6-Methylisoquinoline: More Than Just a Chemical Compound
Historical Development
The story of 6-Methylisoquinoline goes back over a century, rooted in the larger field of heterocyclic chemistry that shaped how we look at the molecular world today. Isoquinoline structures first showed up through coal tar distillation in the late 1800s, when chemists were eager to carve up complex mixtures and understand their building blocks. 6-Methylisoquinoline came out of this wave of discovery, a small change to the basic isoquinoline scaffold that ended up offering a surprising range of utility. Growing up in a family of pharmacists, I watched generations fascinated by how small tweaks to a molecule could translate to big changes in performance—whether for a pharmaceutical task or industrial use. As years passed, researchers shifted from extraction to lab-based synthesis, opening the door to purer and more practical forms.
Product Overview
6-Methylisoquinoline doesn’t make headlines, but dig into research papers and chemistry labs, and you’ll find it quietly powering work in pharmaceuticals, materials science, and even dyes. The reason lies not only in its ring structure but in how that single methyl group changes its behavior compared to its parent isoquinoline. It’s a perfect example of how a little difference can make big ripples across industries—from the search for new medicines to the push for smarter materials. Most people never give much thought to the molecules behind the products they rely on, but for scientists in drug synthesis or pigment creation, this compound has earned its keep across decades of innovation. I’ve seen it used to build blocks for alkaloid derivatives, which often wind up as lead compounds for testing new biological activities or as parts in electronic materials.
Physical and Chemical Properties
Take a vial of 6-Methylisoquinoline off a chemistry shelf, and you’ll find a mostly colorless, oily liquid with a slightly sweet and pungent smell. Its boiling point usually slips just past 230°C, which puts it in a category of fairly stable compounds that don’t evaporate with a quick stir. The extra methyl group doesn’t sound like much, but it increases lipophilicity and changes how the molecule slides into different chemical reactions. My colleagues and I have noticed its relatively high stability in storage, which saves headaches for long-term research projects. The molecule’s basic nitrogen atom gives it moderate solubility in polar organic solvents, which lets chemists play with it in a range of synthetic setups.
Technical Specifications and Labeling
Chemists buy 6-Methylisoquinoline in amber glass bottles, often labeled with CAS number 1837-58-7. Purity matters in R&D, so a certificate of analysis is usually tucked in with the shipment. Attention to details like residual solvents and trace metals makes a difference, especially for pharmaceutical research, where contamination can ruin a whole run of tests. I learned this lesson the hard way, sifting through test results blaming a batch’s off-spec behavior on the hidden impurities. Over the years, regulatory standards—especially in Europe, the US, and Japan—have gotten stricter about what’s allowed in research-grade chemicals, prompting suppliers to keep their processes transparent and documentation thorough.
Preparation Method
Synthesizing 6-Methylisoquinoline starts with either toluene or methyl-substituted benzene derivatives, going through well-trodden routes like Bischler-Napieralski cyclization or Pomeranz-Fritsch isoquinoline synthesis. Each of these methods takes a differently substituted amine or aldehyde through a series of steps to close the ring and install that crucial methyl group at the right spot. What fascinates me is how skilled hands can tweak these reactions to favor the specific 6-position, avoiding byproducts that pop up elsewhere on the ring. I remember a lab supervisor drilling into us the importance of temperature control and careful workup—not only for getting high yield but also for taking home something clean enough to launch into the next synthetic stage.
Chemical Reactions and Modifications
6-Methylisoquinoline acts as a flexible platform for chemical modifications. Its nitrogen atom invites nucleophilic substitution, and the methyl group opens doors to oxidation, halogenation, and coupling reactions. These modifications make it possible to build out whole families of derivatives, which becomes especially important in drug discovery or materials work. Over the past decade, scientists have found clever ways to use it as a precursor for making diazonium salts, hydrogenated derivatives, and even linking larger pharmaceutical fragments. I’ve sat in on enough brainstorming sessions to hear stories of failed coupling reactions turning into the starting points for even bolder approaches—proof that this isn’t just a one-note compound but a real workhorse for synthetic ingenuity.
Synonyms and Product Names
You’ll find it under several names, from 6-Methyl-isoquinoline to 6-Methylquinoline or 6-Methyl-2-benzopyridine. This sometimes confuses new lab techs searching for the right bottle, but old hands learn to treat chemical catalogs and scientific papers with a careful eye for synonyms. As more research goes digital, chemists rely on identifiers like SMILES or InChI Keys to make sure what they’re ordering matches what their experiment calls for. Early in my career, I learned not to trust a label alone—the variety of trade names, spelling variants, and regional preferences can send a project off track unless you double-check molecular structures.
