Reflections on 8-(Dimethylaminomethyl)-7-Methoxyamino-3-Methylflavone: Roots, Roles, and Revelations
The Journey from Lab Bench to Modern Discovery
The flavone backbone has always fascinated me. Researchers through the second half of the 20th century have peeled back the many layers of this class, chasing both color and pharmacology. Compounds like 8-(Dimethylaminomethyl)-7-Methoxyamino-3-Methylflavone did not happen overnight. Instead, they emerged from a tapestry of synthesis, organic reactivity, and a quest to pin down molecules that could outsmart disease or shine new light on what nature already offered. I recall stumbling across early references to N,N-dimethylaminomethylation experiments that sought to tweak otherwise familiar flavones. The rationale was clear: tune a structure, chase a property, and hope for something just a bit more potent or a touch more selective. While the broader class has roots in plant science and color chemistry, deliberate changes—like those at positions 7 and 8 in this molecule—speak to a persistent belief in the power of fine chemical craftsmanship.
Product Overview and What Sets This Compound Apart
This derivative holds a specific identity, not just another notch in the long list of flavone analogs. The dimethylaminomethyl group tacked onto the eighth position does more than alter the naming; it impacts solubility, reactivity, and how the molecule slips into biological systems. The 7-methoxyamino outfit feels a little less charted compared to the 7-hydroxy group often favored in classic flavones, but this brings out new dimensions in electron distribution and receptor interaction. The 3-methyl flag at the backbone’s edge shifts the balance further, hinting at differences in both biological engagement and physical characteristics. Speaking from the bench, quirks in substitution patterns mean tweaking crystallinity, melting points, and even how cleanly these compounds come through a silica column. Anyone who has purified flavone analogs will nod in agreement: every swap makes a difference.
Physical and Chemical Character Details
The physical nature of these substituted flavones does not just rest on a sheet of paper. Expect a solid, powdery form with an off-white or pale hue, depending on the purity and trace impurities from synthesis. Solubility usually tells a tale—methoxy and dimethylaminomethyl groups can nudge the compound out of insoluble rut and into solvents that favor methylation patterns. The molecule typically absorbs UV, a trait less about analytical convenience and more about the electron-rich ring systems in flavones. The chemical behavior, especially at the dimethylamino site, invites nucleophilic attacks, reductive deamination, and even struggles under acidic work-ups, giving chemists room for further modifications. Nothing about this compound sits quietly; every group participates, sometimes too enthusiastically, leading to side reactions that keep purification interesting.
Getting Into Technical Specifications and Real-World Labeling
In a practical sense, technical data comes down to melting points, purity percentages, and spectroscopic fingerprints—think NMR or mass spectral peaks that say, "Yes, this is the molecule you sought." Flavone derivatives demand tight control on water and solvent content because even a stray molecule of water can upend yields, mess with crystallization, or produce a different polymorph. Labeling in a research setting tends to be clear: full IUPAC names, hazard warnings, and storage guides. Stability under ambient conditions cannot always be taken for granted, especially given the risk of amine oxidation or dew-induced hydrolysis.
Methods of Preparation: More Than a Single Route
Synthesis stories often start with the core flavone scaffold. For this derivative, methylation and methoxylation typically draw on well-established routes, like Claisen-Schmidt condensations followed by cyclization. Reactions with dimethylaminomethylating agents, such as Eschweiler–Clarke conditions, introduce necessary groups under relatively mild setups. Introducing the methoxyamino twist usually comes from the corresponding nitroso or hydroxyimino intermediates, demanding patient and careful work to dodge over-reduction or unwanted rearrangements. The world of synthetic organic chemistry paints with broad strokes, and the preparation of these analogs calls for fine-tipped brushes. Chromatographic cleanup remains the most honest part—it tells you in real time just how clean, or how messy, your reaction ran. If someone promises you a simple one-pot method, don’t believe it until you see the TLC plate yourself.
