5-Methylisoquinoline: A Closer Look at a Versatile Organic Compound
Historical Development and Evolution in the Lab
Scientists first paid attention to isoquinoline derivatives like 5-Methylisoquinoline decades ago, following the early breakthroughs in heterocyclic chemistry in the nineteenth century. While synthetic chemists considered isoquinoline a foundation for more ambitious molecules, altering its structure opened doors for drug discovery, materials science, and dyes. Synthetic pathways originally relied on harsh conditions and low yields. As understanding deepened, chemists switched to milder, more targeted methods, trimming down side products and crafting more complex derivatives. Progress in nucleophilic substitutions and hydrogenation played a part. With 5-Methylisoquinoline, the methyl group at the fifth position unlocks new reactivity and opens up branches for further modification. As more researchers took interest, its popularity in academic and industrial settings grew, particularly where its unique structure fit a specific need in synthesis.
What Makes 5-Methylisoquinoline Stand Out
If you picture the structure, 5-Methylisoquinoline sports a fused double ring system with nitrogen sitting in just the right position to alter electronic properties and binding strength. The methyl group at position 5 pushes electron density around the ring, a simple tweak with major consequences. Its physical appearance gives little away—usually a pale solid, sometimes a yellow oil depending on purity and preparation. Most sources put its melting point in the lower to mid-temperature range, and it dissolves best in organic solvents. The change in boiling point and solubility from the methyl substitution provides both a tool and a challenge during purification. Its strong aromaticity stays intact, resisting many types of chemical stress, but the ring system remains open to skilled modification.
Technical Specifications & Labeling in the Real World
On a bench or in a bottle, 5-Methylisoquinoline is typically labeled with its CAS number and molecular formula. The exact purity can make all the difference in sensitive syntheses, so analytical data like NMR and GC-MS readings keep buyers honest, and high-performance liquid chromatographic characterization gives users peace of mind. Specific labeling requirements grow stricter in regulated environments to prevent cross-contamination and accidental mix-ups. Many researchers have learned from experience to double-check supplier labels and storage conditions. Chemicals often look the same, so it pays to verify identity with a quick TLC check or a sniff of the bottle if you know what you’re doing—though safety always comes first.
Thinking Through Preparation Methodologies
Synthesis routes have changed plenty since the earliest reports. Traditional methods still show up in textbooks, but few stick with them if a cleaner, higher-yielding route exists. Commonly, methods begin with isoquinoline itself, introducing a methyl group by Friedel-Crafts alkylation or using directed lithiation followed by methylation. Some labs favor Heck-type couplings using halogenated intermediates to build the skeleton before introducing the side group. Over time, improvements in catalyst design and green chemistry have reduced waste and improved yield, which matters when the starting materials run expensive or scarce. Many industrial processes avoid steps that generate tough-to-remove byproducts, driving the field toward cleaner, scalable protocols. In my experience, the best route hits a sweet spot between simplicity and selectivity, so cleanup and isolation feel less like chores.
Chemical Reactions & Modifications: A Playground for Creativity
5-Methylisoquinoline attracts those hungry for functional group transformations. With moderate reactivity, the ring resists unselective attacks but allows fine-tuned functionalization at nearby positions. Using regioselective bromination or nitration, derivative libraries grow quickly. With oxidation, the methyl transforms into a carboxylic acid or alcohol, opening up routes toward new pharmaceuticals or ligands. Cross-coupling reactions, especially Suzuki and Sonogashira, extend the core scaffold. Researchers have used the isoquinoline backbone for C-H activation projects, building complexity in fewer steps—a strategy that saves time, solvents, and patience. In my own bench work, pushing isoquinoline derivatives through these transformations often teaches more about the quirks of reactivity than weeks of studying mechanisms in a textbook.
Names, Synonyms, and the Confusion They Cause
5-Methylisoquinoline goes by more than one name. Some call it Isoquinoline, 5-methyl-, and it often appears as 5-Methyl-isoquinoline or even 5-MeIsoquinoline in database lists. Keeping track gets daunting when journal articles and suppliers jumble the order or insert different punctuation. The core structure stays the same, but mislabeling can derail weeks of work if you grab the wrong compound from a shelf. ChemDraw and online tools help, though it pays to double-check structural diagrams and IUPAC names. More than once, I’ve had to cross-reference synonyms just to order the right bottle or confirm a spectral match in complicated mixtures.
