Looking Closer at 7-Methylisoquinoline: More Than a Chemical Name
Historical Development
Once upon a time, synthetic chemistry seemed simple, limited to a small collection of basic building blocks. In the world of heterocyclic compounds, 7-Methylisoquinoline started out as little more than a curiosity discovered through coal tar studies and classical organic transformations that shaped the early 20th century. Back then, chemists dug deep into the structures of quinolines and isoquinolines, driven by the possibilities inside pharmaceutical and dye industries. They weren't sure why methylation at the seventh position reshaped biological profiles, but labs from Europe to North America continued to dig. Today, anyone in the field can trace the use of 7-Methylisoquinoline through hundreds of studies, from early spectroscopic proofs through detailed synthesis journals in the later half of the century. Research groups kept chasing new applications as the market evolved, giving this compound a firmly established footprint in both academic and industrial labs.
Product Overview
7-Methylisoquinoline appears simple on paper, but it grabs the attention of chemists because of its compact heterocyclic core with a methyl group at a specific site. This molecule isn't the sort you find on the shelves of every general lab, yet it stands out in the toolkit of anyone working with complex organic synthesis or natural product analogs. In the field, it's valued for its versatility, popping up as a precursor, an intermediate, or sometimes even a final target for specialized applications.
Physical & Chemical Properties
A close look at 7-Methylisoquinoline shows a pale yellow to brownish liquid or crystalline solid, depending on the purity and handling. It produces a subtle, characteristic aromatic smell reminiscent of other polycyclic compounds. Its molecular formula—C10H9N—makes it closely related to other isoquinoline derivatives, and its melting and boiling points sit in ranges common for small heterocycles. In solvents, it reveals its practical nature: soluble in common organics, somewhat sparing in water, and prone to stability over time unless exposed to strong acids or light. Lab experiences tell us this compound responds reliably to heat and pressure during reactions, and it rarely surprises skilled chemists with unpredictable behavior.
Technical Specifications & Labeling
Walking through a chemical supply warehouse, it's easy to spot bottles marked with straightforward labeling: purity grades, lot numbers, hazard signals, and regulatory compliance. 7-Methylisoquinoline comes certified for various research uses, with assurances around purity levels—often 97% or greater—guided by rigorous chromatographic and spectroscopic tests. Responsible suppliers focus on traceability and safety, with clear identification markers. Good labeling and technical transparency do more than tick regulatory boxes; they keep people on the safer side of lab work and build trust in sources.
Preparation Method
Most chemists turning out 7-Methylisoquinoline rely on classical methodologies a century old, sometimes bringing in newer catalytic procedures. Skraup synthesis, Bischler-Napieralski cyclization, or custom cyclodehydration strategies lay the groundwork. Starting from benzylamine derivatives or toluene-based precursors, these reactions push toward ring closure and site-specific methylation. In recent years, some labs have adopted greener, catalytic methods to limit waste and harsh conditions—using palladium catalysts or microwaves to shorten reaction steps. Real-life handling always means balancing cost, time, environmental impact, and reproducibility—elements any working chemist knows to weigh before scaling from benchtop to industrial batch.
Chemical Reactions & Modifications
7-Methylisoquinoline doesn’t stay static in the hands of a creative chemist. It offers reactive sites for halogenation, oxidation, and coupling, giving rise to a library of analogues for pharmaceutical and agrochemical research. Electrophilic aromatic substitution works well, and direct metalation creates an anchor for more elaborate side chains. Derivatization with functional groups like nitro, carboxyl, or sulfonyl opens whole vistas of potential biologically active molecules. In a world where new drug scaffolds are king, this backbone continues to drive structure-activity relationship studies and helps tackle challenging synthetic goals.
Synonyms & Product Names
Not every bottle or catalog refers to 7-Methylisoquinoline by just one name. Depending on the era, supplier, or language, it goes by titles like 7-Methyl-2-benzopyridine or even obscure research codes favored by chemical libraries. The diversity of naming shows up most often in international literature reviews or patent records, underscoring the need for clear CAS registration numbers and proper referencing. Without these, cross-checking data or searching for toxicity results becomes a slow grind through different naming conventions.
