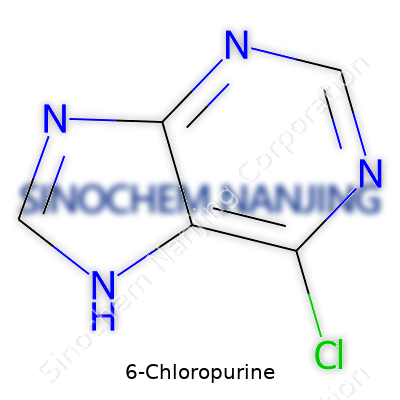
6-Chloropurine: History, Properties, and Applications
Historical Development
Scientists have kept a close relationship with purines since the late 1800s, and 6-chloropurine has played a regular supporting role as researchers explored nucleic acid chemistry. In the early days, chemists like Fischer and Traube opened the doors to purine synthesis, often by methods that would be called impractical now. The deliberate introduction of a chlorine atom at position six on purine came about as researchers searched for new ways to tweak nucleic acid bases, hoping to better understand DNA, RNA, and eventually antiviral and anticancer pathways. Early studies saw 6-chloropurine as a simple intermediate, but it quickly earned a reputation for versatility, becoming a building block for more complex nucleoside analogues. This compound’s roots stretch directly into the heart of medicinal chemistry, linking classic bench science with current research in pharmaceuticals.
Product Overview
6-Chloropurine falls into the category of purine derivatives, setting itself apart with a chlorine atom at the six position. As a fine, off-white crystalline powder, it doesn’t grab a lot of attention at first glance, but don’t let that plain surface fool anyone. Chemists value this material for its ability to participate in critical reactions without unnecessary drama. Take a look at the biopharma world and even advanced organic synthesis: this compound keeps showing up. Researchers rely on it to construct nucleoside analogs that go on to test the boundaries in antiviral, anticancer, and agricultural chemistry. The versatility here stems from a simple, logical structure, giving it an edge in custom drug synthesis and innovative molecular design.
Physical & Chemical Properties
A clear set of traits define 6-chloropurine on the bench. It presents as a fine, often slightly yellowish or off-white crystalline solid. Melting usually lands around 215–217°C. Its odor doesn’t stand out, which in the chemical world can be more blessing than curse. Solubility remains low in cold water, but you’ll see more success in warm polar solvents or with basic pH, making it manageable for those adjusting reaction conditions. Chemically, the presence of the chlorine atom activates the ring toward nucleophilic substitution, which simplifies the path to a range of derivatives. It remains stable under regular lab conditions but can degrade with excessive heat or strong acid. Store it away from moisture, and it will behave for extended periods.
Technical Specifications & Labeling
Product lots marked as 6-chloropurine should show purity levels of not less than 98% by HPLC, with many suppliers routinely testing for common organic impurities and residual solvents. Water content typically sits at less than 0.5%, confirmed by Karl Fischer titration or similar. Identification relies on melting point, NMR, and HPLC-MS spectra. Most commercial packaging includes both chemical and hazard labeling in compliance with GHS and local regulations, and material safety data sheets accompany shipments. Users in regulated labs often scan the batch certificate for any lot-specific variations in purity or physical form, not so much out of concern, but to fine-tune protocols internal to their research.
Preparation Method
The main route to 6-chloropurine starts from hypoxanthine or directly from purine. A common laboratory method relies on chlorination of the appropriate precursor, usually with reagents like phosphorus oxychloride (POCl3) or thionyl chloride. Reaction conditions need controlling—not too much moisture, and don’t push the temperature too high, or you risk side products. More recent technical advances include catalytic variants and flow chemistry adaptations, aiming to increase yields while reducing environmental impact and operator exposure. On a larger scale, plants often use improved solvent recovery and byproduct recycling to keep costs down while reducing waste, an approach rooted in decades of chemical manufacturing lessons.
Chemical Reactions & Modifications
This compound’s defining chlorine atom makes it a go-to substrate for nucleophilic aromatic substitution. Researchers swap in a variety of amine, alkoxide, or thiol groups at C-6 to build nucleoside analogs, pharmaceuticals, and research probes. It’s a favorite jumping-off point for drug discovery work because the reaction conditions don’t need to get extreme, and the by-products remain easy to separate. Even biochemists use it for site-directed mutagenesis in oligonucleotide synthesis. Because 6-chloropurine’s structure keeps the C-6 position reactive while preserving the core purine ring, it has celebrated a long run as a foundational intermediate in medicinal chemistry.