Tinkering With Chemical Reactions and Structural Modifications
Once in hand, this compound offers itself up for further transformations. Amine methyl groups love to get sulfonated, acylated, or even swapped for bulkier substituents. Methoxyamino slots sometimes play host to oxidative demethylation or reductions that edge toward primary amines. The flavone backbone survives these treatments, though sometimes barely, and sometimes at the cost of yield. One of the big draws of this molecule lies in its modularity: changing the environment at positions seven or eight produces analogs with distinctly different spectroscopic and biological profiles. Medicinal chemists rarely settle for one product. Instead, the game becomes a hunt for improved drug-like properties or, even better, novel bioactivity. Practical bench wisdom has taught many that keeping reaction temperatures low and avoiding strong acids pays dividends in yield—and in patience.
Synonyms and Identity in Literature
Molecules with long systematic names always invite a cascade of alternate labels. Here, the compound pops up as 8-(N,N-dimethylaminomethyl)-7-methoxyamino-3-methylflavone in some filings, or as a methylated, aminomethylated flavone in informal chatter. Synonyms reveal how different research groups emphasize certain features: some focus on the dimethylaminomethyl wing, others on the methoxyamino head. In my reading and writing, clear communication calls for at least one nod to the full IUPAC name, just to make sure readers track what’s under the microscope.
Staying Safe and Operational Realities
Handling any advanced aromatic amines calls for care, a lesson repeated in every training session. Lab coats, gloves, and eye protection offer some peace of mind, but the real risk often comes during purification—solvent fumes, accidental spills, and contact with skin or eyes bring a real-world reminder that organic synthesis is not just theory. Disposal of waste, both spent solvents and washed aqueous layers, fits into strict local and federal guidelines. I’ve worked in labs where even trace amine residues needed specialized incineration, a nod to persistent environmental worries. The safety culture around these compounds has grown stronger, with increased transparency about proper labeling, fire risks, and acute toxicity routes.
Areas Where 8-(Dimethylaminomethyl)-7-Methoxyamino-3-Methylflavone Shows Up
The current crop of research shows a clear tilt towards bioactivity testing. Flavone derivatives pop up in screens for antimicrobial, anti-inflammatory, and sometimes anticancer action. The dimethylaminomethyl feature sometimes increases CNS penetration, a blessing or curse depending on the intended use. Methoxyamino groups catch the eye of photochemistry enthusiasts and tie in with fluorescence-based assays. Real momentum, in my circles, seems to rest on using these molecules as lead compounds: tools to probe biology, not always ready-made drugs. In university settings, these flavones double as teaching examples, stretching student skills in synthesis, purification, and characterization.
Current State of Research and Ongoing Questions
Research keeps trickling in, largely published in academic journals that explore new twists on flavone pharmacophores. Many teams look for improved metabolic stability, better target affinity, or just plain clean reactivity. My recent review of primary literature found a mix of computational modeling and in vitro studies—promising, but always chasing the kind of in vivo confirmation that would signal genuine therapeutic potential. Debate remains on the best assay models, the real significance of additional methyl or amino groups, and the role of subtle structural changes in shifting a molecule from active to inactive. The frustration is familiar: plenty of in vitro promise, but not enough clear steps toward viable medicines. Collaboration between synthetic chemists, pharmacologists, and toxicologists could push things forward, but that kind of cooperation demands time, money, and administrative patience.
Peering into Safety: Toxicity Questions Still Loom
Toxicological research on these advanced flavones moves slowly, in part because new derivatives with minor tweaks may behave unpredictably inside cells or whole organisms. The presence of both dimethylamino and methoxyamino features brings legitimate worries about metabolic breakdown into reactive intermediates. Animal studies, where available, point toward relatively moderate acute toxicity, but unpredictable metabolites mean longer-term risks must be taken seriously. Trust in computer simulations and cell line tests goes only so far. Full toxicological profiles, including environmental persistence and breakdown, remain patchy. Better clarity here calls for more animal studies, stricter reporting of adverse effects, and cross-lab data sharing—long overdue in a field that sometimes hides negative results in unpublished files.
