Digging Deeper Into 8-Methylisoquinoline: From Synthetic Curiosity to Real-World Use
Historical Development
Organic chemists have always looked at the isoquinoline family for new possibilities. 8-Methylisoquinoline started showing up in research in the early half of the 20th century as scientists chased after medicinally active alkaloids. Isoquinoline itself made waves thanks to its role in plant biochemistry—think poppy alkaloids, which set the stage for new discoveries. The addition of a methyl group to the eighth position of the isoquinoline backbone may sound like a minor lab trick, but it opened a pathway that expanded the toolkit for chemical synthesis. Once breakthrough methods—especially palladium-catalyzed reactions—became accessible, the compound left the realm of academic curiosity. Its relevance kept growing as researchers realized this small molecule could serve as both a test subject in theoretical studies and as a core scaffold for more complex drug candidates.
Product Overview: What is 8-Methylisoquinoline?
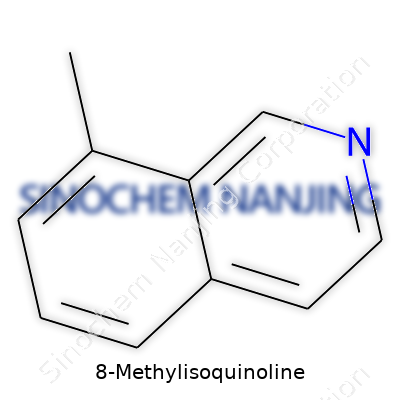
8-Methylisoquinoline is a heterocyclic compound—basically, its structure forms a double-ring system where nitrogen and carbon atoms alternately take their place. What sets this molecule apart is the methyl substituent stitched to the eighth position on the isoquinoline ring. That subtle tweak brings about distinct reactivity and offers new handles for chemists aiming to design building blocks for pharmaceuticals and advanced materials. This compound usually appears as a colorless or pale yellow solid, and in a typical lab, you’d spot it stored away from light and moisture to prevent unwanted decomposition or impurities.
Physical and Chemical Properties: Why the Methyl Group Matters
Chemically, 8-Methylisoquinoline comes with the formula C10H9N. It doesn’t dissolve well in water, but it takes to organic solvents like ether and chloroform. Melting point measurements tend to hover around 60–70°C, though small differences show up depending on purity and how the molecule’s been handled. The methyl group changes the electronic environment, which means the ring’s reactivity shifts compared to straight isoquinoline—something synthetic chemists can exploit. Infrared and nuclear magnetic resonance spectra look different for 8-Methylisoquinoline, providing clear signatures that identify it in a mixture.
Technical Specifications and Labeling in Real Laboratories
Stock bottles on university or industrial shelves don’t just list the name; they show CAS registry numbers and labels warning of possible hazards. The real world demands accurate information—purity above 97 percent, storage below 30°C in dry conditions, and clarity about lingering contaminants. Labels matter just as much as the powder inside, since a misidentified bottle leads to botched experiments or worse. On the safety sheet, expect warnings about skin and eye irritation, flammability, and rough guidelines for handling under a fume hood. Chemists and lab techs care deeply about these details because the work depends on trust in every gram weighed out.
Preparation Method: How Chemists Actually Make It
Making 8-Methylisoquinoline often starts with constructing the isoquinoline ring through a Bischler–Napieralski cyclization or similar strategies, after which methylation steps using reagents like methyl iodide fine-tune the molecule. Sometimes, labs take a shortcut and start with a methylated benzylamine and run the nitrogen through a cyclization process. None of these routes are beginner territory—the steps demand a steady hand, solid glassware, and keen attention to reaction conditions, especially to keep byproducts in check. Yield optimization becomes a balancing act between temperature, time, and choice of solvent. The process may sound routine in print, but it requires years of honed skill to avoid wasted batches or hazardous side reactions.
Chemical Reactions and Modifications: Tuning the Molecule
The true value of 8-Methylisoquinoline shines in its reactivity. That methyl group pulls electrons in just the right way to steer subsequent reactions. Chemists often use it as a building block, tacking on more complex groups at either the nitrogen atom or aromatic rings. N-alkylation, cross-coupling, or even oxidative reactions become more predictable because the methyl group directs activity. In pharmaceutical development, that means new analogs and derivatives keep coming, each with potentially different biological effects compared to the parent isoquinoline. Research often chases after selectivity, aiming the molecule at one enzyme or receptor over another—a task only achieved by smart chemical modification.