Synonyms & Product Names
Different suppliers and researchers often refer to 6-chloropurine using names like 6-chloro-7H-purine, 6-chloropurin, or 6-CP. In some environments, the compound appears under trade names, listed in catalogs and chemical repositories such as PubChem or ChemSpider. Custom order forms for manufacturers or contract research organizations often call out these standard synonyms, so cross-referencing remains important for those navigating broad research partnerships or regulatory submissions.
Safety & Operational Standards
Anyone handling 6-chloropurine in the lab should wear the usual protective gear—gloves, safety glasses, and a coat—because the powder can irritate the skin, eyes, and respiratory tract. It shouldn’t be inhaled or allowed to linger on the skin. Most protocols suggest working in a fume hood, especially with the reactants used to synthesize or modify it, since they can be harsher than the product itself. Disposal guidelines push users to keep the compound and its waste out of the regular trash, usually routing them through chemical waste streams or licensed disposal contractors. As the pressure for green chemistry ramps up, operational procedures keep evolving, but direct contact remains a point of caution, underscored by standard data sheet warnings.
Application Area
6-Chloropurine features in a variety of sectors. Pharmaceutical chemists use it to develop new nucleoside analogs for exploring DNA replication and antiviral mechanisms. Its role as a precursor in synthesizing drugs like mercaptopurine cannot be overstated, and its applications reach into agricultural chemistry, though not as widely publicized. Some fields of diagnostics and molecular biology rely on its ability to form modified bases, which aid in tracking DNA or developing specialized molecular probes. Its fundamental structure offers ways to tune molecular interactions, reflecting a legacy of ongoing refinement.
Research & Development
Research labs continue to push the boundaries of what purine derivatives can do in medicine. Newer studies target improved selectivity in antiviral and antitumor agents, drawing on modifications first made possible by the 6-chloro group’s versatility. Some teams examine faster, cleaner synthesis strategies, shifting toward catalytic processes that produce less waste. The push for “greener” chemistry runs strong, with some success seen in the replacement of harsh halogen sources or more efficient solvent recycling. Academic and industrial groups publish steady reports on streamlined coupling reactions and new methods for modifying purine frameworks. Industry consortia now look toward expanding the structural family for personalized therapies as new biological targets emerge.
Toxicity Research
Laboratory tests show 6-chloropurine can irritate skin, mucous membranes, and lungs when handled without proper precautions. Acute toxicity seems modest compared to many halogenated aromatics, but researchers have noted that chronic exposure can contribute to organ toxicity or deferred hypersensitivity in certain animal models. Regulatory agencies advise against oral, dermal, or inhalational exposure and encourage prompt cleanup of spills. Some newer studies profile its metabolic fate in mammalian cells, helping to clarify potential risks for downstream drug candidates. The big takeaway: treat it with respect, stick to lab safety norms, and track disposal, as with any reactive organic.
Future Prospects
Innovation around 6-chloropurine looks set to accelerate. Pharmaceutical pipelines continue to look for lead compounds with better bioavailability or selective targeting, and this scaffold forms the foundation for much of that synthetic invention. Creative modifications at the chlorine position keep delivering new analogues for therapies once thought out of reach. With automated synthesis, high-throughput screening, and the growth of molecular modeling, there’s every reason to expect 6-chloropurine to keep its place as a quiet classic in chemical research. Coupled with an industry-wide push toward green and scalable processes, it stands poised for an expanding role in the next generation of therapeutics and diagnostics.
What is 6-Chloropurine used for?
What 6-Chloropurine Brings to the Table
Many researchers I know dig around for compounds that let them tweak and build new medicines. 6-Chloropurine looks plain at first glance, but it’s a gateway to a whole mess of other useful chemicals. You’ll see it pop up in lots of textbooks, and for good reason: it acts as a starting chemical for making more complicated ingredients — building blocks for antiviral drugs, cancer treatments, and biochemical tools.
How Scientists Use 6-Chloropurine
Chemists love working with purine bases because our bodies rely on them to form DNA and RNA. Changing up these purines by swapping out specific atoms changes everything. Put chlorine at the sixth position, and you have 6-Chloropurine. That tweak makes it easier to swap in something totally different, like an amino or an alkoxy group. With those swaps, scientists create molecules that block viruses or cancer cells from multiplying.
