inflammatory compounds start as nothing more than basic molecules joined together with the help of reagents like this one. Pfizer, Novartis, and others have patents that include 4-Bromophenylacetyl Bromide during early synthetic stages. Without it, labs lose a shortcut, especially when speed and precision are essential.
Chemical Synthesis Beyond Pharmaceuticals
The presence of a bromine atom means new uses show up each year. Pesticide makers value this compound, too. The pathway from basic raw chemicals to the final crop-protection product includes many steps where selectivity matters. Farmers depend on these chemicals for reliable yields, but back in the development stage, selecting the right intermediate saves both time and money.
Dye and pigment industries also get benefits here. I once spoke with a dye chemist frustrated by unpredictable yields. They found that 4-Bromophenylacetyl Bromide minimized their risks in connecting aromatic rings, which led to purer colors in the end product. Fewer impurities mean better textiles, and the consumer never knows what’s behind that brilliant shade.
Step Stone for Advanced Organic Chemistry
Learning to handle 4-Bromophenylacetyl Bromide gives graduate students and young researchers a chance to practice real-world reaction set-ups. The compound’s reactivity opens up routes for making ketones, amides, and more complicated heterocyclic frameworks. In research, the difference between success and failure often comes down to reagent selection. Both industrial and academic labs appreciate how this molecule streamlines difficult syntheses.
Handling and Safety Challenges
Every laboratory that uses 4-Bromophenylacetyl Bromide needs to address health and safety upfront. Brominated compounds demand strong fume hoods and proper personal protective gear. Even a minor mistake can lead to harmful exposure. Companies prioritize regular training, and students in teaching labs learn to respect its hazards early on. Investing in safe workflows builds both trust and reliability.
Improving Access and Environmental Footprint
Production of this compound relies on bromine and acetyl chemistry, which creates waste streams. Chemical manufacturers now look for greener processes and better recovery systems. Advances in batch continuous flow technologies help cut down on byproducts and energy use. European and U.S. regulators push for cleaner synthesis, and this pressure has led to several innovative projects that reduce waste.
Ultimately, 4-Bromophenylacetyl Bromide supports modern chemistry in ways that stretch far beyond the lab. By improving safety measures, investing in greener manufacturing, and staying alert to new scientific needs, the chemical industry maintains both progress and responsibility.
What are the safety precautions for handling 4-Bromophenylacetyl Bromide?
Understanding the Risks
4-Bromophenylacetyl bromide has a reputation in many labs as a chemical you don’t want on your bare skin, much less in your lungs or eyes. Handling this reagent means you respect the risks that come with powerful acyl bromides, not just for yourself but for anyone nearby. From my days sharing crowded university fume hoods, I’ve watched people underestimate just how aggressive this stuff gets. Even low amounts in the air sting the nose, and one careless spill can eat through gloves much faster than you think.
Personal Protection Pays Off
Latex or nitrile gloves seem like a staple for most bench work, but not all gloves hold up against acyl bromides. I once saw nitrile melt in seconds after catching a droplet during a transfer. Neoprene or butyl rubber gloves handle this product better. It seems like overkill, but full splash goggles and face shields have spared me from some close calls. Even the most careful transfers go sideways sometimes, and a face-level reaction isn’t something you want to risk.
Ventilation Means Everything
No matter how busy or rushed a lab gets, no one should work with 4-Bromophenylacetyl bromide outside a fume hood. I’ve watched colleagues rely on sash doors as a safety blanket, but airflow matters even more. The gas released from these reagents not only irritates the lungs but can trigger full-on respiratory distress at higher exposures. I make sure to use freshly checked hoods and avoid blocking the air intake with bottle caps or flasks.
Storage is a Big Deal
Leaving this chemical out on a benchtop is like inviting trouble. Moisture in the room kicks off hydrolysis, and you end up with hydrogen bromide gas in the air—bad for your health and everyone else’s. Dry, sealed containers inside flammable-proof storage cabinets work well. If possible, stashing it with desiccants helps stop the worst of the degradation, and labeled secondary containment keeps spills from spreading across shelves.
Spills and Disposal Shouldn’t Get Brushed Off
I learned early not to rely on paper towels for chemical spills. For any spill, I reach for prepacked neutralizer pads or spill kits built for acids and bromides. Small amounts require absorbents that won’t react violently. Once cleaned up, I always seal waste in clearly marked containers. Making friends with the hazardous waste coordinator means quick pickup and less chance of a forgotten bottle creating a leak in a seldom-visited closet.
Looking Out for Each Other
One lab lesson stands out more than most: No one should handle tricky chemicals alone. Buddy systems and check-ins helped catch a few near-misses before they turned into ER visits. Quick access to eyewash stations, safety showers, and updated SDS sheets meant fewer surprises after an accident. I keep emergency numbers nearby and make sure everyone knows where to find them. It feels awkward to double-check, but it always beats scrambling in a panic.
