5'-Uridylic Acid Disodium Salt: A Down-to-Earth Look
Historical Development
Digging into the roots of 5'-Uridylic Acid Disodium Salt, it’s easy to see how biochemistry grew as modern tools opened new doors. Back in the twentieth century, isolation and precise identification of nucleotides became possible as chromatography and spectrometry matured. 5'-Uridylic Acid, or UMP-Na2, quickly followed discoveries of DNA, RNA, and the base uracil, leading researchers to explore its role as a building block in metabolic pathways. Nucleotide research helped tie together findings in genetics and cellular signaling, sparking interest not only from chemists but also from those working on therapies for metabolic disorders. Today, its application stretches beyond the laboratory towards fields including medicine and food science.
Product Overview
5'-Uridylic Acid Disodium Salt, commonly known as the disodium salt of uridine monophosphate, plays a crucial part in enzymes’ activity and is often used in biotechnology and food-grade applications. Its structure, made up of uracil attached to a ribose sugar and a phosphate group, keeps it at the center of many biological processes. Many suppliers market it for both research and industrial use, and in my own experience, the powder dissolves well in water, which makes it practical in lab settings. Clear documentation and traceable production have grown in importance as industries demand higher standards.
Physical & Chemical Properties
On the bench, 5'-Uridylic Acid Disodium Salt typically appears as a white to off-white crystalline or powdery substance. It’s highly soluble in water and tends to show a moderate degree of hygroscopicity, which prompts most labs to keep it sealed at low humidity. Molecular formula C9H11N2Na2O9P, with a molecular weight just over 368 g/mol, places it in a range that’s easy to manipulate in solution. Many researchers check purity using HPLC and confirm identity with NMR or IR spectra. Its chemical stability under dry and refrigerated storage stands up to typical lab workflows.
Technical Specifications & Labeling
Industry standards demand tight control on trace metal content and organic impurities. A lot of suppliers promise minimum purities above 98%, and batch-specific certificates usually list heavy metal analysis, microbial limits, and water content. Accurate labeling helps with traceability, so most vials come with lot numbers, manufacturer names, and recommended storage conditions clearly printed.
Preparation Method
Extraction and synthesis of 5'-Uridylic Acid Disodium Salt often starts with yeast RNA. Enzymatic hydrolysis using snake venom phosphodiesterase or mild acid hydrolysis can split RNA into nucleotide monophosphates, followed by purification over ion-exchange resins. Being hands-on with the yield and purity adjustments, I’ve seen how post-extraction filtration and recrystallization steps sort out contaminants, resulting in a powder suitable for downstream work. Industrial-scale synthesis, on the other hand, relies on automated bioreactors and stricter filtration, supporting volumes needed for food and pharmaceutical demand.
Chemical Reactions & Modifications
5'-Uridylic Acid Disodium Salt opens up a range of chemical tweaks that attract researchers. Phosphorylation, methylation, and conjugation with fluorescent probes let scientists track or modify genes and proteins. Coupling reactions, like carbodiimide-based condensation, allow researchers to attach UMP to peptides or other small molecules. I’ve seen teams develop labeled analogs for molecular biology kits, and some modifications enable them to investigate RNA polymerization or editing. Modifying the ribose or uracil rings offers a direct path to new diagnostic and therapeutic research.
Synonyms & Product Names
Besides its IUPAC name, this nucleotide salt is widely referred to as disodium uridine-5'-monophosphate, UMP-Na2, and Uridine-5'-phosphate disodium salt. In distributor catalogs, you might spot designations like “5'-Uridylic acid sodium salt” or “Uridine monophosphate disodium salt.” This variety comes from different naming systems used in pharmacy, food science, and molecular biology. Some markets emphasize “sodium uridylicum” as a legacy name.
