Exploring 8-Methylquinoline: A Deep Dive into Science and Use
Understanding the Journey: The History Behind 8-Methylquinoline
Curiosity about chemicals often leads to interesting discoveries, and 8-Methylquinoline, sometimes called quinaldine, carries its own backstory. Late 19th-century chemists first isolated methylated quinoline compounds during the booming age of coal tar exploration. Researchers weren’t just chasing new formulas; they wanted practical solutions to problems in dyes, medicine, and more. As petrochemistry advanced, laboratories shifted from natural extraction to synthesized routes. Tools became more precise, pathways clearer, and methylquinolines gained attention both as curiosities and as answers to growing industrial needs. The chemical story weaves through an era when basic research meant hands-on experimentation, lacking today’s analytical shortcuts. That grounds modern understanding in a hard-earned scientific tradition.
Getting to Know 8-Methylquinoline: Key Characteristics
8-Methylquinoline falls into the heterocyclic aromatic family—an arrangement known for combining carbon and nitrogen in a stable, ring-shaped structure. Unlike its parent quinoline, this molecule holds a methyl group attached to the eighth position on the ring, which changes how it behaves. The colorless to pale yellow liquid gives off a distinctive odor, often compared to earthy or tar-like notes, something hard to ignore in any lab. Its melting point usually sits just above room temperature, with boiling peaking above two hundred degrees Celsius. This molecule dissolves well in many organic solvents, while showing little interest in water. Chemically, it remains stubborn against mild acids and bases, yet responds strongly to certain oxidation and alkylation processes.
Technical Details: How Labels Reflect What’s Inside
Examining a bottle of 8-Methylquinoline, clear communication matters just as much as chemical purity. Packaging labels must report the CAS number—100-54-9—as a common reference point. In practice, reputable suppliers print purity percentages, batch dating, and detailed storage advice on every container. Some industries apply even stricter standards, setting max limits for impurities down to the parts-per-million level. Tightly controlled processes ensure users know exactly what's entering their synthesis line or research bench. Those details support everything from quality consistency to safe handling, giving professionals the confidence to pursue new applications without second-guessing the basic identity or reliability of their material.
The Making of 8-Methylquinoline: Synthesis and Transformation
Preparation often follows traditional quinoline chemistry. Skilled chemists favor methods like the Skraup reaction, tweaking starting materials and conditions to nudge methyl groups into the right spot. Selective methylation, using reagents such as methyl iodide on precursor compounds, speeds up the process. Over the decades, continuous-flow reactors replaced batch flasks in big factories, shrinking waste and risk. Downstream, chemical modifications start with halogenation or acylation, offering a springboard for building more elaborate molecules used in everything from drug discovery to specialty polymers. Each step demands careful balancing of yield, purity, and cost, since shortcuts can introduce hard-to-remove byproducts. That places practical know-how above textbook knowledge, especially for large-scale runs.
Other Names in the Mix: Synonyms and Trademarks
Chemists and industry professionals call 8-Methylquinoline by different names, depending on context or field. You’ll see quinaldine most often, but systematic registries stick with 8-Methylquinoline or 2-Methylquinoline based on IUPAC rules. Don’t let the mix of names cause confusion; they all point to the same structure. Across old research articles or international trade documents, this mix of nomenclature reflects chemistry’s global reach and evolving standards. For anyone new to the topic, a quick check of structural diagrams helps cut through the jargon and see that all roads lead to a single molecule.
Stay Safe: Handling Standards and Health Considerations
Personal experience in the chemistry lab reminds me that safety protocols make the difference between routine work and costly accidents. 8-Methylquinoline, though not among the most notorious hazards, still poses health and environmental risks. Prolonged skin contact or inhalation of vapors can irritate eyes, respiratory passages, or skin. Most labs require gloves, goggles, and diligent ventilation when pouring or transferring this compound. Storing it away from strong acids, bases, and open flames avoids unwanted reactions. Waste disposal must meet local environmental laws, since improper disposal could lead to lasting soil or water contamination. No matter the setting—university, research center, or factory floor—people benefit from safety briefings and clear instructions. Workers familiar with emergency eyewash stations and spill procedures respond quickly if something goes wrong, keeping minor incidents from turning major.
