5-Tert-Butyl-2,4,6-Trinitro-M-Xylene: A Closer Look
Historical Development
Once the late 19th century welcomed the nitro aromatic compounds boom, chemists began hunting for new molecules that would outperform traditional explosives and dyes. 5-Tert-Butyl-2,4,6-Trinitro-M-Xylene grew from this curiosity. Early research papers from German and British laboratories routinely described the drive to pack more power and stability into smaller molecules, moving on from TNT and dinitrotoluenes. Over decades, the interplay between academic labs and industrial need shaped the way this compound was produced, stored, and studied. The Cold War added fuel to the quest for energetic materials, with research institutions and defense contractors pouring time and resources into synthesis and performance evaluations. Modern scientific advances—refined crystallography, better synthetic routes, and improved safety standards—marked each twist and turn in this story.
Product Overview
5-Tert-Butyl-2,4,6-Trinitro-M-Xylene stands as a dense, yellow solid with properties that set it apart from simpler nitro aromatics. At a glance, you notice the tert-butyl group crowning the molecule—making it less likely to clump or degrade, even in less-than-ideal storage conditions. It has drawn attention across laboratories due to its energetic characteristics and physical stability. Everyone from military engineers to materials scientists keeps an eye on it for new formulations. The strong electron-withdrawing ability from the three nitro groups changes how the molecule reacts when sparked or shocked, pushing it past the boundaries of more pedestrian cousins like TNT.
Physical & Chemical Properties
Shifting from the white powders of basic laboratory chemicals, 5-Tert-Butyl-2,4,6-Trinitro-M-Xylene catches the eye with its light yellow crystalline form. Its melting point hovers above 100°C, higher than many traditional nitro compounds, which hints at the stability added by tert-butyl and xylenic structure. High density—well over 1.5 g/cm³—makes it a powerful material in energy-based applications. Solubility stays modest; most common solvents pull out little of it, so special blends or hot solutions are needed for purifications. Chemical resilience pops out under scrutiny: acids and bases don't break it down easily, and the molecule shrugs off damp air, crucial for long-term storage or transport. Analytical labs see its spectra—a mess of peaks driven by nitro influence—yet reproducible enough that practitioners learn to recognize it with routine scans.
Technical Specifications & Labeling
Manufacturers treat each lot of 5-Tert-Butyl-2,4,6-Trinitro-M-Xylene with precision. Purity often reaches above 98%, checked by HPLC and NMR. Labels list CAS number, molecular formula (C13H15N3O6), molar mass, physical form, and storage details. Safety sheets flag shock sensitivity, recommended storage temperatures, and the need for strict controls around oxidizing agents. Batches destined for research carry lot-specific data so chemists can trace changes in performance or reactivity. For transport, packaging employs sealed glass bottles, often nested in cushioned metal canisters to handle mechanical and thermal shocks. Each label includes emergency handling instructions and hazard pictograms, reflecting lessons learned from earlier, less regulated eras.
Preparation Method
Synthesis starts with m-xylene, then seasoned chemists guide it through electrophilic aromatic substitution. Introducing tert-butyl via Friedel-Crafts alkylation requires dry conditions and a catalyst—commonly aluminum chloride. Next, nitration proceeds in multiple steps, since piling on three nitro groups demands careful control of temperature and acid ratios. Each step faces tight scrutiny, as runaway reactions unleashed in the early 20th century forced a culture of caution. Crude product often contains a mix of isomers and tarry by-products, so teams rely on successive crystallizations and solvent washes. Inquiries from newer labs usually focus on boosting yield, tweaking solvents, or swapping acids for greener alternatives—reflecting both economic and environmental concerns.
