5-Methyl-2-Hexanone: Chemical Realities and Future Paths
Historical Development
From the earliest years of industrial chemistry, researchers explored ketones for solvents, flavorings, and intermediates. 5-Methyl-2-Hexanone started gaining traction decades ago in facilities working with complex organic syntheses. The compound doesn’t have the big-name recognition of acetone or methyl ethyl ketone, yet its story ties to the broader search for performance chemicals suited to tough, multi-stage environments. Its production paralleled advances in selective oxidation and alkylation techniques, at a time when industries demanded molecules that could stand up to increasingly more specialized applications. By the 1970s, as the chemical and coatings industries ramped up, new regulatory frameworks and research pushed companies to review lesser-known ketones. 5-Methyl-2-Hexanone surfaced as a contender in this evolving landscape, offering unique potential where volatility, solvency, and reactivity intersected.
Product Overview
Despite not sitting at the top of solvent sales charts, 5-Methyl-2-Hexanone found its place by solving practical problems. Manufacturers and lab chemists look for compounds that can dissolve stubborn formulations, either in coatings, cleaning agents, or as process intermediates. This ketone gives a balance between moderate boiling point and good solvency, lending itself to formulations where evaporation rate matters. It steps into roles where too-fast evaporation creates defects or where slow-drying leads to operational bottlenecks. In my own work, the frustration with poorly dissolving intermediates disappeared thanks to switching to this ketone, which succeeded where others fell short. Its moderate molecular weight also made it attractive to formulators looking for alternatives during supply disruptions of traditional ketones.
Physical and Chemical Properties
This compound comes with a clear, colorless appearance, offering mild odor compared to many traditional solvents. Its molecular weight sits higher than that of classic methyl ethyl ketone but lower than heavier, sluggish solvents. The boiling point hovers around 143–145°C, which positions it well for applications needing slower evaporation. Its physical behavior aligns with expectations for a branched-ketone; dense enough to sink in water, yet volatile enough for industrial use. The density sits close to 0.81–0.83 g/cm³, and it shows fair miscibility with many organic liquids. With proper storage away from heat and open flame, this ketone remains stable, though it’s worth remembering that exposure to strong oxidizers or acids sets the stage for dangerous scenarios.
Technical Specifications and Labeling
Chemical suppliers tend to grade 5-Methyl-2-Hexanone by assay, water content, and presence of related impurities. Reliable stock maintains a minimum purity above 98%. Labels in laboratory or warehouse environments include hazard symbols for flammable materials and warnings for skin or eye contact. Many facilities evaluate regulatory compliance with standards like REACH or TSCA, ensuring both import and downstream use lines up with safety expectations. Accurate labeling and tracking became more rigorous after mid-2000s changes in hazard communication laws. From personal experience, when safety data sheets improved, both accidental exposure rates and legal headaches dropped—nobody wants a regulatory audit uncovering shoddy paperwork.
Preparation Method
Most commercial routes use condensation and hydrogenation. One synthesis starts by using 2-hexanone as a backbone, followed by methylation in presence of a suitable base and methyl donor. Another technique builds from pentanone derivatives through aldol condensation, then selectively hydrogenates resulting intermediates. Older literature describes Friedel-Crafts methods with aluminum chloride as catalyst, though modern plants tend to avoid such setups due to environmental and handling concerns. High-volume production demands robust recycling of solvents and tighter control of byproducts to minimize both emission and waste, reflecting today’s stricter environmental rules.
Chemical Reactions and Modifications
This ketone shows foundational reactivity typical of its class. The carbonyl group welcomes nucleophilic additions, forming alcohols under reduction with lithium aluminum hydride or sodium borohydride. A Grignard reagent adds at the carbonyl to yield tertiary alcohols, expanding its utility as a synthetic intermediate. Transesterification and acetal formation also fit into its chemical playbook, giving more flexibility for downstream modification. Oxidation generates acids or enolizable products, but excessive conditions risk over-reacting side chains. In the lab, I found that this compound cooperates better with mild bases, while strong acids tend to generate a host of dehydration products best avoided for purity’s sake.
