7-Amino-3-Chloro-3-Cephem-4-Carboxylic Acid Ethyl Ester: Insights from the Ground
Historical Development
Long before the latest wave of synthetic antibiotics, researchers searched for a backbone both strong and flexible, something that could stand up to the relentless mutations of bacterial resistance. The story of 7-Amino-3-Chloro-3-Cephem-4-Carboxylic Acid Ethyl Ester, a mouthful in name and a heavyweight in drug synthesis, stems from the hunt for better beta-lactam antibiotics. It follows the twisting road of cephalosporin discovery—work kicked off in the middle of the 20th century, after one scientist in Sardinia found a fungus that combated typhoid bacteria. As penicillins faced resistance, chemists started using the cephem ring as a basic building block, making modifications to strengthen its resilience. Before the 1980s, options were limited. After the introduction of the ethyl ester group in this compound, manufacturers gained a versatile intermediate, laying the groundwork for many cephalosporin antibiotics we rely on today in both hospitals and outpatient clinics. Every leap in cephalosporin chemistry has traced back to hard lessons from treatment failures, sepsis cases, and the waves of MRSA reports that drove innovation under pressure.
Product Overview
This compound wears multiple hats in the pharma industry. It belongs to the cephem family, acting as a core intermediate in cephalosporin manufacture, especially those needed to fight resistant bugs. Underneath the chemistry, the product’s key role is offering that 7-amino group for further tweaks—think swapping out molecules to produce different generations of antibiotics. Its stability and reactivity under controlled factory conditions let it act as a launchpad for new drugs. Pharmaceutical companies value this product for the way it bridges early raw materials and the finished antibiotic, offering a reliable starting point they can trust for consistent yields and clean chemical reactions.
Physical & Chemical Properties
In the lab, this substance usually appears as a pale, slightly crystalline powder, with a mild odor that doesn’t quite mask its potency. Solubility remains moderate in water, but better in common organic solvents—ethyl acetate, methanol, dimethylformamide. The melting point sits in the range of 160 to 170°C, and it can undergo hydrolysis if exposed too long to moisture. Chemists watch for its sensitivity to acids and bases; under strong conditions, the compound tends to break down or form byproducts. Its molecular structure, with a bulky cephem ring and an ethyl ester arm, helps explain its reactivity and why it’s preferred in large-scale syntheses compared to plainer alternatives.
Technical Specifications & Labeling
Manufacturers set tight benchmarks for this compound’s purity—often topping 98% by HPLC—since impurities can drag down later drug development stages. Labels typically specify molecular formula, batch number, storage conditions, and a shelf life that hinges on dryness and temperature control. Every box carries hazard symbols due to the potential for skin and eye irritation, and chemical safety data scenes printed out for reference. On shipping manifests, the compound’s weight, origin, date, and even humidity readings show up; customers in Europe, Asia, and the US demand that every gram matches international pharmacopeial standards to guarantee traceability and accountability.
Preparation Method
Manufacturing this compound draws heavily on industrial fermentation and precise chemical transformations. Producers usually start with 7-ACA (7-Aminocephalosporanic acid), itself a major product of Cephalosporium fermentation. Chlorination puts the 3-chloro group in place using agents like thionyl chloride or N-chlorosuccinimide under controlled cooling to avoid unwanted side products. Next, esterification brings in ethanol and acid catalysts, transforming the 4-carboxy group into its ethyl ester form. Each step demands scrupulous solvent selection, exact pH controls, and round-the-clock monitoring. Technicians must filter, dry, and sometimes crystallize the product under vacuum to hit the right particle size and moisture mark, ensuring smooth downstream reactions for drug makers.