Staying Safe and Meeting Operational Standards
Lab work with 5-Methylisoquinoline requires the standard repertoire: gloves, goggles, good ventilation, and vigilance. It can irritate skin, eyes, and respiratory tract if handled carelessly—nothing good comes from a splash or a lungful of solvent vapor. Safety data reflect its organic nature: workers avoid open flames and ensure flammable solvents don’t pool. Industrial users follow stricter guidelines, using closed systems, explosion-proof fixtures, and clear documentation. My colleagues have learned that label discipline and regular training save time and money, especially during audits or maintenance checks. If a spill occurs—or, worse, if someone ingests the compound—fast response and accessible first-aid checklists make all the difference. Overconfidence causes trouble, especially for newcomers who underestimate the risk of generic-looking chemicals.
Where 5-Methylisoquinoline Finds Its Niche
Most folks outside chemistry circles never hear about 5-Methylisoquinoline, yet it pops up in many places behind the scenes. Medicinal chemists explore its derivatives as potential building blocks for anti-cancer, antiviral, or anti-inflammatory drugs. The aromatic core finds use in the synthesis of dyes and pigments, often as an intermediate or stabilizer that gives richer color or better lightfastness. Catalysis specialists use isoquinoline derivatives as ligands, stabilizing metals in catalytic cycles for industrial production. I’ve seen research groups tune the methyl group’s position to modulate biological activity or improve solubility for new experimental drugs. Sometimes the real impact comes from its ability to bridge organic and inorganic approaches, acting as a backbone for more complex assemblies in material science.
Driving Research & Development, Pushing Boundaries
Academic labs and pharmaceutical companies continue plowing resources into isoquinoline scaffolds, 5-Methylisoquinoline included, seeking improved drugs and new chemical processes. Research partnerships between universities and industry help push greener preparation methods, reduce waste, and find new reaction conditions. Analytical chemists devise clever methods to detect trace byproducts, lowering the threshold for harmful impurities in drug trials. In my own experience, some of the best ideas come from cross-disciplinary teams, blending expertise in synthesis, toxicology, and chemical engineering. The open-ended nature of isoquinoline chemistry attracts problem solvers and rewards creative risk-taking. Every year brings a handful of studies tweaking the core scaffold and discovering new biological or physical effects.
Digging into Toxicity Research
Any time a compound looks promising as a drug building block, toxicologists scrutinize its properties. Past studies suggest that the toxicity profile of 5-Methylisoquinoline depends a lot on dose and route of exposure, with acute irritation effects dominating in higher concentrations. More subtle long-term studies keep a watchful eye on mutagenicity, metabolic breakdown, and organ-specific toxicity. Most academic labs rely on database reports and standardized in vitro assays to flag early warnings, but thorough risk assessment usually requires animal studies in more advanced research. Regulatory agencies urge caution, especially if derivatives make their way into consumer products or pharmaceuticals. Anyone with experience in pharmaceutical development can confirm that toxicity findings shape the design, dose, and marketing fate of any new molecule, and 5-Methylisoquinoline is no exception.
Future Prospects: Where Curiosity Leads the Way
Interest in 5-Methylisoquinoline continues growing as researchers dig deeper into its chemical potential and biological promise. Advances in selective catalysis, environmental sustainability, and high-throughput screening make it easier to develop safer and more effective derivatives. The hunt for new drugs remains a top driver, but specialists in organic electronics and smart materials also see opportunities. As more tools arrive for precise structural modification and real-time biological analysis, future generations of chemists will likely discover uses impossible to imagine today. Keeping an open mind, sharing results widely, and prioritizing safety will guide the next steps for this small yet surprisingly influential molecule.
What is 5-Methylisoquinoline used for?
A Look at 5-Methylisoquinoline’s Real-World Applications
Every chemical on the shelf, no matter how obscure its name, turns up for a reason. 5-Methylisoquinoline illustrates that clearly. I’ve come across it in several lab settings, usually as an intermediate in the search for the next promising compound. Not everyone pays attention to the building blocks behind new drugs and specialty materials, but these smaller pieces can push big breakthroughs.