In university labs, I’ve watched grad students use 6-Chloropurine as a bridge in synthesizing drugs that fight leukemia. They use it as the root chemical, then add new pieces so the medicine slides into the right spots in a cancer cell’s DNA. A key fact: Allopurinol, a medicine people take for gout, comes from this kind of chemistry. So, even though 6-Chloropurine rarely ends up in a pharmacy, it’s part of more medications than most folks realize.
Why 6-Chloropurine Still Matters
Medicine doesn’t get anywhere without reliable base chemicals to build on. 6-Chloropurine holds a steady spot in pharmaceutical research because it reacts well and gives consistent results. If you’re making antimetabolite drugs—those that trip up DNA copying in fast-growing cells—it’s one of the safest starting points. I’ve seen scientists praise it for how easily they can replace the chlorine, making their lab work more efficient.
In a world where antibiotic resistance and pandemic threats keep rising, the tools researchers use become a big deal. 6-Chloropurine supports antiviral research by helping chemists test new ideas faster. HIV medicines and other antiviral treatments often start with some derivation of purine chemistry like this. A 2022 report in the Journal of Medicinal Chemistry highlighted new 6-Chloropurine derivatives that target hepatitis C by jamming up viral enzymes. That gives real hope for better, faster drug development.
Challenges and Where We Go from Here
Like many chemicals that play well in a lab, 6-Chloropurine comes with baggage. It isn’t safe to handle carelessly. Researchers need to invest in strong safety training, protective equipment, and waste handling. Small labs — often where fresh ideas show up — sometimes lack resources for this level of safety. Fact is, better safety rules and access to clean facilities will bring more breakthroughs.
The future of drug discovery depends on both creative minds and dependable materials. University programs that teach creative organic chemistry with a strong safety culture help push those discoveries out of the lab and into the real world. Funding support for small-group fundamental research can put more insight and safer practices into action. Every medicine we take now stands on the back of earlier work with chemicals like 6-Chloropurine — and it’s worth remembering that progress depends on both brains and the right building blocks.
What is the molecular formula of 6-Chloropurine?
Looking Beyond the Numbers: C5H3ClN4
6-Chloropurine carries the molecular formula C5H3ClN4. That is five carbon atoms, three hydrogen atoms, one chlorine atom, and four nitrogen atoms all tangled into a single structure. The formula looks simple, but the roles that structure has played in real science aren’t always so straightforward. Years back, I spent late nights in a university lab scribbling structures on a whiteboard, and C5H3ClN4 often sparked conversations. This molecule disrupts the usual purine cycle and tells a story that connects lab benches, textbooks, and the promise of better medicines.
Why Chemists Care About This Formula
6-Chloropurine stands out in the catalog of purine analogues. In medicinal chemistry, changing one group can turn a regular nucleobase into something with new powers. Swapping a hydrogen for chlorine at the 6-position doesn’t look dramatic, but it lets chemists create all sorts of purine derivatives. Researchers lean on that formula, C5H3ClN4, because it gives them a flexible stem to attach useful side chains. Drug developers can slip in amino or alkyl groups, opening doors for antiviral or anticancer drugs. It helped lay the groundwork for the synthesis of compounds like 6-mercaptopurine, which still treats leukemia decades after its introduction. In my own projects, I ran into the limitations and the potential of that formula when synthesizing nucleoside analogues in search of antiviral agents.
Problems Facing Chemistry in the Real World
While 6-Chloropurine has boosted drug discovery, its story reminds us that even simple-looking molecules can involve some headaches. Sourcing can be tricky; pure samples come dear because the halogen substitution introduces complexity into the synthesis. Handling chlorinated compounds also asks for respect and safe practice. Over the years, I’ve seen young chemists jump too fast into reactions without thinking about proper ventilation or waste disposal, and chlorine chemistry doesn’t always forgive mistakes. Trace contamination from halogenated intermediates turns up in places you don’t always expect. It eats at yields and sometimes makes scaling up a small lab discovery harder than the original research anticipated.
How the Community Moves Forward
The focus needs to stay on education and sustainable practice. Teaching young chemists about the risks and trade-offs of halogen chemistry isn’t just a box on a safety checklist; it keeps the field moving forward without getting tripped up by preventable accidents. More labs need automation and better ventilation to protect researchers. Sourcing supply from reputable vendors cuts down on impurity headaches, too. Since regulatory standards keep tightening, chemists must constantly review protocols and disposal methods to stay on the right side of environmental law. Collaborative projects among organic chemists, pharmacologists, and regulatory experts build the support systems that research needs.