Chemical Reactions & Modifications
This chemical’s structure gives plenty of room for derivatization. Medicinal chemists often use that 7-amino group as a handle, attaching bulky or hydrophilic groups to tweak how the future antibiotic moves through the body. The 3-chloro site also attracts nucleophiles—chemists swap in thiols, amines, or oxygen-based groups to create cephalosporins that either better resist bacterial enzymes or reach stubborn infection sites. The ethyl ester at the 4-carboxy position acts as a protective group, letting reactions run without unwanted side reactions at the acid site. A couple of familiar drugs—cefuroxime and cefotaxime—get their backbone from this starting point. In my own lab experience, minor tweaks in the amounts of chlorinating agent or the way the ethyl ester is introduced can swing a batch’s quality from pharmaceutical-grade to barely usable, underscoring how tight process controls matter here.
Synonyms & Product Names
The scientific world often shortens “7-Amino-3-Chloro-3-Cephem-4-Carboxylic Acid Ethyl Ester” to jargony acronyms like “7-ACCE-EE” or variants with numbers and ester modifiers. Commercial producers in the chemical trade call it by designated catalog numbers or internal codes. On bulk shipments, security labels often use terms like ‘Cephalosporin Intermediate C’ or reference the ethyl ester to distinguish it from other esters and salt forms. Regardless of the name, seasoned purchasing officers and pharmaceutical chemists recognize it instantly by its role and unique combination of chemical handles.
Safety & Operational Standards
Anyone who handles this stuff day-to-day knows gloves and goggles aren’t optional. Vapors can irritate respiratory passages, and skin contact can trigger rashes. Standard practice means spill kits, sealed storage in cool, dry rooms, and regular audits to spot leaks or expired batches. In big pharmaceutical plants, every technician gets trained on emergency protocols: eye wash stations, chemical fume hoods, and proper waste disposal. Regulations kicked in over the past decades as industry watchdogs noticed links between cephalosporin manufacturing and environmental residues. Today, strict air and water monitoring, along with routine employee health screenings, form routine checks that keep mishaps rare and manageable.
Application Area
In real-world pharmaceutical cycles, this compound pops up nearly every time a new cephalosporin drug is under development. Research teams treat it as a “workhorse” intermediate, pipelining it into new cephalosporin derivatives targeting respiratory tract infections, sepsis, and even experimental treatments for superbugs. Hospitals rely on these downstream antibiotics for critical care, especially as standard penicillins lose their punch. Beyond infection control, the intermediate sometimes shows up in chemical biology research where teams probe ways to use beta-lactam rings for enzyme inhibitors or diagnostics. I’ve seen its legacy firsthand—new cephalosporin launches in different countries all track their lineage back to this early building block.
Research & Development
Pharmaceutical R&D teams throw plenty of intellectual weight into tweaking this molecule. Recent projects test improved synthetic routes—reducing waste, energy use, and solvent toxicity. Collaboration with green chemistry groups has already shaved off several hazardous steps from traditional recipes. Artificial intelligence and modeling predict how to fit even bulkier, smarter side chains onto the core cephem, seeking drugs that can sidestep resistance mechanisms bacteria spin up with frightening speed. I’ve spoken with junior chemists who see promise in using biocatalysts to replace corrosive chemicals, lowering barriers for smaller firms and regions with limited lab resources. Journals brim with new derivatives every year, pointing to the intermediate’s enduring role as a touchstone for antibiotic innovation.
Toxicity Research
No new chemical ingredient escapes scrutiny, especially one destined for drug manufacturing. Animal studies assessed short-term and chronic exposure, looking for liver or kidney impacts with large doses. Skin tests on lab animals reveal that, at industrial concentrations, the compound can prompt contact irritation, occasionally sensitization if workers get sloppy about protection. Despite this, the bigger concern comes from inhalation over time; dust controls matter just as much as gloves. Regulatory agencies in Europe and North America mandate periodic review, especially as more production sites crop up worldwide. Wastewater studies keep an eye on environmental build-up, since antibiotic intermediates spilling into rivers could encourage resistant bacteria. Ongoing toxicology reviews shape future safety rules, nudging plants toward better air filters and effluent controls.
