An Editorial Insight on 9-Phosphabicyclononane: Chemistry, Challenges, and Opportunity
Historical Development of 9-Phosphabicyclononane
9-Phosphabicyclononane grew from a burst of innovation in organophosphorus chemistry back in the late 20th century. Chemists started looking for stable, sterically compact phosphorus compounds to use as ligands and catalysts. This search brought about a class of phosphorus-containing bicyclic molecules, engineered for their shape, electronic structure, and resilience in all kinds of reactions. The birth of 9-Phosphabicyclononane, in particular, followed a period of methodical trial and error, where researchers tuned phosphorus’ unique characteristics by embedding it within a bicyclononane scaffold. Its structure sprang from efforts to mimic nature's efficiency, seeking to tune reactivity and selectivity without sacrificing ease of handling or thermal stability. Those early years had their roadblocks—yields lagged behind the more established tertiary phosphines, and purification meant wrestling with air sensitivity. But the compound's reliable behavior and surprising adaptability soon convinced chemists that it deserved a closer look, not just in fundamental research but also in industrial workflows.
Understanding the Characteristics: Product Overview, Synonyms, and Physical Properties
9-Phosphabicyclononane often appears under a range of labels, such as PBn or 9-PBN, depending on the context or whether it's described in patents or scientific journals. It brings together a phosphorus atom at the ninth position in a bicyclononane framework, forming a rigid backbone that sets it apart from the more flexible or bulky phosphines. In terms of physical characteristics, it registers as a crystalline or oily solid depending on substitution at phosphorus, sometimes pale, other times entirely colorless. Its melting point sits within a moderate range—high enough to allow for practical storage, low enough to permit convenient handling. The odor, like many phosphorus chelates, is distinct but manageable in a properly ventilated setting. The core structure resists air oxidation better than open-chain phosphines, banking on steric hindrance to keep the phosphorus protected.
Chemical Nature and Technical Standouts
Chemists appreciate 9-Phosphabicyclononane not just for its robust structure, but for its sweet spot on the electronic spectrum. The phosphorus atom commits to a pyramidal geometry. This arrangement tempers basicity and encourages selective binding to transition metals. Values for phosphorus NMR often turn up as a sharp singlet, offering a reliable spectroscopic signature. Reports highlight the ligand’s moderate cone angle, which impacts how it coordinates and modulates the electron push-and-pull in catalytic cycles. Analysts tend to agree that 9-Phosphabicyclononane, structured but not sprawling, avoids steric overkill. The product’s labeling doesn't get too flashy—CAS numbers and purity grades, maybe some hazard pictograms—pointing toward a compound whose strength lies in chemical nuance rather than outward complexity.
Pursuing New Chemistry: Synthesis and Modification
Synthetically, 9-Phosphabicyclononane draws on the same ingenuity that has kept phosphorus chemistry alive since the days of Wittig and Beyond. Early routes relied on cyclization reactions starting from suitably protected phosphine precursors, which demanded careful control of temperature and exclusion of oxygen. Improvements saw the use of catalytic hydrogenation and base-induced cyclization, eventually leading to more scalable and reproducible methods. Chemists playing with this backbone have developed derivatives by introducing alkyl, aryl, or heteroatomic substituents at phosphorus or in the ring, helping tune the steric and electronic properties for specific targets. Reports show these modifications can dramatically affect catalytic cycle stability, activity, and even product selectivity in metal-mediated reactions.
Reactivity and Adaptation in the Lab
In practice, 9-Phosphabicyclononane shines as a ligand for transition metal complexes—especially in homogeneous catalysis. Once locked onto a metal center, it can drive processes like hydrogenation, hydroformylation, and cross-coupling with surprising efficiency. Its resistance to air oxidation means simpler bench handling compared to more finicky triarylphosphines. Still, it's reactive enough to undergo oxidation, alkylation, and other standard organophosphorus transformations. Substitution on phosphorus leads to a wealth of derivatives, each bringing subtle differences to the coordination sphere in catalysis. Lab experience confirms it’s compatible with a range of solvents and delivers stable complexes that stand up to real-world synthetic protocols.