Chemical Reactions & Modifications
Chemists explore 5-Tert-Butyl-2,4,6-Trinitro-M-Xylene for opportunities beyond just its parent form. Reduction with metals or hydrides can shave off nitro groups, giving access to amino-derivatives popular in dye chemistry or as intermediates in pharmaceutical research. Mild alkaline hydrolysis sometimes breaks the tert-butyl off, opening a new set of building blocks for high-energy materials. Halogenation and further nitration rarely appeal, as sterics and electronic effects limit the molecule’s flexibility. Researchers hunt for ways to attach the molecule to polymers or blend with stabilizers, always reevaluating how tweaks in the backbone shift thermochemical output. The energetic materials field looks for ways to increase compatibility with plasticizers, easing the path toward safer, more manageable explosive mixtures.
Synonyms & Product Names
Anyone in the field soon bumps into alternative names, which show up on import documents and in catalogues from specialty vendors. “Trinitro-tert-butyl-m-xylene,” “2,4,6-trinitro-5-tert-butyl-m-xylene,” or just “TBNX” find use across colloquial and formal discussion. Product numbers differ from lab to lab, but the root identifiers all circle back to the same core structure. For those handling international trade or compiling literature reviews, keeping tabs on these synonyms prevents expensive, dangerous mix-ups.
Safety & Operational Standards
Working with 5-Tert-Butyl-2,4,6-Trinitro-M-Xylene demands respect. Modern protocols, taught in every decent chemical engineering program, focus on three big risks: accidental detonation, fire, and chronic health effects. Labs install blast shields and automated handling arms, borrowing tactics from the pharmaceutical industry and adapting them for energetic material synthesis. Even small spills or dust clouds mean real risk, so teams drill safety responses and maintain access to first-line fire suppression gear. Regulation, including the ATF’s oversight in the US and REACH in Europe, updates the bar for training, storage, and reporting any incidents. Inspections look for compliance with threshold quantities, to avoid stockpiling beyond safe operational limits.
Application Area
People often associate compounds like this with explosives alone, yet its reach stretches further. Defense industries shape it into main-charge boosters or fuel-air explosives, banking on stability and high detonation velocity. Mining companies dip into these shelves, blending it into specialty charges that slice through seams where dynamite or ANFO struggle. Researchers working in organic synthesis consider its derivatives as stepping stones to dyes and advanced polymers, appreciating the way nitro groups interact with light or boost reactive potential. Small startup labs sometimes court controversy, aiming for “green” oxidizers by manipulating the tert-butyl group, though scalability often kills these dreams in the pilot phase. Law enforcement agencies monitor sales and movement as a matter of course, wary of diversion to illicit use.
Research & Development
Universities and military contractors treat 5-Tert-Butyl-2,4,6-Trinitro-M-Xylene as a testing ground for next-generation energetic materials. Studies compare crystal morphologies, searching for those elusive forms that pack more punch into every gram without tipping into dangerous unpredictability. Computational chemists run simulations on impact and friction sensitivity, connecting molecular structure to field performance. Funding often comes in waves—spiking during geopolitical tension and dipping as regulatory scrutiny intensifies. Each year brings new patents around safer handling, improved syntheses, or couplings with other energetic compounds. Environmental chemists focus on degradation pathways, examining what happens after an explosion or accidental release in groundwater, building risk models to inform policymakers and manufacturers.
Toxicity Research
Toxicologists dig deep into how 5-Tert-Butyl-2,4,6-Trinitro-M-Xylene affects living organisms. Animal studies from the 1980s highlighted disruptions in liver enzymes, blood chemistry, and nervous system responses. More recent data points toward slow breakdown in soil or water. Nitrosamines, well-known carcinogens, can form during some storage or incineration conditions, making disposal a major concern. Regulators review occupational exposure data, tracking cases of skin irritation and respiratory issues. Chronic low-dose exposure doesn’t provoke immediate crises, yet accumulating data signals caution, particularly for pregnant workers or those with preexisting conditions. Industry guidelines push for protective clothing, air handling, and routine environmental monitoring, updating best practices whenever new evidence emerges.