Future Prospects
Looking down the road, the cephalosporin field feels both pressured and promising. Bacterial resistance pushes researchers to the edge, demanding new antibiotics faster and safer than before. As the core ingredient for so many cephalosporin drugs, 7-Amino-3-Chloro-3-Cephem-4-Carboxylic Acid Ethyl Ester sits at the crossroads of chemical reliability and biological possibility. New manufacturing advances promise to cut costs and boost yields, opening antibiotic production to smaller players in the market. Synthetic biology could soon offer biosynthetic routes for the compound, skipping toxic reagents and cutting hazardous waste to trace levels. The chemical’s flexibility gives medicinal chemists a springboard for new drug designs, hinting at combinations that break out from today’s patterns. A stronger grip on safety, greener process chemistry, and a deeper understanding of the molecule’s reactivity all hint at a future where antibiotics built from this foundation can outpace resistance, rather than just play catch-up.
What is 7-Amino-3-Chloro-3-Cephem-4-Carboxylic Acid Ethyl Ester used for?
The Hidden Gear Behind Broad-Spectrum Antibiotics
Most folks outside the pharmaceutical world won’t recognize the name 7-Amino-3-Chloro-3-Cephem-4-Carboxylic Acid Ethyl Ester. The name sounds intimidating, but the reality is pretty fascinating: this compound keeps hospitals stocked with some of the most reliable antibiotics out there. It’s not found on pharmacy shelves, but chemists trust it as a key ingredient in building cephalosporins, a family of antibiotics that have taken over from old drugs like penicillin in many hospital settings.
Antibiotic resistance is a buzzword for good reason. With bacteria outsmarting older medicines, scientists keep hunting for new ways to shut them down. Cephalosporins step in where penicillins sometimes fail, often dealing with bacteria that just shrug off the older drugs. The secret to turning plain chemical building blocks into hospital-grade antibiotics lies with substances like this one — 7-Amino-3-Chloro-3-Cephem-4-Carboxylic Acid Ethyl Ester. In my time around pharmaceutical research, I noticed that most breakthroughs start with tweaking, cutting, or adding to small pieces like this. Tiny changes, like swapping a chlorine atom or using an ethyl ester, can change how a pill works in the body or how strong it gets at wiping out infection.
Here's the science as plainly as it gets. This compound serves as a “core” structure. Chemists build off it, adding other groups to make final antibiotics like cefotaxime or ceftriaxone. Hospitals rely on those to treat everything from pneumonia to meningitis. The medical journal “Clinical Microbiology Reviews” lists cephalosporins among the top lines of defense for tough infections. Without these core compounds, drug makers face hard times finding powerful tools to fight hospital-acquired infections.
Quality and Trust in the Drug Supply Chain
As governments worldwide press for safer drugs, the chemistry stage matters more than ever. During my own graduate research, I watched what happened when supply chain issues cropped up. An impurity at the base ingredient level forced an entire batch to get scrapped. Makers of this chemical must match strict standards, both for purity and for how the molecules line up. Skipping checks could land a batch of potentially lifesaving ceftriaxone in the trash.
Shortcuts in purity or manufacturing traceability don’t just hurt companies. They slow down drug delivery in real world emergencies. I’ve heard from hospital pharmacists who count on repeat orders showing up on time, every time. That means someone somewhere is double-checking the chemistry and the manufacturing paperwork for every bottle of this starting material. Both labs and government regulators keep up that pressure for trustworthiness — an idea that’s no longer negotiable in today’s world.
Looking for Smart Solutions
One answer that jumped out at me after conversations with process engineers: more green chemistry. Some new research points to safer, less polluting ways to make these core pieces, cutting down on toxic byproducts. Open communication between raw material makers and finished drug producers also makes a strong case for digital “track and trace” tools. These already prove effective in flagging weak points in the supply chain long before pills ever reach patients.
As companies and regulators innovate, they rely on the quiet importance of these critical links. 7-Amino-3-Chloro-3-Cephem-4-Carboxylic Acid Ethyl Ester may never become a household name, but the lives changed by its chemistry speak volumes about the value of getting these base ingredients right.
What is the chemical structure of 7-Amino-3-Chloro-3-Cephem-4-Carboxylic Acid Ethyl Ester?
