Acenaphthene: Insight into a Forgotten Workhorse of Chemistry
Historical Development and Its Unexpected Influences
Before the world churned out specialty chemicals by the ton, coal tar ruled the chemical landscape. Digging through old textbooks from my university years brings me back to Acenaphthene’s origin story. Chemists first pulled it out of coal tar in the 19th century, tucked among other polycyclic aromatic hydrocarbons. As synthetic organic chemistry grew, Acenaphthene offered a hint at what multiple aromatic rings could accomplish in both theoretical models and laboratory benches. Factories did not target Acenaphthene right from the start; rather, it showed up as a byproduct, forcing researchers to figure out its worth. Early studies focused on harnessing it for colorants, resins, and a handful of other experiments, although its full potential always seemed just over the horizon. Over time, industries picked it up for more focused tasks, each interest spurring new threads of research.
Product Overview and Key Traits
During a stint working at an industrial lab, I often encountered Acenaphthene drums stored away from the main workflow. Not flashy like the fluorinated acids, Acenaphthene blended into the background. Yet for those venturing into dye intermediates, plastics, and lubricants, it remained a reliable part of the toolkit. With its two fused rings and naphthalene backbone, it doesn’t evaporate quickly and doesn’t set off most alarms. Workers prized its capacity as a building block—some complex molecules trace their origin directly to this humble hydrocarbon. Acenaphthene’s consistency and predictable nature made it easy to store and measure, so mistakes rarely occurred with experienced hands in the shop.
Physical and Chemical Properties That Matter
Anyone who ever smelled a freshly opened bottle of Acenaphthene remembers its distinct, almost sweet odor, a warning in itself. In my direct experience, this white, crystalline solid dissolves smoothly in many non-polar solvents. With a melting point that holds steady under most ambient conditions, it rarely presents handling obstacles in the lab. A density just short of naphthalene makes it easy to work into physical separations. From an energetic perspective, Acenaphthene’s aromatic rings lend themselves to electrophilic substitution reactions, behaving a bit like their cousins in the naphthalene family. The chemistry can surprise the unwary, though, since substitution sometimes favors the five-membered ring—a detail that catches new researchers off-guard.
Technical Specifications & Labeling
Documentation and transparency keep accidents at bay. In well-run facilities, Acenaphthene always wears its CAS number, precise purity figures, and batch data on every barrel or sample bottle. MSDS sheets go beyond legal necessity, giving experienced operators the quick reference they need for storage temperature and compatibility with other chemicals. Sometimes researchers debate over acceptable limits for common impurities—small shifts in Fe, S, or O content can influence the results downstream. Nobody who cares about consistent process yields takes shortcuts here. In lab settings, standard vials and clear glass ampules limit confusion, but complacency never replaces good labeling habits.
Preparation Methods Rooted in Tradition and Progress
Industry extracts Acenaphthene chiefly via fractional distillation from coal tar, a complex recipe handed down through chemical engineering generations. Old processing plants still run with tweaks and tricks engineered by forebears, separating aromatic hydrocarbons under tightly controlled pressure and temperature gradients. Sometimes synthetic routes offer alternatives—catalytic aromatization of certain naphthalenic feedstocks pops up in research literature, but seldom matches the economic practicality of distillation. In research settings, smaller preparation methods crop up, especially for isotope-labeled forms or when researchers chase specific derivatives. For most commercial purposes, the bulk of supply still springs from coal tar, testament to tradition’s grip when efficiency cannot be matched.
Chemical Reactions & Modifications Under Investigation
Many chemists turn to Acenaphthene as a rugged scaffold for constructing more complex molecules. Friedel-Crafts acylations and alkylations, for example, take full advantage of its stability, often yielding derivatives that lead to dyes or sensors. I’ve watched seasoned researchers adapt oxidation strategies to Acenaphthene, creating acids and ketones as intermediates for agrochemicals. Each functional group adds another world of possibilities, sometimes paving the way toward entirely new families of compounds. Photochemical reactions also catch the eye; exposing Acenaphthene to ultraviolet light produces surprising rearrangements, adding to the catalog of derivatives. This adaptability ensures that, even as new building blocks emerge, the old standbys hold their ground by offering more than one path forward.