Safety & Operational Standards
Despite being found in living systems, commercial batches demand careful handling. Most safety data sheets assign minimal acute hazard, pointing mainly to mild irritation with skin or eye exposure, though proper PPE always makes sense. In case of inhalation or accidental ingestion, rinsing and consulting medical help keep risks low. GMP standards help ensure purity and traceability for pharmaceutical and supplement use, while food-grade materials face regulatory review by authorities like the FDA and EFSA. I’ve always found keeping standardized documentation and batch records improves reproducibility and safety in busy labs.
Application Area
This salt doesn’t sit idle on the shelf. Scientists lean on it for RNA synthesis, cell culture media, vaccine formulation, and flavor enhancement in the food industry. As a component in nutritional supplements, it claims to aid cognitive function or support infant nutrition, although these claims require careful scientific scrutiny. Molecular biologists reach for it to study nucleotide metabolism and RNA processing, and it frequently appears in enzyme activity assays. Food scientists use it for umami flavor enhancement, especially in processed foods, where it works alongside inosine monophosphate to intensify savory notes.
Research & Development
Ongoing R&D tracks UMP-Na2 across nutrition, neurology, and gene therapy. Researchers investigate its potential in cognitive health after seeing positive animal study results on brain phospholipid synthesis and memory. Work in gene editing leans on its stable structure, while enzyme studies use it to model RNA transcription. In my experience, recent years have brought more targeted efforts to engineer analogs with different pharmacokinetics and to improve bioavailability in supplement form. Ongoing collaborations between pharma and biotech firms try to clarify which applications translate into clinic or mainstream product use.
Toxicity Research
Toxicological studies show a wide safety window for 5'-Uridylic Acid Disodium Salt at dietary exposure levels. Researchers track acute and chronic effects in animal models, and metabolic breakdown doesn’t produce concerning by-products. At very high doses, some animal studies suggest possible effects on liver enzymes, but these far exceed any likely human consumption. Regulatory panels, reflecting on existing literature, generally grant GRAS or food additive status, pending robust batch testing. Labs monitor for possible allergen content and contamination by nucleases or heavy metals.
Future Prospects
Interest in nucleotides keeps expanding, especially where food technology, medicine, and synthetic biology cross paths. 5'-Uridylic Acid Disodium Salt will likely see use in advanced cell culture formulas and new RNA-based therapies. Testing its value as a cognitive supplement will need more clinical data, but initial work draws attention from nutrition companies and brain health researchers. Growing demand for natural food enhancers positions it as an alternative to MSG and artificial additives. Synthesis improvements may bring costs down and open doors for further uses, spanning from custom biopolymers to next-generation diagnostics. The road ahead depends on strong science, clear regulation, and open dialog between labs and industry.
What is 5'-Uridylic Acid Disodium Salt used for?
The Role in Food Science
Walk into any kitchen and you’re bound to find savory seasonings tucked next to everyday spices. Few realize the science humming behind the taste of umami in broths or bouillon cubes. 5'-Uridylic acid disodium salt plays a quiet but critical role here. Food scientists turn to this additive for its flavor-enhancing abilities. It unlocks that satisfying, rich taste people crave, particularly in soups, sauces, and ready-made meals. Manufacturers rely on it because consumers expect their soups and snacks to deliver both comfort and depth without loading up on salt or MSG. A little of this compound supports a robust flavor, allowing producers to scale back on other, less healthy enhancers.
Use in Research and Molecular Biology
Beyond kitchens and grocery store shelves, scientists and techs loading their pipettes work with 5'-Uridylic acid disodium salt as a research tool. This compound acts as a building block in nucleic acid studies. Its structure helps researchers understand RNA synthesis and degradation. In practical terms, it forms one of the essential components for constructing or breaking down larger RNA molecules in the lab. Deciphering these processes supports breakthroughs in medical diagnostics and treatment strategies for several health conditions. Graduate students may find themselves measuring this salt by the milligram while unraveling the mysteries of viral replication or gene expression.
Pharmaceutical Applications
Drug development relies on a steady supply of reliable raw materials. 5'-Uridylic acid disodium salt shows up in some formulations and analytical methods. Its purity dictates the outcome of pharmaceutical syntheses or quality control steps. Researchers also use this salt in test systems for monitoring enzyme activities. For instance, uridine nucleotides offer insights into how the body digests and utilizes key biomolecules. The value isn’t just academic—it extends into producing safer, more targeted medications.
