5-Nitro-1,3-Xylene: A Detailed Commentary
Historical Development
Industrial chemistry has shaped the world, and 5-Nitro-1,3-Xylene stands as one of those substances with a complex backstory. Early chemical industries explored aromatic nitration reactions out of necessity—dyes, explosives, and intermediates for other chemicals beckoned. During the height of synthetic chemistry advances in the late nineteenth and early twentieth centuries, scientists learned how to manipulate xylene isomers into hundreds of new compounds. The specific nitration at the 5-position on 1,3-xylene (also known as m-xylene) required careful experimentation. Old patents shed light on the willingness of early chemists to tweak reaction gases, acids, and temperatures until the right product emerged. Today, modern manufacturing benefits from that persistent lineage of painstaking trial and error that never saw setbacks as failures, only as signposts toward a safer and more efficient route. Factories in industrial regions refined processes to meet growing demand for nitroaromatic intermediates, which helped scale production and provided local economies a chance to support the expanding synthetic materials sector.
Product Overview
5-Nitro-1,3-Xylene does not come across as a household name, though its footprints show up in countless specialty products. Its main draw comes from its structure—a benzene ring with two methyl groups at the meta positions and a single nitro group hanging off the fifth carbon. This combination brings a balance between hydrophobic and reactive properties, which can be leveraged by chemical manufacturers looking for nitroaromatic intermediates. Large-scale facilities encapsulate the industrial reality behind the laboratory discovery, producing reliable product with consistent attributes. 5-Nitro-1,3-Xylene often enters the stream as a yellowish crystalline solid, and manufacturers stamp out batch after batch to fulfill needs in various sectors. The demand fluctuates as downstream industries call for more or less of this compound, but the chemical’s versatility ensures it remains a staple.
Physical & Chemical Properties
This nitro compound holds onto its aromatic core, making it more stable than you might expect from something bearing a nitro group. The substance tends to crystallize out with a faint yellow color, a physical flag for product verification. Melting points hover near 70°C, and boiling proceeds at much higher temperatures, reflective of the aromatic backbone’s stability and the added methyl groups. Solubility lands low in water, thanks to the hydrophobic ring and methyl groups, while organic solvents dissolve it with ease—especially ether, alcohol, and chloroform. The nitro group at the fifth position doesn’t simply hang on passively; it shapes both the electron density around the ring and how the compound responds to reducing or further substitution reactions. Once in the process stream, 5-Nitro-1,3-Xylene can interact in ways both predictable and surprising, particularly when subjected to strong acids, bases, or reduction systems.
Technical Specifications & Labeling
Manufacturers print key specifics on drums and containers for safety and compliance. Typical quality benchmarks outline purity above 98% and controlled moisture content, since water can influence downstream reactivity. Impurities result mainly from incomplete nitration or side reactions, which experienced labs weed out by distillation or recrystallization. Labels usually feature the CAS number 1510-02-1, hazard identification, and compliance marks in line with regulatory bodies like REACH and OSHA. Packing materials get chosen for chemical compatibility and transport security, as aromatic nitro compounds require safeguards against heat, shock, or accidental mixing. Detailed certificates of analysis stand as common denominators in sales contracts, reflecting customer insistence on reliable, repeatable quality. Shipping papers describe the solid’s features—yellow crystals, strong aromatic odor—and warn handlers of its potential risks in storage and transit.
Preparation Method
Scaling up the nitration of 1,3-xylene brought out the ingenuity of process engineers. The basic approach starts with m-xylene, sending it into a reaction with concentrated nitric and sulfuric acids under controlled temperatures. Pouring acids over an aromatic hydrocarbon would ordinarily sound reckless, but strict temperature profiles and stirring rates have tamed this exothermic step over decades. Engineers learned how to keep side reactions minimal and maximize para-nitration, which is needed for the 5-nitro product. They also dialed in work-up protocols—neutralization of spent acid, precipitation of crude crystals, and filtration steps. Purification usually takes the form of vacuum distillation or repeated re-crystallization, trimming off trace contaminants. Waste management plans and contained reactor systems now reduce hazards that marked early nitrate chemistry, with automated controls reducing risk for those on the factory floor. These advances let producers supply bulk 5-Nitro-1,3-Xylene with uniform quality, batch after batch.
