6-Chloro-M-Cresol: Chemistry Meets Real-World Relevance
Historical Development
Long before “green chemistry” became a buzzword, chemists understood the value of phenolic compounds, especially those tweaked with halogens. 6-Chloro-m-cresol stands out in this family. Folks have been working with cresols since coal tar distillation took off in the late 1800s, and by the mid-20th century, new chlorinated cresols found their way into applications that linked chemistry with daily life and industry. This wasn’t the story of a single eureka moment, but layers of thoughtful work from chemists hunting for antiseptics and disinfectants that balanced effectiveness and safety. Hospitals and water treatment plants demanded compounds that resisted bacterial resistance and held up against harsh conditions, and 6-chloro-m-cresol eventually gained a spot due to its robust properties. Its journey reflects how chemical advancements ripple outward, setting standards and changing practices in sanitation, healthcare, and beyond.
Product Overview
6-Chloro-m-cresol has earned a reputation as a reliable antiseptic and preservative. The compound, with roots in both chemical manufacturing and public health, has carved out space in both industrial and consumer products. Whether used in antiseptic creams or as part of formulations that keep equipment free from bacterial growth, its profile is defined by dependability. Users in both the lab and on factory floors favor it because it doesn’t break down easily, and it stands up to the types of bacteria that challenge many weaker chemicals. Its economic role follows, supporting manufacturers who need a balance between safety, cost, and effectiveness—qualities that only emerge from deep experience and practical testing.
Physical & Chemical Properties
6-Chloro-m-cresol appears as white to off-white crystalline powder under standard conditions. With a melting point in the moderate range and reasonable solubility in alcohols and organic solvents, chemists appreciate its stability in storage and handling. It emits a phenolic odor, which anyone who has worked in a laboratory can recognize from across the room. Its molecular formula, C7H7ClO, points to a small molecule, not too complex and straightforward for synthesis yet nuanced enough to deliver specific activities. The presence of both the methyl and chloro groups on the phenolic ring shapes its reactivity, making it compatible with a wide array of formulation types.
Technical Specifications & Labeling
Experience teaches quickly that product purity has to stay high for consistent performance, particularly when using chemicals for health-related tasks. Standard technical sheets list minimum purity requirements above 98 percent for 6-chloro-m-cresol, because impurities can undermine both safety and activity. It’s essential that packaging carries proper hazard symbols and storage advice: always keep dry, tightly sealed, and away from oxidants. Those facts aren’t just checkboxes—they stem from real setbacks suffered by earlier generations of chemists who saw how mishandled phenolic compounds could corrode containers or become unstable.
Preparation Method
The synthesis of 6-chloro-m-cresol typically involves selective chlorination of m-cresol using chlorine gas or related reagents, keeping the reaction cool and conditions controlled to ensure placement of the chlorine atom at the right spot on the ring. Careful handling matters at every step, especially because unchecked chlorination can produce a mess of unwanted byproducts. Working in an environment that manages air quality and waste streamlines the process. Decades ago, these reactions happened at smaller scale in open vessels, but today, continuous flow reactors cut waste and bring greater safety, reflecting changes around environmental responsibility and worker safety.
Chemical Reactions & Modifications
6-Chloro-m-cresol can undergo a variety of reactions familiar to anyone who’s worked with aromatic compounds. Nitration, sulfonation, and even further halogenation have been explored over the years, both for academic curiosity and for new product development. The hydroxyl group gives it potential as an intermediate in esterification or etherification, while the methyl and chloro substitutions can steer reactivity and solubility. Some manufacturers have explored derivatization to tweak antimicrobial activity, tailoring responses to emerging pathogens or regulatory requirements. The chemical backbone remains, but small tweaks give rise to new applications.
Synonyms & Product Names
This compound shows up under various labels in the literature and industry—4-chloro-3-methylphenol, p-chloro-m-cresol, and sometimes as PCMC. Users switching between suppliers or reviewing global safety data sheets must stay alert to these aliases, as regional differences in naming can slow down procurement or safety checks. The importance of clear nomenclature came home for many companies as they navigated shifting international regulatory landscapes—misidentification causes delays nobody wants when the assembly line or a patient’s care depends on a steady supply.