Quality and Safety Matter
Any story about a compound connected to food and medicine leads back to safety and sourcing. Purity checks, allergen testing, and transparency over origin shape both public trust and effectiveness in use. For companies using this additive, third-party certifications and tight documentation go a long way. There have been recalls in the food industry when suppliers didn’t meet standards, and regulatory bodies stay watchful. Even in research, contaminated or degraded material ruins experiments and wastes precious time. This creates a strong case for transparent sourcing and rigorous oversight along the entire supply chain.
Paving the Way Forward
The importance of 5'-Uridylic acid disodium salt comes down to people’s desire for flavor, convenience, and discovery. There’s room to keep refining extraction and synthesis, reducing the environmental footprint and opening up more sustainable production routes. Further research could spotlight new uses in smart drug delivery or personalized medicine. Companies and labs both have a stake in tracking new findings, prioritizing safety, and sharing data. By focusing on these principles, everyone stands to benefit—from the home cook stirring a pot of soup to the researcher chasing the next breakthrough.
What is the molecular weight of 5'-Uridylic Acid Disodium Salt?
Why Molecular Weight Matters in Science and Industry
Numbers tell stories. The molecular weight of 5'-Uridylic Acid Disodium Salt stands at about 368.17 g/mol. This figure is much more than a routine label on a bottle—it sits at the center of research, manufacturing, and clinical labs that depend on accuracy when making solutions or measuring doses.
From my own work in biochemistry labs, I’ve seen how an incorrect molecular weight can derail an entire research project. Just last year, a miscalculation for a nucleotide salt caused an experiment to misfire, costing ours precious time and funding. Lab teams that double-check the molecular weight during prep steps avoid that headache.
Understanding the Components
5'-Uridylic Acid serves as a fundamental building block in RNA synthesis and cellular signaling. The disodium salt form opens the door for it to dissolve easily in water. With each added sodium ion, the total molecular weight climbs by 23 g/mol. Scientists count on these numbers to make every batch as close to perfect as possible. Even slight miscalculations can affect enzyme kinetics or cause unwanted side reactions. For the standard disodium salt, expect a weight of 368.17 g/mol, based on its chemical formula: C9H11N2Na2O9P.
People blending up these solutions rarely give thought to all the calculations done for them. Students and young lab techs memorize numbers like these early, but the task turns meaningful when running an actual PCR, RNA extraction, or cell signaling assay. The stakes land squarely on their own experiments, not just some recipe in a handbook.
Potential Pitfalls and Smart Steps Forward
Errors in molecular weight go beyond laboratory blunders; they generate confusion across publications and international shipments. A batch produced overseas following the wrong specification can end up unusable or, worse, compromise a whole line of research. Oversights here breed waste—both in money and scientific progress.
I’ve witnessed mismatches between catalog data and package labels that forced our department to pause and call technical support to clarify information. These errors stall real work. This points to a need for consistent quality control and information sharing. Reliable catalogs with full chemical details, batch certificates, and direct web access to updated molecular weights would help prevent such mistakes. Training programs for staff often overlook the simple act of double-checking chemical weights against fresh sources—yet nothing beats forming that habit.
Broader Impacts in Healthcare and Research
Accurate weights on chemicals like 5'-Uridylic Acid Disodium Salt don’t just serve chemists—they stream into hospitals, food labs, vaccine development, and more. In one hospital setting, a reagent’s mislabeling almost delayed a critical diagnostic test. This isn’t only about science but also patient results, regulatory compliance, and the credibility of labs.
With automation and AI entering the lab, it’s easy to assume numbers like 368.17 g/mol never need thinking about again. Experience shows the opposite. Someone has to double-check results, cross-reference supplier documents, and speak up if something seems off. The best teams develop that culture of care. From education and batch checks to lab routines, keeping tabs on something as basic as molecular weight still sets the tone for success.