Making It Useful: Applications Across Sectors
8-Methylquinoline plays a role in more sectors than many realize. Dye manufacturers value quinaldine as a starting material for colorants used in textiles and inks. Agrochemical companies rely on its structure for pesticide intermediates, aiming to boost crop yields or battle persistent pests. Pharmaceutical researchers eye this compound as a launch pad for antimalarial or antitubercular drug prototypes. In many cases, the molecule itself doesn’t end up in the final medicine or product—it serves as a stepping stone, offering unique reactivity thanks to its nitrogen ring and methyl group. More recently, materials scientists incorporate molecules like this in organic electronics, pushing into areas like OLED displays or solar panels. That broad slate of uses means 8-Methylquinoline lives far beyond the laboratory supply store, impacting things we encounter day to day.
Research Directions: What’s Being Explored Now
In the world of research and development, 8-Methylquinoline remains relevant for both practical use and academic study. Investigators probe new catalytic reactions where this molecule acts as a ligand or a building block. Synthetic chemists explore greener methods for making methylquinolines, swapping older, less environmentally friendly reagents for catalysts that waste less and pollute less. Biochemists examine how modifications to the quinoline core change biological effects, hunting for more effective or less toxic drug candidates. Beyond pharmaceuticals, engineers examine photochemical properties, looking to improve the performance of next-generation electronic screens or light sensors. These efforts often involve collaborations bridging organic chemistry, physics, and engineering, showing once again how a single compound can pull together many branches of science towards a common goal.
Learning About Safety: Toxicity and Exposure Studies
Toxicity research draws from a mix of animal tests, cellular studies, and epidemiological observations. 8-Methylquinoline hasn’t grabbed headlines, largely because typical exposures remain low in modern settings, but that doesn’t mean ongoing vigilance should slacken. Studies indicate that high doses, especially over long periods, can harm the liver or nervous system in animal models. Irritation at the site of contact, along with possible sensitization, crops up in concentrated exposures. Experts set safe workplace limits by weighing these findings, often updating recommendations as new results come in. That kind of work can’t run on assumptions; each application—from drug chemistry to materials science—calls for a fresh look at potential outcomes, informed by honest data over wishful thinking.
Looking Ahead: Where 8-Methylquinoline Might Go Next
Attention turns to sustainability, efficiency, and health as industries think about the future. Green chemistry approaches promise less waste and improved energy use in producing 8-Methylquinoline, with research groups racing to design catalysts that cut down on toxic byproducts or tedious purifications. In applications, the push for new pharmaceuticals, advanced dyes, and electronic components puts pressure on the supply chain, encouraging scaled-up production with strict quality controls. As regulations tighten around chemical use and exposure, users will rely on clearer data about ecological impact and long-term safety. Collaboration between academic labs and manufacturers keeps innovation flowing, and funding agencies increasingly look for projects that combine commercial promise with environmental awareness. No single chemical solves every problem, but following the trajectory of compounds like 8-Methylquinoline shows how small molecules continue to shape science, technology, and everyday life.
What is 8-Methylquinoline used for?
A Closer Look at 8-Methylquinoline
8-Methylquinoline might sound unfamiliar to most, but for those spending time in laboratories or behind the scenes in manufacturing, it’s almost a household name. With decades of industrial and chemical research under its belt, this compound steps up as more than just a chemical curiosity. It’s one of those simple molecules that finds a place to shine in a bunch of fields.
Key Uses Across Industries
Pharmaceutical developers see 8-Methylquinoline as a solid building block. Medicinal chemists use it to create new compounds, especially when working on anti-malarial drugs or exploring treatments for infections. Synthetic steps that involve quinoline structures often start or end with this variant, thanks to the methyl group hanging off its eighth carbon. That little tweak changes how molecules fit together or react. As a result, 8-Methylquinoline sets itself apart in constructing drugs and research molecules targeting complex diseases.
This compound walks into dye and pigment labs, too. Some of the most vibrant and stable dyes trace their roots back to quinoline derivatives. Textile experts constantly look for shades that hold up under sun, sweat, and repeated washes. 8-Methylquinoline offers stability and color-fastness chemists appreciate. This matters more than ever, since customers expect their favorite t-shirts and fabrics to last.