A Glimpse Into a Peculiar Core of Antibiotic Chemistry
Peering at the chemical makeup of 7-Amino-3-Chloro-3-Cephem-4-Carboxylic Acid Ethyl Ester gives you more than a mouthful—it’s a ticket into the story behind beta-lactam antibiotics. I’ve spent plenty of late nights bent over a lab bench trying to understand why tiny changes to molecules make or break a medication. This compound isn’t a finished antibiotic, but a key building block, sculpted by chemists for years.
Breaking Down the Name for Clues
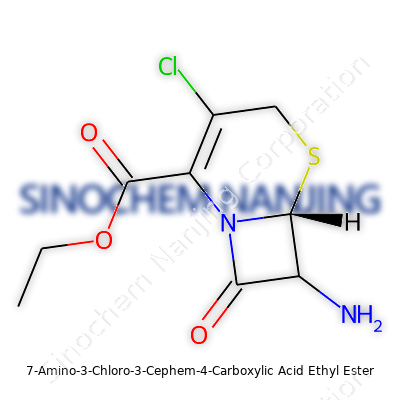
Plenty of folks find chemical names daunting, but every piece gives us a direction. At the core sits “cephem,” a backbone found in cephalosporins, which step beyond penicillin in treating tough bacterial infections. On this core: an amino group at the seventh carbon (7-amino), a chlorine atom at the third (3-chloro), and two more touches—a carboxylic acid group at the fourth position (4-carboxylic acid) and an ethyl ester attached to that acid. Throw all these together, and the resulting structure sets the stage for turning this small molecule into whole groups of useful antibiotics.
Shape Determines Power
Having worked with beta-lactam rings, I know nothing about this class stands alone. The cephem structure forms a four-membered beta-lactam ring fused to a six-membered dihydrothiazine ring. The chimera of an ethyl ester at carbon 4, an amino group at carbon 7, and a chloride atom at carbon 3 brings out a unique chemical signature. Not only does this enable fine-tuning by medicinal chemists, but it also influences how the drug sidesteps bacterial resistance—an issue that’s more urgent than most realize.
Real-World Relevance in Antibiotic Development
Far from being only a classroom curiosity, compounds like this serve as parent molecules, or scaffolds, for cephalosporin antibiotics. Drug developers swap out groups at the seventh and third carbon—making subtle tweaks so the final compound either sticks better to its bacterial targets or dodges attack by bacterial enzymes known as beta-lactamases. I remember reading data showing how one change on that seven position could mean the difference between a useless compound and a reliable hospital antibiotic. This sort of tinkering remains critical in the arms race against superbugs.
Keeping Resistance at Bay
Drug-resistant bacteria do not pause for anyone, and resistance doesn’t take a day off. The ability to synthesize and modify the cephem nucleus means we’re never fighting with blunt tools. Well-constructed beta-lactam scaffolds give chemists the freedom to outmaneuver resistance by shifting groups around until bacteria can’t recognize or neutralize the drug. I’ve watched colleagues adjust functional groups and combine them with new chemical “shields,” leaving bacteria scrambling to catch up.
Challenges and Possibilities on the Bench
Of course, turning this compound into a medicine doesn’t only involve chemistry. Researchers must master purification and scale-up methods that don’t damage the delicate beta-lactam ring, which can break down under stress. The fine balancing act between stability, potency, and manufacturability can stall even the brightest ideas. But by starting with stable, adaptable cores like 7-Amino-3-Chloro-3-Cephem-4-Carboxylic Acid Ethyl Ester, laboratories hold on to the possibility of inventing new life-saving antibiotics—something the world can’t afford to put on pause.
What are the storage conditions for this compound?
Storing Chemicals the Right Way
Anyone who’s dealt with chemical compounds, whether in a lab or at home, knows the importance of proper storage. It isn’t just about following the rulebook. The safety of workers, quality of results, and even the lifespan of the compound depend on where and how a material is kept. Missteps can turn into real hazards — not just lost money, but lives at risk and damaged reputations.
