Unpacking ((5-Thiazolyl)Methyl)-(4-Nitrophenyl) Carbonate: History, Science, and Impact
Historical Development
Chemists first targeted thiazole-based derivatives, like ((5-thiazolyl)methyl)-(4-nitrophenyl) carbonate, in the ongoing hunt for molecules capable of bridging biological and synthetic chemistry. Back in the second half of the twentieth century, heterocyclic chemistry picked up speed. Researchers began exploring structures that combined activity-enhancing units—the thiazole ring for its versatility and the nitrophenyl group for its reactivity. This particular carbonate came out of a tradition where labs sought to improve selectivity in drug development, diagnostics, and advanced material science. The rise of modern organic synthesis opened the possibility of attaching the thiazole ring to different carriers. Interest in thiazole derivatives kept growing with reports of anti-infective, anti-inflammatory, and biochemical reporter applications. Even today, investigators and product developers look back at these early breakthroughs to inspire structure–activity relationship studies and synthetic methods.
Product Overview
((5-Thiazolyl)methyl)-(4-nitrophenyl) carbonate captures attention because it offers a reactive carbonate group fused to a recognizable aromatic nitro compound and a biologically significant thiazolyl moiety. This combination gives chemists a handle for downstream modifications—useful both in pharmaceutical intermediate work and in probing new reactivity patterns. In its pure form, this product appears as a fine crystalline powder or sometimes as aggregated granules. Beyond its immediate chemical profile, this carbonate bridges multiple disciplines, serving as a connector in the preparation of more complicated molecules or as a stand-alone probe in research that seeks to illuminate new chemical mechanisms.
Physical & Chemical Properties
Examining the physical side, ((5-thiazolyl)methyl)-(4-nitrophenyl) carbonate often presents itself as a yellowish to buff crystalline solid due to the nitro group’s deep color. Melt points typically range between 120°C and 150°C, although purity and crystal habit can tip this value up or down. It remains stable under dry conditions but reacts adventurously with moisture, acids, or bases, owing to the reactive carbonate linkage. On the chemical front, the molecule boasts a balanced solubility profile: polar aprotic solvents like dimethyl sulfoxide or acetonitrile dissolve it efficiently, while water barely touches it. The molecule carries a net neutral charge, with the thiazole and nitrophenyl groups balancing electron density. Such features directly affect how labs store, transport, and transform this carbonate in reactions.
Technical Specifications & Labeling
Product batches generally come with a specific CAS number, purity specification (often 98%+ by HPLC), moisture limits, and clear warnings about light and temperature sensitivity. The best practices for labeling rely on the Globally Harmonized System—clear hazard pictograms, concise risk statements, and advice on personal protective equipment. Labels also typically flag the need to keep the material under nitrogen or in sealed containers, plus a reminder to record all handling in lab notebooks for traceability. From my own experience, nothing derails a project like overlooked contaminants, so it pays to insist on full certificates of analysis and batch trace records.
Preparation Method
Lab synthesis usually starts with (5-thiazolyl)methanol, which chemists treat with phosgene alternatives (like triphosgene or carbonyldiimidazole) to generate the corresponding chloroformate. This intermediate reacts directly with 4-nitrophenol under basic conditions, often in dichloromethane or another inert organic solvent, delivering the carbonate product after proper workup and purification. Moisture control plays a critical role throughout, since adventitious water prompts unwanted hydrolysis, and many procedures favor the use of molecular sieves or an anhydrous glovebox. Monitoring by TLC or NMR during scale-up keeps byproduct formation in check and helps labs refine the conditions for higher purity and yield. Scalability emerges mainly from careful reagent addition and rigorous exclusion of environmental contaminants.