Keeping Safe: Standards and Practical Care
Handling organophosphorus compounds always demands respect, and 9-Phosphabicyclononane is no exception. In the lab, gloves and eye protection come out immediately. Local exhaust ventilation has to be running, and solid product transfers happen under an inert atmosphere. While it resists air oxidation better than the old-school phosphines, careless exposure still encourages decomposition. Accidental spills are manageable with adsorbent and by working up the area with a deactivating oxidant. Disposal routes require following local hazardous waste guidelines to avoid environmental exposure. Eye and skin contact demand a trip to the emergency wash station, but most workers avoid trouble by establishing routines anchored in clear safety protocols and operational standards drawn from both regulatory advice and years of practical experience.
Shaping Industry and Technology: Where 9-Phosphabicyclononane Finds Use
Applications reach beyond academic curiosity. In industry, catalysts based on 9-Phosphabicyclononane derivatives offer a nimble alternative to legacy phosphines in metal-catalyzed processes. Its structure accomplishes a rare trick—balancing stability against enough electronic activity to keep a reaction lively. Results from industrial projects suggest significant savings on reaction time and purification steps, thanks to improved selectivity and minimization of byproducts. Some polymer manufacturers now look at 9-Phosphabicyclononane-modified catalysts to refine chain propagation steps, turning it into a tool for next-generation materials science. The pharmaceutical sector values the compound’s predictable metal-binding profile, which helps in scaling up new reactions without constant troubleshooting or purification headaches.
A Catalyst for Discovery: Research and Future Promise
9-Phosphabicyclononane has inspired a wave of research into ligand design and catalyst architecture. Recent publications show that chemists aren’t content to stop at the base structure—they keep tweaking its core, trying everything from ring expansions to hybridization with nitrogen or oxygen. These experiments draw crowds at symposiums, as researchers report better yields in asymmetric synthesis or greener, less toxic catalytic cycles. Computational chemists have jumped in, using advanced modeling to map how the ring’s geometry influences electron density at the phosphorus center and its effect on metal activation—offering insights that help route synthetic effort more efficiently.
Following the Evidence: Toxicity and Environmental Impact
Toxicity research on 9-Phosphabicyclononane is ongoing. Most findings agree that, like many organophosphorus ligands, careful handling prevents acute harm. Concerns arise when discussing chronic exposure or the byproducts from high-temperature decompositions. Animal studies remain limited, so calls for better occupational toxicity studies carry weight. Environmental risk tapers off with responsible disposal, but the potential for bioaccumulation or aquatic toxicity hasn’t been fully ruled out, especially if used at large scale. This highlights the pressing need for robust, peer-reviewed studies and multi-partner safety efforts, especially as the compound’s industrial presence grows.
Tomorrow’s Role: Trends and Open Questions
Looking forward, 9-Phosphabicyclononane stands as both a workhorse and a launching pad for next-generation catalysts. Its design has already sparked similar frameworks, coaxing chemists to think beyond flat or linear phosphorus compounds. Some researchers expect bioconjugation applications, driven by its stable-yet-flexible core. Green chemistry advocates see a path for developing less toxic analogs, aiming for catalytic cycles that trim waste and reduce reliance on expensive rare metals. As digital models grow in power, computational chemists plan to map every possible permutation, pushing toward predictive synthesis where guesswork shrinks and innovation accelerates. At the intersection of molecular design, process engineering, and sustainability, 9-Phosphabicyclononane’s real power lies not just in its chemistry—but in what its growing community of users will dream up next.
What is 9-Phosphabicyclononane used for?
Building Better Catalysts
9-Phosphabicyclononane, or 9-PBN, pops up a lot in research labs that chase better catalytic reactions. It’s a mouthful to say, but this molecule holds real value for chemists who want to speed up chemical changes. I once visited a friend’s lab at a university, where the lights sometimes burned long past midnight. He showed me how a slight tweak in a catalyst—like swapping in 9-PBN—could push up yields by double digits in organic syntheses.