Future Prospects
Global attention to sustainability and the circular economy means compounds such as 5-Tert-Butyl-2,4,6-Trinitro-M-Xylene must justify their footprint. Heavy regulation has trimmed some research budgets, yet demand for high-performance explosives, specialty dyes, and energetic materials remains strong in national security and select industries. Green chemistry initiatives urge the design of derivatives that break down quickly after use—avoiding bioaccumulation or leaching. Efforts continue to boost yields, cut waste, and swap out toxic reagents, aiming to bring life-cycle impact below legacy nitro compounds. Some teams pitch the molecule as a model for teaching advanced organic chemistry, due to its challenging synthesis and distinctive properties. Each push for innovation carries both promise and tension, balancing performance demands against societal and environmental responsibility.
What is 5-Tert-Butyl-2,4,6-Trinitro-M-Xylene used for?
Understanding the Substance
5-Tert-Butyl-2,4,6-Trinitro-M-Xylene, better known in the explosives industry as Methyl-Tert-Butyl TrinitroXylene or simply MBTNX, rarely surfaces in everyday headlines. Its name points squarely toward its chemical roots, but the starring role of this compound belongs to its explosive power.
Key Uses and Importance
MBTNX sits among a group of nitroaromatic explosives. Its unique chemical structure gives it high stability during storage and high energy release on detonation. Factories turn to MBTNX and compounds like it for specialized blasting agents, often in mining, construction, or military applications. The focus here stems from the need for materials that balance stability with raw explosive force. MBTNX’s molecular arrangement resists moisture absorption and handles rough transport better than most alternatives.
Mining crews depend on reliable explosives for controlled demolition. When working underground, every ounce of predictability matters since unpredictable reactions could mean disaster. MBTNX fills in where older explosives either break down in humid shafts or lack the brunt needed for efficient rock breaking. This reliability makes lives easier for engineers and laborers working in some tough conditions.
Groups handling unexploded ordnance take the stability of MBTNX seriously. Few things make a bigger difference to a disposal squad than the confidence that a compound will not detonate from a stray bump or temperature change. MBTNX’s resilience limits accidental detonations, helping safeguard teams in bomb disposal missions.
Environmental and Safety Considerations
The topic of explosives always raises concerns about environmental impact. Nitroaromatic compounds bring risks of soil and water contamination if left unmanaged, and MBTNX does not escape this reality. Some studies have found that residues from explosive compounds linger in ecosystems. Animals and plants can absorb residual chemicals and pass them along food chains, bringing questions about long-term safety. Waste management in the explosives industry has to include careful tracking and neutralization of these residues. Otherwise, rivers and farmlands may suffer from persistent chemical pollution.
On the safety side, working with MBTNX calls for careful oversight. Explosives, by their nature, test the boundaries of workplace safety. Routine exposure to dust or runoff, lack of training, or improper storage can build toward tragedy. Places dealing in MBTNX reinforce strong safety cultures, frequent monitoring, and well-trained staff. The history of industrial accidents has shown that cutting corners leads to injury or worse. The lessons here echo across industries—safety takes hard commitment, not just signs and policies.
How to Address the Challenges
Environmental responsibility starts before mixing or deploying MBTNX. Testing soil and water near blasting sites sets a baseline for later comparison. Investment in containment technology also pays off, whether through lined waste pits or treated effluent systems. Partnering with environmental chemists sharpens insight into how residues behave and degrade after use. Regular training makes workers partners in safety rather than bystanders. Building an honest reporting culture allows everyone to learn from near misses as much as from successes.
MBTNX holds power and risk in equal share. Wrangling those two qualities means ordinary workers, researchers, and managers have to keep pace with science, regulation, and each other. Oversight backed by real knowledge keeps communities and workers safer, and slows down the legacy problems coming from short-term thinking. That’s the path forward with these complex chemicals—steady, careful work, backed by experience and responsibility.
What are the safety precautions when handling 5-Tert-Butyl-2,4,6-Trinitro-M-Xylene?
