5-Methoxytryptamine Hydrochloride: A Close-Up Look
Historical Development
5-Methoxytryptamine Hydrochloride, sometimes called mexamine, comes from a branch of research that began tracking tryptamine derivatives during the mid-20th century. Scientists started noticing that compounds related to serotonin often played roles in both neurological and peripheral processes. For years, doctors and researchers sifted through floral and animal samples, digging into the connection between naturally occurring tryptamines and signaling molecules in the brain. Over time, chemists learned how to modify these compounds, making them more stable and practical for laboratory work. This shift out of natural extraction into bench synthesis shaped the modern methods that now supply consistent, high-purity batches for both research and pilot medical use. In my own experience, these changes made sourcing reliable reagents much less of a gamble; talking to any chemist who worked before the 1990s, you’ll hear stories of unreliable purity and mysterious biological effects from contaminants. The field learned from those missteps, setting the stage for today’s more regimented approach.
Product Overview
5-Methoxytryptamine Hydrochloride stands as a solid, crystalline powder, usually white or slightly off-white. Chemically, it’s an indole-based substance, tracing its structure back to serotonin but altered with a methoxy group at the 5-position. In the lab, this molecule really shines for its clear profile—a reproducible choice for studies probing neurotransmission, pharmacology, and the after-effects of tryptamine analogues. In pharmaceutical settings, it serves as a benchmark for evaluating new treatments that look to harness or modulate serotonin pathways.
Physical & Chemical Properties
The physical side of 5-Methoxytryptamine Hydrochloride often draws interest for its characteristic melting point, usually hovering around 158–162°C. The substance dissolves easily in water and methanol, avoiding the headaches that often come with more hydrophobic tryptamines. Its molecular formula is C11H15ClN2O, and the core structure allows for clear identification on chromatographs, making purity checks straightforward. This transparency helps scientists pinpoint the substance’s role without chasing shadows caused by hidden degradation products.
Technical Specifications & Labeling
Labs or commercial suppliers print technical specifications right on the bottle to help researchers identify the key attributes. You generally see purity standards listed as 98% or higher by HPLC, shelf life attributed to proper storage—dark, dry, below 25°C—and directions for safe handling. The labeling gets reviewed periodically to fit regulations, reflecting ongoing diligence as the list of known impurities grows with testing. Certification through ISO standards adds credibility, serving as a kind of handshake between vendor and buyer.
Preparation Method
Manufacturing 5-Methoxytryptamine Hydrochloride usually begins with 5-methoxyindole, which undergoes a sequence of functionalization and reduction steps. One common route: first, N-alkylation using haloalkylamines, followed by reduction with sodium borohydride under controlled temperatures. The crude product experiences several rounds of recrystallization, often from ethanol or isopropanol, to draw out the pure hydrochloride salt. Technicians stay careful to monitor for side-chain dealkylation and byproduct formation, as each round of reaction risks creating impurities that could affect downstream results. I’ve seen rushed syntheses create batches that look pure by NMR but fall apart under bioassay, so it pays to go slow and check each stage meticulously.
Chemical Reactions & Modifications
5-Methoxytryptamine offers a springboard for many chemical reactions, especially for labs interested in tweaking receptor affinities or metabolic stability. The methoxy group at the five position allows for targeted modifications via demethylation or further alkylation. Other researchers explore acylation of the amine nitrogen or substitution on the indole ring, chasing analogues that show promise as selective receptor ligands or as probes for tracing metabolic steps in living tissue. Plenty of publications chronicle tweaks to this structure, revealing the chemical curiosity surrounding its core scaffold.
Synonyms & Product Names
Scientific literature and catalogs list 5-Methoxytryptamine Hydrochloride under many names: mexamine, 5-MT, 5-methoxytryptaminum chloride and its international nonproprietary equivalents. Some chemical suppliers use catalog numbers tied to their own product lines—a headache for anyone searching databases, but all part of life in chemical research. These synonyms often reflect local language differences or changes in naming conventions over time.