Chemical Reactions & Modifications
Once synthesized, 5-Nitro-1,3-Xylene becomes a crossroads for further chemical manipulation. The nitro group, highly electron-withdrawing, turns the molecule into a candidate for reductions—yielding amines that find use in pharmaceuticals, dyes, and polymers. Catalytic hydrogenation and tin/HCl reduction both lead to aminoxylene derivatives, which open doors to additional functionalization. Beyond reduction, the two methyl groups present distinct functional handles. Chlorination swaps hydrogens for chlorines with the right conditions, and bromination remains selective if process controls stay tight. Chemists sometimes install sulfo or carboxy groups, and those side chains empower even more transformation into downstream specialty molecules. Reactions with formaldehyde or alkali can knit these building blocks into oligomers or set them up for further derivatization. Each successive modification tracks back to the reliability of that original nitration step and feedstock purity.
Synonyms & Product Names
5-Nitro-1,3-Xylene wears several aliases in chemical catalogs. Names like 1,3-dimethyl-5-nitrobenzene, and m-xylene, 5-nitro vary in listings by vendor or region. The CAS number 1510-02-1 brings some consistency across global supply chains. Specialty distributors highlight its status as a nitroaromatic building block. Academic papers reference IUPAC systematic names but tend to revert to shorthand in discussions, reflecting a broader tendency for practicality in the lab. Buyers and sellers come to recognize all major synonyms through experience and context, as the paper trail coming with every shipment reflects these alternate names for quality and safety tracking.
Safety & Operational Standards
Handling nitroaromatics such as 5-Nitro-1,3-Xylene brings responsibilities. Personal experience shows that even seasoned chemists respect the risks that come with exposure to nitro compounds—skin irritation, eye contact concerns, and the potential for more serious systemic toxicity. Facilities train staff to use nitrile gloves, splash goggles, and fume hoods when weighing or transferring solid material. Storage guidelines call for cool, dry spaces away from oxidizers and reducing agents, since inadvertent reactions can lead to product loss or worse, fire hazards. Safety data sheets clarify response actions for spills, inhalation, or accidental exposure, and research labs drill emergency protocols into muscle memory. Environmental management ensures that effluent containing nitroaromatics sees proper treatment, not discharge, since persistence and toxicity make these compounds unwelcome in groundwater or surface systems. Careful labeling and redundant checks during transportation help reduce missteps, and regulatory oversight from agencies such as EPA or ECHA keeps industry vigilant.
Application Area
5-Nitro-1,3-Xylene finds its most steady employment as an intermediate for specialty chemicals. Its nitro group enables further processing into amines, key for synthesizing advanced dyes, pigments, and pharmaceuticals. The xylene backbone, with its two methyl groups, gives derivatives remarkable staying power in both polymer and agrochemical markets. Some uses draw on its value in research—screening analogs for electronics, specialty elastomers, or even certain energetic compounds. The reach of this molecule stretches beyond fine chemistry, as industrial lubricants, plasticizers, and antioxidant precursors all sometimes build on aryl nitro intermediates. Each application builds off the stability of the compound as a starting material and its readiness for tailored modifications, making it a tool for progress rather than a relic of bench-scale lore.
Research & Development
Innovation depends on the raw materials researchers can trust. Over the years in lab environments, I've seen 5-Nitro-1,3-Xylene selected for targeted studies into aromatic reactivity, nitration mechanisms, and the synthesis of novel polymers. Some groups probe the material for potential use in light-absorbing compounds, while others feed it into combinatorial libraries for drug discovery research. As industrial demand pushes for greener and safer nitration routes, R&D efforts focus on reimagining classic processes—solvent swaps, recycling acids, or continuous flow systems. Academic partnerships sometimes yield patents for new downstream derivatives or reaction protocols, which then filter back to commercial labs aiming to improve yield or cut operational hazards. Forward-thinking producers keep an eye on academic preprints and process journals because those breakthroughs often become tomorrow’s production standard.