What Matters About C5H3ClN4
Some formulas unlock more than new chemistry; they’re testaments to what’s possible when the discipline brings people, knowledge, and a dash of safe risk-taking together. As scientists keep tweaking old scaffolds—like 6-Chloropurine—fresh therapies inch closer. The next breakthrough won’t just come from knowing the formula, but from carefully handling what it represents in the wider world of chemistry and medicine.
Is 6-Chloropurine hazardous or toxic?
Understanding 6-Chloropurine
6-Chloropurine finds its way into both research labs and chemical manufacturing. You’ll spot its structure built on the purine core, with a chlorine atom sitting at the sixth position. Chemists prize it as a building block for pharmaceuticals, switching out its chlorine to craft other molecules. But this potential can’t be an excuse to ignore real concerns about safety.
The Risk Side of Chemistry
Ask anyone in a lab about 6-Chloropurine and the first thing they’ll mention is the dust. Crystalline, sometimes bitter-smelling, and notorious for irritating airways. Simple contact with skin can bring on a rash or redness. Eyes sting with just a small amount of exposure. Open a bottle, and you may notice a slight tingle in your nose. Breathing in fine particulates can spark coughing fits. It’s not sensationalism—Merck and Sigma-Aldrich both mark it with hazard warnings, labeling it as “harmful by inhalation, in contact with skin and if swallowed.”
Toxicology studies show that 6-Chloropurine can affect DNA in cell cultures. It’s meant for chemical reactions, not human consumption, but not every workplace has perfect controls. Large-scale production without proper fume hoods or extraction leads to real danger, especially for skin, eyes, or anyone with respiratory issues. The thing is, 6-Chloropurine acts as more than just an irritant. Animal studies reveal it causes acute effects in relatively low quantities. This isn’t a chemical to take lightly. Carelessness can lead to headaches, dizziness, even chemical burns.
Workplace Lessons and Precautions
Safety data isn’t just a formality. In my own years working in academic labs, I’ve seen gloves forgotten and goggles pushed up onto foreheads—a recipe for accidents. Chemical safety works on habit more than anything. Use 6-Chloropurine and good laboratory practice means wearing gloves, working in a fume hood and keeping emergency eyewash handy. Spills need containment; powders can linger in the air, so using wet cleaning methods helps limit dusty clouds. Even the best chemist gets a wake-up call after a single slip-up with something this reactive.
Regulators stress proper engineering controls for a reason. OSHA, the EU’s REACH, and other agencies don’t treat such compounds lightly. Companies moving, using, or selling these materials must track them; safety data sheets require updates alongside new research. Smaller start-ups or colleges might lack expensive ventilation, but even a cheap portable air filter and a lab coat offer crucial protection.
Environmental and Health Perspective
Research shows compounds like 6-Chloropurine could persist in water or soil if disposed of carelessly. Chronic exposure builds up, even for those outside the lab. Environmental impact matters just as much as acute toxicity—it’s not just about what happens in a single day’s work, but what lingers over years. Disposal through chemical waste collection and proper labeling prevents contamination. This concern hits home personally after seeing students learn about chemical safety with substances far less potent, only to later encounter compounds like this with far more at stake.
Moving Toward Safer Practices
Adopting “safety culture” is not about paranoia, but respect for risk. No chemical is just a tool; each one carries a real story about health, responsibility, and common sense. Alternative reagents or automation can cut down on direct handling. Regular safety training sticks best when kept grounded in the reality of daily routine, not just regulations. Sometimes what seems minor—a forgotten glove, a careless rinse—makes the biggest difference with reactive chemicals like 6-Chloropurine.
Beyond the lab, companies owe it to communities to invest in safety, not just compliance. By keeping people informed and engaged, real harm gets prevented. That’s the core value of genuine expertise: knowing not just how a compound fuels discovery, but what it means for the people working with it every day.
What are the storage conditions for 6-Chloropurine?
6-Chloropurine: A Compound That Demands Respect
Every chemist or lab worker comes across tricky, less-forgiving chemicals. 6-Chloropurine belongs in that group. It doesn’t carry the reputation of something extremely hazardous, but it’s far from ‘store it anywhere’ material. Several years ago, I handled this compound during an academic research project focused on nucleic acid analog synthesis. At the time, I learned firsthand that choosing the wrong spot on a cluttered shelf leads to ruined product and unnecessary hazards.