How should 5'-Uridylic Acid Disodium Salt be stored?
What Matters Most for Safe Storage
5'-Uridylic Acid Disodium Salt has a spot in labs and food facilities for a reason. It figures in enzyme studies, RNA research, and food flavor enhancement. But the real challenge for folks handling it often kicks in outside the spotlight—how this powder or crystal sits on a shelf plays a big role in its future usefulness. I’ve seen too many examples where shortcuts led to waste or surprise contamination. The salt’s delicate structure calls for more than tossing it into any old cupboard.
Keep Moisture Out
Leaving open containers or letting dampness creep in wrecks a batch quickly. This material attracts water from the air thanks to its hygroscopic nature, and that dampness encourages clumping, spoilage, and chemical breakdown. A tightly sealed bottle—preferably amber glass or high-grade plastic—protects the contents from humidity in the air. Toss in a desiccant pack, and you improve your chances of a dry, clump-free powder next time you open the lid.
Cool Temperatures Slow Down Spoilage
Heat eats away at quality for nearly every ingredient in the storeroom, and 5'-Uridylic Acid Disodium Salt is no exception. Solid evidence from chemical suppliers and university labs points to refrigeration—anywhere from 2°C to 8°C—helping this salt hang onto its chemical structure and potency. I tell colleagues to keep it out of freezers, since frost or condensation on thawing brings its own set of headaches. In my own work, storing it alongside sensitive enzymes helps keep both stable and consistent across months of work.
Shield from Strong Light
Light, especially UV, can slowly degrade some molecular structures. No dramatic news here, but a routine mistake comes from storing fine powders on open shelving near windows or under fluorescents. I've always favored an opaque or amber bottle and storing the container in a dark cabinet or cold room. Protecting this salt from direct sun and harsh light sources keeps its color and integrity, saving time and money in the long haul.
Simple Labeling Secures Confidence
Every time someone grabs an unmarked jar, confusion follows. Old habits taught me that a clear label—chemical name, date received, date opened, lot number—prevents spoilage and supports traceability if issues arise. This might seem basic, but in multi-user labs, this saves plenty of headaches and failed experiments. Clear labeling also helps quickly spot expired or compromised samples, keeping everyone in compliance with safety rules.
Reliable Handling Equals Reliable Results
Clean scoops, dry measuring tools, and keeping the main container closed except during brief use preserve purity. Spills left in storerooms become sticky invitations for pests or later mistakes. Pharmacy and food-processing standards support these habits, and I’ve found they actually save time by reducing batch failures. Respect for a chemical’s unique storage needs isn’t just about following protocol—it translates to good science and reliable work.
Summary of Best Practices
To get the most from your supply, aim for a dry, cool, dark spot and airtight containers. Include labeling and mindful handling as regular steps, not afterthoughts. In my own experience and across scientific literature, the extra few minutes spent on proper storage returns dividends by preserving the original qualities of 5'-Uridylic Acid Disodium Salt, with less waste and more predictable results every time you reach for it.
Is 5'-Uridylic Acid Disodium Salt soluble in water?
Why Scientists and Industry Care So Much
Ask any biochemist about 5'-Uridylic acid disodium salt and you’ll usually get a quick answer: yes, it dissolves in water. This chemical sits in the toolbox of cellular biologists, food scientists, and pharmaceutical developers. I’ve seen it turn up in a biochemistry lab as a key piece in RNA research. It’s found in certain flavors as a building block for savory food enhancers. Whether for research or manufacturing, folks rely on getting it into solution without a fuss.
Solubility—A Foundation, Not a Detail
A handful of salts like this one come packed with two sodium ions. That matters because these ions tug the sugar-phosphate base to water molecules, pulling the powder into a clear solution. Add a spoonful to a beaker of room-temperature water, swirl, and you’ll see it vanish. No need to heat the water or fiddle with the pH at standard concentrations used in the lab. That stands in contrast to less soluble analogs, which may demand hours of stirring or adjustment to acidic or basic pH values before you see a clear mix.