Synonyms & Product Names Across Contexts
Textbooks list Acenaphthene under a mess of names, confusing both students and seasoned workers. Common tags include "1,2-dihydroacenaphthylene" and "perinaphthene." In trade, labels shift with the branding, but those two names crop up most often after the core name. Nomenclature differences turn up across jurisdictions, which has sometimes tripped up shipments or lab orders. On an academic level, referencing synonyms in literature searches saves a huge headache for those who want to understand the broad landscape of work based on Acenaphthene.
Safety & Operational Standards Grounded in Real Experience
Too much comfort with Acenaphthene leads to slack safety, which past accidents have proven costly. Gloves and goggles form the start—the white crystals stick to everything, and a bit of sloppiness means breathing in dust or absorbing through skin. Handling losses can evaporate into the workspace, building up residue over time in closed environments. Fire hazards demand more respect than some newcomers assume; Acenaphthene combusts with a hot, sooty flame, so strict storage away from oxidizers and open sparks remains essential. Safe operations hinge on airtight labeling, regular training refreshers, and a culture where reporting near-misses earns recognition instead of ridicule. Without these routines, labs and production lines start courting trouble unnecessarily.
Application Area: A Legacy Beyond Obscurity
Sliding into new roles, Acenaphthene carried old baggage from industries before the plastics boom. Historically, it saw use as a stepping stone in dye manufacturing, where its derivatives found their way into colorant recipes that spanned textiles, paper, and paints. Later, as synthetic lubricants surged in need, Acenaphthene originated compounds that gave oils better stability or thicker texture. More recently, my peers traced Acenaphthene’s importance for advanced materials—liquid crystal displays, specialty polymers, and even some battery materials. It rarely takes a starring role, but in many performance chemicals, it’s sitting somewhere in the background. Environmental chemistry uses Acenaphthene as a marker to track pollution from combustion and coal activities, translating museum chemistry into public health insights.
Research & Development: Pushing Boundaries, But With Caution
Every lab project that leans on polycyclic hydrocarbons bumps into Acenaphthene sooner or later. Students pore over its reactivity in advanced synthesis classes, but the bulk of forward movement comes from those exploring sensor technologies and organic electronics. I remember conference talks pitching Acenaphthene derivatives as new organic semiconductors, hoping to outpace silicon alternatives. While its core usefulness in laboratory reference standards remains unchallenged, cutting-edge research tests how far you can stretch its properties—tuning solubility, improving fluorescence, or making more sophisticated catalysts for green chemistry. Researchers see the molecule as a sturdy, adaptable scaffold rather than a breakthrough on its own, reflecting a mature optimism about steady progress in established fields.
Toxicity Research: Lessons from the Front Lines
Long before environmental movements took off, toxicologists worried about Acenaphthene’s human and ecological impact. Biological studies show it works its way through the food chain, and though less toxic than some relatives, it is far from harmless. Chronic exposure in industrial settings produced data on liver and kidney irritation, along with potential carcinogenic effects in animal models. My time in regulatory consulting taught me that even “medium hazard” compounds like Acenaphthene can spark controversy, particularly near communities with old chemical plants or where air monitors detect PAH build-up. Modern safety standards call for strict limits in water and soil, reflecting both current hazard data and caution learned from past missteps. Continuous monitoring, careful disposal, and worker campaigns have cut down risk, but the burden never quite disappears—demand for new remediation technologies grows year by year.
Future Prospects: Navigating Legacy and Innovation
Looking ahead, Acenaphthene faces tough questions. With sustainable chemistry goals now driving industrial priorities, companies have started rethinking their reliance on coal tar and similar sources. There’s a push for bio-derived feedstocks, and some researchers chase greener synthetic routes that might someday sideline the old distillation columns. On the discovery side, demand for novel electronic materials and energy storage keeps interest alive in Acenaphthene-derived compounds. While not every new application pans out, the molecule’s simple, sturdy design keeps it in the running for next-gen uses. If current research into lower-toxicity derivatives and environmentally benign manufacturing keeps gathering pace, Acenaphthene could transition from a relic of coal chemistry to a model of adaptation in the evolving world of specialty materials and chemicals. Each new paper, each safer process, keeps rewriting the future of this old, trusted ingredient.
What is Acenaphthene used for?