Synonyms and Product Names
Many data sheets refer to this ketone by synonyms such as Methyl Isoamyl Ketone or MIAK. The IUPAC name, 5-Methylhexan-2-one, crops up in formal documents or supply contracts. GHS hazard documentation might use the CAS number 110-12-3 for clarity. Chemists in different regions stick to varying nomenclature out of habit or regulatory preference, sometimes creating confusion. I once saw a delayed order simply because a purchaser insisted on the trade name, while the supplier filed the compound under its systematic name—a good reminder to cross-check nomenclature, especially in international settings.
Safety and Operational Standards
Dealing with 5-Methyl-2-Hexanone inside the lab or plant floor means respecting its flammability, moderate toxicity, and vapor behavior. Ventilation with active fume extraction cuts inhalation risk. Skin absorbs liquid ketones surprisingly well, so good gloves and goggles stand as baseline gear. Regulatory standards follow country-specific rules, from OSHA limits on airborne concentration to European workplace exposure directives. Overexposure produces headaches or dizziness, mimicking many common solvents. Fire risk jumps with temperature or in closed environments, and firefighting teams in industrial zones label ketone stores with clear signage and foam-based extinguishers. Every year brings incidents from workers cutting corners, so strict adherence to protocols pays off directly through fewer injuries and legal disputes.
Application Area
The mainstay use for 5-Methyl-2-Hexanone lands in solvent-based coatings and inks, where its drying profile suits resin dispersion and pigment flow. It helps balance rapid evaporators like acetone, reducing surface defects and improving finish smoothness. Polymer manufacturers pick it for its role as a process intermediate or solvent in adhesives and sealants. Cleaning products find benefit due to its ability to break down greases and oils resistant to water-based washes. In the pharmaceutical sector, limited and carefully regulated use for drug synthesis exists, though concerns about exposure restrict its application. The flavor and fragrance fields approached it for synthetic blends, but regulatory hurdles regarding residues and purity kept its use in check outside industrial circles.
Research and Development
Research teams keep turning up new tricks for 5-Methyl-2-Hexanone, particularly in advanced polymer and specialty chemical sectors. Efforts focus on new catalysts that could boost production yield or reduce waste during synthesis. Environmental chemists test biodegradable derivatives, hoping to combine solvency with rapid breakdown after use. Fine chemical houses run trials with structurally similar ketones to tune reactivity or safety profiles for greener production. Novel analytical techniques, like in-line spectroscopy, now help track purity without endless sampling and delays. These kinds of advances sprang from spilled sample vials and delayed results in my own R&D days—a real motivator for process innovation. Academic groups collaborate with industry partners to probe deeper into structure-activity relationships, aiming to broaden the scope or reduce the hazards of existing processes.
Toxicity Research
Toxicologists handle 5-Methyl-2-Hexanone with measured caution, aware that its toxicity profile lands in the gray zone—no outright bans, but plenty of watchful eyes. Animal studies report central nervous system effects after chronic high-level exposure, with symptoms echoing exposure to other branched-chain ketones. Some studies linked extended contact to mild liver or kidney changes, but evidence did not climb to the level seen with more notorious industrial solvents. Strict inhalation limits set by workplace health agencies reflect this middle-ground risk. Human data stays limited, so researchers call for more studies into both acute and chronic exposure. As an industry-standard approach, chemists and operators monitor air, implement proper personal protection, and keep exposure windows short. These safeguards grew out of lessons learned during past decades of working with solvents that now carry outright bans. Regulatory agencies consider cumulative exposure, so shifting usage to automation or closed systems looks like the surest way to lower risk.
Future Prospects
Looking ahead, the path for 5-Methyl-2-Hexanone winds through a patchwork of regulatory, economic, and sustainability trends. Emerging green chemistry principles put pressure on manufacturers to cut down emissions and waste. Startups and established companies both experiment with bio-based synthesis, eyeing renewable feedstocks to maintain compatibility with existing applications. Researchers keep exploring molecular tweaks to cut toxicity without giving up solvency or reactivity. In conversation with process engineers and environmental managers, I’ve heard growing calls to retrofit plants for safer handling, recycling, and monitoring. As global supply chains face disruption, backup molecules like this one edge up in strategic importance. With the right mix of innovation, transparency, and careful risk control, 5-Methyl-2-Hexanone can continue proving its value in an industry always pushing for better solvency, safer operations, and cleaner outcomes.
What are the primary uses of 5-Methyl-2-Hexanone?