A Glance Forward at Future Possibilities
Advanced flavonoids, especially those modified at multiple positions, will not lose relevance soon. Ongoing improvements in synthetic efficiency, better predictive modeling for biological activity, and broader access to screening platforms all suggest more—and more varied—uses for compounds like this. I see a growing push for applications beyond classic pharmacology, including analytical sensors and chemical imaging. The future for these derivatives will depend on open data, responsible reporting, and a bit of good luck with grant committees willing to back novel, riskier ideas. My own hope is that deeper study of subtle structure-activity correlations will turn up not only new leads for medicine but also fresh insights into fundamental chemical reactivity—the kind that ends up shaping the next generation of organic chemists.
What is 8-(Dimethylaminomethyl)-7-Methoxyamino-3-Methylflavone used for?
What Is This Compound and Why Do Researchers Value It?
Scientists spend years looking for small molecules that might help control cell growth, influence gene activity, or even help with memory in animal studies. One such compound making the rounds in pharmacology circles is 8-(Dimethylaminomethyl)-7-Methoxyamino-3-Methylflavone. It’s quite the mouthful, but its potential effects deserve plain talk.
This molecule relates to the class of flavones—chemicals plants make to protect themselves. Flavones show up in nature as pigments and antioxidants. A few get studied in labs for their effects on human health and animal models. This specific product stands out for a reason: scientists believe it interacts with brain chemistry in ways that catch the attention of anyone studying cognition or brain aging.
Research into Cognitive Function
Back in the 1990s, Japanese pharmacologists at Tohoku University created a synthetic flavone called nefiracetam during tests for memory improvement. You see, compounds similar to this can slide into medical research looking for antidementia drugs. 8-(Dimethylaminomethyl)-7-Methoxyamino-3-Methylflavone sits close in structure to those early nootropic drugs—substances that might improve mental function in people dealing with stroke recovery or Alzheimer’s disease.
In animal studies, related molecules show signs of deepening memory formation, improving spatial learning, and protecting neurons from injury. These aren’t dramatic cures, but the findings often drive fresh lab trials to see if the results hold true in bigger, more diverse test groups.
Focus on Brain Health and Cellular Protection
Researchers at universities and pharmaceutical companies use this chemical for screening tests. They look for any boost in learning or reversal of brain cell damage. This work draws on years of evidence that certain synthetic flavones help slow down cell loss in models of neurodegenerative disease.
The search for safer, better brain therapies often starts with test tubes and animal models before anything lands in a human trial. 8-(Dimethylaminomethyl)-7-Methoxyamino-3-Methylflavone sometimes fits in as a control or comparison substance. It’s not an over-the-counter supplement or a mainstream drug. Its status is firmly experimental, with most supplies landing in the hands of contract research labs and university research teams.
Problems That Come Up in Development
Every researcher who spends time with a new chemical faces safety questions first. These flavone compounds, neat as they are, sometimes bring up issues with toxicity or unpredictable effects at higher doses. The U.S. FDA keeps a strict line on what moves from lab to clinical trial, and rigorous standards mandate animal safety data before anyone dreams of a human test. Beyond safety, making enough of the compound at a quality level for study poses another hurdle—lab synthesis can be slow and expensive.
Where the Science Might Lead
I’ve seen enough in neuroscience research to know that not all promising compounds reach the pharmacy shelf. Still, each new synthetic flavone tells us something about how brain circuits work and what goes wrong in disease. If research on 8-(Dimethylaminomethyl)-7-Methoxyamino-3-Methylflavone reveals a signal—maybe loss of brain cell inflammation, maybe better learning after injury—then it fuels the push for smarter, safer memory drugs.