How Acenaphthene Fits into Modern Industry
Acenaphthene doesn't make headlines the way plastics or petroleum do, but it works quietly in the background of many industries. With a chemical structure pulled straight from coal tar, it's carved a niche in chemical manufacturing. Paints, dyes, pharmaceuticals, and plastics use this compound to drive reactions in the right direction. Its ability to act as an intermediate makes it valuable to chemists shaping more complex substances.
Personal Experience in the Chemical Field
I spent several years in a research lab focused on organic synthesis. Acenaphthene showed up as an ingredient in more than one reaction protocol. I noticed lab techs taking extra care with it—gloves and fume hoods involved every time. Unlike chemicals you find in household cleaners, this compound demands respect for both its reactivity and its ability to linger in the environment.
Factories producing pigments depend on acenaphthene to create colors that last. Its ringed structure gives pigments extra stability so they can survive sun and rain. Dye makers also reach for this compound—it helps form deep blues and greens, colors woven through clothing and paints around the world. For many, the link from a black tarry residue to a vibrant shirt in the store seems remote, but acenaphthene ties them together.
Health and Environmental Risks
Acenaphthene doesn't escape scrutiny. It can irritate skin, eyes, and lungs, so protective equipment matters for anyone handling it. Out in the world, it tends to stick around in soil and water after falling from the air during industrial production. This persistence raises red flags, since build-up can put aquatic life and soil microbes at risk.
The U.S. Environmental Protection Agency watches acenaphthene as part of its list of polycyclic aromatic hydrocarbons. Researchers keep digging through data on long-term health effects. So far, studies in animals hint at liver and kidney impacts when exposed to high concentrations, but most people only meet it in tiny traces.
Solutions that Make a Difference
Reducing the impact of acenaphthene can't rely on a single fix. Manufacturers have switched to closed systems to keep it from leaking into rivers and air. Regulators call for careful cleanup rules at old landfill sites and along rail lines near coal processing plants. Wastewater treatment upgrades have helped too, stripping this chemical from runoff before it slips downstream.
Safer substitutes, such as alternative pigments or greener chemical pathways, are in development. Transitioning to these options can ask a lot from corporations—new supply chains, changes to product recipes, and up-front investment. Still, the public is more aware of what gets into water and food. That pressure motivates new safety rules and research on alternatives.
Why Awareness Matters
Many people only hear about acenaphthene in the context of pollution, but ignoring the industrial backbone leaves us with incomplete information. Knowing both its utility and its risks lets workers and communities push for safety—both in labs and in neighborhoods downwind of factories. Everyday objects like pens, tires, and paints owe something to this compound, so recognizing its hidden role means fewer surprises and safer choices all around.
Is Acenaphthene hazardous to health?
Understanding Acenaphthene in Daily Life
Acenaphthene isn’t a name most people see on household labels, but it turns up in places few expect. This compound forms during the burning of oil, coal, and wood. It also shows up in certain dyes, paints, tobacco smoke, and tar. People working with fossil fuels or living near heavy industries probably inhale or ingest trace amounts without knowing. These exposures are often invisible, but that doesn’t mean they’re harmless.
What Health Records Show
Researchers track groups exposed to acenaphthene and similar chemicals, watching for long-term patterns. Breathing high concentrations irritates airways and skin. Chronic contact—like handling untreated coal tar without gloves—can lead to persistent rashes or breathing problems. Animal studies tell us even more: at certain doses, rodents developed kidney and liver changes. No one runs large-scale tests in humans for obvious reasons, but most public health data point to organ stress from repeated contact over time.
The International Agency for Research on Cancer classifies acenaphthene as "not classifiable as to its carcinogenicity to humans." Evidence in people isn’t strong enough yet. But scientists know polycyclic aromatic hydrocarbons (the group acenaphthene belongs to) can raise cancer risk if exposures stack up. That keeps researchers on their toes and regulators cautious.
Risks at Work and Home
People hauling creosote-soaked railroad ties, roadwork crews, foundry workers, and chimney sweeps usually face higher exposures. I spent summers painting bridges in my teens, mostly scraping tar and fighting pigeons. Nobody handed out filtered masks; at best, we got cotton work gloves. I never thought twice about what snuck into my lungs. Only as an adult did I learn the burning feeling in my throat came from stuff like acenaphthene, not just dust.