Small Mistakes, Big Risks
5-Tert-Butyl-2,4,6-Trinitro-M-Xylene, often called Metrinitrobenzene, turns up in lab protocols and industrial formulas, though few folks outside chemistry circles will have heard about it. People use it in explosives research and sometimes specialized chemical testing. If you’ve had any experience around nitro compounds, you've probably heard enough scary stories to know you shouldn’t get casual with them. Once, I watched a seasoned lab tech triple-check every step, never rushing even simple motions. That kind of steady caution matters more here than almost anywhere else.
The Basics Matter Most
Nobody expects an explosion. That’s where people trip up. Lab work trains for clarity—goggles actually over your eyes, gloves sealed at the cuff, apron snapped closed. Metrinitrobenzene can be toxic and shock-sensitive, and it may cause headaches or skin irritation. It also gives off dust nobody should breathe. Use a proper fume hood—not just a cracked window. A good pair of gloves and a splash-resistant coat mean less chance of chemical burns. Even now, I remember the sting from one accidental splash of a milder compound. It set me straight for life; with powerful reagents, you want your skin and eyes covered, and that’s non-negotiable.
Treat It Like It’s Lit
Materials that detonate when bumped or heated need respect. I’ve found people downplay how unpredictable things can get when a routine gets interrupted—a dropped tool, a scraped bottle, an unexpected heat source. Store Metrinitrobenzene in cool, well-ventilated spots, away from direct sunlight and heaters. Use only spark-free equipment—no friction, no static, no open flames. Even the dust over time can get tricky, so regular, disciplined cleaning and clear workspace rules really do make a difference. Keep only as much as you need out; everything else stays sealed, labeled, in approved containers.
Preparation Saves Lives
Training sets most labs apart from garages. People talk about safety routines like burdens, but drills and emergency plans work. In one incident, an immediate steps guide by the fume hood stopped a minor spill from getting out of hand. If you’re ever uncertain about a container, a memory fogs up, or a label starts to peel—stop until you can get clarity. It’s not dramatic, just normal procedure, and it works. Emergency eyewashes and showers never gather dust. Fire extinguishers stand in reach but never feel like clutter.
Smarts over Speed
Good habits keep people working safely day after day. Read every line of safety datasheets, stay patient with protocols, and talk it through any time you feel unsure. Routine almost always breeds space for mistakes, so stay mindful. Nobody learns it all in a classroom, but everyone remembers their first real close call. If you always treat explosives as dangerous and give them room, you lower risks for everyone around. That’s not just good practice—it’s the difference between a career and a cautionary tale.
What is the chemical structure of 5-Tert-Butyl-2,4,6-Trinitro-M-Xylene?
Examining an Unusual Nitroaromatic Compound
Some molecules tell a story just by looking at them. 5-Tert-Butyl-2,4,6-Trinitro-M-Xylene—quite the mouthful—draws plenty of interest, not only from chemists but from folks trying to understand what makes explosives tick. The structure of this compound gives away some clues about how it might behave physically and chemically.
Dissecting the Anatomy of the Molecule
Chemists like to break a complicated name into chunks. Here, “M-Xylene” means a benzene ring with two methyl (–CH3) groups in the meta positions, which are carbons 1 and 3. Add a tert-butyl group at position 5—a bulky appendage just hanging off the ring with three methyl branches. What makes this molecule pop is the trio of nitro groups. Fitting them at positions 2, 4, and 6, it almost starts to look like a spiky little sea urchin—highly substituted, crowded, almost jittery.
Drawing this structure by hand brought back memories of staring at textbooks late at night, realizing how packed molecular space could get. Introducing three nitro groups (–NO2) jams electron-withdrawing functionalities near each other. Nitro groups pull electron density out, making the ring less hospitable to more substitutions, but in this design, every position fights for space and influence. That overcrowding inflicts real effects on physical properties—think melting point, solubility, and, most importantly, chemical reactivity.