Safety & Operational Standards
The importance of chemical safety jumps out every time a spill or mishandling event gets reported. 6-Chloro-m-cresol, like many phenols, demands respect. Direct contact can lead to irritation, and inhalation of dust can harm airways or skin with repeated exposure. Workers receive training on gloves, goggles, and ventilation because companies have learned—sometimes painfully—that shortcuts lead to lost time, injuries, lawsuits, or worse. Regulatory oversight pushes for documented processes, safe storage, and well-marked containers. Only real-world experience—reading incident reports, confronting the aftermath of small errors—drives improvements in training and oversight.
Application Area
Most folks encounter this compound in products that keep our environments hygienic. Medical staff use creams and disinfectants built on its antimicrobial backbone, and engineers work it into water treatment chemicals. In personal care and cosmetics, formulators include it carefully because it protects against bacteria and mold without breaking the bank or harming sensitive users when used correctly. I’ve seen the balance shift as regulatory agencies tighten limits, pushing labs to reformulate and revisit assumptions about consumer safety. Every new application reflects ongoing testing and learning, from shelf-life studies to field reports in healthcare settings.
Research & Development
Chemists and toxicologists keep probing the boundaries of what 6-chloro-m-cresol can do. Academic research often focuses on new derivatives or mixes that boost effectiveness without boosting toxicity. Industry pushes research into new forms for controlled release or lower environmental impact. Over the years, this compound’s story has included a back-and-forth between the laboratory and clinic, with real user feedback guiding what gets funded or shelved. Researchers share a commitment to refining both product safety and performance, crafting new methods for detection, removal, or inactivation, mindful of both immediate public health needs and longer-term ecological effects.
Toxicity Research
Safety isn’t static. Decades of toxicity studies reveal the fine line between useful microbicide and hazardous exposure. Most reputable studies show that while topical or dilute use in products can be safe, concentrated forms carry risks—liver and kidney effects in animal studies, as well as irritation and sensitization potential in humans. Experiences in real clinics and factories repeatedly validate lab data, prompting updates to exposure limits and application guidelines. Ongoing research explores accumulative environmental effects, especially as wastewater treatment plants seek more effective removal strategies for chlorinated organics.
Future Prospects
The story of 6-chloro-m-cresol seems likely to run for many more years. As bacterial resistance grows and demand for safer, greener preservatives climbs, emphasis shifts from just tweaking old formulas to inventing better solutions. Researchers dig for structural modifications that lower persistence in the environment while keeping antimicrobial power. Some are chasing biotechnological routes for synthesis, looking to bypass older, dirtier processes. In safety, better analytical tools and real-world data help regulatory planners recalibrate standards. All this work comes down to a simple thread: a living chemistry, always seeking balance between human health, industry progress, and environmental stewardship.
What is 6-Chloro-M-Cresol used for?
What Is 6-Chloro-M-Cresol?
6-Chloro-m-cresol isn’t a household name, but if you pick up a bottle of antiseptic or look at the fine print on some medical products, you might spot it listed. This compound has gained popularity for its antimicrobial strengths, serving as both a disinfectant and a preservative. Labs and factories use it to tackle bacteria and fungi, keeping everything from ointments to equipment safe for people and pets.
The Power of Infection Prevention
Every time hospitals clean their surgical tools or clinics prep wound care supplies, they wage war on germs we can’t see. Here, 6-chloro-m-cresol steps in as a reliable shield against infection. Imagine a doctor trusting a bottle of antiseptic to stop bacteria from invading a patient’s open wound. This isn’t just theory; studies back up its effectiveness against bugs like Staphylococcus aureus and E. coli. Disinfectants made with 6-chloro-m-cresol help bridge the gap between sterile environments and the messiness of real life. They help keep everyday scratches from turning serious.
Building Safer Personal Care
This chemical has a quiet but important job in soaps, shampoos, and creams. Personal care products usually stay on shelves and in our bathrooms for months. Without something to stop microbes from growing inside the bottle, our cleaning routines could do more harm than good. By fighting off bacteria and fungi, 6-chloro-m-cresol helps keep products fresh and skin safe. Its use as a preservative allows manufacturers to guarantee longer shelf lives and safer experiences for consumers, without relying on harsh or outdated additives.