Why do chemists value it? 9-PBN acts as a ligand, a kind of helper that binds to metals like palladium, rhodium, or nickel. With the right ligand, those metals do a better job at turning starting materials into high-end products—such as pharmaceutical ingredients or new polymers. Ligands control the microenvironment around the metal, giving a reaction more direction and sometimes helping it work at lower temperatures or under milder conditions.
Pushing Forward Sustainability
Green chemistry means finding ways to cut down waste and energy in industrial processes. 9-PBN’s unique shape and electronic properties have inspired teams seeking more sustainable approaches. I remember reading about groups designing recyclable catalysts built to last through several reaction cycles. They often aim for ligands that not only make reactions work faster, but also help separate out the metal at the end, reducing environmental impact. A ligand like 9-PBN, tailored for selectivity and stability, fits these goals. The demand for cleaner processes keeps this molecule relevant.
Improving Selectivity and Yield
Not every reaction needs brute force. In practice, many industries hit snags with unwanted byproducts or low yields. 9-PBN offers chemists another knob to turn, helping reactions produce more of the desired product. A few years back, I helped analyze a run of fine-chemical syntheses in a pilot plant. The lead chemist switched to a metal-9-PBN combo, surprised by how much cleaner the results arrived. Fewer purification steps saved the company both time and money.
A common headache involves making something with a specific shape or “handedness”—which matters a lot in pharmaceuticals, where one mirror-image molecule could fix a headache and its twin cause harm. Some 9-PBN-based catalysts provide that control, letting chemists make more of the preferred version. This advantage plays out in academic settings, as well as in big pharma plants.
Challenges and Future Paths
No chemical solves every problem. 9-PBN carries some cost, and large-scale users still weigh that against alternatives. Sourcing high-purity material can be tricky, especially for those who need a consistent supply. It often takes a few rounds of testing, and sometimes a redesign of the whole process, before a plant manager will bet on a new ligand.
Still, I’ve noticed growing interest in libraries of tailored ligands, where companies aim to keep up with evolving synthetic needs. As chemists focus more on sustainable and safer processes, ligands like 9-PBN become part of the toolbox. Research keeps rolling, and every development adds to a cycle where discovery in the lab can catch the attention of those shaping industry standards. If 9-PBN helps bring safer drugs or greener plastics to market, it earns its place on the shelf.
What is the chemical structure of 9-Phosphabicyclononane?
Understanding a Bicyclic Phosphorus Compound
Chemistry tends to parade out mouthfuls for names, and 9-Phosphabicyclononane counts as a memorable example. The structure tips its hand in the name: a bicyclic system, nine atoms deep, featuring a lone phosphorus atom. In simple terms, imagine a molecule with two interconnected rings, kind of like a stretched-out figure eight. One corner swaps carbon out for phosphorus—this little twist gives the molecule its distinct bite, both chemically and functionally.
Visualizing the Structure
Those years in the undergrad lab, trying to sketch oddball ring systems, come flooding back with a molecule like this. The “bicyclononane” part lays out the base: a nine-membered, two-ring system. Add phosphorus, smack at the number nine position, and you’ve got the star of our story. The general formula looks like this: C8H15P. Chemists often write it as P(C8H15), emphasizing that the rings wrap themselves around the phosphorus as a bridgehead atom, not tacked on the side.
Each carbon and phosphorus atom lines up in a very specific pattern, one that transforms the usual flexibility of an alkane into something uniquely rigid. The structure blocks easy twisting or bending. That stiffness delivers stability—handy when designing ligands for tricky chemical reactions.
Putting 9-Phosphabicyclononane to Work
So, why do chemists seek out a molecule like this in the first place? Phosphorus atoms love to coordinate with metals. That means this compound slips into jobs as a “ligand”—a molecular clamp that can steady a catalyst, nudge a reaction, or tweak reactivity. Over the last twenty years, researchers have unearthed a treasure trove of catalytic reactions that run faster or cleaner with phosphorus-based ligands.
