7-Methylquinoline: More Than a Curious Chemical
Historical Development
The story of 7-Methylquinoline goes back to the late 1800s, when the study of heterocyclic compounds started to change organic chemistry. Early chemists found themselves fascinated by quinoline derivatives, hoping to unlock new dyes, medicines, and lab tools. The push for better synthetic methods mirrored the growing demand for compounds that could serve more than simple academic curiosity. 7-Methylquinoline wasn’t the first methylated quinoline on the scene, but gained attention as researchers learned how methyl groups at different spots on the quinoline ring tweaked its reactivity. By the 1920s, preparation routes relying on classic Skraup syntheses started to take clear shape and opened doors for more deliberate research around this molecule.
Product Overview
This compound shows up as a pale yellow, oily liquid at room temperature. Its structure—a quinoline ring holding a single methyl group on the seventh carbon—might sound forgettable to a casual observer, but it’s anything but that in the right hands. Chemists noticed that shifting that methyl group even one carbon over can lead to a big change in both chemical behavior and industrial usefulness. I learned quickly in my own work that you can’t underestimate the influence of a small side group. It’s a familiar name across research labs, where it plays a supporting role, especially when creating more complex molecules or screening novel reactions.
Physical & Chemical Properties
With a boiling point high enough to make distillation less of a headache and a melting point lower than many related chemicals, 7-Methylquinoline handles like many light aromatic bases. Its unmistakable smell follows you deep into the fume hood. Solubility leans toward organic solvents; it’s stubborn in water, but dissolves well in ether, benzene, and alcohols. The methyl group doesn’t just give a small bump to weight—it subtly nudges the electron cloud, tuning reactivity. This can offer an edge or a hurdle, depending on the type of synthesis underway. And, as with most heterocyclic aromatics, stability comes with caveats—the nitrogen atom opens the molecule to both acid and base attacks, demanding care in storage and handling.
Technical Specifications & Labeling
Lab bottle labels often carry the standard chemical identifiers: CAS number, molecular weight, empirical formula. But for users who want more than bureaucracy, these labels are cues for safety and shelf management. When I grabbed a bottle during my first year as a bench chemist, I realized that such numbers matter most when you’re doublechecking safety data. It’s not about rote compliance; it’s about avoiding mix-ups or harmful accidents. The molecule’s tendency to oxidize under the wrong conditions, or form harmful vapors if overheated, means you have to pay attention beyond technicalities. Certification standards ensure that what’s in the bottle meets purity benchmarks—impurities here can lead to false positives in pharmaceutical testing or unreliable catalytic studies.
Preparation Method
Old textbooks describe the Skraup synthesis as the classic route: a reaction of aniline derivatives with glycerol in the presence of acid and an oxidizer. Modern labs favor tweaks—using different oxidizing agents or catalysts—because cleaner yields matter, especially when chasing trace active ingredients for pharmaceuticals or specialty dyes. Some industry players turn to methylation of quinoline itself, guiding methyl group placement through careful reaction conditions. In the early days of my career, I saw just how stubborn side reactions could be, highlighting the importance of purification steps like distillation or chromatography to get from brownish crud to a usable product.
Chemical Reactions & Modifications
7-Methylquinoline doesn’t just sit on the shelf; it serves as a starting point for countless transformations. The methyl group can undergo oxidation, producing acids or aldehydes, making it valuable for synthetic routes in second-generation alkaloids or advanced materials. Nitration, sulfonation, and halogenation find their place in the toolkit, with the methyl group quietly influencing the direction and speed of these reactions. For researchers studying antimalarial drugs, modifying the quinoline structure with groups at different positions, including the seventh carbon, offers a way to tune biological activity. Working with this compound means balancing reactivity with selectivity, always watching for overreaction or unwanted side products that can cloud analytical results.
Synonyms & Product Names
In journals and catalogs, you’ll see names like 7-Methylquinoline, 7-Quinolinyl methane, or 7-Quinoline methyl. Each label tries to steer the chemist away from confusion. During literature searches, these synonyms pop up. I recommend against relying solely on one name, especially when tracking down older research papers or cross-referencing patents. Such confusion can lead to missed connections and duplicated efforts in the lab—a lesson I picked up after chasing down the wrong methylated isomer for days.