Synonyms and Product Names: More Than One Name in the Database
Getting confused by chemical names comes with the territory. 8-Methylisoquinoline can pop up as 8-Methyl-1-azaphenanthrene in old literature. Catalogs and databases recognize both, and even minor errors in naming can trip up literature searches or ordering processes. Synonyms matter especially as regulations differ—knowing every alternate name ensures compliance and helps avoid duplicate purchases or mismatched stock.
Safety and Operational Standards: In the Lab and Beyond
No one gets to treat lab chemicals casually, and 8-Methylisoquinoline proves no different. Research into its toxicity shows skin and mucous membrane irritation risks. Inhalation of fine powders or fumes brings headaches or respiratory issues, and accidental ingestion leads to far bigger problems. Protective gloves, sealed containers, and well-ventilated workspaces serve as the everyday armor for chemists dealing with the compound. Even disposal requires planning—wasted quantities can’t go into a sink, but must follow strict waste guidelines to avoid environmental spillover. Audits, regular training, and up-to-date safety data keep risk in check. These standards protect not just the scientist, but anyone downstream in waste management or environmental stewardship.
Application Area: From Research Bench to Industry
8-Methylisoquinoline stakes its claim in medicinal chemistry, agrochemical development, and advanced organic synthesis. Drug hunters use its skeleton to mimic biological alkaloids, often looking for anti-inflammatory, anti-cancer, or anti-infective properties. Industrial players see value in its ability to serve as an intermediate for specialty dyes, pigments, or even certain polymers. Analytical chemistry sometimes draws on its distinct spectral signature as a marker molecule in complex mixtures. This flexibility means its importance will grow with each new research breakthrough that links structure to function.
Research and Development: Keeping the Ideas Moving
R&D teams keep finding new angles on this compound. Over the last decade, researchers examined how 8-methyl substitution affects binding to target proteins, opening up entire families of potential therapeutics that didn’t show up using plain isoquinoline. The compound now features in studies on signal transduction and neurochemistry, as well as catalysis. Each published study tends to draw more attention, spurring work on new derivatives, eco-friendlier synthesis routes, or analytics methods that can spot trace levels in biological samples. These innovations rarely unfold in isolation—progress in catalysts, analytical equipment, or even computational chemistry reshapes how scientists test and validate 8-Methylisoquinoline’s potential.
Toxicity Research: Risks and the Push for Safer Science
Toxicology rarely wins headlines, but it’s a constant behind-the-scenes force. Reports in the literature describe mild to moderate toxicity upon ingestion or inhalation. The compound can disturb normal enzyme activity, which sounds small until you factor in chronic exposure risks in industrial settings. Regulatory work chases after these findings, tightening workplace exposure limits and pushing for real-time monitoring in research and manufacturing plants. Some teams look for functional alternatives or tweak the structure further to keep the usefulness while dialing down the hazards. This rich dataset keeps safety front and center, making sure that curiosity doesn’t sideline worker health.
Future Prospects: More Than Just a Building Block
Advances in computational chemistry and biotechnological synthesis may soon make 8-Methylisoquinoline cheaper, purer, and more accessible. As science marches into targeted drug design and green chemistry, this molecule sits primed for expanded use—whether in custom pharmaceuticals, novel materials, or high-sensitivity detection systems. AI-driven retrosynthesis predictions aim to cut down waste and byproducts, bringing sustainable methods within reach. Environmental monitoring and trace detection could soon rely on its distinct chemical footprint. The next lap depends on persistent research, solid regulatory support, and a community of chemists who keep challenging assumptions about what small molecules can do.
What is 8-Methylisoquinoline used for?
The Chemistry Behind the Compound
8-Methylisoquinoline isn’t a name you hear every day, but it holds a special place for chemists. As a derivative of isoquinoline, it features in various organic synthesis paths. In my early years working in a university laboratory, compounds like this often came up, mostly during exploratory phases of new pharmaceutical projects. Scientists have looked at its molecular structure and seen potential for tweaking its chemical backbone to build more complex molecules.