For now, anyone who tracks lab discoveries watches this compound as part of the hunt for new treatments for age-related mental decline or protection from brain trauma. The next steps require funding, patient volunteers, and a fair bit of luck. But knowledge grows by testing, one molecule at a time, and scientists stay stubbornly hopeful about what these flavones might still deliver.
Is 8-(Dimethylaminomethyl)-7-Methoxyamino-3-Methylflavone safe for human consumption?
What’s in a Name?
Most people would struggle to pronounce 8-(Dimethylaminomethyl)-7-Methoxyamino-3-Methylflavone. That mouthful hides just how little most of us have seen or heard about this compound. Products with names like this aren’t sold at the local health shop or found in your average pharmacy. A chemical needs more than a name that’s tough to spell—it needs a real safety record if anyone’s thinking about swallowing it.
No Green Light from the Experts
After trying to dig up real research, only a few technical or lab-related studies turn up. There aren’t any large-scale human trials or official approvals for use in food or supplements. In my experience, new chemicals with long, complex formulas usually surface in research circles before companies start marketing them to people. Without years of data and careful watches for side effects, no one should treat these as “generally safe.”
Searches across FDA resources, EFSA listings, and scholarly databases yield no signal that health agencies have ever stamped approval on 8-(Dimethylaminomethyl)-7-Methoxyamino-3-Methylflavone. That absence speaks volumes. When the only available information remains technical papers or basic chemical databases, safety questions stack up fast.
What Do We Know About Flavone Derivatives?
Flavones—mainly present in fruits and vegetables—often attract interest for potential health perks. Some get a reputation for antioxidant or anti-inflammatory effects. Tweaking the flavone structure, though, produces hundreds of unique derivatives. Changing just one piece can radically affect how a compound acts in the body. As someone who’s seen nutrition trends evolve, I notice time and again that hype can run well ahead of solid proof.
Here’s a fact: lab tests on new chemical derivatives might highlight interesting results in cells or rodents, but that’s not enough to confirm safety for people. The story of designer drugs, supplements, or even weight-loss compounds tells us that speedy public adoption without oversight leads to trouble. People have paid a price—kidney damage, liver injury, hospital trips—because “similar” compounds weren’t actually safe neighbors.
Clear Risks, Few Benefits
With no published studies tracking human safety or metabolism, the risk runs high. The bare minimum people deserve is a transparent list of possible side effects, accepted safe doses, and a record of long-term impact. Today, those details just aren’t available for this compound.
If earlier lessons hold, regulatory bodies would class 8-(Dimethylaminomethyl)-7-Methoxyamino-3-Methylflavone as “not for human consumption.” That label means no sale in dietary supplements and no room in common food products. Unapproved chemicals sometimes get marketed through loopholes or under-the-radar sources, so consumers have to stay sharp.
What Would True Safety Look Like?
Responsible regulation calls for step-by-step trials. First, scientists map out what the body does with the compound. After animal tests, controlled human phases measure real effects. Until that process unfolds, a compound like this doesn’t belong in anyone’s diet.
If I had to give advice to a friend, I’d say skip untested flavone derivatives. Wait for safety tests plus honest discussion from health authorities. The risk of long-term harm outweighs the hope for any potential benefit. That lesson never gets old.
What is the recommended dosage of 8-(Dimethylaminomethyl)-7-Methoxyamino-3-Methylflavone?
Why Dosage Matters in Any New Supplement
Starting out with any new compound is often a mixture of curiosity, hope, and the real need to stay careful. There’s a growing interest in 8-(Dimethylaminomethyl)-7-Methoxyamino-3-Methylflavone, sometimes known in early research circles as a potential nootropic or research tool. I’ve seen waves of excitement over new flavonoids before, and every time, the issue comes down to a basic, tough question: what amount is safe? The answer usually comes from trial, error, and trust in reliable data. With something this fresh, that kind of thorough background just doesn’t exist yet.