Not Just Another Industrial Solvent
Plenty of folks outside chemical labs haven’t come across 5-Methyl-2-Hexanone, but this clear liquid solves more problems in the real world than its name lets on. Industries don’t hunt for odd chemicals just for the thrill—5-Methyl-2-Hexanone handles practical tasks nobody wants to do by hand.
The Go-To Choice in Solvent Blends
In paint shops and factories, paint won’t flow right without the perfect solvent. Many blends wind up too quick, too slow, or too sticky during drying. Add a bit of 5-Methyl-2-Hexanone and suddenly, pots last longer and coatings go on smoother. People working long hours with spray guns count on a finish that doesn’t leave runs or haze. Factories running solvent extraction setups also rely on this tough worker, especially where they need stubborn oils, waxes, or resins gone.
Helping With Resin and Polymer Production
Polymer markets keep growing as everything turns plastic or rubber. Production teams working in small and massive plants alike look for steady results each batch. 5-Methyl-2-Hexanone helps break down raw material clumps and encourages polymers to build just the right chain length. Skipping this step can lead to brittle goods tossed out after failed quality checks, so that solvent’s presence means less waste and more cash kept in the business.
Creating Cleaning Formulas That Actually Work
Janitors, mechanics, and even folks keeping up heavy equipment at parks use degreasing agents thickened with chemicals like 5-Methyl-2-Hexanone. Engines, parts, and greasy workbenches don’t come clean with water and elbow grease alone. This solvent lifts the sort of sticky grime that clings to moving parts and corners. Because 5-Methyl-2-Hexanone works faster than many similar chemicals, it helps workers avoid exposure for longer periods. It won’t make cleaning safe or fun, but shortcuts that cut risk matter.
Why Health and Safety Need Attention
Those managing 5-Methyl-2-Hexanone in large amounts run into safety concerns right away. Inhaling fumes leads to irritation or worse. Handlers in industry put on gloves and ventilate spaces, but even folks at home can stumble onto trouble using strong cleaner blends in a closed garage. The American Conference of Governmental Industrial Hygienists (ACGIH) lists exposure limits for a reason—repeated exposure may affect the nervous system. Proper training, safety sheets, and protective gear keep accidents less likely. Not every shop gets this right, so raising awareness remains crucial.
Moving Toward Better Solutions
Lately, companies that buy and work with industrial solvents ask for greener options. Some have experimented with water-based alternatives, but oil, resin, and paint often call for what water can’t offer. 5-Methyl-2-Hexanone remains popular because it handles the tough jobs reliably. Researchers keep testing safer, less toxic mixes to offer the same cleaning and blending power. Until a true substitute arrives, good ventilation, well-written safety rules, and giving workers the right gear matter more than ever.
Is 5-Methyl-2-Hexanone hazardous to health?
Understanding the Chemical’s Role
5-Methyl-2-hexanone shows up across several industries, especially where solvents are in high demand. Factories lean on it for its ability to dissolve resins, waxes, and coatings. You find it in labs, and occasionally, in paint thinners or cleaning fluids. The smell carries a punch—a sweet, acetone-like hit that seeps into the air, sometimes catching you off guard before you even see the label.
What Science Tells Us About Exposure
Long stretches working around this solvent reveal the truth: it’s far from harmless. Inhaling vapors leads to headaches, dizziness, and irritation in the eyes and throat. Spend enough years breathing that stuff in, and the body might pay a bigger price. The U.S. National Institute for Occupational Safety and Health (NIOSH) flags this substance as a possible threat to the nervous system. People exposed through poorly ventilated workspaces often mention feeling sluggish or having trouble remembering details, and lab tests support these worries.
Skin contact doesn’t make things much better, either. Touching 5-methyl-2-hexanone strips oils from the skin, causing dryness, redness, and sometimes allergic reactions. Sometimes folks avoid gloves, figuring a quick spill poses no real problem, but solvents like this can pass right through the skin to stir up trouble elsewhere in the body. The risk only grows when workers don’t get full training on handling or don’t keep up with personal protective equipment.
Epidemiology and Real-World Stories
Stories from the shop floor prove just as revealing as research papers. Workers in furniture manufacturing, for example, sometimes talk about chest tightness and a chemical taste in the air at the end of the day. In California, a group of workers exposed over months experienced tingling in fingers and forgetfulness long after shifts ended. These aren’t isolated cases. Researchers link repeated exposure to potential nerve damage and, in serious situations, threats to liver and kidney function.