Why Chemists Prefer This Compound
Safety standards in chemical manufacturing keep getting tighter. 8-Methylquinoline, when handled with respect, checks safety boxes compared to other quinoline variants. Users like me have noticed it resists breaking down under normal conditions. Bottles stored in a well-labeled cabinet years ago still do their job as expected. That shelf-stability saves time and cuts down on waste, a big plus from both a business and environmental angle.
The scent isn’t everyone’s favorite—some describe it as “peculiar”—but that hasn’t stopped researchers from coming back to it. Companies chase efficiency, and 8-Methylquinoline usually offers predictable results in multi-step syntheses. Reactions that have ruined weekends with mystery byproducts run with fewer headaches when this compound shows up as a starting piece or intermediate.
Environmental and Safety Concerns
Any time a chemical sees widespread use, some tough questions surface. 8-Methylquinoline isn’t different. Disposing of quinoline-based compounds takes care. Improper handling can harm waterways or air quality. My own years in industrial quality assurance taught me that collecting, neutralizing, and safely destroying residues forms as important a part of the job as making new substances.
Global agencies haven’t labeled 8-Methylquinoline as the worst offender, but best practices call for tight controls and proper gear. Gloves, goggles, and fume hoods all play a role in keeping work environments safe. In my own lab days, labeling and logs kept accidental mishaps rare.
Room for Improvement and Sustainable Growth
We need greener production routes for quinoline derivatives. Teams working on catalysis and waste reduction push for lower-impact processes using renewable resources. Some researchers convert biomass directly into key molecules, bypassing harsher chemistries that dominated the last century. Others tap into water or ethanol-based solvents instead of older, toxic options.
Pharmaceuticals, dyes, and other sectors will keep leaning on known molecules until green chemistry catches up. For now, 8-Methylquinoline stands as a practical compromise: functional, stable, and familiar. Scientists keep pressing for improvements. Cleaner methods, stronger safety rules, and recycling initiatives help this compound fit into a future where performance meets responsibility.
What is the chemical formula of 8-Methylquinoline?
Digging Into 8-Methylquinoline
Chemistry rarely makes headlines, but the story behind a compound like 8-Methylquinoline deserves attention. This molecule goes by the chemical formula C10H9N. Scientists spot a parent structure: quinoline, that double-ring system you find in dyes, pharmaceuticals, and even pesticides. Add a methyl group at the eighth carbon atom, and suddenly you’ve got a different chemical personality—one that shifts the compound’s behavior in synthesis labs and in daily manufacturing.
Down-to-Earth Relevance
I spent enough time hunched behind a chemical hood during university to know why chemical formulas matter. They don’t just describe; they guide choices. Get that formula right—C10H9N—and a chemist knows exactly what kind of beast they’re dealing with. It impacts everything from handling safety to reaction partners. In research circles, mishandling the formula on a bottle means wasted time, skewed data, and sometimes a mess on the lab floor. Multiply that by thousands of labs and chemical companies, and you start to see the scale.
Why Accuracy Counts
Let’s face it—mistakes in formulas mess things up quickly. A methyl group seems tiny, but it tweaks boiling points, alters toxicity, and can influence color in key reactions. Industries depend on these small details for efficiency and safety. The wrong formula creeping into supply chains means pharmaceuticals lose reliability, manufacturing gets expensive, and researchers risk health and funding. That’s not alarmist. The U.S. Environmental Protection Agency makes a point of tracking compounds even more obscure than this, just to keep people safe and products up to scratch.
How Does 8-Methylquinoline Get Used?
This isn’t just a classroom molecule. 8-Methylquinoline pops up in materials science, dye production, and sometimes in drug synthesis. Its structure lets it slip into bigger molecules as a building block. Companies crave efficiency, so a formula that tells them exactly what’s in the beaker saves time, cuts costs, and helps with regulatory reporting. I’ve spoken with manufacturing engineers who value the details, not just because it keeps production humming, but because it guards against regulatory fines and product recalls.