Toxicity Research
The health impacts of nitro compounds never slip far from scientific focus. Toxicologists have scrutinized 5-Nitro-1,3-Xylene for acute and chronic effects—animal studies probe oral, dermal, and inhalation exposures. The compound shows moderate acute toxicity, with potential for methemoglobinemia, a blood disorder that reduces oxygen carrying capacity. Chronic exposure risks get more complicated and hinge on cumulative effects. Regulatory bodies require data on mutagenicity, carcinogenicity, and ecotoxicology before greenlighting commercial scale production or environmental release. Industry routinely runs workplace monitoring, and effluent gets tracked for residual nitroaromatics. Ongoing research investigates better detection methods, blood biomarkers, and the real-world breakdown products in the environment. Risk reductions come from controlling dust, using closed handling systems, and substituting less hazardous intermediates wherever feasible, though specialty markets sometimes preclude simple substitution.
Future Prospects
The path forward for 5-Nitro-1,3-Xylene tracks with the evolving needs of specialty chemical production, sustainability goals, and regulatory shifts. Pressure mounts to refine synthesis with greener solvents and lower energy costs, leveraging new catalytic systems or continuous flow chemistry. Digitized plant control and process analytics improve yields and reduce accidents. Regulatory agencies push for reduced emissions and stricter exposure limits, fueling investments into better waste treatment and process safety measures. Demand in downstream applications will likely remain cyclical but persistent—dyestuffs, pharmaceuticals, and agrochemical intermediates require flexible, reliable nitroaromatics. Expect research to expand in areas like environmental fate, safer derivatives, and even biodegradability. Investing in worker safety, environmental controls, and process innovation squarely aligns with the long-term value this nitro xylene delivers to industry and society alike.
What is 5-Nitro-1,3-Xylene used for?
Chemical Properties and Immediate Uses
5-Nitro-1,3-xylene catches the eye because of its structure: a xylene backbone with a nitro group attached. This compound often makes its way into labs and factories that work with dyes, pigments, and chemicals designed to shift color or improve resistance. Chemists might rely on it as an intermediate step in getting from starting materials to finished goods—turning base chemicals into something that looks or performs differently. One look at industries that regularly reformulate synthetic dyes, and you’ll find this compound somewhere in the process. In my time working near paint manufacturing, folks preferred such intermediates because they deliver reliable changes to chemical color profiles or stability.
Industrial and Scientific Applications
Companies in the dye and pigment sector tend to use the compound to add or tweak colors. These aren’t just for art or clothing. Stable dyes based on organic molecules show up in everything from printer inks to plastics. As industries develop new products, especially where standing out on a shelf matters, chemists behind the scenes rely on versatile building blocks. 5-Nitro-1,3-xylene gets incorporated into the larger framework of molecules that make everything from neon tennis balls to waterproof textiles possible.
On the scientific front, specialty labs see this compound as a starting material. It often takes several steps, but the compound offers predictable reactions. For researchers developing new pharmaceuticals or advanced materials, that kind of predictability matters. Using compounds with clear pathways lowers unexpected detours and streamlines R&D, especially for teams under pressure to innovate.
Potential Environmental and Health Concerns
With every benefit, there’s a catch. 5-Nitro-1,3-xylene doesn’t break down quickly in the environment. Handling it safely takes training and equipment. During my time working in industrial labs, I saw the emphasis put on protective gear and storage. Some derivatives can irritate skin or damage organs over long periods, so companies need strong protocols. Factories that cut corners run the risk of polluting waterways or exposing workers, and you don’t have to look far in history to see the real consequences. Keeping spills contained and waste managed stops bigger problems before they start. Many local regulations require documenting every transfer of such chemicals and monitoring workplace exposure closely.
Balancing Progress and Safety
It’s clear that compounds like 5-Nitro-1,3-xylene help drive industries demanding innovation—coatings, plastics, printing, even pharmaceuticals. As a result, companies continually face the choice: push forward with products that use these molecules and risk potential harm, or take the time to develop safer alternatives and better handling systems. Switching to less hazardous options takes research money and time, both of which run in short supply for many businesses trying to keep up with global competitors.