Safety & Operational Standards
No one pays attention to safety until an accident reminds them why it matters. 7-Methylquinoline can irritate eyes and skin, and repeated exposure to its vapors may resurface as headaches or breathing problems. Proper ventilation and protective equipment count as essentials; I’ve seen how carelessness can spiral into emergency room visits. Labeling must be clear, separating storage away from acids and oxidizers to stave off unwanted reactions. Standardized training and keeping up with the latest SDS updates help prevent mishaps—regardless of how many times a chemist has handled the compound before.
Application Area
Research labs use 7-Methylquinoline for the synthesis of more complex drug precursors, dyes, and optical brighteners. If you’ve ever marveled at how painkillers move through the body or watched a security ink glow under UV light, it’s likely you’ve glimpsed the influence of compounds derived from quinoline rings. Some studies look at metal complex formation with nitrogen heterocycles; these have potential in industrial catalysis or as probes in analytical chemistry. In agriculture, biologists keep an eye on these molecules for their activity as growth regulators or pest deterrents. During my time in chemical development, I learned that demand for such specialized building blocks never really wanes—it just shifts from one application to another as industry challenges grow more sophisticated.
Research & Development
Pushing the limits on what you can make from 7-Methylquinoline attracts plenty of attention. Drug developers sift through libraries of quinoline derivatives, chasing new antimicrobial or antimalarial compounds. The never-ending fight against drug resistance prompts researchers to tweak existing molecules, altering positions like the methyl group to punch through evolving bacterial shields. Material scientists eye quinoline derivatives for possible use in organic electronics, energy storage, and light-emitting devices thanks to their fusion of stability and modifiable properties. Instrument makers refine sensors based on the molecule’s fluorescence, creating better tools for environmental monitoring or sophisticated blood chemistry panels. What feels routine today might blossom into a blockbuster tomorrow, driven by the incremental experiments of scientists committed to stubborn progress.
Toxicity Research
Any compound with promise demands a hard look at how it behaves in the body and environment. 7-Methylquinoline isn’t an outright villain, but toxicity studies show potential risks if mishandled. Repeated lab studies suggest that high doses can produce liver stress or hemolysis in animals. Long-term impact in humans remains inconclusive, largely due to limited widespread use, but this doesn’t mean safety officers can relax. Environmental studies warn about potential persistence in soil and waterways if disposal isn’t handled with care. The right answer isn’t fear, but a habit of good stewardship—favoring closed systems, smart waste management, and transparent reporting of handling practices. I’ve watched small lapses snowball into ugly regulatory headaches; ethical companies put safety and sustainability first, not just because of the law, but because lab workers and local communities deserve respect.
Future Prospects
What excites people about 7-Methylquinoline isn’t just its solo performance. Its ability to serve as a platform for new molecules points toward a bigger story—one about smarter antifungal agents, greener agrochemicals, and more reliable sensors. Synthetic organic chemists sketch out routes for new drugs or advanced optical materials, understanding that each tweak in the quinoline ring could unlock new biological or physical properties. The pace of discovery jumps when collaboration across disciplines replaces solitary silos, with AI-driven modeling helping to predict reactivity and safety profiles long before a drop is synthesized. As someone who has watched waves of fads come and go, I see long-term value in backing substances like 7-Methylquinoline that offer flexibility, scientific interest, and honest potential to make a difference—not through headline-grabbing breakthroughs, but through steady, everyday advances in how we treat disease, track pollutants, and design materials for a complicated world.
What is 7-Methylquinoline used for?
The Role of 7-Methylquinoline in Modern Chemistry
Some chemicals get almost no attention outside labs, yet they quietly power much bigger processes. 7-Methylquinoline counts among them. It shows up in the manufacture of dyes, pharmaceuticals, corrosion inhibitors, and complex organic molecules. This compound doesn’t get front-page coverage, but its fingerprints spread across products that run our lives.
Pharmaceutical Research: Laying Foundations
Drug discovery keeps pushing boundaries, searching for molecules that might turn into relief for millions. The structure of 7-Methylquinoline gives medicinal chemists room to build new drug candidates. It serves as a chemical scaffold, which means other atoms and groups can hook onto it, changing its behavior and effects. Some antimalarial and anti-infective projects use pieces of the quinoline backbone. You may never hear about 7-Methylquinoline on a prescription label, but its core helps researchers design new treatments for serious diseases.