Role in Drug Discovery
Pharmaceutical research runs on building blocks like 8-Methylisoquinoline. Researchers use it to create new molecules that might treat disease. For example, the core isoquinoline ring can be found in several medicines with anti-inflammatory or anti-cancer actions. Chemical companies supply it mainly for this kind of work: designing analogs, screening them against biological targets, trying to figure out which tweaks make a difference in the lab and, possibly, in a hospital someday.
The jump from a chemical scaffold to a working therapy takes years—often decades. Over that span, researchers rely on compounds like this one to map out what changes matter at the molecular level. PubMed and chemical patent databases list hundreds of studies each year on isoquinoline derivatives. A handful use 8-Methylisoquinoline as a starting point to produce new candidates for antibiotics or nervous system drugs. In the bigger picture, these are seeds for tomorrow’s treatments.
Specialty Chemistry and Industrial Applications
Not everything spins around medicine. Chemical manufacturers turn to 8-Methylisoquinoline as an intermediate—a necessary step on the way to other, sometimes much more complex substances, including dyes, solvents, and even agricultural chemicals. Its methyl group gives it unique reactivity, making it a valued input for chemical transformations that would be out of reach using simpler isoquinoline.
Some people might wonder how it makes a difference. Precision matters. Tiny tweaks, like adding a methyl group in the right spot, can change both the practical use and safety of downstream products. In industrial synthesis, reliability trumps flashiness. Here, 8-Methylisoquinoline doesn’t draw a crowd, but for certain steps, nothing else fits as well.
Challenges and Solutions
Safe use always matters. Compounds like 8-Methylisoquinoline demand responsible handling—good ventilation, protective equipment, and strict protocols. In my time around chemical stockrooms, even a rarely used vial comes with its own set of rules. Researchers constantly balance between pushing boundaries and keeping safety front-and-center.
Waste management becomes a real concern with specialty chemicals. Adherence to environmental guidelines already challenges many research teams. Transparent tracking, updated safety data sheets, and wider access to disposal information help mitigate risks. Comprehensive education—covering not just acute hazards but also chronic exposure—remains essential.
Where Curiosity Leads
Science rewards bold questions. 8-Methylisoquinoline illustrates how many small molecules wait in the wings, shaping bigger discoveries. Its everyday uses in labs and factories might not make headlines, but they keep the stream of innovation flowing. Global research teams draw on the same catalog of building blocks—sometimes the simplest ones, like 8-Methylisoquinoline, show the way toward new solutions and a better understanding of the chemical world.
What is the chemical formula of 8-Methylisoquinoline?
Chemistry Meets Daily Curiosity
Most folks don’t wake up thinking about isoquinoline derivatives, but chemical formulas sit at the center of everything from pharmaceutical research to the flavors in food. Take 8-Methylisoquinoline as a case. The formula for this compound is C10H9N, which carries a specific arrangement—a ring structure with a methyl group attached at the eighth position.
Why 8-Methylisoquinoline Stands Out
Building a drug or designing new materials often relies on tweaking these small organic molecules. That methyl group matters. Swap its position and you suddenly get different interactions with enzymes, different shelf lives, and sometimes a completely new function. Chemists learn this fast in the lab: minor differences in structure can push a compound from being useless to something that could wind up in a new medication or specialty chemical.
Having worked with heterocyclic compounds in a college lab, I remember the tedious steps needed to keep impurities away when synthesizing these molecules. A single carbon out of place and the stuff wouldn’t give the right shade under UV—an early sign the reaction had wandered off course. For researchers, accurate chemical formulas remain non-negotiable. Guess wrong and time, resources, and sometimes a week’s work vanish.
Real-World Impact of Getting the Formula Right
C10H9N is more than a jumble of letters and numbers. The formula defines everything: how the compound reacts, the solvents that work, and whether it’ll be stable in storage. In pharmaceutical settings, incorrect formulas can derail clinical trials. In my experience, even small labs double-check these details—trusting a supplier’s label carries real risk if paperwork gets sloppy.
Data points from chemical databases like PubChem and Sigma-Aldrich help keep everyone honest. Each entry for 8-Methylisoquinoline matches the same molecular formula, and researchers cross-check these against spectral data to be sure they’re working with the right molecule.