Chemical Reactions & Modifications
The combination of the carbonate ester and nitrophenyl leaving group opens the door for nucleophilic attack, making this compound a popular choice for forming carbamates or other modified esters. Chemists often use it as an intermediate in peptide coupling protocols or to install custom linkers on small molecules. The thiazolyl side chain confers unique electronic characteristics, sometimes enhancing reaction rates or selectivity in carbon–nitrogen bond formation. My own group has seen this sort of chemistry streamline the assembly of labeled probes for enzyme assays, where the nitrophenyl moiety doubles up as a colorimetric reporter. Even with its reactivity, the carbonate moiety can withstand brief exposure to certain bases, allowing two-step or even telescoped transformations. This versatility makes the compound a backbone component in libraries for medicinal chemistry screening and synthetic biology.
Synonyms & Product Names
Depending on the supplier or research context, the compound might show up as (5-thiazolyl)methyl 4-nitrophenyl carbonate or as carbonate ((5-thiazolyl)methyl)-(4-nitrophenyl). Its structure sometimes gets abbreviated in combinatorial libraries as TMCNP or shown in catalogs under systematized IUPAC names. Knowing the variety of synonyms helps prevent procurement errors and cross-talk in international research collaborations.
Safety & Operational Standards
Working with ((5-thiazolyl)methyl)-(4-nitrophenyl) carbonate calls for careful respect. Contact with eyes, skin, or mucous membranes irritates, and inhalation of fine dust should be avoided through well-fitted masks and good local exhaust. The nitrophenyl component signals the potential for toxic breakdown, and labs always store the compound in flame-resistant, ventilated cabinets, away from acids and bases. Safety data sheets recommend neoprene or nitrile gloves, splash goggles, and procedures handled inside a fume hood. Decontamination after spills means using absorbent pads, alkaline neutralizer, and frequent glove changes. I once saw an inadvertent mix-up with incompatible reagents lead to exothermic decomposition, so incident training and proper labeling always stand as part of the lab’s safety culture. Disposal must follow regional rules, most often incineration in an approved hazardous waste facility.
Application Area
This carbonate’s main role emerges as a coupling or activating agent in organic synthesis, especially for constructing carbamates, ureas, and peptide bonds. In pharmaceutical circles, it serves as a strategic intermediate, linking small drug fragments or introducing masked leaving groups. Biochemical labs use the molecule for installing handles on enzyme substrates and crafting chromogenic probes. The thiazolyl segment holds promise in targeting enzymes like kinases or proteases, echoing its natural role in some cofactors. Researchers in diagnostics turn to the nitrophenyl group’s vivid chromophore for color assays or as a leave-behind tag after enzymatic cleavage.
Research & Development
Ongoing R&D drives the search for next-generation analogs or improved synthetic protocols. Teams investigate enantioselective routes that might give one-handed specialty carbamates, seeking to improve pharmacokinetics or reduce unwanted side effects. Automation and small-volume synthesis emerge as regular features in labs targeting high-throughput chemical modification. Computational chemists run electronic property simulations to screen new analogues, predicting better reactivity or safety profiles. Cross-disciplinary research, blending organic synthesis with computational and biochemical analysis, opens potential for tailored reagents in future drug discovery programs. Each improvement—higher yield, safer reagents, lower-cost synthesis—feeds back into the broader ecosystem of chemical manufacturing and therapeutics development.
Toxicity Research
Toxicological evaluation anchors any new molecule’s path to broader application. Early in-vitro work flags risks associated with the nitrophenyl functionality, linked historically to methemoglobinemia and organ toxicity. Animal studies often track metabolic breakdown, looking for bioaccumulation or reactive metabolites. While the thiazole ring doesn’t usually register as a high-alert moiety, the carbonate link and nitrophenyl components require close examination under simulated physiological conditions. In labs, researchers measure thresholds for skin, eye, and inhalation exposure, feeding this information into standard protocols. Regulatory agencies ask for robust chronic and acute toxicity data, pushing the field to explore safer alternatives and improved analogues. Transparency in sharing toxicity data within the scientific community, as well as public registries, ensures responsible use and motivates the design of more benign chemical variants.