Safety and Operational Standards
Working safely with 6-Methylisoquinoline means respecting its chemical nature. This compound has an acute vapor, and like most heterocyclic aromatics, it needs good ventilation and protection against skin or eye exposure. In my experience, a good fume hood and gloves are non-negotiable, given the compound’s tendency to cause irritation. Waste handling draws on established industry standards—using dedicated containers and attention to compatibility, as reactions with acids or oxidizers can create unexpected hazards. Safety data sheets keep folks grounded in the basics, but in a busy lab setting, the living memory of close calls and best practices often defines real operational standards even more than what’s printed on a label.
Application Area
The full value of 6-Methylisoquinoline shows itself through its diverse uses. Researchers treat it as a building block in synthesizing pharmaceutical intermediates, such as precursors for alkaloid-based drugs. Teams working in agrochemicals have turned to it for developing pesticide derivatives, while pigments and dyes manufacturers find its framework ideal for color-stable compounds. Electronics researchers see promise in the molecule’s stable aromatic system for creating advanced materials, including some used in OLEDs and organic semiconductors. Over the years, I’ve also seen it surface in experimental polymer chemistry and in testing anti-cancer or neuroactive compounds, a testament to its reach across many specialties.
Research and Development
Academic and corporate labs keep circling back to 6-Methylisoquinoline for new research. Drug development pipelines have cast a fresh eye on its possible role in synthesis, looking to leverage isoquinoline frameworks for candidate molecules targeting everything from infectious diseases to inflammation. Materials scientists are getting more creative with how they tie these compounds into frameworks for light-emitting structures and sensors. Data mining journal databases shows a clear uptick in papers exploring both fundamental chemistry and applications—driven both by the demand for sustainable synthesis routes and by regulatory pressure to find greener solvents and processes. I see colleagues exploring ways to cut down the number of steps in its making, or swapping out hazardous reagents, finding efficiency gains that are as much about practical problem-solving as they are about idealistic "green chemistry" principles.
Toxicity Research
Scrutiny over toxicity has ramped up along with the compound’s broader use. Animal studies have helped map out its acute and chronic toxicity, revealing mostly moderate risk profiles at laboratory concentrations. The methyl group seems to draw little additional hazard compared to isoquinoline itself, but anyone working with it still faces the risk of skin, eye, or respiratory irritation. Chronic exposure through improper handling can’t be brushed off, given evidence of bioaccumulation in environmental systems. From my own stints working closely with such compounds, the key is always paying attention to updated literature, watching for signs of regulatory reclassification, and never falling into comfortable routines that ease up on safety standards.
Future Prospects
Future innovation pushes the boundaries set by compounds like 6-Methylisoquinoline. I’ve seen predictions placing it at the core of new antiviral and anticancer drug research, and materials scientists seem set on using its backbone for next-generation organic devices. The quest for more sustainable and less wasteful manufacturing processes could open fresh doors, as pressure grows on the chemical industry to leave a smaller environmental footprint. I expect younger chemists, equipped with better computational tools and new types of automation, to keep expanding what’s possible with this underappreciated but highly adaptable molecule. The way things are going, 6-Methylisoquinoline looks set not to fade quietly into the background, but to continue offering up new surprises for those willing to experiment and dig deeper.
What is 6-Methylisoquinoline used for?
Chemists' Quiet Helper
6-Methylisoquinoline rarely grabs headlines, yet this compound quietly powers more than a few breakthroughs behind the scenes in labs. You find it in the toolbox of synthetic chemists who are working to build other, more complex molecules. With its distinct structure, this chemical acts as a backbone for developing bigger compounds, from drug candidates to new polymers. It doesn’t show up in your daily life the way aspirin or vitamin C might, but some of the medications and materials we use start with this little piece of chemistry.
Key Ingredient in Medicinal Chemistry
Pharmaceutical researchers frequently look toward substances like 6-Methylisoquinoline when designing new drugs. Its ring structure offers a stable foundation, making it easier to add other pieces and create molecules that might one day treat illnesses. Many cancer therapies and treatments for infectious diseases trace their roots to the isoquinoline family. In my own chemistry classes, projects involving these building blocks gave students a sense of how one small tweak can change the action of a medicine entirely.