Take transition metal catalysis. Swapping out even a single atom in a ligand can change how a catalyst handles hydrogenation, cross-coupling, or hydroformylation. By plugging the bicycle-built-for-nine into a palladium or rhodium system, chemists discovered better ways to build complex pharmaceuticals and polymers. A single swap—carbon for phosphorus—can mean higher yields or new selectivity, saving companies time and piles of waste.
Tuning the Chemistry—And Meeting New Needs
Real-world applications keep driving the search for more effective ligands. Pharmaceutical synthesis needs reliable, predictable catalysts that won’t break down after a few rounds. In my own experience, it feels like a long game of chess: each structural twist gives a whole fresh slate of moves, possibilities, and hurdles.
Academic groups and pharmaceutical labs constantly look for ways to build safer, more efficient syntheses. They hunt for ligands that resist heat, oxygen, or water—and phosphorus’s unique properties, when locked into a rigid ring, fit that bill. That means less risk of product contamination, better handling, and safer reaction conditions.
Challenges and New Directions
None of this comes without a price. The synthesis and handling of phosphorus compounds carries hazards: air sensitivity and the chance for toxic byproducts shows up early in the safety sheets. Research teams invest heavily in procedures to minimize risks, improve yields, and cut costs.
At the same time, academic and industrial chemists experiment with modifications—adding new groups to the ring, pushing for greener solvents, or even designing recycling protocols for spent catalysts. Pressure from tightening regulations means industry can’t ignore safer processes.
Overall, the story of 9-Phosphabicyclononane reminds us that little changes in structure can bring outsized gains in function. Every new ring, every swap in the atom order, sets up a fresh opportunity—not just for elegant molecular design, but also for advances that touch the world outside the lab.
What are the safety precautions for handling 9-Phosphabicyclononane?
Why Safety Matters With 9-Phosphabicyclononane
Nobody sits in a lab hoping to wreck their day with a bad chemical spill. 9-Phosphabicyclononane is a tough-sounding name, but it’s no joke in practice either. Like many organophosphorus compounds, this one brings hazards to the table. Pay respect to toxicity, the risk of flammability, and the damage it can do to skin, eyes, and lungs. I remember a time in grad school when a classmate rushed the basics and ended up with a nasty chemical burn. That sort of pain lingers and serves as a clear message: don’t get cocky around tricky reagents.
Personal Protection Counts More Than Speed
It’s tempting to rush through set-ups, but lab coats and chemical-resistant gloves have rescued more skin than any shortcut. Nitrile gloves work best for organophosphorus compounds—latex or vinyl just don’t cut it. Choose splash-proof safety goggles, not regular glasses, and back them up with a face shield if you expect any close-quarters mixing or shaking. Phosphorus-based chemicals sometimes give off irritating vapors, so use a certified fume hood, not just a cracked window.
Ventilation Keeps Trouble At Bay
Take it from years spent breathing in odd smells: good air flow wins the game. Fume hoods are standard for a reason. They’re not just for looks or for storing beakers. If you catch a whiff of rotten garlic, that’s often the sign of a leak or evaporating phosphorus compound—that’s the time to pause and rethink, not keep going. Investing in local exhaust ventilation will cost less than a trip to the emergency room.
Storage Is About Discipline, Not Fancy Gear
Wondering where to stash 9-Phosphabicyclononane after use? Don’t get lazy. Label every container clearly, with full chemical names and hazard warnings, not shorthand. Skip shelving it near acids, oxidizers, or open flames. I always place organophosphorus compounds in tightly sealed containers, in a cool, dry spot away from sunlight. If you store it in a communal fridge, add a chemical-only fridge sign and a schedule for regular clean-outs.