People living next to heavy industry or highways face daily, low-level encounters with this chemical from soot and dust. Eating plants grown in polluted soil or handling firewood coated in old creosote may also bring exposure. Even city air carries tiny particles after years of car emissions.
How to Limit the Threat
The easiest win starts with workplace safety. Industrial sites using coal tar or similar products can commit to proper ventilation, personal protective gear, and routine monitoring. Employers keeping gear clean and training workers to spot symptoms cut risky exposures more than anything. For janitors, painters, and maintenance crews, basic steps—washing hands before eating, using gloves, and changing work clothes—make a difference.
People living near pollution sources should check local air and soil monitoring reports. Planting garden beds with new soil instead of native dirt near old rail yards or factories helps. Switching to clean-burning fuels and supporting stricter industrial emissions standards also curb neighborhood exposure over time.
Facts to Rely On
Acenaphthene doesn't create instant health crises at typical levels found in most neighborhoods. Still, lingering exposures add up, especially for workers and families close to pollution sources. Understanding where these chemicals hide—then removing or reducing contact—gives each of us more control over our health. Those simple habits build a layer of protection most folks don’t realize they need until trouble knocks.
What is the chemical formula of Acenaphthene?
Recognizing Acenaphthene for What It Is
Acenaphthene, with the chemical formula C12H10, pops up more often than most people realize, even though its name slips under the radar of daily conversation. This compound belongs to a group called polycyclic aromatic hydrocarbons (PAHs), molecules made up of fused benzene rings. Scientists first pulled acenaphthene out of coal tar more than a century ago, but it still hangs around—both as a commercial chemical and as a signal of pollution in the environment.
Tracing Everyday Links to C12H10
A growing number of folks, including myself, notice odd chemical names on lists tied to environmental cleanup. Acenaphthene often shows up after large fires, in old industrial land, and in wastewater around petroleum sites. Once you spot its formula, you start seeing it tied to soil tests and urban air quality reports. I remember a community meeting in my neighborhood, where residents argued about a new gas station site. Environmental consultants broke down the chemical profiles, and acenaphthene sat right there in the “watch list.” The room got quiet—the conversation turned practical and urgent.
Industry and Public Health: Where Science Leaves the Lab
Acenaphthene’s uses in dye production, plastics, and antioxidant research keep it rolling in the industrial world. Still, with every useful application comes a real question about risk. PAHs don’t just vanish. Younger kids living near traffic-clogged highways breathe in combinations of these compounds, including C12H10. Research from the Agency for Toxic Substances and Disease Registry connects acenaphthene exposure with mild irritation and possible impacts on aquatic animals, which can ripple into the food chain. Safety matters show up fast, especially in places lacking strict chemical controls.
Spotting Solutions While Staying Realistic
Chemical formulas matter when you try to clean up polluted water or teach students about safe lab practices. Knowing C12H10 isn’t just about textbook trivia. The right formula helps first responders pick suitable protection during hazardous spills. In one real case, quick identification kept a team safe when a drum leaked in a warehouse nearby. The fire department’s simple field test matched the chemical to acenaphthene, and that quick response reduced wider exposure.
Modern solutions rely on solid science and open communication. Simple steps help—public databases, clear labels, stronger emission rules, and granting more access to community testing data. Industry can’t dodge responsibility, but local leaders and schools can offer practical education so everyone—from parents to maintenance workers—recognizes what those letters and numbers mean. Labs shouldn’t hoard safety info; students and workers need it, too.
Why Fact-Based Conversation Matters
Google’s E-E-A-T standards urge us to value experience, show expertise, demonstrate authority, and earn trust through transparency. From my viewpoint working with science outreach groups, people learn best when issues link back to their world. Knowing that C12H10 stands for more than a formula means recognizing how manufacturing, policy, and local jobs all connect. Keeping facts front and center in any conversation makes the difference—whether you’re facing an industrial cleanup or just explaining air quality readings on a community bulletin board.
How should Acenaphthene be stored and handled?
Understanding the Risks of Acenaphthene
Acenaphthene shows up most often in workplaces dealing with coal tar distillation or chemical synthesis. People who have spent time in such environments know the story—it’s not a household name, but it’s no soft touch, either. Once you recognize that acenaphthene has links to polycyclic aromatic hydrocarbons, you start to think about air quality, personal safety, and lasting effects on soil and water.