Future Prospects
The coming years will likely witness even more creativity in designing and deploying thiazolyl–nitrophenyl carbonates. As green chemistry matures, milder reagents and solvent systems will push synthesis toward sustainability, reducing both hazard and environmental footprint. Machine-learning tools, already present in screening new molecular candidates, could accelerate the identification of safer, more effective analogs for both research and industry. Fields like targeted drug delivery and molecular electronics may draw from the toolbox provided by these multifunctional molecules. From my perspective, the next leap comes not just from better molecules, but from end-to-end transparency in manufacturing processes, open-access data on reactivity and safety, and robust interdisciplinary collaboration. Such efforts promise not just safer labs, but real benefits for medicine, diagnostics, and materials science.
What is the primary application of (5-Thiazolyl)Methyl-(4-Nitrophenyl) Carbonate?
Chemistry Unlocks Progress
Things in a chemistry lab often look ordinary, but certain reagents tip the scales in research. (5-Thiazolyl)Methyl-(4-Nitrophenyl) Carbonate doesn’t show up on pharmacy shelves or in supermarket cleaners, yet it gets plenty of attention behind the scenes. Chemists use this compound mostly for something called peptide synthesis. Peptides, in simple terms, are shorter chains of amino acids. Labs need them for drug research, protein mimicry, and medical diagnostics.
Direct experience from time at the bench reveals just how tedious peptide building can become. Molecules need to stick together at specific spots. Protection and deprotection steps, which keep reactive groups from getting too friendly too soon, take quite a bit of clever planning. Here’s where (5-Thiazolyl)Methyl-(4-Nitrophenyl) Carbonate excels. It acts as a coupling agent, helping the peptide chemist attach amino acids together more smoothly and predictably than some traditional reagents. Fewer side-reactions mean purer peptides and less wasted money.
Research Shapes Future Therapies
Big names in drug development—think cancer immunotherapies and diabetes treatments—turn to peptide structures for targeted therapies with fewer side effects. The value here trickles down quickly. For instance, drugs built from peptides can imitate natural hormones or intercept signals in the body, treating disease at the root. Success in this kind of research means patients get better therapies and fewer long-term complications.
Synthetic workhorses like (5-Thiazolyl)Methyl-(4-Nitrophenyl) Carbonate enable these discoveries by making the process of joining amino acids more reliable. The compound’s specific chemical structure helps activate the carboxyl group on one amino acid, encouraging the formation of a peptide bond with another. This results in fewer by-products and greater yields. Researchers at places like Novartis and academic medical centers often report how switching to more efficient coupling reagents saves them months of troubleshooting and thousands in material costs.
Safety and Accessibility Remain Challenges
Of course, using specialized reagents brings responsibility. I’ve seen small labs struggle with safe storage, since carbonate esters, especially those containing nitrophenyl units, can be sensitive to light and moisture. Handling risk seems minor to outsiders, yet spills or improper disposal put both people and the environment at risk. Smart training remains key. Chemical manufacturers can step up with clearer instructions, practical workshops, and regular safety reviews.
Supply chain hitches have shown up now and then. Researchers tell stories of sudden shortages that stall entire projects. Having multiple suppliers and partnerships between academic labs and specialty chemical companies helps cushion shocks. If funding agencies and research institutions support smaller companies who synthesize such compounds domestically, the load spreads out better. Not every lab can afford high costs or long shipping delays.
Looking Ahead: Smarter Chemistry, Broader Impact
As targeted therapies keep growing, demand for quality reagents like (5-Thiazolyl)Methyl-(4-Nitrophenyl) Carbonate won’t drop. Better education for researchers, stronger safety focus, and more robust global supply networks can ease growing pains. In the end, this humble molecule—rarely in the spotlight—helps keep medical innovation moving forward. Progress in the lab builds on small, reliable steps, and every efficient reaction brings the next breakthrough a bit closer.
What is the chemical purity and grade of this product?