Dye and Pigment Production
Everyday items — from clothes to car paint — depend on improved dyes that last and look better. 7-Methylquinoline acts as a building block for many dyes, especially those in the quinoline family. By tweaking the structure, manufacturers get richer colors or more stable pigments. Textile makers and coating producers push for these changes because brighter, longer-lasting color sells products and cuts replacement costs. It keeps textiles looking new and cars shining under the sun.
Corrosion Inhibitors and Industrial Fluids
Few people get excited about rust, but it eats away at bridges, cars, and factory machines. 7-Methylquinoline features in research on corrosion inhibitors. These compounds slow down or stop metal from breaking down under harsh conditions. Factories and municipal water plants rely on inhibitors made with chemistry like this to push back expensive repairs and keep infrastructure safe. Every extra month that a pipeline lasts means less disruption and lower costs for everyday services.
Organic Synthesis: Solving Complex Problems
Chemists see 7-Methylquinoline as a handy intermediate. It fits into several synthetic pathways, helping scientists build new molecules for everything from crop protection to advanced materials. These reactions demand reliability and consistency, or the whole process can go sideways. That’s why high-purity 7-Methylquinoline gets close attention from manufacturers and labs. Impurities or mistakes in its synthesis slow down entire research programs.
Weighing Health and Environmental Considerations
People working with this chemical have to pay close attention to safety. The quinoline family can carry health risks if handled incorrectly — inhaling or swallowing these compounds needs to be avoided and skin contact ruled out. Factories control emissions tightly, following environmental rules that protect workers and neighbors. Responsible chemistry means combining innovation with oversight, making sure useful materials don’t become hazards. Everyone benefits from strict traceability and open reporting on chemicals that move through our supply chains.
Next Steps in Chemical Innovation
University labs and corporate research centers keep investigating how to dial up the usefulness of 7-Methylquinoline while reducing its downsides. Green chemistry pushes for cleaner ways to make it, using less energy and generating less waste. New applications in electronics and imaging keep appearing as material science advances. Tackling these challenges takes expertise and persistence, along with accountability for the impacts outside the lab. Those at the front lines learn to recognize both the rewards and the limits of even the most useful molecules.
What is the chemical formula of 7-Methylquinoline?
Grasping the Basics
7-Methylquinoline goes by a few names—sometimes it’s called baurine in pharmaceutical circles. At its core, the chemical formula is C10H9N. You get this by taking the quinoline structure (C9H7N) and swapping in a methyl group at the seventh position on the ring. Chemists care about that little twist because swapping just one group on a molecule can change everything about how it acts in the world—with drugs, with industrial reactions, even in nature.
Why These Formulas Matter Beyond the Lab
Most folks haven’t heard about 7-methylquinoline—unless you’re knee-deep in chemistry, or tinkering with pharmaceuticals and dyes. But having been in and out of academic labs and small manufacturing facilities, I can say: the fine details of a formula like C10H9N show up everywhere. Just pulling information off a bottle isn’t enough. You look at the structure, and suddenly you understand why it smells pungent, why it interacts better with certain metals, why technicians reach for it when making certain sensitive fluorescent dyes.
With chemistry, you’re always trying to carve out a more direct path—from raw material to final product. I’ve seen how grabbing a molecule with the right formula, and the right arrangement of atoms, means trimming down waste and fewer side reactions. That’s time and resources saved, but there’s more at stake than just efficiency.
Human Exposure and Practical Risks
7-Methylquinoline isn’t something the average person encounters daily. Still, it finds its way into the workplace. I remember working with older ventilation hoods. Even when using chemicals like this in small amounts, you start to notice eye or skin irritation. Toxicological data tells us that similar quinoline compounds can damage organs over time, so paying careful attention to the molecules at hand matters. Chemical formulas aren’t dry facts—they point to real risks or safety advantages.
I’ve always trusted the databases curated by major health and environmental groups. Reliable toxicology reports, along with regulatory updates, keep everyone honest and safe. Companies have to keep up with the latest best practices, swap out old solvents and build better training into their workflows. All this comes back to understanding the formula and what it represents.
Pushing Ahead: What Can Change?
Researchers working on these compounds can push boundaries for greener chemistry. One solution: use alternative solvents that break down more easily in the environment, or develop ways to recycle chemicals more effectively. The discussions I’ve had with environmental engineers point to another fix—full transparency about which compounds end up in waste streams and how they’re managed.