Role in Dye and Material Production
This compound also finds its way into chemical processes that lead to pigments and specialty materials. Textile dyes and even organic electronic devices sometimes owe part of their properties to derivatives of 6-Methylisoquinoline. In factories, workers blend it with other ingredients to tailor the color or functionality of products. The fact that you can change it slightly to get something entirely new gives chemical makers a lot of flexibility.
Research Applications on the Rise
Scientists have not stopped exploring what else these molecules can offer. The search for greener chemistry often involves tweaking structures like this one to make them less harmful to the environment or easier to break down after use. For students stepping into the world of research, working with a reliable platform such as 6-Methylisoquinoline opens doors to both traditional and cutting-edge projects—think new antifungal options, or advanced coatings for sensors.
Challenges and Next Steps
Despite its value, access to 6-Methylisoquinoline sometimes gets tricky due to limited suppliers or shifting regulations about precursors. Safer handling and ethical sourcing of starting materials will keep getting more attention as demand in research and manufacturing grows. I’ve seen discussions at academic conferences where supply chain transparency comes up regularly, pushing teams to either develop alternatives or push for clearer production standards.
Solutions and Moving Forward
Collaboration between companies, regulatory agencies, and the scientific community could smooth out some of these bumps. More shared data on sourcing and environmental impact supports smarter choices all the way down the line. As for chemists and students, learning responsible use and disposal of this compound not only helps protect the environment but also ensures continued research benefits. I’ve always found that promoting open dialogue and practical training produces better results than locking down access or hiding methods.
Looking Past the Molecule
What matters most ultimately extends beyond the formula itself. 6-Methylisoquinoline, like so many chemicals, tells a story about the power—and responsibility—in discovery. Supporting both innovation and stewardship stands at the core of making safer drugs, smarter materials, and a cleaner future. Keeping an eye on both the science and the ethics protects not only human health, but also the potential for chemistry to solve tomorrow’s problems.
What are the physical and chemical properties of 6-Methylisoquinoline?
The Structure Behind the Name
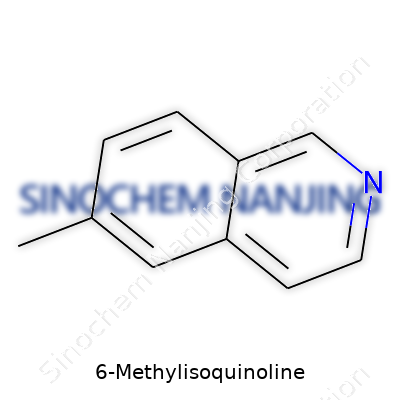
6-Methylisoquinoline draws its identity from a simple chemical twist: it’s an isoquinoline ring with a methyl group stuck at the number six spot. To chemists, this seemingly small modification means a heap of changes to how the molecule performs. Anyone who has ever smelled an aromatic chemical, mixed a batch in the lab, or watched a substance change color under heating knows how those little shifts alter everything from smell to melting point.
Physical Properties That Count
Old-school chemists would spot 6-Methylisoquinoline by its look—a pale-yellow crystalline powder, tucked away in small vials on a shelf. Its melting point hovers close to 38–42 °C. That might not seem impressive if you’re used to icy winters or scorching tea kettles, but it tells you a lot: the compound slips from solid to liquid with just the warmth of your hand. The boiling point clocks in at about 256–257 °C, so it needs serious heat to vaporize.
Relative density gives a clue about how it stacks up in a mix—1.05 g/cm³ at room temperature, which means it’s only a hair heavier than water. The pale color and slightly earthy, organic smell probably wouldn’t be out of place in some sharp-edged old textbooks. That odor, by the way, reminds me a bit of how libraries used to smell, back before everything went digital.
Chemical Behavior in Practice
This methyl group on the sixth position doesn’t just sit there as window dressing. It tweaks the electron cloud, changing reactivity in organic reactions. 6-Methylisoquinoline can jump into a range of transformations: Friedel–Crafts acylation, electrophilic substitutions, and oxidation-reduction reactions, to name a few. In the right hands, and with the right knowledge, it steps up as a building block in bigger jobs, like pharmaceuticals or specialty dyes.
On the solubility front, toss 6-Methylisoquinoline into water and not much happens—it barely dissolves. But drop it into organic solvents—think ethanol, chloroform, or ether—and it blends right in. That’s no small matter in a lab, where extraction techniques live and die by what dissolves and what won’t. It cooks up well under standard lab conditions, and doesn’t fuss or fizz unless provoked by serious acids or oxidants.