Calls for Safer Practices and Alternatives
Companies sometimes cut corners on safety to keep costs down—but the aftermath carries a higher price. Engineering controls like local exhaust ventilation can pull fumes away from breathing zones. Gloves made of nitrile or neoprene dampen skin contact. Simple awareness—putting up prominent warning signs and holding regular safety briefings—can remind crews to stay alert. Regular air monitoring inside the workspace helps track vapor levels. None of these steps break the bank, but they make a big difference for everyone working in the area.
Some manufacturers already started the shift toward greener alternatives. Solvents that break down naturally or don’t put so much stress on the nervous system now sit on the market. The move toward safer chemistry doesn’t just reduce long-term health costs; it makes the workday smoother for those who once suffered through constant headaches or nausea.
Building a Smarter Future for Workers
Listening to workers’ reports and backing them up with research leads to smarter regulations. If companies, regulators, and workers each do their part—updating safety data sheets, giving real-world training, responding to near-miss incidents—everyone stands a better chance of heading home healthy. A few changes now can shape a safer workplace, while letting businesses benefit from the strengths of skilled, healthy teams.
What are the storage and handling requirements for 5-Methyl-2-Hexanone?
Why Chemical Storage Deserves Attention
Many industries rely on chemicals that rarely make the headlines, despite their impact on production, worker safety, and even the environment. 5-Methyl-2-Hexanone fits into this group. It shows up in paints, coatings, adhesives—jobs that power construction, automotive, and manufacturing. In these places, people can’t afford mishaps linked to careless storage or handling.
Physical Properties That Dictate Requirements
This liquid boils around 130°C and flashes at roughly 40°C. Those numbers stick out if you’ve ever worked near a loading dock or paint booth. The substance evaporates more quickly than water on hot concrete, filling a work area with its heavy odor. Flammable vapors demand regular ventilation, far away from ignition points. Smokers, open flames, sparks—anyone familiar with solvents understands how a simple spark can change lives in a second.
Safe Storage Starts with Common Sense
Experience proves that people often trust chemical drums and cans to “just behave.” With 5-Methyl-2-Hexanone, pushing luck has consequences. Store it in tightly sealed containers—metal or high-quality plastic—on floors with strong secondary containment. Temperatures shouldn’t push above room temperature or dip too low; both extremes mess with pressure and viscosity. I’ve seen leaky storage spurred by wild temperature swings because someone parked a drum in afternoon sun, thinking nothing of it.
Separate flammable liquids from oxidizers, acids, and other reactive stocks. People sometimes cut corners, stacking everything in a single storage room for convenience, but it only takes one mistake to create a headache for a whole operation.
Real-World Handling Prevents Accidents
Gloves, splash goggles, and aprons might feel inconvenient during a busy shift. Yet, there’s no replacement for skin and eyesight. Even if someone’s lucky enough to avoid a trip to the emergency room, short exposure can lead to dizziness or irritation. Anyone using the chemical should work with solid ventilation, or better yet, work outdoors where vapors disperse.
If you move these drums or containers with forklifts or dollies, take it slow. Rushing tends to bungle more jobs than it helps. Spills are more expensive than patience, not to mention the cleanup and possible reporting to authorities.
The Environmental Piece
Runoff from spills has a way of sneaking into drains and soil, where it lingers. Most local regulations require spill kits with absorbents and neutralizers on hand, plus clear labeling on every storage unit. I’ve worked with operators who thought labels were a formality—until hazmat teams showed up during an inspection.
Safe disposal matters even more than storage. Most municipalities ask for collection by licensed hazardous waste companies. Pouring solvents down the drain remains shockingly common, but the fines could easily shut down a small business.
Building a Culture Around Chemical Respect
Safe storage and mindful handling of 5-Methyl-2-Hexanone help businesses avoid medical bills, lost production, and legal headaches. Crew training, clear procedures, and regular checks keep slips from turning tragic. The cost of doing things right leaves plenty of value on the table—especially for anyone hoping to stay in business for the long haul.
What is the chemical structure and formula of 5-Methyl-2-Hexanone?
Chemistry Made Approachable
Chemistry sometimes feels like a secret language, but anyone working in labs, manufacturing plants, or even high school science class knows a little curiosity goes a long way. Take 5-Methyl-2-hexanone for example. This organic compound shows up in industrial solvents and occasionally in discussions about workplace exposure standards. The real question most folks face: what’s hiding behind that name, and why should anyone care?