Safety & Operational Standards
Every organic chemist knows not to judge a substance by its bland name. 7-Methylisoquinoline comes with its own handling advice. Labs enforce fume hood protocols because of its modest vapor pressure and potential irritant qualities. Technicians wear gloves, goggles, and lab coats as a simple, effective barrier. Regulatory guidance focuses on skin, eye, and respiratory protection. Spills and off-gassing aren’t frequent, but swift reporting and careful cleanup matter. In facilities where good culture beats written rules, people share handling wisdom face-to-face, reducing near-misses that never make it into published documentation.
Application Area
The uses of 7-Methylisoquinoline run longer than most would expect. In medicinal chemistry, it's an anchor point for synthesizing new pharmacologically active agents—sometimes analgesics, sometimes antimicrobials. Researchers in agrochemicals look at its base structure as a launching pad for new herbicides or growth regulators. A handful of advanced materials projects dig into its photophysical properties, teasing out applications in organic electronics or fluorescent probes. The link tying these fields together comes from the molecule’s adaptability. If it performs well in one area, word spreads, and other teams get curious enough to experiment for themselves.
Research & Development
Study after study tells us the same story: no single molecule can claim a monopoly on innovation. Still, 7-Methylisoquinoline keeps showing up in R&D pipelines across different institutions. Drug discovery projects lean on it for modifying core scaffolds, chasing new activity against bacteria, tumors, or parasites. Students learn the ropes by testing new synthetic routes or optimizing catalysis around this molecule. Funding bodies appreciate tangible research impacts, leading to patent filings and published structure-activity relationships. A few R&D groups push for computational chemistry input, mapping likely reaction courses or docking predictions before rolling out pilot reactions. The feedback loop between benchtop, in silico, and pilot plant keeps shifting the baseline forward.
Toxicity Research
Lab experience teaches respect for the unknown. 7-Methylisoquinoline, like most aromatic amines and heterocycles, deserves careful toxicological study. Early animal work flagged irritation and mild systemic toxicity at higher exposure. Data collected from acute and chronic assays shape the guidelines that modern occupational health teams rely on. Today’s chemists encounter high-quality hazard assessments in international regulatory agencies’ reports, giving labs good data to build their safety protocols. Any new analogs synthesized off the core molecule send researchers back to the drawing board for fresh toxicology screens. In cases of accidental exposure, on-the-ground medical teams respond according to up-to-date chemical health records, helping keep workers safe and operations moving.
Future Prospects
If the past is any guide, 7-Methylisoquinoline has plenty of chapters yet to come. The push for green chemistry may inspire more sustainable preparation routes, like biocatalysis or solvent-free protocols. The pharma pipeline rarely rests—novel therapeutic uses for isoquinoline analogs are a near certainty as chemical libraries expand. The world of advanced materials remains hungry for organic semiconductors and sensors based on unique aromatic backbones. In classrooms, the molecule serves as a lesson in both what chemistry has already achieved and what remains on the table for future researchers. Every new discovery traces its lineage back to molecules like this—reminding us that sometimes, history, creativity, and careful work combine to move science forward in unexpected ways.
What is 7-Methylisoquinoline used for?
Understanding the Value of Chemical Building Blocks
7-Methylisoquinoline doesn’t land on the typical grocery list, but it matters a whole lot in labs and pharma companies worldwide. For chemists, this compound has gained traction because of how useful it is as a starting point. Its structure serves as a base for making bigger, more complex molecules. Over the years, I’ve seen a steady rise in requests for rare building blocks like this among researchers hunting for new medicines.
Pharmaceutical Research and Drug Discovery
Researchers want to find compounds that act on disease pathways without causing problems elsewhere. It sounds simple. In practice, it takes thousands of test runs before hitting a breakthrough. 7-Methylisoquinoline often features in early-stage experiments because its skeleton mimics segments found in some promising drug classes. I’ve chatted with industry scientists who use it to design agents that might fight cancer or act as muscle relaxants. Early data points out that isoquinoline rings play a role in cardiovascular drugs and some painkillers. It's easy to see why researchers keep this molecule close at hand.