Building Better Habits
I’ve seen the difference between labs with strong safety cultures and ones that cut corners. Training, routine drills, and a willingness to call out risky moves—these go further than any posted sign. When people feel responsible for group safety, they handle dangerous reagents with the respect they deserve. Proper handling of 4-Bromophenylacetyl bromide doesn’t just protect the person in gloves; it keeps the whole team working and learning together, safely.
What is the storage condition for 4-Bromophenylacetyl Bromide?
What Makes 4-Bromophenylacetyl Bromide Special
Anyone who’s worked with fine chemicals quickly understands that mistakes in storage can lead to serious headaches or even emergencies. 4-Bromophenylacetyl bromide stands out as a reactive chemical, often found in pharmaceutical labs and academic research. It isn’t one of those harmless shelf-sitters; this compound reacts strongly with moisture and decomposes to produce highly irritating or corrosive fumes like HBr. Ignoring the right storage conditions easily turns a useful tool into a hazard.
Real Risks of Poor Storage
I’ve seen a bottle of brominated acyl agents get left on a bench near a window. Over a summer week, even without direct sunlight, condensation crept in through a loose cap. By the time anyone noticed, the container had popped and the lab reeked of acid gas. That incident didn't just waste product. It affected everyone in the room. Eyes stung, people coughed, and we all scrambled to evacuate and ventilate the space. People get complacent after seeing so many powder jars sitting quietly, but acyl halides demand more respect.
Essentials for Safe Storage
You can’t just toss 4-Bromophenylacetyl bromide on the nearest shelf. The main thing is to keep it bone dry: water triggers decomposition, so secure caps and good seals matter. Glass bottles with Teflon-lined caps make a real difference compared to regular plastic. If a container is in any way damaged, it’s best to transfer the material to new packaging under a fume hood.
Temperature comes next. Cool storage, preferably at 2–8°C, slows degradation. Standard refrigerators in chemical storerooms work as long as they keep the airflow clean and prevent condensation inside. Dedicated desiccators or refrigerators with clear hazard labeling offer extra insurance. I’ve always used containers with moisture indicators; it’s an extra line of defense when someone forgets to reseal a cap.
Ventilation and Location
Separation from incompatible materials—alcohols, amines, water, and even some bases—matters a lot. I once saw a spill go from minor annoyance to full-blown panic after someone stacked halides beside acids. HBr gas built up and we needed a full hazmat clean. Small labs without vented cabinets can create similar risks. A flammable storage cabinet with chemical-resistant shelving keeps things under control, but ventilation should pull any accidental vapors away from busy work areas.
Labels and Training
Clear, durable labeling avoids confusion. People working in academic settings—especially new grad students—sometimes misjudge the risk. Having visible hazard signs and clear notes about “moisture sensitive, corrosive” helps everyone. Adequate training about emergency procedures and chemical properties should be part of every lab’s culture.
Simple Solutions Go Far
Storing dangerous chemicals like 4-Bromophenylacetyl bromide safely doesn’t require huge budgets. Regular integrity checks on containers, a dry and cool environment, solid labeling, and good communication about hazards protect both the people and the research. Everyone makes mistakes, but with thoughtful storage, one slip is less likely to become a disaster.
Author Experience
After fifteen years in chemical research, I’ve learned to respect the small details. Saving a few minutes on storage never beats the peace of mind that comes from knowing you, your colleagues, and the work you’re doing stay safe. Storage routines become second nature, and the payoff is fewer accidents and more time focusing on results instead of cleanup.
Is 4-Bromophenylacetyl Bromide available in different purities or quantities?
Different Purities, Different Projects
Walking into any lab, each scientist knows that purity isn’t just a number. Small impurities can cause whole batches to fail. With a reactive chemical like 4-Bromophenylacetyl Bromide, every decimal on a purity certificate tells you something about risk, reliability, and reputation. Some suppliers advertise 98% purity, others claim 99%—and the price gap can bounce widely. In organic synthesis, chasing that last percent can make all the difference between a clean result and weeks of troubleshooting unexplained noise in your data.
My days in a university research group, pooling together grant money, gave me a quick education in compromise. Higher-purity chemicals hit the budget hard, so every order sparked a debate: do we trust our technique enough to scrub away the last remnants, or do we need to pay a premium for substance that fits straight into the protocol? It’s not just about chemical greediness—using high-purity materials matters when your publications, patents, and grants ride on reproducibility.
Buying in Bulk? Think Storage, Think Safety
Talking about quantity, the major divide falls between research-scale and industrial-scale orders. A professor’s cabinet and a pharma warehouse couldn’t be more different. In small research labs, one has to count every milliliter. A supplier selling 5-gram or 25-gram bottles meets that market—anything bigger turns into a safety liability, attracting regulatory headaches and extra paperwork.
Industrial buyers pull out the stops for kilos. Their world turns on logistics: sealed drums, batch consistency, robust documentation. A bulk order cuts per-gram cost, but that opens doors to regulatory scrutiny. 4-Bromophenylacetyl Bromide isn't exactly household bleach—importing it triggers paperwork that most folks never see in a lifetime of shopping. I've seen colleagues stuck for weeks untangling customs and safety measures, all so a drug development project keeps moving forward.