Getting to the Heart of the Molecule
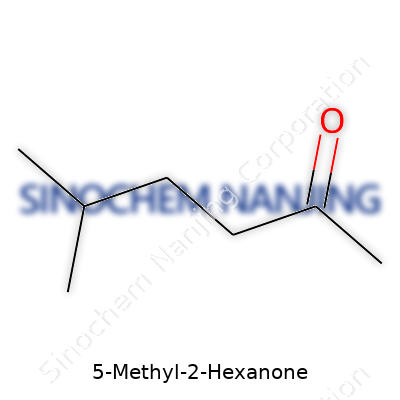
The chemical formula for 5-Methyl-2-hexanone is C7H14O. It starts with seven carbon atoms, fourteen hydrogens, and a single oxygen. Pretty standard for a small molecule found in various workplaces. The name gives away its main features. The “2-hexanone” hints at a six-carbon chain with a ketone group on carbon number two. Throwing a “5-methyl” into the mix means there’s a little arm – a methyl group – tacked onto the fifth carbon of the main chain.
The structure itself looks like this: you have a straight chain of six carbon atoms. The second carbon sports the oxygen double-bonded to it (that’s your ketone group). The fifth carbon reaches off to the side holding a single methyl group. In organic shorthand, it’s written as CH3-CO-CH2-CH2-CH(CH3)-CH3. Calling it “methyl isobutyl ketone’s sibling” usually brings nods from chemists, since they share that familiar backbone but branch out in slightly different places.
Importance in Industry and Research
Solvents like 5-Methyl-2-hexanone play a heavy role across paint shops, chemical plants, and even electronics manufacturing. You find it as a cleaning agent and a chemical intermediate. Its balance of volatility and solvent power makes it valuable. Safety data matters because most solvents move through air fast, and inhaling them on the job isn’t a recipe for long-term health.
Researchers years back noted a rise in workplace complaints tied to certain ketones. The National Institute for Occupational Safety and Health (NIOSH) and similar organizations list exposure limits, and those numbers mean something every time you crack open a drum of this stuff. Small changes in molecular structure create big shifts in health risks, flammability, and transport rules.
Safer Handling and Smarter Choices
I once watched a maintenance team swap out a cleaning agent after someone started experiencing dizziness at the end of each shift. Simple substitution dropped exposure to these ketones significantly. It’s not only about swapping chemicals, though—ventilation, respirators, and regular air tests are all part of the process. Labs using 5-Methyl-2-hexanone often double down on containment and storage training, especially in smaller facilities where oversight can slip.
As regulations keep shifting, companies face pressure to document every chemical’s structure and potential effects. Understanding the backbone of molecules like 5-Methyl-2-hexanone makes it easier to predict hazards, argue for better alternatives, or simply explain why that odd-smelling bottle in the storeroom can’t be ignored. As people keep digging into safer formulations and greener processes, knowing your chemical structures pays off on the frontlines of work and health.
Are there any environmental regulations for disposing of 5-Methyl-2-Hexanone?
Understanding the Substance
5-Methyl-2-Hexanone is no harmless leftover. Anyone who has handled it in a lab or a factory knows that sharp, solvent smell and sees the safety data sheets warning against careless contact. The chemical belongs to the family of ketones and carries risks for health and the environment. It acts as an irritant and its vapors can affect the nervous system. The EPA lists it as a hazardous waste, and OSHA outlines industrial exposure limits. This is chemical territory that invites close attention, especially by those dealing with disposal.
Disposal: What the Law Expects
All across the US, the EPA’s Resource Conservation and Recovery Act (RCRA) sets the rules for hazardous waste. RCRA counts 5-Methyl-2-Hexanone among substances that need strong controls. You can’t pour it down the drain, let it evaporate, or mix it with regular trash. Municipal wastewater plants will not tolerate organic solvents like this entering their system, and state environmental agencies fine heavily when they find offenders.
From years working in labs, I’ve watched technicians sealing spent solvent in clearly marked, compatible containers, kept away from heat and sunlight, and logging each day’s waste track. Hazardous materials get picked up by licensed haulers who understand compatibility and the dangers of mixing organics with other volatile wastes. They know the transport and disposal rules in the Code of Federal Regulations (CFR). Any slip-up lands the generator in hot water, not just the waste vendor.