Why the Shape and Stresses Matter
This molecule’s arrangement matters far beyond the drawing board. Take 2,4,6-trinitrotoluene (TNT), a relative. TNT earned its dangerous reputation partly because of the way its nitro groups destabilize the benzene core and make it ready to release energy. Add more bulk with a tert-butyl and another methyl? That changes how heat and shocks ripple through the material.
Laboratories, including some I’ve visited, handle trinitro aromatics with respect and sometimes trepidation. Bulky substituents like tert-butyl can create unexpected safety profiles—sometimes more friction-sensitive, sometimes less. The structure dictates critical qualities like density, crystal packing, and sensitivity. Data from technical literature suggest that tert-butyl groups in aromatic nitro compounds lower density and slightly dull sensitivity, though every structure tells its own tale.
Sharing Knowledge and Encouraging Responsible Chemistry
Not many people walk into work, wondering how the structure of a nitroaromatic compound impacts public safety, but plenty benefit from that science. Unintended exposure to energetic materials can lead to disaster; thoughtful research and training keep labs and communities safe. In my own work with students, I’ve seen how recognizing the role of big, clunky groups like tert-butyl helps build a healthy respect for chemical unpredictability.
Moving forward, open dialogue and clear guidelines matter more than isolated know-how. Push for transparency in chemical databases, rigorous training on physical properties, and access to modern structure-activity data. That’s how responsible chemistry grows and protects all sorts of people, not just those wearing lab coats.
How should 5-Tert-Butyl-2,4,6-Trinitro-M-Xylene be stored?
Hands-On Risks Call for Respect
If you’ve worked with energetic chemicals, you recognize the importance of treating each compound with respect, especially those with “trinitro” in the name. 5-Tert-Butyl-2,4,6-Trinitro-M-Xylene falls right into the category of materials that demand cautious handling and thoughtful storage. The experience of opening a cabinet and spotting cracked jars or odd-smelling residues reminds you that shortcuts have real consequences. All it takes is one overlooked factor—humidity, temperature swing, or friction—to ignite disaster with compounds like this.
Straightforward Storage Practices
The compound deserves storage because of its explosive nature and sensitivity to environmental triggers. Let’s cut to the basics. You need a cabinet rated for explosives. These cabinets provide physical barriers and isolate the chemical away from everyday workspaces. Thick steel, grounded surfaces, and space between each container help prevent accident escalation. A locked, durable storage unit set in a low-traffic, monitored area sets a strong foundation for safety.
Conditions inside the cabinet directly affect shelf stability. Heat and sunlight speed up decomposition. Humidity creeps in and can react with chemical residues. Both need to stay low—ideally under 25°C for temperature and close to 0% relative humidity. Desiccants work well for drawing out moisture; I favor using fresh silica gel packs. Always close containers tightly; air sneaks in easier than you might expect, especially in glass bottles with old lids.
Container Choice Makes All the Difference
Pick containers designed for reactive solids. Glass works, but consider thick-walled types that resist pressure. Some labs lean toward certain plastics, yet plastic can develop static, which sparks serious trouble. Avoid storing in metal unless the material is treated to avoid unexpected reactions or friction. Don’t fill containers to the brim; a little headspace helps manage expansion and avoids messy leaks after bumps or temperature swings.
Labels stay visible, bold, and clear, with backup logs outside the storage area. In practice, nobody wants to wonder what sits behind a faded label months down the line. Everyone I’ve worked with keeps a chemical inventory and updates it every time they add or remove material. This may sound like extra paperwork, yet it’s the step that stops you from stumbling onto forgotten leftovers later.
Minimizing Storage and Maximizing Safety
Keep only what current work demands. Ordering bulk quantities isn’t safer or cheaper if half the material sticks around in storage. Smaller, clearly dated batches move out before instability creeps in. If possible, rotate stock, using older supplies first and never mixing different ages in the same jar. Many dangerous incidents in chemistry labs began with old stock someone forgot about, sealed up for years.