Potential Issues and Thoughts on Solutions
The most pressing problem traces to communication. Mislabeling or copying errors trickle down. One missed number can scramble batches across multiple labs. Chemical education also faces gaps—students should understand how to draw and verify structures from a formula rather than memorize meaningless sequences. I’ve seen projects stall because a member didn’t know how to check their own work—basic skills make or break bigger efforts.
Updating digital tools holds promise. Software that flags inconsistencies between drawn structures and formulas could prevent a lot of wasted time. Open-access chemical registries now help, but keeping them accurate requires vigilance from the community.
Building Stronger Foundations in Chemistry
Working knowledge of chemical formulas drives progress across chemistry. In the case of 8-Methylisoquinoline, those ten carbons, nine hydrogens, and one nitrogen unlock plenty of opportunity. Mastery here leads to safer drugs, better materials, and fewer headaches in the lab. Mistakes get costly, both in dollars and hours, so clear understanding and solid tools remain the best defense.
Is 8-Methylisoquinoline hazardous or toxic?
Understanding 8-Methylisoquinoline
Life often puts us around chemicals we’ve never heard about. 8-Methylisoquinoline is one of those names that pops up in chemical supply lists and research projects. It belongs to the family of isoquinolines, which turn up in both natural sources and manmade products. You probably won't find it on a grocery shelf, but those working in labs or in manufacturing might cross paths with it.
Hazards: What We Really Know
Hazard data on 8-Methylisoquinoline doesn’t stack as high as it does with some better-known chemicals. That doesn’t mean it’s harmless. Research points to isoquinoline compounds acting as moderate skin and eye irritants. Inhalation can lead to headaches, dizziness, and respiratory irritation. I’ve worked with aromatic compounds, and their fumes tend to stick in the air longer than most people realize. Sometimes it just takes a sniff before you know you shouldn’t be inhaling that stuff.
Animal studies involving related isoquinolines show effects on the liver and kidneys after prolonged exposure, like we see with other nitrogen-containing aromatic compounds. The lack of conclusive human studies might sound reassuring, but in my experience, a thin research base only means we haven’t studied the dangers deeply enough.
Handling and Personal Safety
I’ve seen chemists don gloves, goggles, and a good lab coat just to handle materials with unknown risks. That says something. Material safety data for 8-Methylisoquinoline calls for solid personal protection. Even if the acute effects are mild, nobody wants an irritating rash or burning eyes from a careless splash. Ventilation keeps those fumes out of your lungs—no one wants surprise headaches or worse, after a day in the lab.
Chemical suppliers label 8-Methylisoquinoline as harmful if swallowed, inhaled, or when it meets bare skin. Signs remind workers to treat it with respect: avoid eating around it, wash hands, don’t splash or breathe the vapors. Basic, but these are steps too many ignore when things get busy.
Environmental and Long-Term Considerations
Disposal raises another challenge. Aromatic chemicals and nitrogen compounds often persist in the soil or water. Even though there isn’t a lot of data on this exact compound breaking down, related substances show up in waterways down the line and build up in fish or wildlife. That can mean headaches for environmental health teams if disposal gets sloppy. Sending waste to proper hazardous disposal routes stops problems before they start.
Building Stronger Practices with Chemicals
Most chemists I know lean toward caution when the science has holes in it. Strict safety gear, tidy lab benches, and careful records can go a long way. When I started out, veterans drilled an idea in my mind: if the MSDS doesn’t tell you enough, act like it’s worse than you think. That advice holds up.
It’s tempting to skip safety routines, but the memory of a coworker’s chemical burn snaps you back to reality. Nobody wants to find out the hard way that a lesser-known compound has outsized effects. Contact with authorities on chemical hazards, transparent reporting when accidents happen, and calls for deeper studies all push us toward safer workplaces.
Small steps—like swapping in less hazardous alternatives, improving air systems, and spreading word about new findings—do more than regulations on paper ever could. The story of 8-Methylisoquinoline reminds us that in chemistry, uncertain risk is best treated like certain risk, until we know more.
How should 8-Methylisoquinoline be stored?