Challenges and Health Considerations
Effective cleaning comes with a responsibility to consider the health impact of the tools we use. 6-chloro-m-cresol stands out for its broad antimicrobial action, but it does require careful handling. High concentrations can cause skin irritation, and mixing it with the wrong chemicals can spark toxic fumes. Regulations direct producers to keep concentrations at safe levels, and these rules didn’t come out of nowhere. Safety studies led to these guidelines, so people can benefit without adding risk.
I’ve watched workplaces cut corners with cleaning supplies, but the best teams stick to the science. They train staff to respect the instructions on chemical labels and always demand proper ventilation. These small choices make big differences when handling chemicals like 6-chloro-m-cresol. The risk drops, and so does the chance that someone ends up at urgent care from a preventable exposure.
Safer Production and Environmental Questions
Manufacturers who work with 6-chloro-m-cresol face tough rules about handling waste and keeping workers safe. Many countries demand airtight processes to keep spills and fumes contained. In recent years, factories have moved to better ventilation, sealed packaging, and strict disposal plans. People want products that deliver protection and don’t harm the planet, driving more oversight on these chemicals’ life cycles.
Better Solutions Through Education
Knowledge about 6-chloro-m-cresol isn’t just for chemists or factory managers. Shoppers are savvier than ever, scanning ingredient lists and asking questions about what goes on their skin. Schools can do more to teach about the chemicals in common products, giving people the power to make safer choices. Companies can step up as well, offering transparency and supporting health-focused product development. With a little effort, there’s room for smarter innovation—products that work hard against germs and treat people and the planet with respect.
What are the safety precautions when handling 6-Chloro-M-Cresol?
Why Safety Matters With 6-Chloro-M-Cresol
6-Chloro-M-Cresol keeps cropping up in labs and factories thanks to its biocidal properties. It pops up in disinfectants, and some personal care products. But a small lapse with this chemical can send you running to a sink or ward.
Working around chemicals like this for years, you get two instincts: one for spotting shortcuts, and another for what goes wrong. You learn that accidents aren’t always spectacular—they start with a little skin contact or one careless whiff. Add up the effects: irritation to your skin, eyes, and breathing passages, and the risks are never worth pushing your luck.
Personal Protective Equipment Sets the Barrier
Gloves stop the first line of contact. Not every glove cuts it; disposable latex will break down fast, so industrial nitrile or neoprene gloves work better. People sometimes shrug off goggles, but a splash from a rogue droplet will change their mind quick. Full-face shields stop the finest splashes, especially during transfers.
Lab coats and aprons block spills from reaching your arms and body. For bigger jobs, long sleeves and closed shoes turn a brush with danger into a forgettable non-event.
Ventilation Makes a Huge Difference
6-Chloro-M-Cresol gives off fumes that shouldn’t hang around. In stuffy rooms, those fumes will sting your throat and nose, and could make you dizzy. Always make sure you’re in a space with a reliable fume hood or solid exhaust fans running. Never trust an old fan stuck in the window.
Storage, Labeling, and Clean-Up
Labels fade over time, but someone using a mystery bottle is how bad days start. Always check the label or print a new one before moving it onto a crowded shelf.
Keep it away from heat sources, sparks, and open flames. 6-Chloro-M-Cresol isn’t the most flammable chemical, but storing it with careless neighbors—like strong acids or oxidizers—can create tricky reactions. Lock it up, not just in any cupboard, but somewhere with chemical-grade shelving that won’t corrode.
Spills demand a level head. Absorb small spills with activated carbon or spill pads, bag the mess, and throw it in proper hazardous waste. Sweep up every granule. Skip the paper towels—those do more harm than good.
Train and Watch Your Colleagues
Watching someone new with chemicals always brings back the memory of my first day. It’s easy to miss a step or rush through a safety check. Regular training keeps memories fresh and makes good habits stick. It pays to ask each other questions, or to flag shortcuts, because teamwork carries more weight than any rulebook.
Every good workplace has a well-marked emergency shower and eyewash station. Practicing with that equipment, even dry runs, pays off when things get messy. It’s not just about knowing where the equipment stands—it’s about reaching it blind and panicked.