Facts from Research and Use
Reference any major biochemistry text or supplier catalog: typical descriptions say 5′-uridylic acid disodium salt reaches concentrations above 10 grams per 100 mL water at room temperature. For perspective, sodium chloride maxes out near 36 grams per 100 mL at the same temperature. Every time I prepared buffers for enzyme studies with this compound, it dissolved so quickly that no residue remained. The smooth behavior in water becomes a relief in any setting where you don’t want clumping or undissolved powder at the bottom of your flask.
Why It Matters in Practice
Think about what happens if your ingredient won’t dissolve. Laboratories face wasted hours coaxing solutions or filtering out stubborn particles. In food production, gritty texture or cloudy finish turns up in the end product—never good when customers expect creamy soups or savory sauces. For RNA research, any undissolved bits risk throwing off results, especially in delicate enzyme assays.
Because 5′-uridylic acid disodium salt practically launches itself into solution, quality control becomes easier. Less time, lower costs, smaller risk of losing batches. That earned trust across labs and production lines worldwide.
Issues and Practical Considerations
Sometimes, suppliers sell this chemical with tiny amounts of moisture or minor caking. If you run into slow mixing or see a haze, it points to water loss or clumping during storage, not a problem with the inherent solubility. Solution: tightly sealed containers, silica packs, and storing in cool, low-humidity rooms. I’ve seen teams keep backup supplies in small aliquots for this very reason, a small inconvenience compared to rebuilding a failed experiment.
Transparency and Reliable Sources
Reliable documentation backs up these claims. Look past promotional blurbs and read certificates of analysis or peer-reviewed studies. Data sheets from leading chemical suppliers offer clear numbers on solubility ranges, purity, and batch consistency. That kind of transparency gives scientists and manufacturers confidence—nobody needs last-minute surprises when scale or trust are at stake.
What Could Improve Accessibility
Some smaller suppliers skimp on updated certificates or don’t provide storage tips in plain language. Routine training could help new users avoid mistakes that threaten solubility. A little more communication from suppliers about best storage practices and stability under different climates would help minimize any avoidable issues.
What is the purity of 5'-Uridylic Acid Disodium Salt offered?
Why Purity of Biochemicals Matters
Most folks outside of a lab don’t spend much time thinking about the little codes and percentages attached to bottles of chemicals. Anyone in research—or food science, for that matter—knows those small details either drive results or turn them upside down. 5’-Uridylic acid disodium salt, commonly used as a flavor enhancer, molecular biology reagent, or buffer ingredient, stands as a good example. Purity isn’t just another line on the spec sheet—it’s what keeps experiments reproducible, data honest, and food safe.
Common Ranges You’ll See
Reputable suppliers usually state a purity of 98% or higher for 5’-uridylic acid disodium salt. Some push for 99% and top producers will share batch certificates with their numbers proved by HPLC or similar robust assays. This high level of purity builds confidence, letting researchers rule out contamination-related surprises. The remaining percentage—less than 2%—normally sits within the range of water or common salts, not pesky organic solvents or heavy metals.
Why the Number Matters
In molecular biology labs, a contaminated or impure sample can torpedo weeks of work. Say you’re amplifying RNA: even a little bit of degradation or residue from synthesis ups the background noise, drowns out genuine results, and empties your grant money faster than you’d like. Food producers and flavor chemists feel the crunch, too. An unexpected impurity could mess with taste panels or, worse, safety tests. Just because an additive passes the taste test doesn’t mean the process went by the book.
Verifying Supplier Claims
Not every supplier follows the same playbook. While big names in reagents have a strong track record and clear documentation, others cut corners to drive the price down. Sometimes, you’ll see the percentage up front but no mention of the methods used to determine it. The best suppliers send along a Certificate of Analysis (CoA) with each batch, including purity figures, heavy metals content, and more. Labs I’ve worked in won’t use a new lot without checking these documents and, for the high-stakes stuff, they’ll run a small in-house test as double insurance.