Understanding What’s Really Inside the Bottle
Questions about chemical purity and grade go far beyond a label on a bottle. I’ve seen labs make mistakes because someone trusted a glossy product sheet instead of digging into real test data. On the surface, chemicals look similar: fine powder, clear liquid, neat packaging. But there’s a world of difference between chemicals for industrial cleaning and chemicals used in making medicine. The difference starts with purity.
Purity: Not as Simple as a Number
Purity sounds like a single, simple figure—98%, 99.9%, 99.99%—but that number hides a lot of detail. Let’s say you buy sodium chloride listed at 99.5%. That missing half-percent tells a story: metals, moisture, maybe even other salts. I once watched a student prep a solution using “pure” salt from the hardware store, only to discover subtle contaminants that skewed the results. For some applications, those trace impurities won’t matter. For pharmaceutical, semiconductor, or food work, even tiny residue can destroy a whole batch or endanger users.
Grades Actually Matter in Real Life
Grades aren’t just for report cards in chemistry class. There's reagent grade, ACS grade, USP grade, food grade, technical grade—and each aims for a different kind of use. A chemical labeled as “lab grade” might be perfect for cleaning glassware, but not for making a vaccine. Actually, I’ve seen manufacturers quietly use food-grade ingredients in process water, then run into problems when a customer pushes for pharmaceutical-grade documentation. Documentation matters as much as the material itself, especially in regulated industries.
Trust But Verify: Reading Beyond Product Labels
Sellers throw around terms like “high purity” without real data to back them up. Certificates of Analysis and Safety Data Sheets provide the hard proof. For a chemical to meet USP or ACS standards, it goes through independent verification and testing. Scientists waste hours—and sometimes months—when a supply house delivers a different standard than what the project requires. Trust gets built when suppliers back up claims with recent, batch-specific certificates, not just catalog promises.
Why Source and Chain of Custody Matter
Where a chemical comes from makes a difference. I remember a time someone in the lab used the same compound from two sources, only to find wildly different impurity profiles. Some manufacturers use old storage techniques or packaging that allows moisture, clumping, or cross-contamination. Good quality control starts before shipping and doesn’t stop until that bottle’s used up.
Solutions: Education and Better Verification
One way forward is closing the communication gap between buyers, lab techs, and suppliers. Site visits and supplier audits weed out unreliable sources. Training lab staff to check batch records, analyze certificates, and understand the regulatory meaning of “grade” prevents expensive mistakes. Labs that keep documentation organized and publicly traceable don’t just meet audits—they keep projects on track. It pays to call the supplier when something seems off. A simple question about actual testing practices or batch quality can save a lot of trouble down the line.
Every Grade Has Its Place, But Not Interchangeably
Picking the right chemical grade is less about chasing the highest number and more about understanding the risk for each job. Sometimes a lower grade suits an industrial process just fine. But for health, high-tech, or food-related work, a single unnoticed contaminant can cause disaster. Experience shows that peace of mind comes from matching product claims with proof, not just reaching for whatever looks cheapest or says “pure” on the box.
How should (5-Thiazolyl)Methyl-(4-Nitrophenyl) Carbonate be stored?
Core Principles and Daily Lab Life
Laboratories brim over with bottles and powders, but some chemicals command extra respect. (5-Thiazolyl)Methyl-(4-Nitrophenyl) Carbonate is one where good practice isn’t just a checkmark—it’s a barrier between routine work and regret. Many researchers will recognize those vivid yellow warning labels, calling out potential hazards that demand more than absent-minded shelving.
Heat, Moisture, and Light: The Trouble Trio
Heat in a storage area might sound innocuous until you remember the stories of compounds gone unstable, giving off fumes or worse. This carbonate belongs in a refrigerator or a dedicated cold cabinet, best held to temperatures below 8°C. Anyone skimping on refrigeration hopes for luck rather than relying on science. Moist air creeps through loose seals and turns shelf-stable chemicals into sludge or something unpredictable. Desiccators, packed with fresh silica gel, answer this challenge.