Having the correct chemical formula in hand, and knowing what it means, lets everyone—chemist, manager, regulator—make smarter choices. In my experience, simple things like putting up accurate chemical signage, reviewing supplier data sheets every quarter, and investing in up-to-date handling protocols make all the difference. Only with solid knowledge can you balance innovation, safety, and responsibility.
Is 7-Methylquinoline hazardous or toxic?
Looking at the Risks
Ask anyone who works with chemicals, and they’ll tell you: even modest changes to a molecule can flip the story from safe to sketchy. 7-Methylquinoline doesn’t steal headlines, but dig beneath the jargon and it becomes clear why people keep asking about its hazards. Most folks see a complex name and wonder if they're dealing with a stubborn stain or a serious health risk.
The Science Says: Handle with Respect
7-Methylquinoline comes from the quinoline family, and if you’ve ever spent time reading up on aromatic amines or their derivatives, you know that some can sting or irritate, some cause nasty blisters, and others even carry cancer risks. In lab settings, this stuff can irritate eyes, skin, and the inside of your nose and throat. A single splash can cause redness and maybe even burns. Breathing in its vapor doesn’t do your lungs any favors either.
The Globally Harmonized System (GHS) classifies this compound as hazardous, typically listing warnings about skin and eye irritations, possible organ toxicity, and threats to aquatic life. Most safety data sheets put a clear spotlight on the dangers: this isn’t a chemical to handle in shorts and a tank top.
Long-Term and Environmental Impact
People working with quinoline compounds have reported headaches, dizziness, and impacts on liver or kidney health after repeated exposure. The CDC and International Agency for Research on Cancer check for cancer links in chemicals like this one. While quinoline itself has drawn scrutiny, studies on 7-Methylquinoline are less conclusive. That doesn’t mean it gets a free pass. Researchers often flag it as a substance to keep an eye on—especially for long-term and high-level exposures.
Aquatic impact draws even more concern. This molecule tends to stick around in waterways. Fish and other aquatic creatures can’t always break it down, so it accumulates, moving up the food chain. The Environmental Protection Agency puts chemicals like 7-Methylquinoline on watch lists because of how slow they degrade and how easy it is for them to reach streams and rivers during spills or through poor storage.
Why Workers and Home Chemists Should Pay Attention
I remember a time in the lab, years back, when a new student got a whiff of quinoline and turned green. That episode stuck with me. It’s easy to underestimate how quickly a headache can become a hospital trip in poorly ventilated rooms. One slip, a missed glove, or a carelessly sealed bottle can mean trouble. For small operations and home chemists, losing focus causes accidents that trained technicians try hard to avoid.
Most householders won’t ever run into this chemical. Yet it turns up in research, pharmaceutical steps, and sometimes in dye production. Folks in those industries know the drill: gloves, goggles, lab coats, and a properly vented fume hood aren’t up for debate. Safe storage in tight containers and scrupulous labeling make all the difference. Shared spaces, like university labs, crack down on solo late-night experiments for good reason—no one wants to get caught off guard.
Better Ways Forward
Chemistry never stands still. Companies and research teams search for safer replacements whenever possible, or at least set strict rules. Dilution can be friend and foe; it lessens risk but only if chemical waste still ends up in the right container. Disposal through certified waste management stops 7-Methylquinoline from slipping into drains where it could wreak havoc downstream.
What truly shifts the needle? Widespread training, not just on paper but as habits, drilled every day. Regular inspections and open dialogue keep teams sharp. For lab workers, reporting problems and small spills keeps everyone accountable. Home chemists, hobbyists, and small businesses often need clearer, more accessible safety guides—that’s a point regulators and educators can work harder on.
How should 7-Methylquinoline be stored?
Paying Attention in the Storeroom
There’s a particular smell that comes from working around quinoline compounds. Anyone who’s spent time in a chemical storeroom knows it. That sharp, characteristic odor tells you right away this isn’t just another bottle on the shelf. 7-Methylquinoline gets used for all sorts of research purposes, from dye synthesis to pharmaceutical projects. This compound doesn’t scream danger as loudly as some, yet storage mistakes can still wreck budgets, ruin experiments, or risk health. I’ve seen wasted material and near-misses from casual attitudes, so storing it right earns some real attention.