Improving Chemical Safety Every Day
Mistakes happen less when everyone takes responsibility, from storing reagents right to reporting broken hoods. Extra training, open conversations, and updated safety sheets build a climate where mistakes shrink and trust grows.
People work better when they feel respected and looked after. Chemical safety is part of that respect—a habit, not a hurdle.
What is the chemical structure and formula of 6-Chloro-M-Cresol?
Understanding 6-Chloro-m-Cresol
6-Chloro-m-cresol goes by another name: 6-chloro-3-methylphenol. This compound plays a consistent part in disinfectants and antiseptics. The way atoms are organized inside this molecule tells the story of how it works, how safe it is, and why it keeps winding up in so many cleaning products.
Structural Details
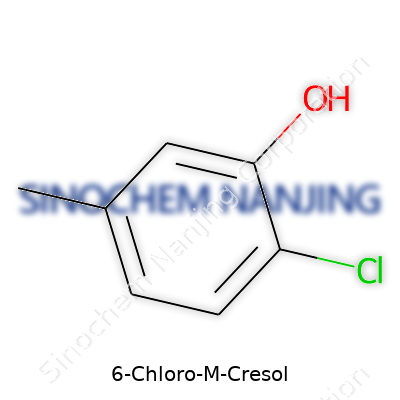
Scientists use the name “6-chloro-3-methylphenol” because that’s exactly what’s glued to the benzene ring: a chlorine atom and a methyl group, along with a hydroxyl group. The full chemical formula is C7H7ClO. Breaking this down, you get seven carbons, seven hydrogens, one chlorine, and one oxygen. It looks simple at first glance, but the real power comes from where those groups stick onto the ring.
The structure of 6-chloro-m-cresol isn’t random. There’s a benzene ring core. Attached to the number 1 position is a hydroxyl group (–OH). The methyl group (–CH3) sits at number 3, and the chlorine is clamped onto position 6. Chemists would draw it out as follows:
- Benzene ring core
- Hydroxyl group (–OH) at carbon 1
- Methyl group (–CH3) at carbon 3
- Chlorine atom (–Cl) at carbon 6
In real-world terms, this setup means the molecule blends the antiseptic punch of phenol with a slight boost from chlorine’s chemistry.
Real-World Importance
Listen to any infection-control expert, and you’ll find reference to compounds like 6-chloro-m-cresol. Hospitals, clinics, and even households lean on these molecules for surface and skin cleaning. The chlorine atom isn’t here for decoration; it increases antimicrobial action, according to several peer-reviewed studies, including work published by the American Society for Microbiology. The placement of the methyl and chlorine groups helps target a wider range of bacteria and fungi compared to basic “m-cresol.”
A lot of folks take these products for granted. Decades ago, my neighbor who worked in a veterinary lab showed me, hands-on, the persistent job these antiseptics do. It wasn’t about just killing germs. It was about making sure bacteria don’t adapt too quickly, and specialized structures like 6-chloro-m-cresol fill that gap when traditional phenolic disinfectants start to lose their edge. This matters not only in sterile environments but also in places where people just want to keep their homes safe.
Potential Concerns and Solutions
No chemical comes without questions of safety. 6-Chloro-m-cresol has well-recognized antimicrobial properties, but improper use can lead to skin irritation or worse. The molecular structure makes it persistent—great for fighting microbes, not so great if someone overuses it or disposes of it incorrectly. Responsible education goes further than a warning label. Manufacturers can step up by using clear labeling and simple instructions.
Regulators and scientists have choices. They can support ongoing research into alternatives that keep up antimicrobial power without building up in the environment. Green chemistry looks for replacements that break down faster. Until such advances become routine, 6-chloro-m-cresol’s unique structure gives it a solid, though sometimes controversial, place in disinfectant products.
How should 6-Chloro-M-Cresol be stored?
Understanding 6-Chloro-M-Cresol’s Risks
6-Chloro-M-cresol doesn’t sound friendly, and it certainly isn’t. In the lab, even a small spill comes with big problems: respiratory irritation, strong odors, skin reactions. With industrial chemicals like this, one quick slip-up can mean scrambling to run safety showers or call poison control. The long shelf life, the common use as a preservative, and the risk of hazardous by-products—these all demand respect.