Light can be forgotten, yet a sudden color shift or degradation means a compromised bottle—so amber vials or wrap-around aluminum foil give simple, no-nonsense protection in any lab I’ve worked in. In a place with direct sunlight or fluorescent bulbs, skipping this step is an invitation for problems.
Away from Acids, Bases, and Strangers
Everyone shoves bottles together on crowded shelves at times, but mixing chemicals out of convenience puts people at risk. (5-Thiazolyl)Methyl-(4-Nitrophenyl) Carbonate doesn’t play well with strong acids or bases. Storage on a segregated shelf, ideally lined with chemical-resistant material, gives peace of mind. I’ve seen what happens when someone stores everything alphabetically—the emergency room sees more chaos from that than you’d like to believe.
Curiosity brings people to science, but some chemicals only belong in the hands of trained staff. Lab managers use locked cabinets for good reason, tracking every gram with a logbook or barcode. In universities, graduate students find it easy to cut corners, but locked access gives everyone a sense of accountability—which can mean the difference between routine work and a lab evacuation.
Labeling: Simple but Underrated
It’s easy to scribble a formula or shorthand on a bottle with a drying marker. That’s not enough. Full, up-to-date labels including hazard pictograms, concentration, and date of receipt keep everyone on the same page. In collaborations, clear labeling prevents unfortunate surprises for colleagues unfamiliar with old stock or experiments-in-progress.
Disposal and Spill Response
Any experienced chemist knows that disposal matters just as much as storage. Leftovers or leaks from these bottles could mean toxic byproducts—so every person working with this carbonate should know their institution’s disposal protocol. Chemical spill kits don’t sit in the corner for show; dry absorbents and proper PPE can snuff out trouble before it grows. I’ve been part of safety audits that lagged behind changeover, with expired or missing spill kits—a risk no one wants to take.
Moving Toward Better Practices
Better storage keeps accidents far away. Refrigeration, low humidity, protection from light, safe stacking, clear labeling, and secure shelving represent the basics. These aren’t abstract rules—they’re daily habits molded from the experiences of every researcher determined to make it home after a long day in the lab.
References for Peace of Mind
Institutions like the CDC’s NIOSH and OSHA, the ACS, and manufacturer safety data sheets offer invaluable guidelines. Their advice isn’t just legalese, but a distillation of lessons learned from real accidents. Checking for the most recent SDS and reviewing institutional procedures means less guesswork and more security for everyone who steps into the lab.
Is there specific handling or safety information for this compound?
The Real Risks Lurking in a Lab or Workshop
Most folks grab a bottle or container off a shelf and don’t think twice about its dangers. Experience in chemistry labs tells a different story. Whether you’re mixing cleaning solutions at home or working in pharmaceutical research, chemical safety cannot be shrugged off. I once opened a container of sodium hydroxide because the label had faded. My gloves started to smoke, and it could have been a trip to the emergency room. Incidents like these drive home the lesson: knowing how to handle a compound can’t just be an afterthought.
Trust, But Verify: The Label Won’t Tell You Everything
Most chemical containers list a common name, maybe a hazard symbol, and not much else. Some don’t mention incompatibilities or how fumes can irritate the lungs or eyes. The Material Safety Data Sheet (MSDS) or Safety Data Sheet (SDS) goes much further—it describes routes of exposure, recommended storage, and details on what to do if someone spills or ingests the substance. OSHA statistics show that accidents related to poor chemical handling top the list of workplace incidents. That’s not because people ignore directions, but because the hidden dangers can catch anyone off guard.
Not All Gloves Are Equal—Or Lab Coats, Or Goggles
Lots of people believe one size fits all for protective gear. That couldn’t be further from the truth. Nitrile gloves don’t stand up to acetone. Latex gloves can disintegrate if exposed to strong acids. The right barrier depends on the specific chemical. I learned fast that wearing the wrong type means putting your skin and eyes at risk. From hydrochloric acid burns to solvent spills, the right prep means knowing what gear stands between you and a trip to see the campus medic or, even worse, permanent damage.