Moving New Medicines Down the Pipeline
Big discoveries often start small. Chemists will tweak the structure of 7-Methylisoquinoline, adding or swapping side groups. These changes can lead to new compounds with unique effects. More than once, I’ve seen research projects begin by altering a structure like this one and later uncover molecules that could block enzymes tied to tumors or inflammation. Scientific literature lists several papers where derivatives from this molecule exhibit antibacterial or antifungal activity, which feels particularly important with antibiotic resistance climbing worldwide.
Beyond Medicine: Other Scientific Uses
Not every use involves human health. 7-Methylisoquinoline also finds a place in the synthesis of dyes, pesticides, and specialty chemicals. A friend working in agrochemicals mentioned they leverage such compounds to fine-tune how pesticides break down. In this context, having control over structure transforms how a product behaves outdoors. Fine chemical synthesis often relies on these small molecules as stepping stones towards bigger, more application-ready materials.
Access and Quality: A Real-World Challenge
For all its potential, obtaining high-quality 7-Methylisoquinoline isn’t always straightforward. Many smaller labs struggle with access, which slows progress. Reliable sourcing means better reproducibility and ensures experiments can actually lead somewhere. Quality control remains crucial, as even minor impurities can ruin days of work or lead to misleading findings.
Supporting Scientists and Safer Medicines
Increasing support for chemical research matters. Grants, open-access databases, and more collaborative programs between universities and businesses help push scientific projects forward. By making rare compounds more widely available, we strengthen the frontlines in drug discovery and technology development.
Looking Ahead
7-Methylisoquinoline proves important not because it's flashy, but because progress often depends on having the right puzzle piece at hand. Whether it’s the next painkiller, a better pesticide, or a basic dye, this compound’s versatility sets the stage for new discoveries. Investment in infrastructure, education, and open sharing helps maximize the benefit from building blocks like these.
What is the chemical structure of 7-Methylisoquinoline?
Breaking Down the Basics
Take a close look into any field of life sciences, and sooner or later, the significance of nitrogen-containing aromatic compounds shows up. 7-Methylisoquinoline isn’t just another ringed molecule tucked away in a pharmacist’s catalogue—its core structure represents a crucial type within organic chemistry, boasting both versatility and real-world applications. As someone who has worked in chemical research, I’ve seen how molecules like this often underpin early-stage medicinal chemistry, powering everything from new painkillers to trial-and-error processes in drug discovery.
Structure: More than Just a Name
At the center is an isoquinoline scaffold: two fused rings, one benzene ring alongside a pyridine ring. In the case of 7-Methylisoquinoline, a methyl group (CH3) hangs on at the seventh carbon of the aromatic core. This spot gives the compound unique properties, setting it apart from other methylated derivatives. Position on the ring impacts how it behaves; for instance, the seventh carbon site influences not only reactivity but also how the molecule interacts with other substances—think hydrogen bonding, stacking, or fitting into biological “locks.”
Chemically, the formula lands at C10H9N. Picture a flat ring, six carbons in one ring, four plus a nitrogen in the second, with that extra methyl parked on the upper edge (the seventh spot). This orientation drives several downstream effects, like solubility, or how enzymes in the liver might break down this molecule if it passes through a body.
Significance in Real-World Contexts
What does this structure mean beyond a textbook drawing? In pharmaceutical research, such subtle changes as adding a methyl group oftentimes make or break a promising drug’s profile. I remember an old project where swapping a similar methyl group altered the entire behavior of an antihypertensive candidate, flipping how the compound interacted with cell receptors. In flavoring agents and dyes, methylated isoquinolines adjust physical properties like boiling point or the ability to dissolve in different solvents—key in manufacturing safe, stable products.
These seemingly minor changes also cascade into safety profiles or metabolism. A methyl at the seventh position might either protect the molecule from certain enzymes or leave it prone to breakdown by others. This isn’t hypothetical; small tweaks like these often draw attention during early-phase toxicity testing or synthesis planning, such as reducing unwanted side products that complicate scaling a drug for the market.