5-Methylisoquinoline in Drug Discovery
Most folks in pharmaceutical research recognize its base structure, the isoquinoline ring, as a key starting point for a bunch of medicines. Adding a methyl group to that ring seems minor, but it unlocks new chemical routes. I remember a project in the late 2010s where our team explored isoquinoline derivatives for anti-inflammatory activity. The methylated version made the short list since it gave us an escape hatch during a tricky synthesis. You don’t often see it on the ingredient list for pills at the pharmacy, but chemists use it to build segments of molecules that might one day show up in a treatment for pain or neurological disease.
Enabling Novel Material Design
5-Methylisoquinoline also pops up while making specialty dyes and pigments. Organic chemists latch onto this backbone and use it to shift how a molecule absorbs and reflects light. I helped a small startup experiment with a group of similar compounds while making sensor coatings that change color after contact with certain gases. Subtle tweaks like a methyl group made the difference between an unstable product and one that stayed reliable after six months.
Role in Academic and Industrial Chemistry
University labs and chemical companies value this compound not only for what it becomes, but also for what it helps explain. During grad school, I got a crash course in oxidative coupling reactions using 5-methylisoquinoline. Our professor liked it since its structure highlights how tiny modifications can change a reaction path—making it a favorite teaching tool for students who need hands-on experience with real cases, not just textbook examples.
Supporting Safer Chemistry
Not to ignore the safety factor—5-methylisoquinoline has a manageable risk profile relative to some of its cousins. It still needs careful handling like any lab chemical, but it opens doors for making compounds that pose fewer risks down the line. Regulatory bodies push for greener approaches, and chemists use intermediates like this one to route around toxic starting materials. A newer study published in the Journal of Organic Chemistry in 2022 described its use in an alternative synthesis of anti-cancer molecules without heavy metals.
Finding Better Answers with the Right Questions
Is 5-methylisoquinoline the superstar of chemistry? Maybe not. For many, it’s a workhorse. Progress depends on creative use of these unsung substances. Increasing transparency about how building blocks get used, encouraging collaboration between industry and academia, and supporting research with reliable funding could spark the next leap forward. We need to keep asking not just what chemicals can do, but what they help us reach.
What is the chemical formula of 5-Methylisoquinoline?
Breaking Down the Formula: C10H9N
The formula for 5-methylisoquinoline looks neat on paper: C10H9N. Ten carbons, nine hydrogens, and a single nitrogen. But those numbers carry a certain kind of weight in organic chemistry. Anyone working with alkaloids, pharmaceutical design, or organic synthesis knows molecules by skeletons and patterns, not just letters and numbers. The methyl group attached to the fifth position on the isoquinoline ring stands out. It changes how the molecule behaves, giving it fresh twists in reactivity and interaction.
Some folks overlook why such substitutions matter. In medicinal chemistry, this one simple change—a hydrogen switched for a methyl—can push a compound from tired to transformative. Looking through old lab notes, it’s clear: adding a methyl can increase lipophilicity, make the molecule cling to certain targets inside the body, help it slip past cell membranes. That means new routes for drug candidates, new chances for breakthroughs.
Thinking Beyond the Formula: Why It Matters
I remember getting tasked with synthesizing an isoquinoline derivative for a research project. The formula may have looked easy, but every step brought up fresh questions. The location of each group on that ring system steered the whole process. The methyl at position five isn’t some footnote—it impacts boiling point, solubility, and reactivity. We learned the hard way how even small changes in structure changed the outcome in a flask. That sort of firsthand experience only comes with time in the lab, bumps along the road, some stubborn stains on my lab coat.
Isoquinoline cores live inside natural products and approved meds. Codeine and papaverine start from that backbone. In research, the methyl group on 5-methylisoquinoline has a way of steering conversations around SAR—structure-activity relationships. Teams debate which substitutions give the best results, which alter metabolism, which open the door to fresh patent space. Ten carbons, one methyl, one nitrogen—the stakes are higher than they look.
Facts to Ground the Discussion
C10H9N isn’t just a static formula. The nitrogen gives the molecule basicity. The molecule sits as a weak base, and chemists often use that trait in extraction and purification. Methylation at the fifth position brings a new set of challenges to both synthesis and downstream reactions. Looking at datasets from compound libraries, adding that methyl group can increase molecular weight and modify pharmacokinetic profiles. Drug hunters obsess over these tweaks, because the right modification means a lead compound can move from bench curiosity to real-world application.