The Science of Stability
Every compound carries its own personality. Some love calm, dry places; others break down in the light. A lot of folks ignore label instructions, thinking one-size-fits-all. Moisture sneaks in, temperature goes up, or sunlight streams through a window, and chemical changes get underway. For example, sodium hypochlorite—better known as bleach—quickly loses its punch in hot, bright conditions. Light sends it from powerful disinfectant to weak saltwater. On the flip side, something like lithium metal gets dangerous if moisture reaches it. Boom. Out in most labs, even steady room temperature swings between 20-25°C. Heating or chillier corners of a room make a difference when weeks or months add up.
More Than Just a Storage Location
Every label often lists the basics: cool, dry place, keep container closed. But experience taught me it matters to do more. Desiccators keep things bone dry. Flame-sensitive compounds do better in metal cabinets, away from electrical sparks or open flames. Volatile organic solvents, such as acetone or diethyl ether, wind up in explosion-proof refrigerators, far from sparks and heat sources that could set off fires or worse.
One bad mix-up stuck with me. A colleague put a peroxide-forming chemical in a clear glass jar, tucked in regular storage. That jar received more sunlight than anyone realized near the window. Tiny crystals started forming on the lid after just a couple of weeks. We caught it in time, but cleaning up peroxide crystals is a nerve-wracking job nobody signs up for twice. You can’t just move chemicals around at random, even if shelf space runs low.
Quality Drops Fast Without Care
Medications, reagents for testing, and even what goes into our food or cleaning products all play by the same rules. Vitamin C, for example, loses strength when exposed to air and heat. That’s why, even at home, keeping vitamins capped tight and away from ovens or bathroom steam gives them a longer shelf-life. Chemists and pharmacists see the difference right on test results: accuracy plummets fast when storage guidelines get ignored. Fake readings cost time, money, and sometimes, health.
Better Solutions Make All the Difference
Fixing the problem means more than building new shelves. Digital temperature and humidity monitors give instant feedback. Color-changing silica gel packs flag rooms getting too damp. Storeroom audits, even quick check-ins every month, catch issues early. Labeling goes beyond writing the name—add dates, include hazard warnings, and move older stock to the front. Everyone from students to industry pros gets a safer workplace and more reliable results.
Genuine expertise grows from a mix of training, sharp senses, and respect for both science and people. Good storage is a habit, not just a rule. Treat every compound like its quirks really matter—because they do. Small steps save resources, prevent health risks, and protect everyone who relies on what comes out of the lab or the warehouse.
Is a certificate of analysis (COA) available for this product?
Digging Into the Details
Standing in a supplement shop or browsing online, it’s easy to get lost in bold promises and flashy packaging. Folks often ask if there’s a certificate of analysis for a product. It sounds technical, but the question points straight at trust. The COA serves as a snapshot from a laboratory: here’s what’s in the product, and here’s proof. It goes beyond glossy advertising and dives into what you’re actually putting into your body or using in your work.
The Reality Behind the Label
Many brands toss around buzzwords—“pure,” “potent,” “natural”—but these claims become empty without hard evidence. I’ve walked into health stores and seen bottles priced five times higher than others, boasting unique benefits. Once I started asking for COAs, the story shifted. The best products put the proof right up front. Others stumbled or shrugged, suggesting their purity is more marketing than substance.
The supplement industry has had its share of scandals. An investigation in 2015 revealed that several major retailers sold herbal supplements that didn’t contain the main ingredients they claimed. This debacle shook public trust and underscored the hidden risks of skipping over real testing. Scientific studies point to consumer safety risks ranging from missing ingredients to dangerous contaminants, especially in supplement markets with less regulation. A COA provides a line of defense—clear results from a reliable lab that confirm what’s inside the bottle.
Why Transparency Builds Confidence
Asking for a COA doesn’t make you a difficult customer—it shows you care. Reputable companies invest in high-quality testing because they stand behind what they make. When a vendor shares a current COA, they help you check for heavy metals, microbes, allergens, or other possible concerns. If a product’s COA isn’t readily available, I don’t buy it. This habit has led me to brands that prioritize honesty, and those brands usually have a loyal following, too.