In my experience, clear communication keeps everyone on track. Let coworkers know any time you transfer or open a jar. Store emergency gear nearby: spill kits, firefighting supplies, and personal protective equipment. Quick response only happens if you plan ahead and share that plan.
Waste Disposal: Don’t Wait Until It’s an Emergency
Disposing of expired or surplus material closes the safety loop. Contact hazardous waste disposal professionals; never dump or dilute trinitro compounds yourself. I’ve seen teams stall, hoping the issue disappears. It never does. Disposal experts know how to handle every step, giving you more confidence each time you open the door to the storage cabinet because nothing inside has turned into a lurking hazard waiting for the next mistake.
What are the potential health hazards of 5-Tert-Butyl-2,4,6-Trinitro-M-Xylene?
A Closer Look at a Lesser-Known Chemical
5-Tert-Butyl-2,4,6-Trinitro-M-Xylene belongs to a group of chemicals known for their use in explosives and specialty chemicals. This isn’t a substance found on neighborhood store shelves. Most folks don’t know the name, but the risks linked to handling or exposure should not get ignored—especially for anyone working in labs, manufacturing, or storage sites where it might turn up. Chemicals in the same family—think TNT—already carry heavy baggage, with documented effects on both people and the environment.
Risks to People Handling or Exposed to the Substance
Anyone in a facility where this compound is manufactured or used taps into a world where personal protective equipment isn’t optional. Inhaling dust or vapors can irritate the throat and lungs, trigger headaches, and, over time, might even harm blood and liver function. Some nitroaromatic compounds cause anemia by damaging red blood cells; these kinds of health troubles do not usually announce themselves with a warning sign in the early stages. Skin contact brings similar risks—dermatitis, redness, and, in rare cases, long-term effects that nobody wants as a surprise later in life.
Eye exposure causes burning, tearing, and temporary vision changes. If I’ve learned anything from safety work in old industrial labs, it’s that accidents can turn risky with just a small spill or splash. Folks who do this work every day know the uneasy feeling when handling something that could put their health on the line because proper storage, ventilation, and barrier clothing matter a lot more than luck.
More Than Physical Health at Stake
Many overlooked risks trace back to repeated or long-term exposure. Studies on related chemicals—including other nitroaromatics—link chronic contact to risks like carcinogenicity and nervous system effects. OSHA and the EPA spell out clear standards for substances in this family; those workplace posters aren’t there for decoration. Proper labeling, restricted access, and rigorous training all cut down on the likelihood of serious consequences.
One nagging worry: environmental exposure. Chemicals in this class linger in soil and water, breaking down slowly and threatening organisms far beyond the plant gates. Local mishaps can lead to fish kills or crop damage down the line. A simple mistake, like dumping leftover material or not checking leaks, can ripple through local communities.
Coming Up With Better Protections and Solutions
Engineering safer working conditions stands higher on the list than bracing for bad outcomes. This means strong ventilation, automated transfer systems that cut down open handling, and gloves and goggles that turn away both splashes and dust. Regular health monitoring gives early warning for anyone showing early symptoms from low-level exposures, which is crucial—better to catch issues early than face long-term fallout.
Some companies now use less toxic substitutes for nitro compounds wherever possible. Finding greener alternatives is often expensive or slow, but it adds up to fewer hazards for workers and the world outside. Some basic steps—finding leaks, training new staff, and using spill kits right away—lower the odds that anyone has to find out about the chemical’s worst effects the hard way.
Responsibility Grows With the Risk
From experience, I see that no checklist covers every angle—sometimes you only get one shot at getting safety right. Everyone in the chain, from management to new hires, plays a role in keeping these hazards in check. Treating 5-Tert-Butyl-2,4,6-Trinitro-M-Xylene with the care it demands keeps risk in its place and makes sure the worst-case scenarios stay on paper, not in real life.