Opportunities and Responsible Handling
Anyone working with organic compounds knows the value of precision, both in theory and practical application. Understanding 7-Methylisoquinoline’s structure pays off in targeted synthesis—reducing waste, maximizing yields, and supporting green chemistry efforts by choosing the shortest, cleanest pathway to the finished product. Reliable data from research groups around the world, including crystal structure information and reactivity trends, inform these decisions. Literature and peer-reviewed sources play a role here, supporting safe handling, regulatory compliance, and ethical use in laboratory and industrial settings.
7-Methylisoquinoline’s chemistry offers lessons in the importance of detail. The work doesn’t end at knowing a name or recognizing a ring—seeing how tiny adjustments steer a molecule’s fate gives scientists and engineers the toolkit to develop better medicines, safer materials, and more responsible technology.
What are the storage conditions for 7-Methylisoquinoline?
Understanding the Basics: What Really Matters
7-Methylisoquinoline, a compound that often floats through research labs and chemical libraries, comes with its own set of practical storage rules. Anyone who has spent time working with nitrogen-containing aromatics knows: a little common sense—paired with scientific respect—goes a long way. This isn’t just about “best practices;” it’s about protecting your work, your investment, and sometimes your team’s safety.
Stay Cool—Literally and Figuratively
Heat doesn’t care about your deadlines. It speeds up reactions, promotes decomposition, and messes with the purity of pretty much any compound over time. In my early days at the bench, I once underestimated humidity and ended up with compounds that degraded faster than I could log the inventory. It doesn’t take long for 7-Methylisoquinoline to lose its edge if the temperature creeps up. Most sources, including Sigma-Aldrich and official datasheets, recommend room temperature—but in practice, keeping the vial stored away from direct sunlight, heat vents, and those notorious lab window sills works best.
Sealing Makes All the Difference
Oxygen and moisture aren’t friends of many heterocyclic compounds. Over time, air will attack sensitive chemicals slowly but steadily—sometimes robbing you of yield, sometimes tossing unwanted oxidation products into your next synthesis. I learned to always check that the vial cap is screwed tight. Parafilm, though decidedly low-tech, has saved more analytes than most folks admit. Store this stuff under an inert atmosphere if you're holding onto it for months, and always keep open containers capped or in a desiccator. Practical chemistry isn’t just about reaction setups; it’s these small routines that make or break a project.
Keep It Dry—Moisture’s Not Your Friend
Water, even in trace amounts, can render your precious sample into a headache waiting to happen. Dry boxes might seem like overkill, but for compounds intended for sensitive analytical work, you want to minimize exposure to humidity. I’ve worked in basements where humidity ruined entire sample batches. Once, a multi-gram order arrived with the seal half-broken, and in days flat, the compound looked clumpy—no confidence left in its analytical purity. Silica gel packs inside storage cabinets became a lab staple after that.
Label, Track, and Date Everything
Nothing’s worse than reaching for what you think is a fresh reagent, only to find a faded label or a mystery substance decades old. Documenting the receipt date and storage location does more than satisfy compliance; it keeps your workflow smooth. I always add the opening date on each vial—over the years, this habit saved my group from accidental mix-ups. Chemical safety demands discipline, not just paperwork.
Looking Out for Long-Term Success
Effective storage doesn’t need heroic solutions. Clear labeling, tight seals, and a dry, stable environment: these habits add up. In my experience, if you respect these simple principles for 7-Methylisoquinoline, you’ll sidestep most headaches and keep your chemistry moving forward. Safeguard your investment with care, and the science will take care of itself.
Is 7-Methylisoquinoline hazardous or toxic?
Getting Real About Chemical Safety
Learning whether a chemical poses harm takes more than a quick internet search. All too often, chemical names blend together in a haze of unfamiliar syllables. That’s especially true for something like 7-Methylisoquinoline. The name barely rolls off the tongue, but the risks aren’t so easy to shrug off. This is a story about transparency, knowledge, and responsibility.
What Is 7-Methylisoquinoline?