Heat, Light, and Air: The Big Three to Watch
Over years of lab work, three things have stood out as key troublemakers: heat, direct light, and exposure to open air. 7-Methylquinoline stays stable at room temperature—no need to freeze or chill it. Still, storing it in a cool, dry place helps keep it from breaking down over time. Moisture and warmth just invite trouble. Humidity leads to clumping and can mess with chemical purity, and heat speeds up unwanted reactions.
Transparent bottles might look fine, but direct sunlight or harsh lab lights can slowly degrade a lot of organics, including methylquinolines. Opaque, amber, or other darkened storage bottles help put a lid on light exposure. I’ve seen the yellow tinge in open bottles disappear much faster under bright light. Stick the bottles where sunlight can’t sneak in and you avoid unnecessary loss.
Sealing the Bottle Really Matters
Quinoline derivatives often let off a wisp of vapor, and the pungent smell makes its way through weak caps. Some compounds degrade more in air than others, but all of them benefit from a tightly closed bottle. I’ve found that containers with ground glass stoppers do a better job, but modern screw caps with chemical-resistant liners usually work, as long as you screw them down firmly.
Letting bottles sit open for a while on the bench practically guarantees contamination. Dust, moisture, even careless pipetting can sneak molecules into the bottle that shouldn’t be there. Writing the opening date on every bottle keeps surprises to a minimum—old stock turns unreliable quickly.
Labeling, Spacing, and Spill Avoidance
The most careful chemist still makes mistakes without clear labeling. Good labels go beyond the name and date—adding hazard info, emergency contacts, and any special storage instructions stops confusion before it starts. Shelves in our storeroom never get crowded for one simple reason: separation makes spills easier to control. Methylquinolines carry some risk for skin and respiratory irritation, so nobody wants solvents or acids nearby to compound the danger.
Personal Experience and Small Labs
Small academic labs often skip on fancy flame-proof cabinets or up-to-date ventilation. Even so, storing methylquinoline away from sources of ignition proves essential. Most chemical safety sheets point out its flammability. I’ve seen small fires start from splashes near electrical outlets or hot plates. Fire risk never feels immediate—until it happens.
Teaching assistants sometimes get assigned stockroom duty, and experience matters. Training new folks to check seals, replace faded labels, and monitor expiration dates forms one layer of defense. No substitute exists for a regular, thorough sweep of chemicals—especially in aging buildings with unreliable climate control.
Simple Solutions Make a Difference
It doesn’t take a high-tech lab to treat 7-Methylquinoline right. Tight seals, a cool, shaded shelf, good labeling, and spacing beyond arm’s length from anything flammable keep things safe. Respecting how organics behave, not just what’s printed on the MSDS, makes a chemical storeroom work for everyone. I’ve learned through tough lessons and tight budgets—paying attention on the shelf protects the science and the people doing it.
What are the physical and chemical properties of 7-Methylquinoline?
Breaking Down 7-Methylquinoline
7-Methylquinoline belongs to a family of compounds called quinolines, which have long been valued in research labs and industries alike. This yellowish liquid has a distinct, sharp smell that reminds some people of the earthy odor in certain pharmaceuticals. What sets it apart is the methyl group attached at the 7-position. This little difference can change how it interacts with other chemicals and even how it behaves in daily lab routines.
Physical Features That Matter
The stuff you notice right away with 7-Methylquinoline: it's usually a yellow to slightly brown liquid at room temperature. Its boiling point hits around 243 °C, which tells you it doesn't evaporate as easily as water or alcohol. A chemist dealing with this liquid won’t find it thick and sticky; it flows fairly well, with a viscosity close to water’s. Its melting point lands below standard room temperature, so solids rarely show up unless you’re working in a pretty cold environment.
The compound isn’t fond of water. Pour it into a beaker of water and you'll watch it separate itself, hinting at its hydrophobic nature. In my own experience, I’ve seen this property really count during purification steps—being able to easily pull the chemical into an organic solvent helps during extraction or washing procedures in organic synthesis.