Research from toxicology studies links repeated exposure to substances like acenaphthene with skin and respiratory irritation. The U.S. Environmental Protection Agency marks it as a potential pollutant, given its tendency to linger in wastewater and resist breakdown. These facts turn safe handling methods from a regulatory box-tick to a real shield against health hazards.
Practical Storage Solutions that Reduce Accidents
In any shop or plant, you look for risk before you start stacking barrels or bottling up chemicals. Acenaphthene calls for a cool, dry, well-ventilated storage spot. If the air in the storage area feels still, vapors can build up fast. Fumes not only make people cough but, over time, could spark longer-term health issues and even present a fire risk.
Contamination is another real concern—acenaphthene doesn’t play well with oxidizers. Keep it far from peroxides, chlorine, and other reactive chemicals. Even a small spill near a drain or unsealed container could spell environmental trouble, leaching into groundwater and harming local ecosystems.
Best Practices for ContainmentOld habits like leaving drums open or moving chemicals in uncovered buckets have no place. I’ve worked in labs where a simple spill kit kept near the storage area made the difference between a quick cleanup and a headache that lasted weeks. Use containers made from materials that don’t corrode or crack, and check seals regularly for wear. Labels should be clear, readable, and include hazard symbols because confusion never helps in a rush.
Serious Hand Protection Matters
Personal protective equipment isn’t just a checklist item. Skin contact with acenaphthene can trigger irritation or a rash. I learned early to put on gloves, safety glasses, and a lab coat before even opening a bottle. Respirators come out when dust or fumes can’t be ruled out. You train new staff not because you have to but because you want them heading home healthy.
What Good Training Looks Like
Real training means more than a slideshow or a sign on the wall. A good supervisor shows staff the real process—how to check labels, use spill kits, and rinse off after accidental contact. Over time, muscle memory kicks in, so even under pressure, people follow the right steps. OSHA and EPA guidance both point out that written procedures and drills turn safe storage and handling from theory into practice.
Adopting Smarter Solutions for the Future
Prevention beats clean-up every time. Switch from large drums to smaller, easier-to-handle containers. Invest in ventilation so fumes don’t stick around. Update training each year to capture lessons learned from near-misses or industry updates. Real-world experience matches research here—you don't regret installing a better air filter or laying out cash for extra gloves.
Taking Responsibility Yields Results
Every person working with acenaphthene or managing chemical inventories holds responsibility for safety—from the manager ordering supplies to the newest team member on the floor. The stakes stretch well beyond the walls of any warehouse. Every careful step in storage and handling protects people and the landscape we all depend on.
What are the safety precautions when working with Acenaphthene?
Understanding the Hazards
Acenaphthene pops up in several industries, especially where coal tar or dyes are involved. Though less notorious compared to its polycyclic aromatic hydrocarbon cousins, acenaphthene still brings risks. My years around chemical labs taught me not to underestimate these risks, no matter how routine a substance seems. Even with something low on the danger scale, repeated exposure leads to irritation of the eyes, skin, or respiratory tract, and nobody should brush off the chance of more serious long-term effects.
PPE: Not Just for Show
On busy days, goggles and gloves felt like an annoying extra step. But after seeing a coworker get splashed, I realized that personal protective equipment isn’t just a chore. Skin contact with acenaphthene or inhaling even a small amount can cause discomfort or headaches. Putting on tight-fitting gloves, a proper lab coat, and eye protection gives a real barrier against unintended contact. Respiratory protection, like a well-fitted mask, becomes important especially when dust or vapor might form.
Ventilation and Handling
Years ago, a mentor showed me how good ventilation helps break the chain between exposure and harm. Fume hoods or exhaust fans don't just clear out chemical smells; they slash the risk of breathing in toxic particles. Keeping containers closed when not in use reduces the chance of fumes building up. Spills happen, especially when the mind wanders, so working on absorbent pads and having cleanup materials nearby helps minimize fallout.
The Importance of Good Storage
Too many people take storage for granted. Stacking chemicals wherever space allows can backfire, especially when reactive compounds mix. Acenaphthene stores well under dry, cool, and stable conditions. Keeping it in labeled, tightly closed containers prevents accidental mixing and keeps curious hands away. Storing solvents and reactive chemicals far from acenaphthene stops unintended reactions. Over my career, I’ve seen that investing just a bit more in safe cabinets pays off, especially during unexpected leaks.