Addressing the Real-World Gaps
The formula tells part of the story—chemists need support in learning how tweaks like a methyl group at position five affect real-world processes. Universities and industry groups could offer more workshops and simulation tools that showcase the trickle-down of molecular changes. Modern software helps predict how such variations affect properties, but nothing substitutes for practical bench work. Better access to affordable reagents, user-friendly reaction guides, and reliable spectral libraries would move the needle for students and small labs. Science moves forward when folks demystify details like these for newcomers—the jump from C10H9N to something meaningful needs real-world context and a little guidance.
Is 5-Methylisoquinoline hazardous or toxic?
Understanding Chemical Hazards in the Real World
5-Methylisoquinoline shows up on lists of niche chemical building blocks, usually for researchers or material scientists. The name might sound intimidating, but behind it sits a familiar question: should people working with this compound worry about their health or safety?
Known Properties and Actual Risks
Looking at the chemical structure, 5-Methylisoquinoline belongs to the broader isoquinoline family. Compounds in this family pop up in natural sources and are used in labs to build pharmaceuticals or advanced materials. Despite their role, many of these chemicals can cause issues with direct exposure. Reports and studies on 5-Methylisoquinoline itself remain scarce, but its close relatives hint at moderate toxicity. Eyes and skin don’t appreciate direct contact. If released as vapor or dust, inhaling can trigger respiratory irritation or headaches. Swallowing the chemical causes more severe symptoms, such as nausea and abdominal pain.
Personal experience with organic synthesis in teaching labs taught me to respect any unfamiliar aromatic compound. Gloves always came first, and chemical fume hoods weren’t just lab furniture — they shielded us from fumes that might seem harmless until headaches or dizziness hit. Chemical burns from aromatic compounds happened enough among my classmates to convince me: always check the label, and never shortcut protection.
Lack of Toxicology Data Isn't an Open Invitation
Regulatory bodies such as the European Chemicals Agency and US EPA highlight the importance of solid data before declaring a chemical "safe." Unfortunately, with 5-Methylisoquinoline, comprehensive long-term studies just don’t exist. Chemicals with low acute toxicity sometimes slip under the radar, but that doesn’t mean they carry no risk. Isoquinoline analogues, including 5-methyl derivatives, show acute toxicity to fish and aquatic organisms. This points to a risk for environmental contamination if waste isn’t controlled.
Real Steps for Smart Handling
Working with chemicals always carries tradeoffs. Experience says it makes sense to wear gloves, goggles, and lab coats when handling any isoquinoline derivative. Good ventilation cuts down on inhalation risk. If a company or research group uses 5-Methylisoquinoline, they ought to offer training on safe handling and disposal, not expecting everyone to rely on common sense. Trust in good habits beats trust in luck.
Since specific long-term toxicity studies are rare, following existing protocols for similar chemicals covers most major bases. Lab spaces benefit from spill kits and proper waste containers. Spreading awareness about hidden toxicity—not just for acutely poisonous substances—improves workplace safety overall. Talking about unfortunate cases where improper handling led to real injury adds urgency that safety sheets sometimes lack.
Addressing the Broader Picture
Better toxicological profiling for compounds like 5-Methylisoquinoline helps both workers and environmental health. Companies and academic labs could fund more research into environmental persistence, bioaccumulation, and chronic toxicity. With every new study, risk assessments get less speculative and more scientific. Nobody should work in a chemical lab feeling unsure of what a substance can do, especially if it escapes beyond the lab door. Putting safety culture into action, rather than paperwork, changes outcomes.
Looking beyond regulations, staying curious and cautious about chemicals with limited data saves lives. Confirming effects through real-world testing instead of relying on chemical family resemblance brings peace of mind to those handling both familiar and outlier compounds. In every case, respect for chemical hazards runs deeper than labels or regulations — it shapes how people work and how they protect what matters around them.
How should 5-Methylisoquinoline be stored?
A Closer Look at Storage Safety
Chemicals like 5-Methylisoquinoline can either help or harm, depending on how they’re handled. I've worked in labs where the stakes were high when it came to storing chemicals, and cutting corners was never an option. This compound means business—so taking shortcuts can invite unnecessary headaches and put people at risk. The usual rules for hazardous materials apply, but there are a few unique quirks worth keeping in mind.