Why 8-Methylisoquinoline Demands Caution
Working with chemicals asks a lot more than mixing and pouring. 8-Methylisoquinoline, a building block for pharmaceuticals and fine chemicals, carries its own risks. This compound is known for being flammable and giving off possible toxic fumes if mishandled. I’ve seen small mistakes with storage—wrong containers, poor ventilation—turn into emergencies. Proper storage might sound basic, but it’s where real safety begins.
Know the Properties, Shape Your Storage
8-Methylisoquinoline comes as a pale yellow liquid or sometimes as a solid, depending on the room’s temperature. It gives off a pungent smell. If you let it near heat or open flames, it won’t hesitate to catch fire. Direct sunlight can mess with its stability, possibly making it break down faster, producing unknown byproducts. Keep this chemical anywhere near strong oxidizers and you set yourself up for a chemical reaction you absolutely do not want indoors.
Practical Steps: Doing it Right Day-to-Day
Keep it cool and shaded.Heat and light speed up decomposition or reaction. A dry, cool area, ideally below room temperature, works well—think 2-8°C but check if your supplier gives a specific range. Wrap bottles in aluminum foil if storage is near a light source and you can’t avoid some daylight.
Depend on sturdy, clearly labeled containers.Use glass or high-density polyethylene containers with airtight seals. Don’t leave it in generic bottles or containers you can’t trust for chemical use. Labels matter—no one in the lab wants to play guessing games, especially if someone else pulls that bottle off the shelf months later. List the full chemical name, and include hazard warnings right on the label.
Ventilate the storage spot.Fumes happen even from closed bottles, especially over time. Use cabinets with direct ventilation or labs with good air changes per hour. A flammable storage cabinet with flame arrestors brings extra peace of mind. In smaller labs, I’ve seen people rely on simple open shelving, but spills and fumes turn costly fast. Better to invest in the right cabinet up front.
Real Risks: Fires, Health Setbacks, Regulatory Fines
I’ve seen what happens when a lab shoves flammable chemicals together on one shelf. One spark and everything goes up in smoke. Inhaling 8-Methylisoquinoline’s vapors can irritate the nose and throat—and chronic exposure might cause longer-term health problems not immediately visible. Fire marshals do check chemical storage. A surprise inspection can crush a small company with fines or force a shutdown. Sticking to safe storage isn’t an academic exercise—people’s health, jobs, and even the future of your facility are at stake.
Smart Solutions, Real Habits
Good practice comes down to habits built over years. Keep an updated chemical inventory. Rotate stocks, so old bottles don’t sit and become mysterious hazards. Train staff to check for leaks, cracks in lids, and signs of contamination during every use. Personal protective equipment—gloves, goggles, lab coat—should never be optional. Regularly scheduled cleaning prevents hazardous messes from building up unnoticed.
Keep Respect for Every Chemical
People sometimes forget that safety starts with respect— not just for the rules but for the chemicals themselves. Treat 8-Methylisoquinoline with caution, keep your storage area secure, and stay on top of labeling and inventory. One overlooked step turns into tomorrow’s crisis. Treating proper storage as an everyday priority is what separates safe labs from the ones that make the evening news.
What are the physical properties of 8-Methylisoquinoline?
The Look, the Feel, the Basics
Glance at a sample of 8-Methylisoquinoline and you’re likely to see a pale yellow liquid, sometimes described as having a touch of oiliness. The color hints at purity and gives an easy reference point if you’re working in a lab where you need to sort through dozens of similar compounds. Over the years in research settings, having an eye for these subtle differences in color and viscosity helps prevent mix-ups, which can cost both time and resources.
This compound has a molecular formula of C10H9N. That gives it a relatively small molecular weight—143.19 g/mol. It’s light, not as heavy as some complex organics, and that matters when it comes to handling and storage.
Handling Temperatures and Storage
8-Methylisoquinoline boils at about 254°C and melts near 18°C. So, in most room temperature labs, you find it as a liquid, but it can edge toward solid when left in the fridge. This melting point might not seem important at first glance, but it makes a difference for transport and long-term storage. If you store chemicals in a colder room in winter, you can come back to a bottle full of solid 8-Methylisoquinoline, which is tough to pour or measure.
Its boiling point shows you don’t have to worry about this compound evaporating or degrading quickly during most lab work. For chemists who need to run reactions at slightly higher temps, this property saves some hassle.