Safety Matters and Real-World Concerns
Every chemist learns early to respect their tools. 6-Methylisoquinoline follows the typical pattern for nitrogen-based aromatics. If you work with it, don’t ignore your gloves or fume hood. Skin and mucous membranes can get irritated, and any inhalation is best avoided. There’s no household use here; its playground is the research bench and the shop floor.
Years of study have shown that these heterocyclic compounds can poke at biological receptors. Some analogues serve as drugs, others as potential toxins. No surprise—handling and disposal rules for 6-Methylisoquinoline match up with its status as a specialty chemical, not a common industrial feedstock. I remember carefully labeling similar bottles in grad school, knowing a small spill could lead to a ruined experiment or an uncomfortable rash.
Where Chemistry Meets Utility
Many researchers chase the isoquinoline core because it unlocks new directions for drug discovery, pest control, and pigment creation. Push a methyl group onto position six and a molecule can block an enzyme, color a dye, or step into some new role after a few quick modifications. In competitive industries like pharmaceuticals, knowledge of these fine property differences saves time and resources.
There’s always room for better methods: green chemistry can cut down on the use of toxic reagents and improve yields. Synthesis from sustainable feedstocks, enhanced purification using chromatography, and using computational models to predict reactivity can streamline how scientists put 6-Methylisoquinoline to use.
Is 6-Methylisoquinoline hazardous or toxic?
Looking at the Hazards: Facts and Realities
6-Methylisoquinoline probably isn’t a household name. Still, it lurks in the background of industrial labs, chemical manufacturing, and a handful of specialized applications. The bigger question is: Does this chemical cause harm?
People in research and manufacturing often handle chemicals that come with long data sheets and complicated hazard stamps. I’ve spent years reviewing, storing, and working with both benign and harmful substances. Materials that seem obscure at first glance can sometimes pack real punch—so not underestimating them is key.
What Studies Show About Toxicity
Let’s get right to it: 6-Methylisoquinoline isn’t as deeply studied as many common solvents, but the basic chemical backbone—isoquinoline—rings a few bells in toxicology circles. Some studies connect isoquinoline derivatives to possible risks, especially with long-term exposure or high doses. Animal data on similar compounds reveal depressant effects on the central nervous system and some liver stress when ingested in large quantities. With 6-Methylisoquinoline, precise human data is hard to pin down, since it falls into a category of “use with caution, handle with sense.”
A few chemical safety resources point to signs of skin and eye irritation. And inhaling its dust or vapor could lead to headaches, nausea, or worse if you work with it day after day. Emergency response guides urge quick action if someone gets splashed or breathes too much in. These aren’t signs of an innocent substance.
Comparisons and Proper Handling
If you compare it to known offenders like benzene or formaldehyde, 6-Methylisoquinoline doesn’t make health headlines. But nobody should take its risk lightly. Most manufacturers involved in chemical synthesis advise gloves, goggles, and a fume hood. I’ve seen people ignore that advice and get rashes or spend hours dealing with lab headaches that disrupt concentration.
Reviewing standardized sources—like the European Chemicals Agency (ECHA) and the National Institute for Occupational Safety and Health (NIOSH)—gives the same message: no casual exposure, keep it contained, and treat spills with full PPE. These guidelines reflect decades of lessons from chemical incidents, even if 6-Methylisoquinoline hasn’t caused famous disasters on its own.
Why Transparency and Training Matter
Many chemical incidents happen when people don’t know what they’re dealing with. Labels might slip off bottles. Older labs sometimes keep mystery jars with faded writing. That’s where accidents find their opening. I urge every lab to maintain up-to-date inventories and hazard sheets—never assume a rare compound is less dangerous just because few folks use it daily.
Trust in the safety information provided by regulatory bodies serves people better than guesswork. I’ve called poison control after someone spilled a less familiar solvent, only to realize the incident could’ve been avoided by simple attention to documentation and basics like gloves and working in a fume hood.
Alternatives and Moving Forward
Anyone searching for safer alternatives should start with the use case. Can a similar reaction proceed just as well with a less hazardous compound? Green chemistry principles suggest swapping in chemicals with clear safety records. Every industry has its own constraints, but the question deserves time before every order and every experiment.
6-Methylisoquinoline isn’t the worst chemical to pass through a lab, but its risks are real. Handling it with respect and vigilance, backed up by science-based protocols, protects workers and the communities around them. At the end of the day, the story always circles back to training, transparency, and the willingness to ask questions before opening that next bottle.