Temperature Control: Not Optional
6-Chloropurine fares best away from extremes. Room temperature might sound safe, but unpredictable labs—prone to overheating in summer—may put the material at risk. Look for a spot with consistent, moderate temperature, typically between 15°C and 25°C. Avoid keeping it in direct sunlight; slow heat buildup degrades the powder and can even change its appearance. Fluctuations from heating or cooling vents also stress the chemical and shorten its usable life.
Humidity: The Silent Enemy
One rainy season in an old lab, careless humidity control nearly ruined an entire bottle of 6-Chloropurine. Moisture creeps into containers, especially when lids aren’t tight. This compound doesn’t dissolve right away, but even a little water changes purity. Stash it in a dry place, using silica packs or inside a desiccator if possible. Moisture in the bottle creates clumps and can trigger low-level hydrolysis reactions, which interfere with experiments.
Light and Air: Keep Both Out
Many who work with nitrogen-rich heterocycles already know they dislike light and oxygen. 6-Chloropurine fits the bill. Standard amber glass keeps out much of the light, and frankly, it signals to users that direct exposure isn’t acceptable. Air-tight sealing matters; oxygen tweaks purity over time and can cause mild oxidation. The safest route always feels routine: tightly sealed bottles, in original containers, and limited exposure during sub-sampling.
Organization Matters: Chemical Compatibility
It’s tempting to shelve everything alphabetically or by personal convenience. In practice, it works better to think about nearby chemicals. Acids, bases, and oxidizing agents sit in their own space—mix-ups invite disaster. I once witnessed cross-contamination between purines and a strong base; the cleanup taught everyone in our group the importance of careful placement. 6-Chloropurine prefers a shelf among other purine derivatives or nucleic acid analogs, well-removed from reactive neighbors.
Labeling and Documentation
Always keep clear labels. Even in experienced labs, hand-written or fading labels make life harder for anyone coming after you. Include the date received and opened on the bottle. Good practice includes logging storage details and periodic checks. It prevents the letdown of finding degraded material after months of careful work.
Safer Practices
Some labs teach best practices from day one; others let newcomers learn the hard way. In either world, 6-Chloropurine rewards old-school discipline: reliable temperature, dryness, protection from light, careful documentation, and separation from incompatible chemicals. Carelessness wastes time and money, but organization and respect for these details build safer, more productive labs.
Better Labs, Better Results
Chemistry moves forward by learning from what works and what fails. Proper storage cuts frustration, keeps results reproducible, and prevents a long list of headaches. Everyday habits—like double-checking closures and writing clear labels—matter more than expensive equipment for protecting compounds like 6-Chloropurine. Over time, these habits just feel like the right way to work.
Can 6-Chloropurine be used in pharmaceutical synthesis?
A Building Block Worth Knowing
6-Chloropurine stands out in organic synthesis for anyone deeply involved in research or drug development. Chemists across both academia and industry keep turning back to it. That often tells you something valuable sits upstream in a synthetic route. This molecule brings a mix of reactivity, cost-efficiency, and versatility that stays relevant as new projects keep cropping up.
Why 6-Chloropurine Catches the Eye
Pharmaceutical projects call for clever shortcuts. There's always a push to find building blocks that shave weeks from timelines. 6-Chloropurine does exactly that. Its chlorinated purine core can be modified in many ways, opening up options for nucleoside analogues, antiviral compounds, and even anti-cancer agents. Chemists prefer molecules that let them swap out the chlorinated position for a wide variety of functional groups using common chemistry. That brings down barriers for rapid idea testing and process development.
In my own experience running small-scale synthesis for early-stage drug candidates, 6-Chloropurine stayed on our shelves. Swapping chlorine for amine groups or triazole rings using simple methods saved labor hours and delivered clean products. That’s rare with core-modified purines; not every heterocycle proves this reliable under routine lab conditions. It’s also worth noting suppliers rarely ran short on 6-Chloropurine, with purity hovering at levels trusted for most downstream steps.
What Makes It Useful in Drug Design?
The best-known application focuses on anti-viral and anti-tumor nucleosides. Medicinal chemists often use 6-Chloropurine as a scaffold to create analogues targeting enzymes or disrupting DNA replication. For example, a lot of the early work on acyclovir analogues and synthetic intermediates for drugs like mercaptopurine or thioguanine began here, then moved toward more complex modifications. This scaffold adapts because the chlorine atom sits at a place where swapping in other nucleophiles still keeps the purine ring intact.