Chemical Behavior and Reactivity
Chemically, 7-Methylquinoline offers a stable aromatic ring system. What’s interesting here is the interplay between the nitrogen atom in the ring and the extra methyl group. Both affect electron distribution, making it just a bit less reactive to acids than plain quinoline. This subtle tweak matters if you’re using it as a building block for making other substances. I’ve seen synthetic chemists lean on this compound because it resists getting torn apart by mild acids, holding up well in reactions that require stability under heat or pressure.
The molecule doesn’t shy away from getting involved in electrophilic substitution reactions, like most aromatic compounds. Its methyl group makes it a touch more reactive in certain spots, which can speed up lab work. I remember an instance in a university lab where that speed saved us hours during a multi-step synthesis project that required methylation—our output was higher because this compound played along just as the textbooks predicted.
Why These Properties Matter
The combination of a high boiling point and low water solubility means 7-Methylquinoline can stay stable through pretty rigorous processing steps. That counts for a lot in the real world, especially if you’re working in pharmaceuticals or dyes—two areas where quinoline compounds have carved out an important role.
Handling this compound comes with standard lab safety measures. It’s flammable and gives off fumes that could irritate eyes and skin, so proper ventilation and gloves are more than just a suggestion. I’ve always stressed to my students the importance of this; forgetting a fume hood can turn a routine experiment into an uncomfortable mess.
Improving Lab and Industrial Use
Moving forward, better containment and greener solvents could address lingering concerns around waste and exposure. Researchers keep pushing for alternatives that soften the blow to both workers and the environment.
Overall, the personality of 7-Methylquinoline—how it looks, how it moves, and how it reacts—shapes how scientists use it and where its strengths show up. Getting the details right keeps outcomes consistent, whether we’re developing new drugs or working on next-generation pigments.
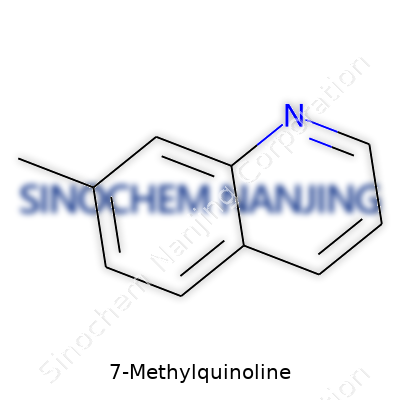
| Names | |
| Preferred IUPAC name | 3-Methylquinoline |
| Other names |
7-Methylquinoline benz[b]picoline 7-quinolyl methane quinaldine 2-methylquinoline |
| Pronunciation | /ˌsɛv.ənˈmɛθ.ɪl.kwɪn.əˌliːn/ |
| Identifiers | |
| CAS Number | 611-34-7 |
| Beilstein Reference | 95615 |
| ChEBI | CHEBI:34587 |
| ChEMBL | CHEMBL32074 |
| ChemSpider | 13101 |
| DrugBank | DB03792 |
| ECHA InfoCard | ECHA InfoCard: 100.012.924 |
| EC Number | 207-528-7 |
| Gmelin Reference | 89096 |
| KEGG | C01767 |
| MeSH | D015883 |
| PubChem CID | 7007 |
| RTECS number | GN8575000 |
| UNII | HD995L4B9N |
| UN number | 2811 |
| Properties | |
| Chemical formula | C10H9N |
| Molar mass | 143.19 g/mol |
| Appearance | Yellowish liquid |
| Odor | strong; quinoline-like |
| Density | 1.083 g/mL at 25 °C(lit.) |
| Solubility in water | slightly soluble |
| log P | 2.57 |
| Vapor pressure | 0.0651 mmHg (25 °C) |
| Acidity (pKa) | pKa = 4.85 |
| Basicity (pKb) | 9.73 |
| Magnetic susceptibility (χ) | -81.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.649 |
| Viscosity | 1.220 mPa·s (25 °C) |
| Dipole moment | 2.11 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 217.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 142.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3876.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H411 |
| Precautionary statements | P264, P280, P305+P351+P338, P337+P313, P501 |
| Flash point | 113°C |
| Autoignition temperature | 630°C |
| Lethal dose or concentration | LD50 oral rat 533 mg/kg |
| LD50 (median dose) | LD50 (median dose): **380 mg/kg (oral, rat)** |
| NIOSH | MNQ6600000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 20 mg/m3 |
| Related compounds | |
| Related compounds |
8-Methylquinoline Quinoline Lutidine Isoquinoline |