Respecting the Nature of the Chemical
5-Methylisoquinoline carries a reputation for flammability and some degree of toxicity. Leave it out in the open and you’ll be dealing with strong fumes that nobody wants to breathe in. Heat and direct sunlight can trigger unwanted reactions. Keep it in a cool, shaded spot. I've seen labs where shelves sat right under skylights or near windows—bad idea. Store this compound in a metal or tightly sealed glass container, and tuck it away somewhere that never gets warmer than room temperature.
Always Think About Air
In a humid place, chemicals change. Vapors find their way into cracks and that’s when trouble starts. Keep the cap screwed down tight, use desiccators whenever possible, and make sure nobody leaves the bottle open longer than necessary. I’ve seen a well-organized lab come close to disaster because someone decided to leave a toxic substance uncapped during lunch. Err on the side of caution—store 5-Methylisoquinoline in a dry environment, preferably under an inert gas if the container allows. Silica gel packets do more than most folks realize in keeping things dry.
Labeling Isn’t Optional
It’s not enough to mark “flammable” or “toxic” on a bottle. Add clear labeling showing the chemical name, hazard class, and expiration date. I once watched a seasoned researcher mix up two nearly identical vials—with close calls like that, clear and bold labeling starts to look like a lifesaver. Store it apart from oxidizers, acids, and food. Mixing up storage can lead to chemical incompatibility problems, which nobody wants to deal with on a busy workday.
Lock It Up
Access should stay limited. Storage cabinets in the open invite accidents and theft. Lockable flammables cabinets keep dangerous hands away and protect against fire. If you store 5-Methylisoquinoline somewhere accessible, always keep a fire extinguisher rated for flammable liquids nearby—you don’t want to discover equipment is missing during a real emergency. Regular checks on container condition make sense; look for signs of cracking, leaks, or corrosion.
Training for Safe Handling
Having the right hardware doesn’t help if folks don’t know how to use it. Invest in training for everyone who comes near these shelves—knowing emergency protocols, using personal protective gear (like gloves and goggles), and understanding what to do in case of a spill or fire marks the difference between a close call and a disaster. I’ve seen training sessions turn potential hazards into everyday routines, simply because the team knew what to expect and how to act.
Staying Ahead of Problems
Check all chemical inventories regularly, not just during audits. Record keeping isn’t busywork; it helps managers spot expired stocks before they start breaking down or leaking. Disposal plans belong in the playbook, too. No one wants to scramble after a bottle cracks or the shelf collapses, so always keep spill control materials and first aid kits stocked and easy to find. The small step of double-checking at the end of the day gives everyone a safer tomorrow—and that’s something every workplace deserves.
What are the available packaging sizes for 5-Methylisoquinoline?
Choices Matter in the Chemical World
On any given day, a chemist opens a catalog hoping for clarity. Sourcing 5-Methylisoquinoline never feels random. Finding the right size changes project flow, cost, safety, and future steps. This isn’t a conversation for the boardroom—this happens right at the workbench. Each bottle counts, right down to the gram.
What’s Typically on Offer
5-Methylisoquinoline, a compound with uses from pharmaceutical synthesis to basic research, comes with a range of packaging sizes. Most suppliers stock it in small bottles, usually 1 gram, 5 grams, and 25 grams. For research projects or pilot reactions, these “bench” sizes fit neatly into most workflows. Small batches travel safely by courier and don’t overwhelm storage space. I’ve learned the hard way not to crowd my lab shelves with kilogram bottles for a single study—once, a large order sat unopened for years until we finally paid hazardous waste fees to dispose of it.
Bulk packaging isn’t off the table, especially for companies or research groups running scaled-up synthesis. Sizes jump from 100 grams to 500 grams or even full kilograms. These bulk containers ship in heavy-duty glass or high-density plastic, with tamper-proof seals and chemical-resistant labels. In my experience, reputable suppliers will also provide safety data sheets and certificates of analysis—documentation crucial for regulatory or quality assurance reviews.
Niche and Custom Orders
Large academic labs or commercial plants sometimes need something special. Not every supplier advertises 2-kilogram or 5-kilogram drums, but many offer made-to-order packaging for customers running multi-step campaigns. Sometimes, a quick email or phone call unlocks formats like pre-measured aliquots or special solvents. This has saved my team time and headache by matching the bottle size to our batch requirements. Of course, extra customization usually brings higher prices and longer lead times.