Chemists slot 7-Methylisoquinoline into a much bigger family of isoquinolines. It shows up most as a building block in labs, sometimes in research that explores its potential for new drugs or advanced materials. Most people won’t run into it at home. But researchers, students, and workers in chemical labs need to treat every substance as if it can bite.
Sizing Up the Risks
I’ve looked at dozens of material safety data sheets in past research jobs, and it doesn’t take long to spot trouble. For 7-Methylisoquinoline, official documentation rarely goes deep. There’s a hole in our collective knowledge—at least in public records. The US National Library of Medicine and some specialty chemical providers note that isoquinoline derivatives often cause skin, eye, and respiratory irritation. Some isoquinolines also show up in studies as potential toxins or even carcinogens, but these generally tracked much more common cousins of this molecule.
No major regulatory agency, including OSHA, NIOSH, or the EPA, lists 7-Methylisoquinoline on its hazard sheets as a unique threat. But that shouldn’t > lull anybody into carelessness. Labs always have that old rule: If you don’t know what it does, wear gloves, goggles, a lab coat, and work under a fume hood. Symptoms like dizziness, headache, or nausea may crop up with exposure to chemically related compounds. That gives chemists a pretty strong reason to take safety seriously until hard proof sets minds at ease.
The Gaps in Research
The real story is the uncertainty. Modern chemical databases lean on animal studies or accidental exposure reports to paint a full picture of risk, and there’s barely any such data for this specific molecule. Wait long enough, and someone somewhere might fill in the blanks, but right now, the shade of “unknown” covers a lot.
Why That Matters
People working with chemicals like 7-Methylisoquinoline trust their health to procedures, training, and—in the best cases—clear science. The gaps can put young lab assistants and curious undergrads in a tricky spot. Many accidents don’t happen because a liquid is especially deadly, but because someone underestimates a plain-looking, little-discussed bottle. I’ve seen a careless splash burn a hole in a favorite pair of jeans, and watched as someone learned the hard way to respect even an “ordinary” vial.
What Can Be Done
Researchers could push for more open science by publishing toxicity tests and detailed handling protocols. Transparency expects companies and labs to share not only the warnings but the real experiences—the close calls, the fixing-up, and the tests that turn question marks into clear answers. Universities could do their part by teaching new students to take every chemical seriously, not just the ones with scary names.
Staying safe around 7-Methylisoquinoline boils down to habit. Wear protective gear, label everything, ask questions if the data looks thin. A healthy respect for the unknown brings us closer to safer labs and a world where nobody has to gamble with their health, even around chemicals that rarely make the news.
What is the purity specification of 7-Methylisoquinoline offered?
Purity Specification Shapes Results
Jumping into any lab work, I pay close attention to the purity of the chemicals on my bench. With 7-Methylisoquinoline, most suppliers list the purity at 98% or above, using techniques like HPLC or GC to confirm that level. This number isn’t just paperwork: it has real effects on experiments. Even a percentage point can change the way a reaction unfolds or the reliability of an assay result. In drug research or advanced material synthesis, trace contamination isn’t just a flaw—it’s a risk to the work’s credibility.
Where the Rest of that 2% Hides
Every batch that comes in with “98% purity” leaves scientists eyeing the remaining bits. In my experience, those slivers can be leftover solvents, reaction byproducts, or even environmental contaminants. Sometimes they don’t show up until a critical step—setting off alarms when a chromatography column clogs or a yield drops for no clear reason. So that label is a warning as well as a promise.
Choosing the Right Purity for the Task
Quality standards depend on what you want to achieve. When synthesizing reference compounds, researchers often reach for materials at or above 99% purity. Industrial work, such as producing intermediates for dyes, often runs on less stringent specs, possibly 95% or even lower, if the end product will go through more purification. Cutting corners, though, sometimes brings hidden headaches—a lesson learned after a few ruined batches.
Analytical Backing: Proving the Grade
Suppliers put purity numbers front and center, but real trust comes from asking about the methods behind those numbers. Analytical reports—with results from NMR, HPLC, or MS—offer the transparency customers want. Working in a regulated environment, I’ve seen how vital it is to match not just technical needs but also documentation standards. Compliance teams and auditors always ask for proof, not promises.