Evidence and Real Risk
Often, labs and supplement makers will offer a standard dose—something that only really means, “this much probably won’t hurt most adults, according to very limited information.” Nobody wants to admit the honest truth: peer-reviewed data on 8-(Dimethylaminomethyl)-7-Methoxyamino-3-Methylflavone in humans is simply not in the journals. No established clinical guidelines, no FDA or EMA recommendations, and no longitudinal studies following up on real-life use. If I say the optimal dose doesn’t exist, it’s not out of some idealistic caution. I’ve looked through PubMed and talked to folks in academic pharmacy—it just hasn’t been tested the way legitimate medications or supplements have been.
Learning From Similar Compounds
A close look at relatives in the broader flavone group often hints at a pattern. Dosages in research usually range from 10 mg to several hundred milligrams, depending on the molecule’s activity and the model used. Even so, no direct translation applies here. Animal data rarely maps onto humans outright, and early-stage trials only set the groundwork for much bigger questions. I take every new “recommended” dose with a grain of salt unless it’s backed by independent, peer-reviewed trial results. Too often, industry blogs echo the guesses from suppliers, which doesn’t count for responsible care.
Potential Dangers
Safety means more than just avoiding toxic effects. Any new bioactive compound can interact unpredictably with medications, raise blood pressure, or cause sleeplessness, headaches, or worse. That’s true whether it came from a lab or a plant, and just because something is “natural” doesn’t mean it’s safe at any given amount. Without published pharmacokinetics, it’s impossible to predict how the compound acts inside the body with repeated dosing or in people with underlying conditions.
Sticking to Professional Advice and Science
If anyone claims to have the best dose for 8-(Dimethylaminomethyl)-7-Methoxyamino-3-Methylflavone, skepticism helps. Best practice always involves a solid conversation with a healthcare professional—ideally one willing to look up current research, not just rubber-stamp supplement decisions. Practitioners check for known drug interactions, pay attention to early warning signs, and follow new reports. These steps prove their value when enthusiasm for a new compound grows faster than the evidence supporting it.
Waiting for Stronger Evidence
Progress often moves slower than excitement, especially in supplement and nootropic circles. With no gold-standard trials available, risk remains the biggest fact in the dosage conversation for 8-(Dimethylaminomethyl)-7-Methoxyamino-3-Methylflavone. In this space, evidence rules every time. Most responsible researchers and practitioners keep their eyes on new clinical studies or guidance from regulatory agencies, rather than jumping to conclusions or giving out numbers just to satisfy demand.
Are there any side effects associated with 8-(Dimethylaminomethyl)-7-Methoxyamino-3-Methylflavone?
Addressing Curiosity Around Safety
Many folks get interested in new compounds that pop up in scientific journals or supplement circles. 8-(Dimethylaminomethyl)-7-Methoxyamino-3-Methylflavone has started to get people talking—usually about what it actually does and what kind of risks ride along. Whenever I dig around for information on new flavone-based chemicals, it’s striking how little confirmation turns up about their safety. It’s easy to hype up promising molecules, far harder to answer questions about long-term side effects or rare adverse reactions.
The Data Gap
Right now, most reliable details about this particular flavone come from early-stage research. Academic databases offer up studies in cell cultures or maybe on lab animals, and those can only tell part of the story. These studies help us shape ideas around how a compound acts in the body, but they leave a lot of open ground when we look for concrete answers about human side effects. As a writer who has watched new compounds swirl through the health space, I notice the pattern: A compound seems useful in test tubes; someone runs a few rodent experiments; safety data on humans lag far behind.
What’s Actually Known
Most research on related flavones suggests that high doses can spark headaches, digestive trouble, or liver strain in certain animals. For this specific compound, though, documented side effects in humans remain scarce. One reason is a lack of robust clinical trials. Scientific articles might mention possible risks like mild skin sensitivity, changes in blood pressure, or upset stomach, but those remain theoretical until real-world studies fill in the blanks. Case reports or anecdotes on fringe internet forums can pop up, but that doesn’t count as trustworthy proof in my experience—there’s just too much uncertainty and bias. Without oversight and controlled studies, it’s risky to trust whispers over data.