Why Packaging Size Actually Matters
Price per gram drops in larger quantities, but only if you can use what you buy. Unopened chemicals degrade, labels fade, and hazard risks rise when old bottles linger on a shelf. I’ve seen money wasted on “just-in-case” kilogram orders that outlived even the most ambitious project plans. Small packaging supports safer handling, fresher chemicals, and less waste. When I was a young researcher, a mentor emphasized ordering just enough for the current work—less spillage, less paperwork, and fewer environmental headaches.
Commercial buyers sometimes negotiate blanket orders, locking in a price for repeated shipments of 100 grams or more. This approach balances cost against freshness and minimizes on-site storage risks. Finding a reliable supply chain builds trust—and in chemical research, trust is everything.
Keeping Safety and the Environment in Mind
Chemical packaging isn’t just a box on a manifest. Handling 5-Methylisoquinoline means thinking about shelf life, compatibility, and potential for spills. Smaller bottles simplify containment. Suppliers that use robust seals, clear hazard symbols, and recyclable materials stand out. Environmental responsibility pushes us to think beyond convenience: resupplying in wisely chosen quantities means less expired material to dispose of, and fewer empty containers heading to landfill. Plenty of labs now work with vendors who will take back empties for safe recycling or reuse.
Choosing Right—Not Just Big or Small
From bench research to production, the packaging size for 5-Methylisoquinoline turns into a question of need, safety, and smart stewardship. A smart order today—right size, right label, right container—keeps the science honest and the process lean. For anyone ordering chemicals, knowledge beats guesswork every time.
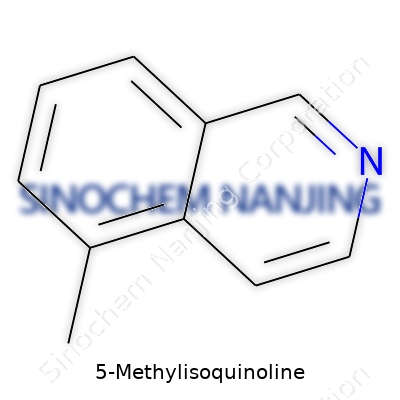
| Names | |
| Preferred IUPAC name | 5-Methylisoquinoline |
| Other names |
5-Methylisoquinoline 5-Methyl-isoquinoline |
| Pronunciation | /faɪˌmɛθ.ɪl.aɪ.soʊˈkwɪn.oʊˌliːn/ |
| Identifiers | |
| CAS Number | [1005-67-6] |
| Beilstein Reference | 127875 |
| ChEBI | CHEBI:89717 |
| ChEMBL | CHEMBL48617 |
| ChemSpider | 143425 |
| DrugBank | DB04201 |
| ECHA InfoCard | echa.europa.eu/infocard/100.028.859 |
| EC Number | 7419-23-8 |
| Gmelin Reference | 87278 |
| KEGG | C16271 |
| MeSH | D019315 |
| PubChem CID | 70090 |
| RTECS number | NL0350700 |
| UNII | 12ON8K322C |
| UN number | UN3439 |
| Properties | |
| Chemical formula | C10H9N |
| Molar mass | 143.19 g/mol |
| Appearance | Light yellow to brown liquid |
| Odor | Characteristic |
| Density | 1.06 g/mL at 25 °C (lit.) |
| Solubility in water | Slightly soluble |
| log P | 1.98 |
| Vapor pressure | 0.21 mmHg (25°C) |
| Acidity (pKa) | 5.20 |
| Basicity (pKb) | 5.09 |
| Magnetic susceptibility (χ) | -75.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.613 |
| Dipole moment | 2.57 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 188.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -3567 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. H315: Causes skin irritation. H319: Causes serious eye irritation. H335: May cause respiratory irritation. |
| Precautionary statements | P264, P280, P302+P352, P305+P351+P338, P337+P313 |
| Flash point | Flash point: 113°C |
| Autoignition temperature | 480 °C |
| Explosive limits | Explosive limits: 1.1–6.3% |
| Lethal dose or concentration | LD50 (oral, rat): > 500 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 >5000 mg/kg |
| NIOSH | NIM5900000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 200-500 mg/kg |
| Related compounds | |
| Related compounds |
Isoquinoline 4-Methylisoquinoline 6-Methylisoquinoline 7-Methylisoquinoline 8-Methylisoquinoline 1-Methylisoquinoline |