Investing in better waste management and transparent reporting offers another layer of protection. In regions where regulators and industries work together, community health outcomes often improve. In my experience, companies that prioritize open communication with workers and residents about chemicals on site build trust and avoid many headaches down the road. There’s real value in keeping both jobs and neighborhoods healthy.
Looking Ahead
There’s no denying the significance of compounds like 5-Nitro-1,3-xylene. As industries keep chasing brighter, tougher, and more resilient materials, the demand for specialty chemicals like this one isn’t slowing down. Pushing for safer alternatives, better education, and research partnerships creates a pathway to a future that doesn’t sacrifice progress for safety. Experience and common sense tell us it’s never only about the molecule—it’s about how we use, manage, and respect the impact of every step along the way.
What are the safety precautions when handling 5-Nitro-1,3-Xylene?
Understanding the Risks
5-Nitro-1,3-Xylene grabs the attention of anyone familiar with chemical safety. Over the years, I have handled a range of aromatic compounds in the lab, and every time I see a nitro group in the name, alarms go off. Nitro compounds demand respect. 5-Nitro-1,3-Xylene isn’t just another chemical you keep on a shelf — it carries health risks and physical hazards that deserve thoughtful precautions. Exposure can irritate skin and eyes, harm the respiratory tract, and, in high enough amounts, impact the blood. Dust or vapors create real problems, and accidental spills need to be treated like fire drills.
Personal Protective Gear Matters
Starting with the basics, I never touch 5-Nitro-1,3-Xylene without a sturdy pair of nitrile gloves. Latex often won’t cut it. Splash-resistant goggles protect eyes from accidental sprays, and a lab coat shields my arms and torso. In situations where powders or vapors might drift, I use an N95 mask or cartridge respirator. Skin and mucous membranes pick up chemicals without much warning, so there’s no excuse for shortcuts. I’ve seen colleagues regret skipping simple gear after a quick spill left them with redness and burning sensations on the skin.
Ventilation Makes a Difference
Good air movement isn’t optional. I stick to handling this chemical inside a well-functioning fume hood, with the sash dropped low. This approach prevents vapors from reaching breathing zones and stops accidental dust from floating around the room. One time a fan failed during a routine weighing session and you immediately notice how fast odors build up. It's uncomfortable, and it’s unsafe. After that experience, I never rely on luck—always check ventilation before starting any work with this compound.
Smart Storage Habits
Safe storage stops accidents before they even start. Keeping 5-Nitro-1,3-Xylene in tightly sealed glass or compatible plastic bottles blocks moisture and limits vapor release. I choose cool, dry, well-labeled cabinets, always away from open flames or sources of heat. Nitro aromatics can react or even ignite if mixed with strong oxidizers or other incompatible materials, so keeping clear separation makes sense. It takes only one incident of mislabeling or overcrowding for containers to topple, and nobody forgets the clean-up that follows. Easy-to-read hazard labels, clear signage, and a neat environment work far better than a memory test when time matters.
Preparedness and Emergency Response
I’ve learned the value of knowing exactly where safety showers, eye wash stations, and spill kits sit in the lab—not just in theory, but with practice. For 5-Nitro-1,3-Xylene, having chemical absorbents and neutralizers within reach speeds up response to spills. Immediate flushing with plenty of water serves as the first step after skin or eye contact. For inhalation exposure, getting to clean air fast can limit symptoms.
Why Routine Training Matters
Newcomers often trust posted protocols, but regular refreshers keep safety top of mind. Running drills, reviewing material safety data sheets, and talking through hypothetical scenarios give teams a real edge. I’ve watched teams perform far better after annual spill simulations than after years of just reading guidelines. Walking through what to do makes everyone more confident and keeps hesitation at bay.
Looking Ahead
Labs and factories use compounds like 5-Nitro-1,3-Xylene for important synthesis work, but no task justifies cutting corners with safety. Each layer of precaution, from gloves to fume hoods, reflects hard experience earned by those who have learned from close calls. Simple routines and good equipment shape an atmosphere where everyone leaves work the same way they arrived: healthy and safe.
What is the chemical structure and formula of 5-Nitro-1,3-Xylene?