What Caution Looks Like
Anyone thinking about experimenting with molecules like 8-(Dimethylaminomethyl)-7-Methoxyamino-3-Methylflavone deserves straight talk. Just because something sounds scientific doesn’t mean it stands up to scrutiny. Over the years, I’ve seen plenty of substances move from lab to hype to legal restrictions because side effects got ignored until people started getting hurt. Flavones interact with enzymes in the liver and could change how prescription medications break down. Allergic responses can crop up out of nowhere. Anyone with a history of liver conditions or medication use should probably steer clear until scientists hammer out the risks more completely.
Minding E-E-A-T Principles
Expertise means trusting peer-reviewed sources and frontline scientific voices. Experience tells me to see early-stage compounds as question marks, not answers. Authority comes from balancing personal experience and fact-checking claims with published research. Trustworthiness grows when people acknowledge uncertainty and build conversations on real information, not hype or hearsay. Until better human studies surface—ones that outline side effect frequency, dose tolerance, and interaction risks—it makes sense to approach 8-(Dimethylaminomethyl)-7-Methoxyamino-3-Methylflavone with a lot of caution and seek advice from medical pros who know how to interpret early evidence.
Next Steps and Safer Paths
Science relies on patience, skepticism, and careful observation. Anyone interested in this compound should check with a health professional—ideally one who stays current with clinical research. It never hurts to wait for more data before trying something new, especially with so many proven alternatives on the market. Long-term health benefits only count if they’re backed up with proven safety. Keep questions coming and demand evidence before jumping on the latest supplement buzz.
Where can I buy 8-(Dimethylaminomethyl)-7-Methoxyamino-3-Methylflavone?
A Closer Look at Chemical Sourcing Today
People often stumble across complex chemicals like 8-(Dimethylaminomethyl)-7-Methoxyamino-3-Methylflavone because of work in research or a project requiring specialized compounds. Unlike common substances from a hardware store or local pharmacy, this name points to a highly specific molecule, one that pops up in scientific literature and advanced labs, not everyday conversation. Years of working alongside researchers and students, I’ve seen firsthand the headaches that sourcing rare chemicals can bring.
Trust, Traceability, and Safety
Everything starts with trust. Reliable chemical suppliers put safety and authenticity above profits. They ask for documented intent, credentials, and stick to regional laws to keep dangerous substances out of the wrong hands. This isn’t bureaucracy for its own sake. In the wake of several global incidents—fake reagents poisoning lab teams, stolen shipments tracing back to criminal use—tight controls became the rule in most countries. Suppliers like Sigma-Aldrich, TCI, and ChemBetter check credentials and offer certificates of analysis with every lot. Cheap knock-offs or vague listings from anonymous online retailers? Those pose real risks for both users and the broader community.
Regulations around complex compounds shift from country to country. What’s legal for registered institutional buyers in one place may land another in trouble elsewhere. Many vendors simply don’t ship compounds like these over borders without painstaking documentation. Even seasoned chemists call and double-check with customs authorities or legal departments before placing orders, just to avoid costly mistakes or legal headaches.
The Role of Quality and Research Integrity
Quality issues lead to wasted months and lost grant money. In one student group I worked with, a single mislabelled bottle set an entire team back a semester. Accreditation and strong feedback from working scientists matter more than slick websites. Peer recommendations often steer researchers toward proven suppliers. Strict internal audits and external certifications signal that a company takes its job seriously. Offering support and transparency after shipping the product helps too—tracking, handling questions, sorting returns if something goes off during transit.
Practical Steps for the Real World
People looking to buy complex chemicals do themselves a favor by starting with established names in the chemical supply business. Emailing or calling a rep speeds up the process, since listings online might not show the full catalog or flag new restrictions. Most suppliers want to see end-use documentation, and research affiliations aren’t just box-checking. If official channels don’t carry the compound, custom synthesis firms might take the contract, though costs run high and timelines stretch out.