Smell and Safety
Anyone who’s spent time with aromatic heterocycles knows they can carry a distinctive odor. 8-Methylisoquinoline brings a faint, almost earthy chemical smell—never pleasant, but usually not overwhelming. Sometimes, you can spot a small spill by simply noticing a shift in the room’s odor before instruments even pick it up.
The fact that this compound isn’t highly volatile keeps the vapor in check, so the risk from inhaling fumes remains fairly low under normal use. Still, gloves and safety glasses belong on your hands and face when you handle any heterocycle. Accidental splashes or contact with skin, though uncommon, still pose risks because even mild organic irritants can cause lasting sensitivity.
Mixing, Dissolving, and Working with 8-Methylisoquinoline
In practical terms, 8-Methylisoquinoline dissolves well in many organic solvents—think ethanol, chloroform, and ether. This makes it useful for synthetic chemistry because you don’t have to fight with solubility issues that bog down some other nitrogen-containing rings.
It doesn’t play well with water, though. Like most isoquinolines, it stays mostly separate from aqueous phases, which is a detail that matters during purification or extraction. Getting it out of an organic reaction mixture requires the right choice of solvent, and knowing its preferences helps avoid headaches, especially in a busy lab or production setting.
Dealing with Real-Life Risks and Solutions
Spills happen, often when a researcher is rushing or near the end of a long workday. Because 8-Methylisoquinoline isn’t highly flammable and doesn’t fume aggressively, absorbent pads usually take care of liquid spills without much fuss. Its relatively high boiling point helps you avoid many risks associated with volatile organics, but you don’t want to get lax. Consistent training around chemical hygiene and spill response goes a long way, especially for new staff or students handling these compounds for the first time.
Documented safety profiles advise keeping containers tightly closed and handling them in ventilated spaces. Not all labs follow best practices, but building these habits early can prevent chronic exposure or mishaps. Supportive supervision and easy-to-follow signage help maintain safe working environments. Regular audits, plus offering refresher sessions on chemical properties and handling tips, can make a tangible difference, driving real improvements in lab safety and efficiency.
| Names | |
| Preferred IUPAC name | 8-Methylisoquinoline |
| Other names |
8-Methylisoquinoline 8-Methyl-isoquinoline |
| Pronunciation | /ˌeɪtˌmɛθ.ɪl.aɪ.soʊ.kwɪˈnoʊ.lin/ |
| Identifiers | |
| CAS Number | 3438-33-7 |
| Beilstein Reference | 0177813 |
| ChEBI | CHEBI:87274 |
| ChEMBL | CHEMBL151602 |
| ChemSpider | 114388 |
| DrugBank | DB04208 |
| ECHA InfoCard | ECHA InfoCard: 100.018.708 |
| EC Number | 211-469-2 |
| Gmelin Reference | 643813 |
| KEGG | C14541 |
| MeSH | D008620 |
| PubChem CID | 70412 |
| RTECS number | NL9275000 |
| UNII | Y5L15A82T3 |
| UN number | UN3439 |
| Properties | |
| Chemical formula | C10H9N |
| Molar mass | 143.19 g/mol |
| Appearance | Light yellow to yellow liquid |
| Odor | amine-like |
| Density | 1.102 g/mL at 25 °C |
| Solubility in water | Insoluble |
| log P | 2.4 |
| Vapor pressure | 0.0323 mmHg (25°C) |
| Acidity (pKa) | 5.14 |
| Basicity (pKb) | 6.20 |
| Magnetic susceptibility (χ) | -77.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.117 |
| Viscosity | 1.051 cP (25°C) |
| Dipole moment | 3.25 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 203.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -6.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4821 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | Precautionary statements: P261, P264, P271, P301+P312, P304+P340, P312, P330, P405, P501 |
| Flash point | Flash point: 113°C |
| Autoignition temperature | 510 °C |
| Lethal dose or concentration | LD₅₀ (oral, rat): >2000 mg/kg |
| LD50 (median dose) | LD50: 530 mg/kg (mouse, oral) |
| NIOSH | NIOSH: NQ6475000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 200 mg |
| Related compounds | |
| Related compounds |
Isoquinoline 2-Methylisoquinoline 5-Methylisoquinoline 4-Methylisoquinoline 6-Methylisoquinoline 7-Methylisoquinoline Quinoline |