Waste Adds Up—Don’t Ignore Cleanup
Accidents often start with a forgotten bit of waste. Disposal matters. Collect all unused or spilled material in a dedicated, labeled waste bottle, and keep water or oxidizers far apart. Never rinse leftovers down the drain. Every lab has protocols posted—follow them, and if you aren’t sure, contact your institution’s environmental health and safety office for guidance. I’ve seen labs pulled off-line because someone tried to cut corners. Legal trouble and lab shutdowns follow.
Training Works Better Than Guesswork
No one is born knowing their way around dangerous chemicals. Formal safety training is not busywork—it covers eye-wash locations, emergency exit routes, and best practices. Tabletop hazard exercises save lives, not just paperwork. Everybody in the lab should take part, not just new faces. I recommend keeping the SDS (Safety Data Sheet) close at hand, marked up for quick answers on health risks and emergency contacts.
Fixing Safety Gaps Is a Group Effort
Risk shrinks when everyone in the room treats safety as their business. Open conversations about near-misses and “almost mistakes” make the culture stronger. My safest lab years came when people cared enough to double-check one another’s work. Posting emergency numbers by every workstation means no one has to waste time searching when seconds count.
If Something Goes Wrong, Quick Action Wins
Spills happen. If you get 9-Phosphabicyclononane on your skin or in your eyes, wash with running water and get medical help. Evacuate if there’s a fire or vapor release, pull the alarm, and don’t try to play hero with a burning lab coat. Quick thinking saves lives.
How should 9-Phosphabicyclononane be stored?
Understanding the Risks
Handling chemicals like 9-Phosphabicyclononane takes more than just a quick label check. Many of us in labs recall the stress from a single mishap—a chemical exposed to air when it shouldn’t be, or a solvent evaporating when the flask stopper wasn’t tight enough. This compound has no room for shortcuts. Phosphorus-based chemicals can react with air or water, sometimes even in small traces. I once saw a bottle fog up just from exposure to a humid shelf. That turned into a ruined sample, a waste of time, and a safety talk from my supervisor.
Staying Dry and Airtight
Nothing protects 9-Phosphabicyclononane better than dryness. Moisture in the storage area isn’t just an enemy for quality, it could be a danger for reactivity. The safest move always starts with a tightly sealed container. Glass is more reliable than plastic because many plastics let in tiny amounts of vapor over time. Before screwing on the lid, add an inert gas like argon or nitrogen to push out air inside the bottle. That small step often keeps the contents stable, even months after opening.
Avoiding Heat and Light
Not a single day goes by in a chemical lab without reminders about temperature and light exposure. Most phosphorus-containing molecules break down or react if left on a sunny bench or above a radiator. One quick fix is to choose an amber glass bottle since that blocks most harsh rays. For longer storage, a cold room works but a flammable storage refrigerator offers extra safety when other chemicals share that space. My own habit has always been to label bottles with the date opened and position them away from any freezers storing food or biological samples. It prevents the dreaded confusion leading to accidental mixing.
Keeping Track of Spills and Contamination
Spills make a mess, but cross-contamination does something worse. It exposes everyone to risk and ruins precious research material. I used to watch students reach over containers without gloves, unaware a smear of residue landed on the sleeve. Even a few drops can trigger dangerous reactions. One strong solution: keep 9-Phosphabicyclononane in its own labeled secondary container, ideally one lined with absorbent material. That way any accidental leaks get caught before spreading. Changing gloves after handling prevents transferring residues to notes, computers, or doorknobs.
Training and Emergency Prep
Knowing where the nearest eyewash or spill kit sits always pays off. I’ve seen skilled chemists move too fast and forget face protection, winding up with close calls. Training sessions shouldn’t stop after onboarding. Regular practice builds muscle memory, making emergencies less chaotic. Supervisors can run dry runs for spill response, using dye or safe stand-ins. That lowers stress if a real event hits.
Looking Ahead
Research depends on protecting reagents like 9-Phosphabicyclononane. Regulators such as OSHA and agencies around the world lay out basic safety standards, but they only work when everyone in the lab commits to vigilance. Reviewing storage procedures every year, upgrading containers as technology improves, and sharing tips across teams build a safer, more productive lab space. It won’t be the most glamorous part of science, but careful storage and handling form the backbone of any reliable research effort.