How should 6-Methylisoquinoline be stored?
Understanding the Real Risks
Anyone who’s spent time around chemical supply rooms knows the work doesn’t stop at the label. Dealing with compounds like 6-Methylisoquinoline isn’t just about keeping things neat; it’s about keeping everyone safe. This chemical, often found in fine chemical labs and occasionally in pharmaceutical research, carries more weight than its modest name lets on. It has a sharp odor and vapor that can catch the sinuses. The science says it may irritate eyes and skin, and researchers have logged its flammability. Once you open that bottle, you can’t just tuck it anywhere.
Choosing the Right Storage Location
A cool, dry, and well-ventilated space remains the best bet. 6-Methylisoquinoline won’t thank you for leaving it near heat sources, sunlight, or moisture. Throw it in with oxidizers, and you’ve got a disaster waiting to happen. Flammable liquids cabinet? That’s where it belongs, not just because of codes on the wall, but because anyone who’s seen a minor spill turn into hours of cleanup knows how quickly these vapors stretch through a room. My own run-in with improperly stored chemicals in grad school proved the wisdom of shelving what reacts or volatilizes out of sight and away from foot traffic.
Container Choice and Chemical Integrity
The bottle the manufacturer provides doesn’t only protect the contents; it also helps everyone recognize danger at a glance. Glass containers with secure, sealed tops keep vapors contained. Metal might corrode, and plastic sometimes interacts with organics, so if the shipping container is glass, stick with it. Tamper-evident seals or parafilm matter more than they seem. Over time, tight seals mean less evaporation and fewer chances for someone to catch a whiff and end up coughing.
Labeling the container with the date you opened it brings another layer of defense. Chemicals don’t live forever, and degradation might mean new risks or even false results in a synthesis. In my experience, dated labels saved coworkers from reaching for aged chemicals that had lost their punch or picked up impurities.
Protecting People, Not Just Paperwork
Regulations matter, yes. But following best practices saves more than just compliance headaches. As someone who’s seen too many near misses, it’s clear that no amount of paperwork beats hands-on vigilance. Use appropriate signage. Store 6-Methylisoquinoline where only trained folks go. Emergency eyewash and spill kits, at arm’s reach, turn accidents into manageable blips instead of full-blown incidents.
Keeping an Eye on Inventories
Small steps like regular inventory checks keep chemicals from becoming ghosts on the shelves. Chemicals tucked away and forgotten breed all sorts of trouble, from expired stock to containers that build pressure or leak. Running a simple audit every couple of months, combined with a rotation system (use the oldest container first), keeps everyone honest. The fewer forgotten bottles, the lower the risk.
Building Good Habits
Sharing knowledge among coworkers leads to safer handling. Telling stories from the field makes a point better than just rattling off rules. Getting real about mistakes and what close calls taught us reinforces how even minor lapses can have big consequences.
Final Thoughts
6-Methylisoquinoline isn’t something to treat casually. With the right space, solid containers, sharp labeling, and steady routines, the lab stays safer for every pair of hands and eyes working within it. Experience, not just instruction, forms the best foundation for chemical storage—one lesson at a time.
What are the available packaging sizes for 6-Methylisoquinoline?
Choosing a Reliable Supply
For anyone working in chemical research or pharmaceutical development, the details around sourcing reagents like 6-Methylisoquinoline often matter as much as the actual benchwork. This aromatic heterocycle draws attention for its potential in alkaloid synthesis, drug discovery, and advanced materials. Yet, the form it arrives in—the bottle you open, the drum you haul into the lab—shapes the pace and cost of each project.
Available Packaging Options in Today’s Market
Manufacturers usually target multiple user groups for 6-Methylisoquinoline, from bench chemists to scale-up facilities. Smaller academic labs order the substance in amber glass bottles of 5 grams, 10 grams, or 25 grams. These sizes match the scale of pilot reactions and give easy storage in lab drawers without hogging shelf space. I've learned over the years how a 5-gram bottle can sometimes keep an entire summer project afloat, sparing the hassle of managing leftovers or disposal.
Once synthesis moves to a larger stage, suppliers provide 100-gram, 250-gram, and 500-gram bottles. These mid-range packaging formats work well for repeated pilot runs, chemical intermediates, or contract research where cost and minimizing waste trade off with lab throughput. In my own work, the jump from 25 grams to a 100-gram bottle serves as a clear signal—a project is gaining momentum, and so is its need for reliable sourcing.