Mixed Compounds: Small Mistakes, Big Consequences
Some chemicals look harmless on their own but become hazardous when mixed. Mixing bleach and ammonia produces toxic chloramine gas. Laboratories and factories use strict labeling, color coding, and clear signs so staff never mistake two similar-looking jars. Home users regularly mix drain cleaners or cleaning agents, unaware that some combinations can destroy airways in minutes. Simple solutions like permanent, readable labels and lockable storage keep those risks out of reach. The EPA and CDC both press the importance of separation by chemical hazard class.
The Role Of Training And Vigilance
Proper chemical handling starts with real-world training. Watching a video or reading a manual helps, but hands-on supervision makes the lessons stick. At my first lab job, routine safety audits weren’t just red tape. Supervisors walked through common mistakes: how to hold containers, how to pour, how to clean spills. Institutions and companies can encourage this mindset with refresher courses and safety incentives. The payoff comes in lower incident rates and workers who trust each other to follow protocols.
Taking Personal Responsibility
Safety doesn’t always depend on the boss, supervisors, or government rules. Each person handling a compound shares the load. Skipping goggles or thinking a fume hood isn’t needed leads to risks that pile up. Small habits—like double-checking labels, never eating near chemicals, and washing hands before leaving the room—add up to a culture where people watch out for each other. It’s not just about avoiding trouble; it’s about showing respect for coworkers and the environment.
Building Better Systems
Companies and schools can make safety guidelines more visible and easier to follow for newcomers and veterans alike. Clear emergency instructions, readable safety signs, and accessible spill kits all help. The compound itself may never change, but safety information and habits do. If you ever doubt whether a substance needs special handling, assume the answer is “yes” and find out exactly what precautions belong in the toolkit.
What is the molecular weight and CAS number of this product?
Understanding the Basics: Why Molecular Data Matters
Choosing chemicals for research or production brings up two pieces of data over and over—molecular weight and CAS number. Plenty of folks glance right past these lines on the technical sheet, but these details shape everything from safety protocols to experimental results. Anyone who’s spent time at a lab bench knows the headache of grabbing the wrong bottle because a label seemed similar, only to discover that the reaction doesn’t work or, worse, a new hazard walks into the room. That’s not just a paperwork snafu; it can mean lost time, wasted money, and safety risks.
Why Care About Molecular Weight?
Grams, moles, concentrations—these calculations depend on one thing: knowing the weight of each molecule. I remember my first year out of college, running bioassays, and underestimating how much one digit off on molecular weight can mess up an experiment. If the compound’s molecular weight isn’t right, dosages get thrown off. The cells or animals don’t respond as expected. Results lose their value. With a correct molecular weight, scaling up batches for a pilot might go smoothly, saving headaches on the production floor. Getting it wrong leaves a trail of inconsistent data and irritated colleagues.
CAS Number: Cutting Through Confusion
Forget hoping the chemical’s trade name makes sense from lab to lab. The CAS number saves time and confusion. Each chemical—no matter the supplier, purity, or odd name—gets one unique identifier. I’ve worked with teams across continents, and nothing clears up confusion quicker across language or vendor differences than a CAS number. You can call it by the old name or a new brand, but if the CAS number matches, you’re holding the right molecule. That’s peace of mind, especially with regulatory needs tightening year by year.
Industry Facts: Data that Drives Safety and Compliance
Global regulations use these numbers as checkpoints. Safety data sheets, transport documentation, and compliance records all look for accurate molecular weight and CAS numbers. Without them, shipments get delayed or blocked. The European Chemicals Agency’s REACH program, for example, demands precise reporting of both details, or the product stays out of the market. Inconsistent data can even lead to large fines or recalls.