Solving the Communication Gap
One problem is old. People mistake similar-sounding chemicals for each other. Just a single number off—a misplaced methyl or ethyl—and suddenly the properties change. I remember a story about a technician using the wrong quinoline variant. It cost the lab extra hours and introduced contamination in a batch meant for medicine research. The solution starts at the basics. Teach formula literacy in academic labs. Push chemical suppliers to stick tight to international standards. Digital inventory systems ought to flag near-match names and prompt verification before shipping or use.
Keeping It Real
Clear communication around formulae like C10H9N keeps labs safe and businesses productive. As research grows more global and supply chains longer, the details matter more than ever. Chemicals don’t have to be mysterious. They just demand respect—and a close look at the little numbers and letters that describe them.
What safety precautions should be taken when handling 8-Methylquinoline?
Understanding the Risks
8-Methylquinoline isn’t a common household item. As someone who's spent years in chemical research, I can say it has its place in specialty lab work and some industrial processes. Its low flash point means it can catch fire even below room temperature. The vapors irritate your nose and throat, and if you breathe it in regularly, it can start to wear you down fast. So, anyone working with this chemical needs to respect the risks from day one.
Choosing the Right Gear
No one serious about lab safety walks into a room with 8-Methylquinoline wearing shorts and open shoes. Gloves made from nitrile keep you protected—latex hasn’t held up in solvent work from my trials. Safety goggles are a must; you do not want any splashes near your eyes. Where the work could splash or spill, I trust a chemical-resistant apron or lab coat more than thin scrubs. A good face shield helps if mixing or pouring, since even a small drop can turn into a big problem.
Ventilation Matters
Fume hoods make the difference between a safe routine and a visit to occupational health. Standard ventilation doesn’t cut it; you need airflow designed for organic solvents. I once watched a colleague try to shortcut by using a regular fan near an open beaker. Within minutes, the lab filled with sharp fumes and everyone had to evacuate. After that, fume hoods became non-negotiable, no exceptions.
Storage and Handling
8-Methylquinoline stays stable in a cool, dry container. It can react with acids and oxidizers, so it never sits next to bleach, nitric acid, or strong cleaning agents in our chemical cabinet. All bottles carry clear hazard labels. Sealed glass or high-density polypropylene containers work; never trust makeshift packaging from another solvent or food product, no matter how tight the cap seems.
What Happens in an Emergency
Spills don’t have to turn into disasters. My team always has absorbent pads and neutralizing agents like sand and sodium bicarbonate ready to go. For skin contact, a thorough rinse at the eyewash station is more effective than trying to wipe it off—long sleeves won’t help if you skip immediate washing. Fire safety calls for a CO₂ extinguisher. Regular water won’t put out chemical fires and could spread the spill.
Protecting People New to the Lab
Not everyone who handles 8-Methylquinoline has a chemistry degree. Training newcomers means more than handing over a manual. I show them where the nearest eyewash and shower are, walk through mock drills, and make them practice donning and doffing PPE until it’s second nature. I’ve seen well-written safety sheets ignored during crunch time; hands-on instruction sticks much better, especially for students or techs just starting out.
Building a Safety Culture
Organizations do best when people support each other. I learned after a lab incident that reporting near-misses and discussing what went wrong (without blame) helps everyone avoid future trouble. Regular audits and gear checks become routine, not an afterthought. If you see someone cutting corners, stopping and helping them make it right pays off more than hoping nothing goes wrong.
Improving Lab Safety Industry-Wide
Clear labeling with QR codes for instant-access safety data, widely available personal protective equipment, and ongoing education keep labs safer for everyone. Open communication between labs and regulatory agencies leads to better guidelines—ones grounded in real-world experience, not just theory. Every year, sharing stories and lessons learned sharpens how the whole field handles hazardous materials, making everyone’s work not just productive, but much safer.
How should 8-Methylquinoline be stored?
Understanding What You’re Handling
8-Methylquinoline doesn’t show up in everyday life unless you work with chemicals or research materials. My own introduction came during a stint in a paint formulations lab, where this compound’s sharp, musty odor made it instantly memorable and not terribly pleasant. Recognizing that it is both flammable and toxic, nobody wants to mess up storage—human Health depends on good habits, not luck. It only takes one careless moment with volatile organics to turn a safe bench into a hazard zone.