Storage Conditions: What Matters Most
From experience, just stacking drums of 6-Chloro-M-Cresol in a corner brings trouble. Damp spots or open sunlight play havoc with chemical stability. Dry, well-ventilated rooms keep exposure risks manageable. Climate control matters—temperatures above room level send volatile compounds into the air, which is not what anyone wants in workspaces or warehouses.
I see too many stories of containers swelling up or leaking because the wrong seals were used. Manufacturers recommend tight lids with chemical-resistant linings, and that isn’t a suggestion to skip. Even a small crack lets fumes escape or contaminants sneak in. Properly labeled containers, stored off the floor, bring peace of mind during inspections or emergencies. Clear signage—hazard warnings, storage instructions—makes life easier for everyone stepping into the storage area.
Fire Safety Worries
Organic compounds often bring fire hazards, and 6-Chloro-M-Cresol isn’t different in that respect. Flammable vapors could build up in enclosed spaces, which means ignition sources have no place around storage. Static discharge from plastic drums causes problems if chemicals get splashed or transferred carelessly. Storing absorbents and fire extinguishers close by is not just a formality. Training teams on emergency procedures feels tedious until a spill or flame starts. Local regulations call for compatible fire suppression systems and safe storage distances from oxidizers.
Regulation and Environmental Concerns
Regulatory guidelines, like those from OSHA or local health authorities, don’t just exist for paperwork. Following these rules keeps hefty fines and legal headaches off your to-do list. Inspections usually check if containers are labeled clearly and if logs show exact storage dates and supplier batches. Disposal regulations ask for records too—treating waste streams seriously protects drinking water, soil, and nearby communities.
Realistically, environmental mishaps start small—a leaky container, poor ventilation, missed alarm on the detectors. These build up, sometimes unnoticed, turning a simple oversight into a public complaint or government lockout. Extra training, safety data sheets available at a glance, and secondary containment systems offer the strongest shield. Neighborhoods care about fumes and runoff, and a business that has a record for safe practices gives itself a solid reputation.
Adding Layers of Safety
Relying on one method for chemical storage invites problems. Double-barrier systems stop leaks, but periodic checks prevent silent failures. Separate chemicals with incompatible hazards, like acids or oxidizers, so there’s no nasty surprise during a storm or power outage. At past jobs, simple things like spill kits by every aisle paid off many times over. Emergency response drills hold value, too—even for long-timers who think they’ve seen it all.
In the end, good storage for 6-Chloro-M-Cresol means treating it with respect. The more effort put in upfront, the fewer surprises down the line. Consistent attention, strong protocols, and a healthy respect for health and safety laws make daily operations smoother—and keep the workplace safe for everyone.
Is 6-Chloro-M-Cresol toxic to humans or the environment?
Understanding the Chemical
6-Chloro-m-cresol pops up on the ingredient list in more products than most people notice. It comes from a family of chemicals called chlorocresols, found in disinfectants, preservatives, and even some personal care routines. While the word "chloro" triggers alarms because of its connection to many hazardous substances, it's not enough to judge this compound by its name alone.
Direct Effects on Humans
Handling 6-chloro-m-cresol means recognizing risk and safety. Skin contact can cause irritation or allergy. Some folks react with redness or swelling after exposure during cleaning or lab work. In my time working around cleaning solutions, I learned quickly to respect the caution labels and glove up. Ingesting or inhaling significant amounts—something that would usually happen only in a workplace accident or unsafe handling—can hit the central nervous system, the liver, or the kidneys, sometimes with pretty nasty effects. The U.S. Agency for Toxic Substances and Disease Registry identifies it as “moderately toxic,” and the European Chemicals Agency notes it can lead to acute health issues at high concentrations. Smaller, everyday exposures, especially from diluted products, seem unlikely to produce major long-term harm. Still, each body reacts differently, and repeated exposure can make allergy or sensitivity more likely.
Environmental Impact
Disinfectants and preservatives get flushed, rinsed, or tossed. Eventually, they reach waterways, soil, and possibly local wildlife. Studies show 6-chloro-m-cresol doesn’t break down quickly in the environment. Bacteria and sunlight do degrade it, but not before aquatic life faces the consequences. Fish and small organisms often react to this chemical with problems ranging from stunted growth to disrupted reproduction at moderate concentrations. Flora near outflows from manufacturing plants or poorly managed landfills can suffer stress or die outright.