Reducing Risk and Streamlining Communication
Mix-ups can happen anywhere from ordering to documentation. Relying on molecular weight and CAS number, rather than product nicknames or assumptions, cuts down costly errors. It helps customer service track ingredient questions, assists in avoiding allergens or restricted substances, and supports audits or recalls. As someone who’s trained junior lab staff, I’ve seen reliance on names and catalog numbers go sideways. Insisting on CAS numbers in our internal systems solved those headaches fast.
Real Solutions: Making Data Work for You
Adopting strict data recording early helps avoid hassle later. Reputable suppliers display CAS and molecular weight front and center—if the numbers aren’t obvious, look elsewhere. Digital lab notebooks and order forms should require these fields. Software can flag discrepancies or missing IDs, while barcoded inventory keeps everything streamlined. Training staff to check CAS and molecular weight, not just packaging, creates a culture that respects accuracy as more than a suggestion. Ultimately, these numbers aren’t just technical trivia—they’re foundations for trust, safety, and scientific progress.
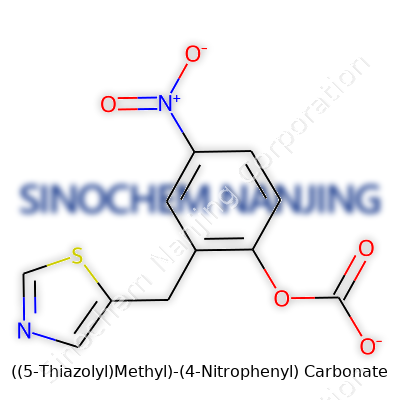
| Names | |
| Preferred IUPAC name | (4-nitrophenyl)\[(5-thiazolyl)methyl\] carbonate |
| Other names |
4-Nitrophenyl ((5-thiazolyl)methyl) carbonate ((5-Thiazolyl)methyl) 4-nitrophenyl carbonate 5-Thiazolylmethyl 4-nitrophenyl carbonate |
| Pronunciation | /ˈfaɪ-θaɪəˌzɒlɪl ˈmɛθɪl ˈfɔːrˈnaɪtroʊˌfɛnɪl ˈkɑːrbəˌneɪt/ |
| Identifiers | |
| CAS Number | 1372305-45-7 |
| Beilstein Reference | 3724535 |
| ChEBI | CHEBI:131967 |
| ChEMBL | CHEMBL3184917 |
| ChemSpider | 34255755 |
| DrugBank | DB08240 |
| ECHA InfoCard | echa.europa.eu/substance-information/-/substanceinfo/100.152.085 |
| EC Number | EC 626-520-2 |
| Gmelin Reference | 866256 |
| KEGG | C14369 |
| MeSH | D000077237 |
| PubChem CID | 139631785 |
| RTECS number | XN8225000 |
| UNII | KS6B9QY72B |
| UN number | UN3334 |
| CompTox Dashboard (EPA) | DTXSID2040573 |
| Properties | |
| Chemical formula | C11H8N2O5S |
| Molar mass | 321.28 g/mol |
| Appearance | Yellow solid |
| Odor | Odorless |
| Density | 1.5 g/cm³ |
| Solubility in water | Slightly soluble in water |
| log P | 1.98 |
| Acidity (pKa) | 5.2 |
| Basicity (pKb) | 10.2 |
| Refractive index (nD) | 1.613 |
| Viscosity | Viscous liquid |
| Dipole moment | 3.97 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 319.5 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | J04AK03 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS07, GHS08, GHS09 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P264, P280, P302+P352, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 1-2-1-🛢️ |
| Flash point | Flash point: 273.4 °C |
| LD50 (median dose) | LD50 (median dose): 500 mg/kg (rat, oral) |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for ((5-Thiazolyl)Methyl)-(4-Nitrophenyl) Carbonate is not specifically established by OSHA or other major regulatory agencies. |
| REL (Recommended) | 0–8°C |
| Related compounds | |
| Related compounds |
4-Nitrophenyl chloroformate 4-Nitrophenol 5-Methylthiazole Thiazole Phenyl carbonates |