Health and Safety Rely on Purity
Impurities aren’t only a technical problem. Some can introduce unknown toxicity or provoke unexpected reactions. In pharmaceutical research, a tiny contaminant could mean the difference between a promising lead and a failed trial. From personal experience, I’ve found that investing in cleaner materials cuts hassle later. Clean chemicals protect both the science and the researchers who handle them.
Improving Access to Better Purity
High-purity chemicals don’t just appear—they need steady investment in manufacturing and processing. Companies that focus on better purification technology set themselves apart and attract more careful buyers. Collaborating with reputable suppliers proved to be a simple way to raise the standard in my own projects. Feedback, clear requests for detailed specifications, and following up on analytical data push the whole market forward.
Next Steps for Researchers and Industry
Laboratories and purchasing departments gain a lot by asking about purity and backing data before the order goes in. Buyer vigilance, tough quality checks, and partnerships with established chemical producers help maintain the reliability of every experiment. The work gets easier when everyone values the same level of care from product design to delivery.
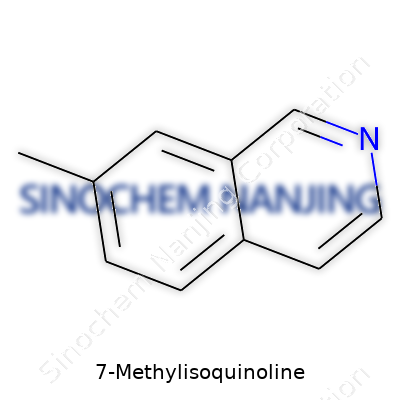
| Names | |
| Preferred IUPAC name | 7-Methylisoquinoline |
| Other names |
7-Methylisoquinoline 7-Methyl-isoquinoline Isoquinoline, 7-methyl- |
| Pronunciation | /ˈsɛvən ˌmɛθɪlˌaɪsəʊkwɪˈniːl/ |
| Identifiers | |
| CAS Number | 1837-58-3 |
| Beilstein Reference | 120924 |
| ChEBI | CHEBI:89672 |
| ChEMBL | CHEMBL434047 |
| ChemSpider | 112679 |
| DrugBank | DB08395 |
| ECHA InfoCard | 13c6f8e6-2e97-4e72-ab3d-17b0db2499a2 |
| EC Number | 7279-21-2 |
| Gmelin Reference | 8217 |
| KEGG | C16237 |
| MeSH | D02.886.590.700.560 |
| PubChem CID | 70129 |
| RTECS number | NL4375000 |
| UNII | 36NRV5E2F2 |
| UN number | UN3439 |
| Properties | |
| Chemical formula | C10H9N |
| Molar mass | 143.19 g/mol |
| Appearance | Yellow to brown liquid |
| Odor | aromatic |
| Density | 1.08 g/cm3 |
| Solubility in water | Slightly soluble |
| log P | 1.92 |
| Vapor pressure | 0.0674 mmHg at 25 °C |
| Acidity (pKa) | 5.14 |
| Basicity (pKb) | 5.10 |
| Magnetic susceptibility (χ) | -73.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.107 |
| Viscosity | 2.1 mPa·s |
| Dipole moment | 1.73 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 176.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -3958.8 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | Precautionary statements: P261, P280, P302+P352, P304+P340, P312 |
| NFPA 704 (fire diamond) | NFPA 704: 2-2-0 |
| Flash point | Flash point: 113°C |
| Autoignition temperature | 480 °C |
| Explosive limits | Explosive limits: 1.2–7.5% |
| Lethal dose or concentration | LD₅₀ (oral, rat): 520 mg/kg |
| LD50 (median dose) | LD50: 320 mg/kg (mouse, intraperitoneal) |
| NIOSH | RN8220000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 100 mg |
| Related compounds | |
| Related compounds |
Isoquinoline 6-Methylisoquinoline 5-Methylisoquinoline 8-Methylisoquinoline 1-Methylisoquinoline 2-Methylisoquinoline 3-Methylisoquinoline 4-Methylisoquinoline |