Hidden Costs: Legality, People, and the Environment
Volume and purity don't surface as simple choices. Behind the label sits a web of regulations. Countries treat this molecule with caution. This isn’t overkill—4-Bromophenylacetyl Bromide finds use in sensitive processes that can link to controlled substances. Any order draws questions about intent and end use. Even the smallest mishap can bring legal inquiries or audits, not just for buyers but for sellers, too.
Disposal threads itself through this conversation. Once a bottle empties, responsibility doesn't vanish. Disposing of leftover, even tiny amounts, demands following rules that keep local water supplies clean and workers out of harm’s way. Once, I watched a junior colleague forget a simple dilution step—corrosive fumes ruined an afternoon and left two people with headaches. Safety training means more than paperwork: in the wrong hands, high-purity chemicals invite accidents.
Solutions: Partnership, Vigilance, and Transparency
Open dialogue with suppliers solves more problems than secrecy ever could. Lab managers who ask for detailed batch documentation, real-time support, and technical data sheets walk away with fewer regrets. Those from my circle who build these relationships always get better service—and are quicker to detect potential supply chain problems early.
Training doesn’t stop after the first year. Refresher courses on hazardous material handling, plus strict inventory checks and disposal processes, give every researcher a line of defense. I’ve seen this culture prevent disaster more than once—someone double-checking a lot number caught a shipment error that could have derailed a project for months.
Better transparency, not more red tape, helps everyone along the supply chain. Reliable data, honest labels, and swift recall processes go further than handwaving claims of quality. In the end, quality materials empower good science—and protect people, profits, and the planet.
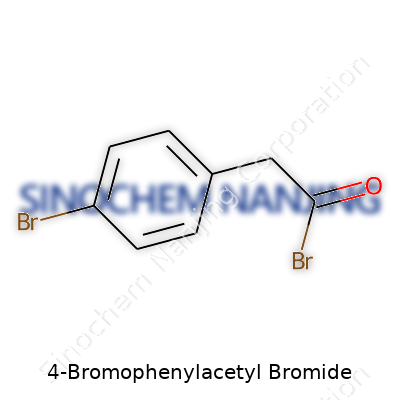
| Names | |
| Other names |
4-Bromophenylacetyl bromide 2-Bromo-1-(4-bromophenyl)ethan-1-one p-Bromophenylacetyl bromide 4-Bromobenzyl bromide α-Bromo-p-bromophenylacetyl bromide |
| Pronunciation | /ˈfɔːr broʊmoʊˈfiːnɪl əˈsiːtɪl ˈbroʊmaɪd/ |
| Identifiers | |
| CAS Number | 2372-58-9 |
| 3D model (JSmol) | `Brc1ccc(cc1)CC(=O)Br` |
| Beilstein Reference | 1205777 |
| ChEBI | CHEBI:136432 |
| ChEMBL | CHEMBL415585 |
| ChemSpider | 16215144 |
| DrugBank | DB03785 |
| ECHA InfoCard | 06c65eaf-2f0c-43c0-b45f-79a25a1e8158 |
| EC Number | 211-663-3 |
| Gmelin Reference | Gmelin Reference: 81894 |
| KEGG | C18367 |
| MeSH | D017956 |
| PubChem CID | 1558570 |
| RTECS number | CU1400000 |
| UNII | AW47K9Y0C2 |
| UN number | UN3265 |
| CompTox Dashboard (EPA) | 4-Bromophenylacetyl Bromide CompTox Dashboard (EPA) ID: **DTXSID3057298** |
| Properties | |
| Chemical formula | C8H6Br2O |
| Molar mass | 291.93 g/mol |
| Appearance | White to light yellow crystalline powder |
| Odor | Pungent |
| Density | 1.7 g/cm³ |
| Solubility in water | Reacts with water |
| log P | 1.83 |
| Vapor pressure | 0.3 mmHg (25°C) |
| Acidity (pKa) | 14.08 |
| Magnetic susceptibility (χ) | -74.0e-6 cm³/mol |
| Refractive index (nD) | 1.626 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.93 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 362.9 J·mol⁻¹·K⁻¹ |
| Hazards | |
| Main hazards | Causes severe skin burns and eye damage. May cause respiratory irritation. Toxic if swallowed, in contact with skin or if inhaled. |
| GHS labelling | GHS05, GHS06 |
| Pictograms | BrC1=CC=C(C=C1)CC(=O)Br |
| Signal word | Danger |
| Hazard statements | H302, H314, H318, H335 |
| Precautionary statements | Precautionary statements: "P260, P261, P264, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P312, P363, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | 82°C |
| Lethal dose or concentration | LD50 (oral, rat) 980 mg/kg |
| NIOSH | DH3675000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL (Recommended): 10g |
| IDLH (Immediate danger) | NIOSH: Unknown |
| Related compounds | |
| Related compounds |
Phenylacetyl bromide 4-Bromophenylacetic acid 4-Bromobenzyl bromide 4-Bromobenzoyl chloride Benzyl bromide |