Digging Into the Chemistry
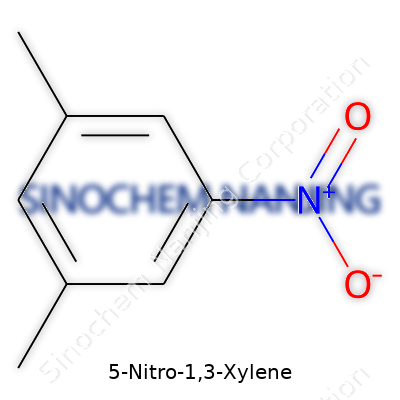
Take a look at 5-Nitro-1,3-Xylene, and you'll see how small changes in a chemical’s structure push its behavior in very different directions. The core of this compound is a benzene ring, a six-carbon ring famous for its stability. For 1,3-xylene, attach two methyl groups (–CH3) at positions 1 and 3 on that ring. Then stick a nitro group (–NO2) onto the fifth carbon. The formula, C8H9NO2, comes straight from counting up all those pieces.
Dive into the structure and you spot a pattern common in aromatic chemistry. The methyl groups give the ring a kick in terms of reactivity, each offering a slight nudge for electrons to wander across the ring. The nitro group pulls electrons in the other direction, acting like an anchor. Chemistry gets interesting in the tug-of-war between these groups. That's not just textbook material. In the real world, the behavior of compounds like this influences things from how we dye clothes to how we treat pesticides.
Applications Touching Real Life
5-Nitro-1,3-xylene doesn’t show up on every grocery store shelf, yet it plays a backstage role in bigger stories. Aromatic nitro compounds carry the legacy of dyes, explosives, and industrial solvents. Each functional group doesn’t just hang around for show—it decides how the compound gets used. The methyl groups make it less prone to combustion than cousin molecules like toluene, a favorite in traditional solvents and fuels. That little nitro group adds potential for further transformations in the lab. Chemists harness it to build drugs, pigments, and even ingredients in electronics.
Chemicals like 5-Nitro-1,3-xylene highlight the subtlety in engineering molecules. Back in the classroom, learning about electron-withdrawing and -donating groups felt abstract. Suddenly, in the lab or factory, those details start paying off if you’re searching for specific reactivity or stability. For example, the nitro group draws the eye of anyone looking to trade up for an amino group, a classic move in pharmaceutical chemistry. Even small changes—a nitro on carbon 5 instead of carbon 4—mean reactions run differently. You don't have to see the chemistry firsthand to appreciate just how much hangs on these little tweaks.
What Safety and Environment Demand
Handling nitroaromatics like this compound reminds us that chemistry never floats free of impact. Nitro groups signal red flags for toxicity and persistence. We’ve seen how production waste from similar molecules contaminated water for decades. Some countries set tight rules around aromatic nitro compounds in the workplace. Safer substitutes are always a hot topic, and green chemistry keeps nudging researchers toward less toxic or more biodegradable alternatives. Nobody wants aromatic nitro residues turning up in groundwater—or food.
On the industrial side, smart engineering lets companies capture, recycle, or neutralize most waste. Not every factory has the funding or oversight. International collaboration and regulations must push for cleaner practices. Universities keep pushing students to weigh reactivity against safety; older workers know the cost of shortcuts. Responsible use and disposal stand as the baseline for trust, not just for regulatory paperwork but for the peace of neighborhoods near chemical plants.
Knowledge Rooted in Purpose
Understanding the chemical structure of 5-Nitro-1,3-xylene doesn’t stop at memorizing formulas for a test. Each functional group tells a story that reaches from theory to daily life. As labs and factories keep innovating, reflecting on risks and benefits reminds us what matters most—using our tools and knowledge not just to generate value, but to protect the world we're part of.
How should 5-Nitro-1,3-Xylene be stored?
Why Proper Storage Matters
5-Nitro-1,3-Xylene isn’t just another laboratory chemical. Inhaling its dust or fumes can cause anything from headaches to more serious lung irritation. Handling mistakes can lead to spills or leaks, raising safety risks for everyone in the area. I’ve worked in research labs where a lack of clear labeling and poor storage has led to close calls with harmful substances. Simple steps could have avoided those problems.