Facts that Shouldn’t Get Ignored
Regulatory agencies like the FDA and European Food Safety Authority publish specifications for food additives, touching on things like microbial load, toxins, and specific measurable impurities. Adhering to those isn’t a suggestion—it's mandatory before the product can hit the market. Many academic publications also require detailed supplementary data on reagents. I once had a paper delayed for weeks while my team tracked down purity documentation for a single nucleotide.
How to Make a Smart Choice
Anyone ordering 5'-uridylic acid disodium salt—whether for an academic, industrial, or culinary application—should keep a short checklist: purity percentage above 98%; documentation from the supplier; and, for mission-critical work, a willingness to check samples in house. Building trust with vendors by demanding transparency is what keeps both experiments and food supplies reliable. In a world where budgets have limits, resisting the temptation to cut corners on purity saves far more time and money than it costs.
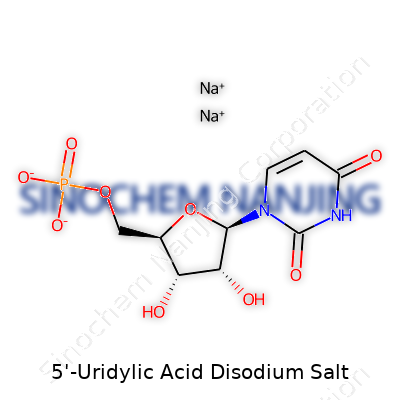
| Names | |
| Preferred IUPAC name | disodium uridine 5'-phosphate |
| Other names |
DISODIUM URIDINE 5-MONOPHOSPHATE UDPNA Uridine 5-monophosphate disodium salt Uridylic acid disodium salt Uridine monophosphate disodium salt |
| Pronunciation | /faɪv juːˈrɪdɪlɪk ˈæsɪd daɪˈsoʊdi.əm sɒlt/ |
| Identifiers | |
| CAS Number | 6159-34-2 |
| Beilstein Reference | 2672991 |
| ChEBI | CHEBI:61448 |
| ChEMBL | CHEMBL12384 |
| ChemSpider | 10215 |
| DrugBank | DB01883 |
| ECHA InfoCard | The ECHA InfoCard of product '5'-Uridylic Acid Disodium Salt' is: **03c3b834-8c8d-43ad-a2cf-8afc09cedb51** |
| EC Number | 2.7.7.6 |
| Gmelin Reference | 92251 |
| KEGG | C00299 |
| MeSH | D014538 |
| PubChem CID | 23667676 |
| RTECS number | YV9625000 |
| UNII | 3KX376GY7O |
| UN number | non-regulated |
| Properties | |
| Chemical formula | C9H11N2Na2O9P |
| Molar mass | 700.18 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 1.64 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -3.9 |
| Acidity (pKa) | 2.0 |
| Basicity (pKb) | 11.33 |
| Magnetic susceptibility (χ) | -59.5e-6 cm³/mol |
| Viscosity | Viscous oil |
| Dipole moment | 8.97 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 344.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −1964.6 kJ/mol |
| Pharmacology | |
| ATC code | A11HA30 |
| Hazards | |
| Main hazards | May cause irritation to the eyes, skin, and respiratory tract. |
| GHS labelling | GHS labelling: Not a hazardous substance or mixture according to the Globally Harmonized System (GHS). |
| Pictograms | GHS07, GHS09 |
| Signal word | Warning |
| Hazard statements | Non-hazardous according to GHS classification. |
| NFPA 704 (fire diamond) | NFPA 704: "Health: 1, Flammability: 0, Instability: 0 |
| LD50 (median dose) | LD50: >5000 mg/kg (oral, rat) |
| NIOSH | Not Listed |
| PEL (Permissible) | Not established |
| REL (Recommended) | 15 mg/m³ |
| Related compounds | |
| Related compounds |
Uridine Cytidine Adenosine monophosphate Guanosine monophosphate Thymidine monophosphate |