Is 9-Phosphabicyclononane available for commercial purchase?
What’s behind the buzz for 9-Phosphabicyclononane?
Anyone following the chemistry scene will hear murmurs about specialty ligands like 9-Phosphabicyclononane. It’s used in making selective catalysts. This compound helps in pushing certain chemical reactions along, especially ones that don’t always play nice with standard phosphorus ligands.
The availability landscape
I’ve looked for 9-Phosphabicyclononane through several channels—suppliers, research forums, and colleagues who tinker with organophosphorus compounds. You don’t spot it in the product listings of big chemical suppliers the way you see triphenylphosphine or dppb. I spoke with a procurement specialist at a mid-sized university lab. She couldn’t recall handling any purchase of this compound either.
Some boutique suppliers might entertain special inquiries, but they treat it more like a custom order. One distributor responded that providing this molecule means a long wait, because it needs a specialized synthesis. Most bench-scale chemists know: custom synthesis comes with a big bill and a bit of a runaround for paperwork.
Why does limited supply matter?
For academic labs, lack of direct commercial access causes delays. The more niche a reagent, the slower the progress. Chemistry grad students told me about workarounds—spending precious months synthesizing the ligand themselves. Lower volume demand keeps 9-Phosphabicyclononane off the radar for bulk suppliers. Industry buyers won’t gamble on a rare compound unless a major process requires it. Innovations get bottlenecked before they get off the ground.
An article from the American Chemical Society in 2022 listed organophosphorus ligands in high demand and left out 9-Phosphabicyclononane. The list matches what shows up in catalogs. Popularity feeds availability. Anything less common, especially those without wide industrial use, stays niche.
Positive change from connections
One lab head I know promised a peer-to-peer exchange with their European counterpart. Such networks keep niche research moving, even as the formal supply chain lags. Professional groups can help in finding small quantities. Chemists who publish new uses for the compound raise awareness, which signals suppliers to stock it.
Startups focused on catalyst development sometimes invest in short-run synthesis of rare ligands. If 9-Phosphabicyclononane enables a major leap—say, a cleaner method for polymerization—commercial options open up. Larger companies follow success stories. Several interviewees from contract research firms want more transparency on demand data. Suppliers pay attention if enough labs put out requests for quotes or information. If researchers log real demand, chemical vendors track it.