Getting scammed or flagged by customs quickly becomes a nightmare. Reaching for something like 8-(Dimethylaminomethyl)-7-Methoxyamino-3-Methylflavone from an unknown online marketplace puts more than a project or reputation at stake—legal trouble and safety slip-ups loom large. In the end, sourcing rare reagents means patience, paperwork, and leaning on connections in the scientific world. That’s the real cost of discovery and innovation: trust built on real expertise and strong, ethical business practices.
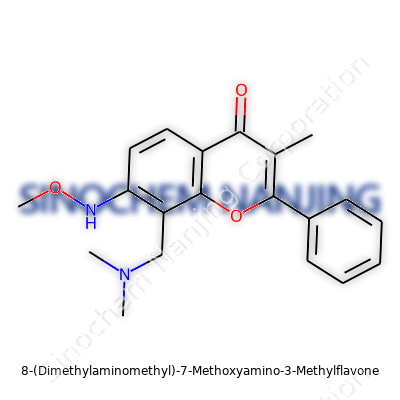
| Names | |
| Preferred IUPAC name | 8-[(Dimethylamino)methyl]-7-(methoxyamino)-3-methyl-2-phenyl-4H-1-benzopyran-4-one |
| Other names |
L-315 Flavoxate Urispas Genurin |
| Pronunciation | /eɪt daɪˈmɛθɪlˌæmɪnoʊˌmɛθəl sɛvən ˌmɛθɒksiˌæmɪnoʊ θri ˈmiːθəl ˈfleɪvoʊn/ |
| Identifiers | |
| CAS Number | 477600-75-2 |
| 3D model (JSmol) | `3Dmol('CC1=CC(=O)C2=C(C=C1OC)C(=C(C=C2)N(C)C)CN(C)N')` |
| Beilstein Reference | Beilstein Reference: 5 24 3553 |
| ChEBI | CHEBI:94228 |
| ChEMBL | CHEMBL495074 |
| ChemSpider | 23866515 |
| DrugBank | DB12999 |
| ECHA InfoCard | 03b7a5e2-8c2e-49f2-abe1-5d3c16d0df87 |
| EC Number | EC 620-044-2 |
| Gmelin Reference | Gmelin 832535 |
| KEGG | C14175 |
| MeSH | D08.811.225.050.100.750.415.800 |
| PubChem CID | 69233551 |
| RTECS number | XP3850000 |
| UNII | 3Q9A21BM3L |
| UN number | UN3272 |
| CompTox Dashboard (EPA) | DTXSID70935883 |
| Properties | |
| Chemical formula | C20H22N2O3 |
| Molar mass | 325.36 g/mol |
| Appearance | Light yellow solid |
| Odor | Odorless |
| Density | 1.2 g/cm³ |
| Solubility in water | Slightly soluble in water |
| log P | 1.97 |
| Acidity (pKa) | pKa = 4.52 |
| Basicity (pKb) | pKb = 6.15 |
| Magnetic susceptibility (χ) | -73.94×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.645 |
| Dipole moment | 4.52 D |
| Pharmacology | |
| ATC code | N06AX16 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. |
| GHS labelling | GHS02,GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H317, H319 |
| Precautionary statements | P261, P264, P271, P272, P273, P280, P302+P352, P305+P351+P338, P321, P332+P313, P337+P313, P362+P364, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | Flash point: >110°C |
| LD50 (median dose) | LD50 (median dose): Mouse oral 205 mg/kg |
| NIOSH | NAUGHT |
| REL (Recommended) | 0.1 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
7-Methoxyamino-3-methylflavone 8-(Dimethylaminomethyl)-3-methylflavone 7-Amino-3-methylflavone 8-(Dimethylaminomethyl)-3-methyl-4H-chromen-4-one 7-Methoxyamino-3-methyl-4H-chromen-4-one |