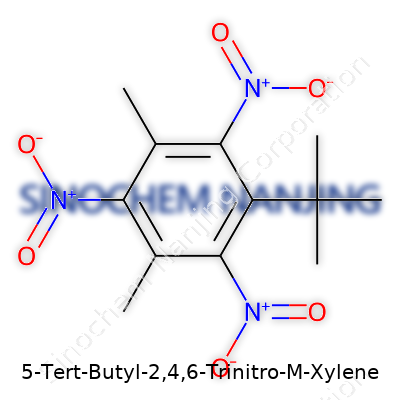
| Names | |
| Preferred IUPAC name | 2-tert-butyl-3,5-dimethyl-1,4,6-trinitrobenzene |
| Other names |
Musk Xylene Musk Xylol Fixolide Musk Ambrette Xylene 6-Tert-Butyl-3,5-dimethyl-2,4,6-trinitroanisoIe 6-Tert-butyl-2,4,6-trinitro-3,5-xylenol |
| Pronunciation | /ˈfaɪ tɜːrt ˈbɜːtɪl ˈtuː fɔːr sɪks traɪˈnaɪtroʊ ɛm ˈzaɪliːn/ |
| Identifiers | |
| CAS Number | 131-73-7 |
| 3D model (JSmol) | `3DModel_JSmol:5-Tert-Butyl-2,4,6-Trinitro-M-Xylene` |
| Beilstein Reference | 3647583 |
| ChEBI | CHEBI:131747 |
| ChEMBL | CHEMBL2007613 |
| ChemSpider | 15341017 |
| DrugBank | DB13981 |
| ECHA InfoCard | 100.013.899 |
| EC Number | 616-107-0 |
| Gmelin Reference | 313225 |
| KEGG | C19275 |
| MeSH | D038792 |
| PubChem CID | 159750 |
| RTECS number | XZ2000000 |
| UNII | NZ8B10CMOA |
| UN number | UN1321 |
| CompTox Dashboard (EPA) | `DTXSID5047279` |
| Properties | |
| Chemical formula | C13H16N3O6 |
| Molar mass | 317.26 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 1.37 g/cm³ |
| Solubility in water | Insoluble |
| log P | 1.706 |
| Vapor pressure | 1.8E-7 mmHg at 25°C |
| Acidity (pKa) | pKa = 6.76 |
| Basicity (pKb) | 6.88 |
| Magnetic susceptibility (χ) | -0.00048 |
| Refractive index (nD) | 1.605 |
| Viscosity | Viscous yellow oil |
| Dipole moment | 3.89 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 523.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -291.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -6546 kJ/mol |
| Hazards | |
| Main hazards | Explosive; harmful if swallowed; causes skin and eye irritation; may cause respiratory irritation. |
| GHS labelling | GHS02, GHS06, GHS09 |
| Pictograms | GHS01,GHS02,GHS03,GHS07 |
| Signal word | Danger |
| Hazard statements | H201, H302, H332, H315, H319, H335 |
| Precautionary statements | P210, P220, P240, P250, P260, P264, P270, P273, P280, P283, P284, P301+P310, P302+P352, P304+P340, P305+P351+P338, P306+P360, P308+P311, P332+P313, P333+P313, P337+P313, P362, P363, P370+P378, P371+P380+P375, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-3-4-✕ |
| Flash point | 110 °C |
| Lethal dose or concentration | Oral rat LD50: 700 mg/kg |
| LD50 (median dose) | LD50 (median dose): 600 mg/kg (rat, oral) |
| NIOSH | NA0450000 |
| PEL (Permissible) | PEL (Permissible exposure limit) for 5-Tert-Butyl-2,4,6-Trinitro-M-Xylene: Not established |
| REL (Recommended) | Not established |
| Related compounds | |
| Related compounds |
2,4,6-Trinitrotoluene 1,3,5-Trinitrobenzene 5-Tert-butyl-2,4,6-trinitro-m-xylene monohydrate 1,3-Dimethyl-2,4,6-trinitrobenzene |