Why the Rules Matter
Industrial solvents like this one break down slowly in the environment and pollute groundwater. A single careless pour by a janitor or maintenance worker can contaminate drinking water or threaten wildlife. Chronic exposure impacts people, too: headaches, dizziness, and over longer periods, more serious nervous system disorders.
From an environmental perspective, there’s no safe amount of illegal dumping. State data shows that contaminants like 5-Methyl-2-Hexanone show up in soil at sites slated for Superfund cleanup. These cleanups cost taxpayers millions and stretch on for decades. The EPA tracks these sites in every state. When companies follow proper procedures, that mess never happens.
Fixing Gaps in Practice
Rules alone can’t prevent every problem. Too many smaller businesses believe they can skip hazardous waste training, or they rely on outdated paperwork. My advice: keep waste disposal logs, label everything, and don’t skimp on onboarding. Contracting reputable waste handlers also helps. They know the latest federal and state regulations and how to tell real from counterfeit disposal certificates. On the plant floor, regular audits and unannounced inspections catch bad habits before they get embedded in culture.
Switching from hazardous chemicals to safer substitutes, wherever possible, makes a big difference. Industry groups share best practices on solvent alternatives and emerging green chemistry. State grants sometimes support businesses willing to invest in safer technology. I’ve worked with a few companies using these incentives to upgrade their processes, which paid off for both safety and reputation.
Community Responsibility
Every person who works with dangerous chemicals has a role. Reporting leaks or unsafe handling, seeking out updated training, and speaking up about unsafe instructions all keep communities safer. Public agencies post disposal requirements for hazardous wastes online, and local government environmental health offices answer questions directly. Transparency helps everyone keep the water and soil clean.
The rules around 5-Methyl-2-Hexanone aren’t just bureaucratic red tape—they reflect years of lessons from pollution, illness, and hard-won public victories. Taking shortcuts puts people and ecosystems at long-lasting risk, and in my experience, the best companies treat environmental compliance as a core value, not just another part of paperwork.
| Names | |
| Preferred IUPAC name | 3-Methylhexan-2-one |
| Other names |
Methyl isobutyl ketone Isobutyl methyl ketone MIBK Hexan-2-one, 5-methyl- Isopropylacetone |
| Pronunciation | /ˈfaɪˌmɛθɪl tuː ˈhɛk.səˌnoʊn/ |
| Identifiers | |
| CAS Number | 110-12-3 |
| Beilstein Reference | 1209247 |
| ChEBI | CHEBI:88219 |
| ChEMBL | CHEMBL156151 |
| ChemSpider | 7664 |
| DrugBank | DB02254 |
| ECHA InfoCard | 100.011.204 |
| EC Number | EC 203-640-2 |
| Gmelin Reference | 92465 |
| KEGG | C06355 |
| MeSH | D004582 |
| PubChem CID | 7907 |
| RTECS number | MP5425000 |
| UNII | Q60LO468R6 |
| UN number | UN1230 |
| Properties | |
| Chemical formula | C7H14O |
| Molar mass | 114.19 g/mol |
| Appearance | Colorless liquid |
| Odor | sweet; fruity |
| Density | 0.809 g/mL at 25 °C |
| Solubility in water | 4.2 g/L (20 °C) |
| log P | 1.98 |
| Vapor pressure | 0.89 kPa (at 20 °C) |
| Acidity (pKa) | pKa ≈ 20 |
| Magnetic susceptibility (χ) | -63.2e-6 cm³/mol |
| Refractive index (nD) | 1.411 |
| Viscosity | 1.04 mPa·s (25 °C) |
| Dipole moment | 2.75 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 359.2 J/mol·K |
| Std enthalpy of formation (ΔfH⦵298) | -302.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3914 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Precautionary statements | H225, H319, H336 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | Flash point: 46°C (115°F) |
| Autoignition temperature | 515 °C |
| Explosive limits | 1.2% - 8.0% |
| Lethal dose or concentration | LD50 oral rat 2100 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 1600 mg/kg |
| NIOSH | GY8225000 |
| PEL (Permissible) | PEL: 100 ppm (410 mg/m³) |
| REL (Recommended) | 50 ppm |
| IDLH (Immediate danger) | 100 ppm |