Choosing the Right Environment
Chemicals like this one demand a dry, cool, and well-ventilated space. Heat can nudge volatile organics toward unwanted reactions, upping the odds of a spill or even fire. Humidity opens doors to clumping or decomposition. Many labs rely on dedicated chemical storage cabinets, designed to prevent both moisture buildup and accidental ignition. General-use shelves or open storage areas often lead to trouble: substances can leak, labels can fade, and the chance for mix-ups rises fast.
Avoiding Incompatible Substances
Storing 5-Nitro-1,3-Xylene next to oxidizers, acids, or strong bases carries real danger. Unplanned reactions could release heat or toxic gases. In practice, most well-run labs separate chemicals into clear categories, sometimes by color-coded cabinets or locked sections. I’ve seen accidents traced directly to someone shelving incompatible compounds together. Those events can upend an entire workday, contaminating equipment and risking everyone’s health. Keeping strict separation avoids those headaches.
Labeling Counts
Clear, legible labels make the difference between safe handling and mistakes. Details like date received, hazard warnings, and chemical name allow anyone to recognize what’s in the container—even years later. One overlooked step can mean someone else uses the chemical wrong, or worse, confuses it with something less dangerous. My own team started running short labeling audits after nearly mixing up similar-looking powders. Small habits like this build a safer lab.
Container Choice and Maintenance
Never transfer 5-Nitro-1,3-Xylene into cracked or soft-plastic containers. Tough, chemical-resistant glass or HDPE containers prevent leaks and accidental absorption of vapors. Screw-cap lids with intact gaskets keep air and moisture out. Once, I uncovered an old batch of solvents in deteriorating containers. The sharp scent and deformed plastic flagged a problem long before testing equipment joined the scene. Tossing old, damaged containers prevents unpleasant surprises.
Fire Safety and Ventilation
Fire hazards come with the territory. Storing this chemical near heat sources, sunlight, or electrical panels spells trouble. Facilities should have working smoke detectors, accessible fire extinguishers, and exhaust fans. Labs that lack proper ventilation risk buildup of fumes, harming both air quality and worker health. Years of experience taught me to double-check that air vents remain unblocked and fire safety gear hasn’t expired. Simple routines shield people from bigger risks.
Professional Oversight and Training
Routine inspections keep bad habits from creeping in. Trained staff check storage spaces for leaks, corrosion, or clutter. Spotting problems early puts a stop to unsafe trends. Teaching staff and students about the dangers of each chemical—including 5-Nitro-1,3-Xylene—builds a workforce that looks out for itself. My own background in chemical safety reminds me no shortcut pays off: investing in proper storage and education pays dividends every day.
What are the hazards and toxicity levels associated with 5-Nitro-1,3-Xylene?
What Is 5-Nitro-1,3-Xylene?
5-Nitro-1,3-xylene stands out for its bright yellow color and use in making dyes, pigments, and some specialty chemicals. If you work in a lab or industrial facility, that familiar whiff says you’re around aromatic nitro compounds—a category with a rough reputation.
Why 5-Nitro-1,3-Xylene Raises Red Flags
Workers handling this compound often have concerns that go beyond spilled powder or messy glassware. Even a few drops on your hands or a forgotten smear on a workbench can haunt you later in the day. Studies show nitro-aromatic compounds like this one can seep through the skin, not just rest on it. The real trouble starts with repeated exposure, where headaches, nausea, and blue-tinged skin (a sign of methemoglobinemia) pop up, and those symptoms point straight to blood and nervous system involvement.
The molecular structure of 5-nitro-1,3-xylene lets it interact with metabolic enzymes in the liver, creating byproducts that can bind to proteins and DNA. For people with years around these chemicals, scientists have spotted increased risks of certain cancers and chronic organ damage. That’s not scaremongering—multiple occupational health studies (including NIOSH toxicology profiles) draw a clear connection between exposure and lasting harm.
Inhalation, Ingestion, and Absorption
Taking a breath around spilled 5-nitro-1,3-xylene isn’t the same as sniffing paint thinner, but the long-term outcome might hurt you more. Dust or vapor isn’t common in everyday situations, but it’s a real threat during mixing or cleanup. The inhaled stuff slowly sticks around in fatty tissues and can interfere with oxygen transport in blood. Ingestion, though rare, can trigger gut corrosion, vomiting, and an ugly kind of systemic toxicity you won’t soon forget.