There’s a practical benefit here, too. The conditions needed to substitute the chlorine don’t usually require exotic reagents. A straightforward nucleophilic substitution or catalytic reaction will cover most needs. This means graduate students aren’t stuck wrestling with temperamental reactions at their benches, and industrial chemists keep production lines moving with fewer surprises. The known toxicology of purine derivatives also helps regulatory filings later in the drug life cycle, so suppliers and process chemists trust it during scale-up.
Challenges and Solutions in Real-World Use
Making the switch from lab-scale to manufacturing brings fresh pain points. Many purine analogues turn out to be finicky when extra oxygen or moisture sneaks in. You watch out for hydrolysis during storage or handling. Since 6-Chloropurine can sometimes release traces of hydrochloric acid, industrial chemists tackle this by sticking to dry, air-free techniques or smart packaging. Investing in better containers pays off, especially where long-term storage matters or when shipping between continents.
Some scientists question whether better starting points exist. Sure, if another compound gives easier downstream chemistry, they’ll shift focus. Still, 6-Chloropurine’s wide compatibility with nucleoside chemistry keeps it valuable. Researchers keep exploring green alternatives and flow synthesis methods to shrink waste and boost yields. This trend speeds up drug discovery while making it easier on both budgets and the environment. More efficient purifications and solvent recovery help facilities avoid bottlenecks and keep regulators happy, too.
Looking Forward
Demand for novel drug scaffolds shows no signs of slowing. Whether someone works in university labs or at a pharmaceutical powerhouse, 6-Chloropurine deserves a spot in their toolkit. As new automated synthesis, greener procedures, and robotic workflows catch on, the practicality and cost-effectiveness of this tried-and-true molecule keep it ready for next-generation discoveries.
| Names | |
| Preferred IUPAC name | 6-chloro-7H-purine |
| Other names |
6-Chloropurine 1,6-Dihydro-6-chloropurine 6-Chloro-7H-purine 6-Chloro-9H-purine |
| Pronunciation | /sɪks-ˈklɔːrəˌpjʊəˌriːn/ |
| Identifiers | |
| CAS Number | 87-42-3 |
| 3D model (JSmol) | `3D Model (JSmol) string for 6-Chloropurine: C1=NC2=C(N1)N=CN=C2Cl` |
| Beilstein Reference | 136961 |
| ChEBI | CHEBI:28384 |
| ChEMBL | CHEMBL14061 |
| ChemSpider | 13338 |
| DrugBank | DB04115 |
| ECHA InfoCard | ECHA InfoCard: 100.004.225 |
| EC Number | 205-393-9 |
| Gmelin Reference | 192805 |
| KEGG | C06587 |
| MeSH | D015520 |
| PubChem CID | 6804 |
| RTECS number | UR8225000 |
| UNII | Y9D6Q828L2 |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C5H3ClN4 |
| Molar mass | 151.55 g/mol |
| Appearance | White to almost white crystalline powder. |
| Odor | Odorless |
| Density | 1.61 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 0.85 |
| Vapor pressure | 1.55E-7 mmHg at 25°C |
| Acidity (pKa) | 8.85 |
| Basicity (pKb) | 13.06 |
| Magnetic susceptibility (χ) | -85.0 x 10^-6 cm³/mol |
| Refractive index (nD) | 1.668 |
| Viscosity | 1.52 (mPa·s, 25°C) |
| Dipole moment | 1.89 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 155.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 81.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2588 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Irritating to eyes, respiratory system and skin. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | Hazard statements: "H302, H315, H319, H335 |
| Precautionary statements | Precautionary statements: P261, P264, P271, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P332+P313, P337+P313, P362+P364, P501 |
| NFPA 704 (fire diamond) | 1-2-1-0 |
| Flash point | > 153.8°C |
| Lethal dose or concentration | LD50 oral rat 760 mg/kg |
| LD50 (median dose) | Oral rat LD50: 1200 mg/kg |
| NIOSH | SN2000000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 6.4 mg/m³ |
| IDLH (Immediate danger) | N/D |
| Related compounds | |
| Related compounds |
Adenine 2,6-Dichloropurine 2-Chloropurine Hypoxanthine 6-Mercaptopurine Guanine |