Storage Basics: Simple Steps, Serious Stakes
Storing this compound calls for a dedicated, airtight glass container. Plastic reacts, metal corroded. Once, I saw a tech use an old soda bottle. After a week, the cap cracked; fumes filled the cupboard. Using proper vessels is not about following a rulebook. It’s about avoiding headaches and protecting your health.
Chemicals like 8-Methylquinoline break down or become extra dangerous when left in sunlight or a humid, warm spot. Heat speeds up reactions; moisture can corrode, especially with traces of acid often left in lab air. Every label or safety data sheet calls for a cool, dry place, out of direct light. A simple steel cabinet lined with chemical-absorbing mats kept my lab safe for years. A regular fridge doesn’t work—it belongs in a dedicated flammables fridge, with spark-proof switches and tight seals. Fire inspectors and insurance adjusters check for this, and nobody enjoys writing incident reports late at night.
Labeling Prevents Chaos
I learned the hard way that ignoring good labeling fuels mistakes. Not long ago, I visited a workshop cluttered with jars, most marked in faded Sharpie or not at all. It’s tempting to cut corners; it feels faster. Yet accidents rarely come from one blatant act, but a tumble of little lapses. Every container ought to carry the full chemical name, date received, hazard warnings, and the contact for the responsible person. It sounds fussy. It’s actually just common sense.
Ventilation and Handling: Respect the Fumes
Fumes creep up and linger. They don’t care if you’re a rookie or a veteran. Use proper ventilation, even with the lid closed most of the time. Chemical fume hoods cost money, true, but you only have one set of lungs. Absorbent pads near storage minimize risk from drips and leaks. Regularly inspect and swap out anything that starts looking grimy or soaked—it beats dealing with an insurance claim after an unnoticed spill.
Emergency Prep: Setting Expectations
No one plans to have a spill, but being unprepared makes a mess worse. Store absorbent material, goggles, gloves, and a basic spill kit nearby—not ten rooms away. Post emergency numbers on the cabinet. A simple quarterly walk-through of your lab or storage room will remind you to check expiration dates and catch early signs of wear on containers. Old habits slip under pressure; checklists help keep things real.
Why All This Actually Matters
In a shared workplace, protecting each other isn’t about bureaucracy, but respect and realism. Every chemical tells a story. How you store it says a lot about your standards and your priorities. Diligence isn’t dramatic, but it keeps people out of hospitals, workplaces open, and research running. That’s where real value shows up—every day, without headlines.
What are the physical properties of 8-Methylquinoline?
Getting to Know This Compound
8-Methylquinoline lands on the chemist’s radar for more than just its name. It’s a derivative of quinoline, marked by a single methyl group sitting on the eighth carbon. That subtle tweak throws off a handful of its physical traits, which matter every time a researcher weighs out a sample or a manufacturer decides which solvent to use. The stuff looks like pale yellow crystals, not all that different from table salt at a glance. But breathe near it, and you sense a strong, pungent odor—hard to ignore in any workspace.
Appearance Tells Its Own Story
Every bottle of 8-methylquinoline in a storeroom glimmers slightly under the lights. The way it forms in crystalline shapes gives you a hint about how its molecules stack together. Chemists use melting point as a quick test for purity, and this one has a melting point of about 66°C (151°F). So it doesn’t need much heat before turning into a liquid. That trait makes for easier handling during synthesis, but also demands a stable storage space. If the room gets hot, you end up with a sludgy mess instead of neat crystals.
Solubility and Why It Matters
Anyone who’s worked with organic compounds knows that how a substance dissolves shapes the whole research process. 8-Methylquinoline shows solid solubility in organic solvents like alcohols and ethers. Try tossing it in water, and it quickly shows its apathy for H2O—hardly budging, and stubbornly clumping to itself. This tells you something about its structure: a mostly nonpolar surface, making it fit better with other nonpolar molecules than with polar ones like water. Picking the right solvent isn’t a trivial decision in a pharmaceutical or agrochemical lab, and 8-methylquinoline’s solubility profile narrows those choices.