Industrial and Bulk Packaging for Scale-Up
Producers don’t just stop at bottles. Companies handling process chemistry or supplying fine chemicals order 6-Methylisoquinoline in 1-kilogram or 5-kilogram containers. These larger tubs typically use high-density polyethylene with tight-fitting caps to combat both moisture and contamination. For kilogram quantities, the stability of the packaging becomes a concern, particularly around air exposure and ease of dispensing. Factories dealing with even bigger needs often request metal drums, lined fiber containers, or custom tote sizes up to 25 kilograms or more. By speaking with purchasing managers and quality assurance staff, I’ve seen how a few tweaks in container choice can reduce product loss and keep things under budget.
There’s also the growing practice of offering custom packaging. Research consortia and pharmaceutical partners sometimes negotiate private-label solutions if their protocols demand unique container types, inert atmosphere fills, or special labeling for regulatory compliance. These arrangements help organizations handle volatile or high-purity stocks with more confidence, with logistics tailored to shipment cycles and on-site storage rules.
Safety, Traceability, and Compliance
Size isn’t the only issue at stake. Labels on 6-Methylisoquinoline packaging offer batch numbers, shelf life, hazard warnings, and regulatory citations. Overlooking these details invites supply chain headaches or even safety incidents. During my time in chemical inventory management, I spotted firsthand how many issues can be traced back to ambiguous labeling or mismatched shipping documents. Having a clear record of packaging types and traceability codes has helped many labs avoid mix-ups and maintain compliance during audits.
Weighing Up Your Options
Demand for 6-Methylisoquinoline spans several fields, so the variety of packaging available helps researchers and manufacturers stay agile. The right size means less waste, more predictable costs, and a workflow tuned to the realities of day-to-day science. For those unsure about which option fits best, speaking directly with suppliers helps clarify lot availability, lead times, and any extra services like inert-filling or post-shipment support. It all comes down to understanding your project’s timeline, scale, and compliance requirements.
| Names | |
| Preferred IUPAC name | 6-Methylisoquinoline |
| Other names |
6-Methylisoquinoline 6-Methyl-isoquinoline |
| Pronunciation | /ˌsɪksˌmɛθɪlˌaɪsəʊkwɪˈnoʊliːn/ |
| Identifiers | |
| CAS Number | 1849-30-1 |
| Beilstein Reference | 1462348 |
| ChEBI | CHEBI:89679 |
| ChEMBL | CHEMBL15636 |
| ChemSpider | 15385 |
| DrugBank | DB02174 |
| ECHA InfoCard | 100.068.040 |
| EC Number | 220-706-2 |
| Gmelin Reference | 8886 |
| KEGG | C14533 |
| MeSH | D008920 |
| PubChem CID | 70171 |
| RTECS number | NL3675000 |
| UNII | R3G7V88V6F |
| UN number | UN3439 |
| CompTox Dashboard (EPA) | 6YY5UWM2BL |
| Properties | |
| Chemical formula | C10H9N |
| Molar mass | 143.19 g/mol |
| Appearance | Light yellow to yellow crystalline powder or solid |
| Odor | Aromatic |
| Density | 1.08 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 2.38 |
| Vapor pressure | 0.0467 mmHg at 25°C |
| Acidity (pKa) | 5.14 |
| Basicity (pKb) | 6.04 |
| Magnetic susceptibility (χ) | -77.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.103 |
| Viscosity | 0.962 cP (20°C) |
| Dipole moment | 2.60 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 322.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 93.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3756 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H302, H315, H319, H335 |
| Precautionary statements | Precautionary statements: P261, P264, P271, P301+P312, P304+P340, P312, P330, P405, P501 |
| NFPA 704 (fire diamond) | NFPA 704: 2-2-0 |
| Flash point | 113°C |
| Autoignition temperature | 898°F (481°C) |
| Explosive limits | Explosive limits: 1.1-6.7% |
| Lethal dose or concentration | LD50 (oral, rat): 380 mg/kg |
| LD50 (median dose) | LD50: 516 mg/kg (rat, oral) |
| PEL (Permissible) | Not established |
| REL (Recommended) | 30 mg |
| Related compounds | |
| Related compounds |
Isoquinoline 5-Methylisoquinoline 7-Methylisoquinoline 8-Methylisoquinoline 6,7-Dimethylisoquinoline |