Transparency fuels a safer market. People want to trust what they buy, especially when health is on the line. Public access to test results helps root out those in it for a quick buck. My own experience has taught me to read the COA—not just look for its existence. Spotting an outdated document, or one missing key information, can be a red flag.
Helping Both Sides—Manufacturer and Consumer
Quality control goes both ways. I’ve worked with small producers who initially hesitated over the cost and hassle of third-party testing. Still, once they made room for it in their process, they noticed fewer complaints and better reviews. They even picked up new wholesale partners that demanded proof of quality. The upfront investment in testing often led to higher sales and a stronger reputation. Regulations from agencies like the FDA and international counterparts increasingly call for documentation anyway, so being proactive just makes sense.
Making Smarter Choices
People deserve the truth about what they’re buying. With stories of tainted products and mislabeled ingredients floating around, a COA has become more than a box to check—it's a right. Anyone can ask for it. Seeing that document lays the foundation for informed choices every time you buy, no matter if it’s a vitamin, food additive, or other product. Demand the proof, read it, and expect brands to back up their promises.
What is the purity level of 7-Amino-3-Chloro-3-Cephem-4-Carboxylic Acid Ethyl Ester?
Why Purity Matters in Beta-Lactam Chemistry
Folks working on beta-lactam antibiotics know the role of pure chemical intermediates. Take 7-Amino-3-Chloro-3-Cephem-4-Carboxylic Acid Ethyl Ester, often called 7-ACCE. This compound plays a huge part in crafting third-generation cephalosporins. If you ask any chemist what makes or breaks a drug’s effectiveness, purity ranks near the top. Even the most innovative process or advanced lab equipment cannot compensate for mediocre raw materials.
Purity Benchmarks and Industry Realities
Manufacturers set benchmarks high for 7-ACCE. Most pharmaceutical suppliers publish purity specifications—look around and you’ll see them list numbers like 98% or even 99%. This isn’t just a badge of pride. High-purity 7-ACCE reduces the risk of side reactions during synthesis. Impurities, even trace solvents or isomeric byproducts, can clog reactors, slow enzyme steps, and drive up purification costs later. Anyone who has spent time in QA labs will recognize the headache of failing a batch on HPLC or LC-MS because something slipped past the checks.
Peeking into published monographs and supply catalogs, 7-ACCE with 98% to 99% purity exists across major suppliers in China, India, Europe, and the US. Documentation often goes further, specifying residue solvent levels, water content, heavy metals, and related substances. Precise numbers are not academic, either. For a drug master file or an ANDA, regulators expect raw materials supporting the declared impurity profile of the final antibiotic.
What Low Purity Can Cost Labs and Patients
I've seen raw 7-ACCE sourced from off-brand suppliers at 95% or below. The cost savings on the invoice shrink pretty fast when downstream yields drop. Lower yields mean more waste, extra cleaning cycles, and sometimes entire batches scrapped. Stories circulate among process chemists about mystery peaks on chromatograms leading to expensive recalls. In the worst scenario, undetected impurities sneak into the medicine itself, raising safety flags or triggering regulatory holds.
How to Ensure High Purity—And Why It’s a Team Sport
It’s tempting to see purity as just the supplier’s responsibility, but oversight goes beyond a purchasing checklist. Analytical teams need validated, sensitive methods—HPLC, NMR, or even chiral chromatography—to pick up not just obvious contaminants but enantiomeric impurities. Pharmacists and formulation scientists should push for certificates of analysis with every lot. I remember working long nights verifying batch records, knowing a slack review could ripple through the entire drug chain. Lab culture matters. One shortcut—from skipping an extra filter step to using tired glassware—can bring avoidable contamination.
Where Solutions Begin: Open Data, Solid Relationships, and Direct Communication
Labs cannot build trust by hiding methods or batch records, and suppliers helping troubleshoot problems prove the most reliable in the long run. Some pharma companies follow up every new source of 7-ACCE with comprehensive testing before scaling up. Regular audits, transparent supplier relationships, and open access to test results pay off.