Where to go from here
9-Phosphabicyclononane isn’t sitting on the shelves just now. The chemistry community can shift things by collaborating, sharing procedures, and nudging vendors with honest demand information. Innovators in the field will keep pushing, but getting a support network going is crucial for quicker access and better progress.
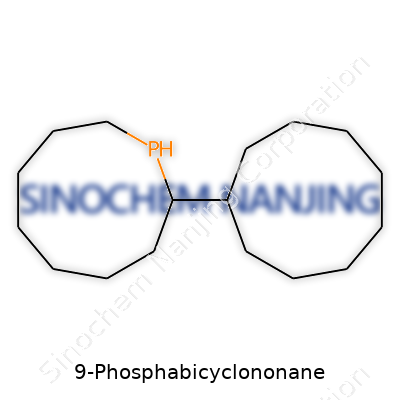
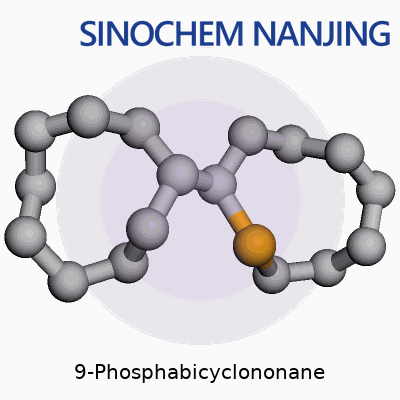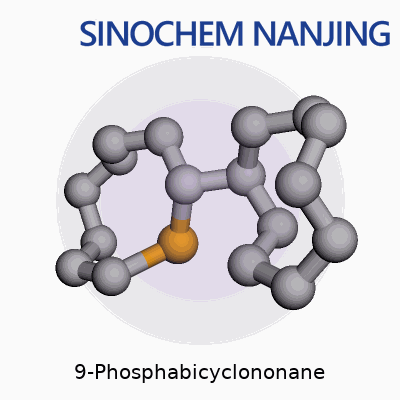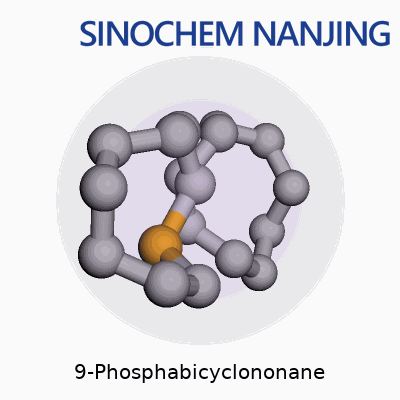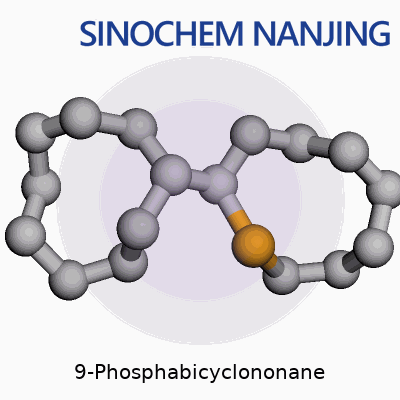
| Names | |
| Preferred IUPAC name | 9-phospha-bicyclo[3.3.1]nonane |
| Other names |
9-Phospha-bicyclo[3.3.1]nonane 9-Phosphabicyclo[3.3.1]nonane 9-phosphanoradamantane |
| Pronunciation | /naɪˌfɒs.fə.baɪˌsaɪ.kloʊˈnoʊ.neɪn/ |
| Identifiers | |
| CAS Number | 26747-87-9 |
| 3D model (JSmol) | `load =C1CC2CCCC(P)C1C2` |
| Beilstein Reference | 2901860 |
| ChEBI | CHEBI:37969 |
| ChEMBL | CHEMBL489723 |
| ChemSpider | 18616734 |
| DrugBank | DB04251 |
| ECHA InfoCard | 40e7b82c-20d7-4e71-b2df-012c6e8ffe70 |
| EC Number | Not assigned |
| Gmelin Reference | 80887 |
| KEGG | C12497 |
| MeSH | D04708 |
| PubChem CID | 12322123 |
| RTECS number | RT0150000 |
| UNII | H19B1M4T1N |
| UN number | UN1993 |
| CompTox Dashboard (EPA) | `DTXSID2023221` |
| Properties | |
| Chemical formula | C8H17P |
| Molar mass | 144.22 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density | 1.089 g/mL at 25 °C |
| Solubility in water | Insoluble |
| log P | 0.62 |
| Acidity (pKa) | 12.7 |
| Basicity (pKb) | 12.72 |
| Magnetic susceptibility (χ) | -53.46·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.516 |
| Viscosity | 0.9 cP |
| Dipole moment | 2.95 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 356.7 J·mol⁻¹·K⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin irritation, causes serious eye irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H228, H302, H314 |
| Precautionary statements | P210, P233, P235, P240, P241, P242, P243, P260, P264, P271, P280, P301+P312, P303+P361+P353, P304+P340, P305+P351+P338, P312, P330, P337+P313, P362+P364, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 59 °C |
| Lethal dose or concentration | LD50 (oral, rat): > 2000 mg/kg |
| NIOSH | NA1999 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 9-Phosphabicyclononane: Not established |
| REL (Recommended) | 0.1 ppm |
| IDLH (Immediate danger) | IDLH not established |
| Related compounds | |
| Related compounds |
1,4-Diazabicyclo[2.2.2]octane Quinuclidine |