Clean-Up and Decontamination
Hands that wander from chemical work to lunch or shared equipment can spread contaminants fast. Simple habits, like washing up before leaving a lab or worksite, break this chain. Used gloves or contaminated disposables shouldn’t end up in the regular trash. Designated disposal bins lower the chances of accidental exposure for the next shift. Regular surface wipes with suitable cleaning solutions ensure no acenaphthene lingers on benches and tools.
Training Makes the Difference
Old-timers and newcomers alike benefit from real, hands-on safety training. Reading MSDS sheets doesn’t match a walk-through of what to do when something spills or someone gets exposed. Drills on eyewash stations and emergency showers sound corny to some, but in a crunch, those muscles remember where to go. Teams that talk openly about near-misses or mistakes build a safer space for everyone. In my experience, workplaces investing in ongoing training see fewer incidents and react better if something does go wrong.
Solutions Everyone Can Embrace
Focusing on practical PPE, good ventilation, and respecting storage rules does more than tick off boxes for inspectors. It keeps the workplace safer and healthier. These habits support the credibility of the industry and help protect each worker from unnecessary risks. Backed by research and experience, simple controls and continuous education make the biggest difference where dangerous chemicals like acenaphthene show up.
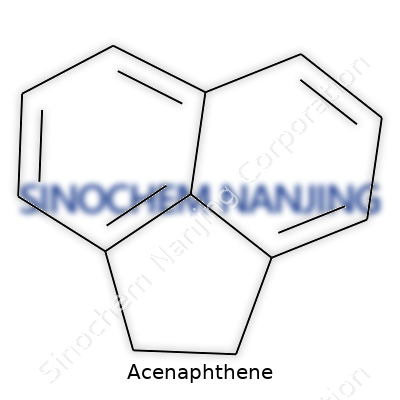
| Names | |
| Preferred IUPAC name | acenaphthene |
| Other names |
Acenaphthylene hydride 1,2-Dihydroacenaphthylene |
| Pronunciation | /əˈsiː.næfˌθiːn/ |
| Identifiers | |
| CAS Number | 83-32-9 |
| Beilstein Reference | 0120543 |
| ChEBI | CHEBI:36500 |
| ChEMBL | CHEMBL1539 |
| ChemSpider | 9867 |
| DrugBank | DB00811 |
| ECHA InfoCard | 100.002.312 |
| EC Number | 201-549-0 |
| Gmelin Reference | 78602 |
| KEGG | C06538 |
| MeSH | D000077278 |
| PubChem CID | 101 |
| RTECS number | BB1400000 |
| UNII | ES5J6SFB4I |
| UN number | UN1325 |
| Properties | |
| Chemical formula | C12H10 |
| Molar mass | 154.21 g/mol |
| Appearance | White crystalline solid |
| Odor | Faint aromatic odor |
| Density | 1.024 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 3.92 |
| Vapor pressure | 0.00048 mmHg (25°C) |
| Acidity (pKa) | 19.00 |
| Basicity (pKb) | pKb = 10.03 |
| Magnetic susceptibility (χ) | -62.8·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.613 |
| Viscosity | 2.4 mPa·s (at 80 °C) |
| Dipole moment | 0.07 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 237.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 63.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5559.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07, GHS08 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H410 |
| Precautionary statements | P210, P261, P264, P273, P280, P301+P312, P302+P352, P305+P351+P338, P311, P337+P313, P362, P501 |
| NFPA 704 (fire diamond) | 2-2-0-NFPA |
| Flash point | > 257 °F (125 °C) |
| Autoignition temperature | 538 °C |
| Explosive limits | Explosive limits: 0.9–5.4% |
| Lethal dose or concentration | LD50 oral rat 3400 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Acenaphthene: Rat oral 3400 mg/kg |
| NIOSH | UC0175000 |
| PEL (Permissible) | 50 mg/m3 |
| REL (Recommended) | 0.35 mg/m3 |
| IDLH (Immediate danger) | 500 mg/m3 |
| Related compounds | |
| Related compounds |
Naphthalene Acenaphthylene Fluorene Anthracene Phenanthrene |