Today, as regulatory bodies like the FDA and EMA demand not just high purity but traceable proof, it’s time to move beyond just chasing numbers on a spec sheet. Maintaining 7-ACCE at 98% or above purity secures clinical outcomes and protects everyone along the medicine’s journey—from the chemist at the bench to the person relying on a finished antibiotic at their pharmacy.
| Names | |
| Preferred IUPAC name | ethyl (6R,7R)-7-amino-3-chloro-7,8-dihydro-6H-cephem-4-carboxylate |
| Other names |
7-ACA Ethyl Ester Ethyl 7-amino-3-chloro-3-cephem-4-carboxylate |
| Pronunciation | /ˈsɛv.ən əˈmiː.noʊ θri ˈklɔː.roʊ θri ˈsɛ.fɛm fɔːr kɑːrˈbɒk.sɪl.ɪk ˈæs.ɪd ˈiː.θəl ˈɛs.tər/ |
| Identifiers | |
| CAS Number | 68401-81-0 |
| 3D model (JSmol) | `3Dmol.dataURI("data:model/xyz;base64,CgogICAKMS45MzAwIDAuNjEyIDAuMDU3IE5IMwouMjc4LjEyMjEyIEsuMjQzLjAwMzAgQ0guNDM3IC4zNDcgQ0wuNDY2IC40MzUgR0UuMTIyIC4wNTUgQ0wuNDM4LjQ2OCBDTC40NjIgLjQyOSBLS0wuMTU1IC4wNTUgQ0guNTE3Ti40MzUgS0kuMjI3IC4xMjEK")` |
| Beilstein Reference | 1627001 |
| ChEBI | CHEBI:131918 |
| ChEMBL | CHEMBL1231157 |
| ChemSpider | 2299324 |
| DrugBank | DB08226 |
| ECHA InfoCard | ECHA InfoCard: 100.108.123 |
| EC Number | 61969-27-1 |
| Gmelin Reference | 132197 |
| KEGG | C06835 |
| MeSH | Demiester 7-Amino-3-Chloro-3-Cephem-4-Carboxylic Acid Ethyl Ester does not have a specific MeSH (Medical Subject Headings) term assigned. If relevant, the closest MeSH heading might be "Cephalosporins". **string:** "Cephalosporins |
| PubChem CID | 10430760 |
| RTECS number | AT9652000 |
| UNII | 5Y0D8KUO3C |
| UN number | 3276 |
| CompTox Dashboard (EPA) | DTXSID20819948 |
| Properties | |
| Chemical formula | C11H13ClN2O3S |
| Molar mass | 401.84 g/mol |
| Appearance | White or almost white crystalline powder |
| Odor | Odorless |
| Density | 1.56 g/cm³ |
| Solubility in water | Slightly soluble in water |
| log P | 0.5 |
| Acidity (pKa) | 2.5 |
| Basicity (pKb) | 11.03 |
| Magnetic susceptibility (χ) | -47.2×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.639 |
| Dipole moment | 3.47 Debye |
| Thermochemistry | |
| Std enthalpy of formation (ΔfH⦵298) | -204.5 kJ/mol |
| Pharmacology | |
| ATC code | J01DB53 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation, may cause respiratory irritation |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS05, GHS07 |
| Signal word | Warning |
| Hazard statements | Hazard statements: H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P272, P273, P280, P302+P352, P305+P351+P338, P308+P313, P332+P313, P337+P313, P362+P364, P501 |
| LD50 (median dose) | LD50 (median dose) of 7-Amino-3-Chloro-3-Cephem-4-Carboxylic Acid Ethyl Ester: "LD50 > 5000 mg/kg (oral, rat) |
| NIOSH | AN9926000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | '0-8°C' |
| Related compounds | |
| Related compounds |
7-Amino-3-chlorocephalosporanic acid Cephalexin Cephalosporin C 7-Aminocephalosporanic acid (7-ACA) Cefadroxil |