Boiling Point Shapes Its Use
Boiling points are far from random trivia. With 8-methylquinoline, that number lands at around 253°C (487°F). This means it hangs around as a liquid for a pretty wide range of temperatures, but once it gets hot enough, watch for vapors. Ventilation becomes crucial in bigger processes. If you’ve ever worked in scale-up production, you know that controlling vapor is necessary both for safety and quality. It also influences how the compound can be purified—distillation works, but watch the thermometer closely.
Density and Handling
Density rarely gets the headlines, but it determines how you package and move a compound. 8-Methylquinoline packs in at about 1.07 g/cm3 at room temperature. So it’s a bit denser than water, but not by much. Pouring and measuring are straightforward, but always keep its volatility in mind. Spills may turn into fumes before you know it, especially under warm conditions.
What Stands Out for Safety?
No one wants to take a whiff of 8-methylquinoline longer than needed. Its odor signals some risk—vapors can irritate your airways, and skin contact isn’t harmless. Any lab handling this stuff should keep extra nitrile gloves and goggles close at hand. A fume hood isn’t just optional; it’s urgent for anyone working with it beyond small drops. Fact: data from the National Center for Biotechnology Information shows that inhalation and skin exposure can trigger acute symptoms, so safety protocols aren’t just red tape.
Practical Steps Forward
8-Methylquinoline’s physical properties dictate everything from storage to safe disposal. More research on greener, safer solvents could reshape how this compound is used, especially if regulations start clamping down on volatile organics. Switching to closed-system synthesis, deploying better ventilation, and using real-time monitoring for vapors would make workplaces safer. These steps don’t just tick boxes—they build trust for anyone handling the compound, from students in teaching labs to industrial chemists running overnight reactions.
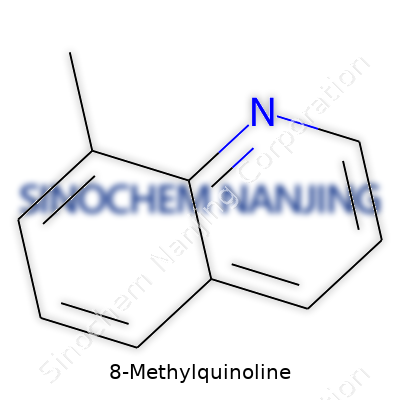
| Names | |
| Preferred IUPAC name | 8-Methylquinoline |
| Other names |
8-Quinolinol 8-Methyl-1-aza-naphthalene 8-Methylquinoline Lepidine |
| Pronunciation | /ˌeɪtˌmɛθɪlkwɪˈnəʊliːn/ |
| Identifiers | |
| CAS Number | 1008-72-6 |
| Beilstein Reference | 120918 |
| ChEBI | CHEBI:34558 |
| ChEMBL | CHEMBL16022 |
| ChemSpider | 15438 |
| DrugBank | DB02444 |
| ECHA InfoCard | DTXSID0522057 |
| EC Number | 202-807-2 |
| Gmelin Reference | 678139 |
| KEGG | C14803 |
| MeSH | D000437 |
| PubChem CID | 7006 |
| RTECS number | GN8575000 |
| UNII | PI1O9D94ZL |
| UN number | UN2656 |
| Properties | |
| Chemical formula | C10H9N |
| Molar mass | 129.18 g/mol |
| Appearance | Yellowish liquid |
| Odor | strong amine |
| Density | 1.023 g/cm3 |
| Solubility in water | slightly soluble |
| log P | 2.7 |
| Vapor pressure | 0.09 mmHg (25°C) |
| Acidity (pKa) | 5.14 |
| Basicity (pKb) | 9.76 |
| Magnetic susceptibility (χ) | -70.7·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.735 |
| Viscosity | 2.24 mPa·s (at 20 °C) |
| Dipole moment | 1.74 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 182.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 92.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4867 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P337+P313, P332+P313, P362+P364 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | 113°C |
| Autoignition temperature | 535 °C |
| Explosive limits | Explosive limits: 1.1–7.1% |
| Lethal dose or concentration | LD50 (oral, rat): 740 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 670 mg/kg |
| NIOSH | SN 2100000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.1 |
| Related compounds | |
| Related compounds |
Quinoline 2-Methylquinoline 4-Methylquinoline 6-Methylquinoline 7-Methylquinoline 8-Hydroxyquinoline Lepidine Quinaldine |