Authorities, like the Environmental Protection Agency, keep a close watch on how much finds its way into wastewater. In some places, regulations focus on limiting discharges. It’s a race to keep concentrations below levels proven to harm insects, fish, and amphibians.
Practical Lessons from Experience
Decades in cleaning and maintenance showed how easy it is for overlooked chemicals to build up in our surroundings. Staff rarely checked dilution ratios. Leftover disinfectant often got dumped outside. Even those little shortcuts slowly carved out bigger issues, especially for streams neighboring large facilities. Colleagues with asthma or sensitive skin always guessed wrong about which cleaner would spark a flare-up.
Real protection comes from habits, not just legal limits. Workers need honest hazard training and strong protective rules. At home, people should watch for the chemical’s name on product labels and handle products in well-ventilated spaces, keeping them away from water sources.
Solutions for Safety and Sustainability
Manufacturers can keep risk down by choosing less persistent or less toxic alternatives, especially for products likely to go straight down the drain. Investing in advanced filtration at wastewater facilities can cut contamination after use. Inside industrial plants, spill containment and chemical recycling make a difference, shrinking the toxic load that ends up in nature.
People play a part just by following directions and not pouring unwanted chemicals outside or down the sink. Advocacy pushes for updated labeling and greater transparency, letting consumers decide what's right for them and their neighborhoods. Protecting the world outside factory gates means reading up, speaking out, and choosing products that balance cleaning power with environmental health.
| Names | |
| Preferred IUPAC name | 3-Chloro-5-methylphenol |
| Other names |
4-Chloro-3-methylphenol 4-Chloro-m-cresol 4-Chloro-3-cresol PCMC p-Chloro-m-cresol 4-Chloro-3-methylphenol Chlorocresol |
| Pronunciation | /ˈsɪksˈklɔːroʊɛmˈkrɛsɒl/ |
| Identifiers | |
| CAS Number | [86-60-6] |
| Beilstein Reference | Beilstein Reference 2040416 |
| ChEBI | CHEBI:81817 |
| ChEMBL | CHEMBL48903 |
| ChemSpider | 13649 |
| DrugBank | DB14060 |
| ECHA InfoCard | 100.008.467 |
| EC Number | 204-385-8 |
| Gmelin Reference | 8906 |
| KEGG | C01742 |
| MeSH | D002772 |
| PubChem CID | 79213 |
| RTECS number | GO5950000 |
| UNII | K9TY0N24U6 |
| UN number | UN3077 |
| CompTox Dashboard (EPA) | DTXSID3020347 |
| Properties | |
| Chemical formula | C7H7ClO |
| Molar mass | 156.61 g/mol |
| Appearance | White to pale yellow crystalline solid |
| Odor | phenolic |
| Density | 1.3 g/cm³ |
| Solubility in water | Slightly soluble in water |
| log P | 2.7 |
| Vapor pressure | 0.0014 mmHg (25°C) |
| Acidity (pKa) | 10.2 |
| Basicity (pKb) | 8.55 |
| Magnetic susceptibility (χ) | -0.0008 |
| Refractive index (nD) | 1.587 |
| Viscosity | 350 mPa.s (20°C) |
| Dipole moment | 2.61 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 216.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -159.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3845.3 kJ/mol |
| Pharmacology | |
| ATC code | D08AE05 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin irritation, causes serious eye damage, may cause respiratory irritation. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H318, H411 |
| Precautionary statements | P280, P305+P351+P338, P337+P313, P261, P304+P340, P312 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | 113°C |
| Autoignition temperature | 138°C |
| Lethal dose or concentration | Oral rat LD50: 1830 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 1830 mg/kg |
| NIOSH | GG9275000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL (Recommended Exposure Limit) of 6-Chloro-M-Cresol is "5 mg/m3". |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
4-Chloro-m-cresol 3,5-Dichloro-2-methylphenol 2-Chloro-m-cresol 6-Chloro-o-cresol Phenol p-Chloro-o-cresol |