The absorption risk might be the quietest issue in the room. I remember a mentor warning new staff that gloves and lab coats mean little if sweat mixes with dust on your arms. Cleanup protocols matter a lot more in these scenarios, and you’ll notice experienced technicians never take shortcuts.
Environmental Risks and Emergency Handling
5-Nitro-1,3-xylene doesn’t break down quickly in soil or water. Environmental scientists worry about its persistence—every spill has a chance to travel downstream and find its way up the food chain. Aquatic organisms exposed to trace levels often lose their ability to grow or move; some studies show reproductive failure in common freshwater fish. Wildlife biologists track these nitro-aromatic builds up in animal tissue, raising the threat of chronic exposure not just for those who work in factories, but for anyone living nearby.
Responding to spills or leaks calls for more than urgency—you need containment, forced air ventilation, and neutralizing agents. Anyone tasked with cleanup gets full-face respirators, heavy-duty gloves, and disposable coveralls. Clinics in chemical plants stock methemoglobinemia antidotes and run regular blood checks for technicians assigned to nitro compound projects.
Pushing for Safer Practices
Regulators like OSHA and EPA set exposure limits and require regular monitoring for good reason. Accidents have led to permanent disability in some workers, and legal settlements reflect the true cost of ignoring safety rules. From my experience, the safest labs are where workers respect the threat enough to double-check containment and storage tools, not just sign the OSHA training checklist.
Switching to less toxic alternatives and automating processes can slash the risk dramatically. Ventilation and real PPE—not just a paper mask—make accidents less likely to stick around in your memory or in your bloodstream. If your workplace stores 5-nitro-1,3-xylene, treat it as you would an open flame: respect it, understand its risks, and never let down your guard.
| Names | |
| Preferred IUPAC name | 1,3-dimethyl-5-nitrobenzene |
| Other names |
1,3-Dimethyl-5-nitrobenzene NSC 41032 3,5-Dimethylnitrobenzene |
| Pronunciation | /ˈfaɪ ˈnaɪtroʊ waɪˈliːn/ |
| Identifiers | |
| CAS Number | 99-08-1 |
| Beilstein Reference | 1368736 |
| ChEBI | CHEBI:131437 |
| ChEMBL | CHEMBL153402 |
| ChemSpider | 21404991 |
| DrugBank | DB07636 |
| ECHA InfoCard | 100.011.031 |
| EC Number | 210-849-4 |
| Gmelin Reference | 137430 |
| KEGG | C14352 |
| MeSH | D000073307 |
| PubChem CID | 88676 |
| RTECS number | ZE8225000 |
| UNII | 60K2J203JU |
| UN number | 1669 |
| Properties | |
| Chemical formula | C8H9NO2 |
| Molar mass | 150.16 g/mol |
| Appearance | yellow crystalline powder |
| Odor | aromatic |
| Density | 1.17 g/cm³ |
| Solubility in water | Insoluble |
| log P | 1.98 |
| Vapor pressure | 0.0075 mmHg (25 °C) |
| Acidity (pKa) | 7.51 |
| Basicity (pKb) | pKb = 10.6 |
| Magnetic susceptibility (χ) | -68.6·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.583 |
| Viscosity | Viscous liquid |
| Dipole moment | 3.73 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 267.6 J mol⁻¹ K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -5.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3813.4 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P210, P261, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-3-1-爆 |
| Flash point | 79°C |
| Autoignition temperature | 540°C |
| Explosive limits | 2–7.5% |
| Lethal dose or concentration | Lethal Dose (LD50) oral (rat): 1200 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1590 mg/kg (Oral, Rat) |
| NIOSH | SN 2100000 |
| PEL (Permissible) | PEL (Permissible): Not established |
| REL (Recommended) | 0.1 ppm (0.58 mg/m3) |
| Related compounds | |
| Related compounds |
1,3-Xylene 2-Nitro-1,3-xylene 4-Nitro-1,3-xylene 5-Nitro-m-xylene Nitroxylenes |