Safety & Operational Standards
Handling 5-Methoxytryptamine Hydrochloride calls for the usual precautions you’d expect with laboratory chemicals: gloves, goggles, attention to hood ventilation, and careful waste disposal. The chemical, while not especially volatile or reactive, still falls under the rules for tryptamine derivatives due to possible bioactivity. MSDS documents highlight acute toxicity studies and set limits for inhalation or skin contact. For operations outside the lab, adherence to environmental disposal rules means collecting even dilute aqueous solutions instead of pouring them down the drain. Safety data evolves as regulators gain new information from accumulated toxicity data and metabolite tracking, keeping practitioners on their toes.
Application Area
In research, 5-Methoxytryptamine Hydrochloride feeds studies on serotonin regulation and sleep, as well as pharmaceutical development aimed at psychiatric disorders. Its structural similarity to melatonin and serotonin turns it into a versatile molecule for unraveling the subtle dance of neurotransmitters in the brain. Pharmacologists use it to examine receptor affinity profiles, check for signal transduction triggers, and screen for side effects in developmental-stage drugs. A variety of medical and veterinary researchers include it in animal models that simulate sleep cycle disruptions or mood disorders, giving it more reach than most molecules its size.
Research & Development
Interest in this compound shows no signs of slowing. Ph.D. students the world over rely on it as a baseline for studying biogenic amines and neurotransmitters, using its clear-cut profile to anchor complicated experimental setups. Some biotech firms, eyeing the growing market for sleep and mood disorder therapies, examine derivatives as drug prototypes. Many experiments revolve around synthesizing analogues and mapping their pathway through human or animal metabolism. A growing field tracks the molecule’s fate in the environment, helping chart how research chemicals might leak into broader ecosystems.
Toxicity Research
Toxicology studies dig deep into acute and chronic exposure in both laboratory animals and cell cultures. Doses well above routine lab use show mild neurotoxic effects in animal models, especially in offspring during gestational exposure. While short-term contact appears manageable under strict lab safety, evidence continues to accumulate about the impacts of sustained exposure—not so much that it would raise alarms for a trained chemist, but more than enough for regulatory agencies to monitor it carefully. I’ve seen protocols updated after journals published long-term bioaccumulation risks; researchers adapt quickly, but people new to the field do best taking these warnings seriously from the start.
Future Prospects
5-Methoxytryptamine Hydrochloride still holds plenty of promise for neuroscience and pharmaceutical innovation. Modern imaging and fast chemical detection tools let researchers map its action in live animals and cultured tissues with precision nobody imagined twenty years ago. Emerging fields—such as neuroinformatics and chemical genetics—draw on this compound’s reliability as a reference tool or building block for custom ligands. As startups and large drug developers race to capture new serotonin-based therapeutics, interest in novel tryptamine analogues only expands. The next few years look set to bring even tighter controls, richer data about metabolic breakdown, and perhaps new therapeutic classes branched from its familiar structure.
What is 5-Methoxytryptamine Hydrochloride used for?
Digging into the Basics
5-Methoxytryptamine Hydrochloride often goes by 5-MT. Its chemical family includes melatonin and serotonin—two heavy hitters in the fields of sleep and mood research. At a glance, 5-MT doesn't make daily headlines, but labs and universities put a lot of time into studying it. This compound shows up in research for some pretty big questions about mental health, circadian biology, and the chemistry behind our feelings and sleep cycles.
Exploring How It Helps Science
Most studies using 5-MT focus on how it interacts with serotonin receptors in the brain. For anyone talking about neurotransmitters, serotonin comes into the conversation right away. It's about more than just feeling good. Serotonin influences how we manage stress, deal with anxiety, and sleep at night. Researchers use 5-MT to look at how tweaking these pathways might shift brain chemistry. That matters for conditions like depression or sleep disorders, which affect millions.
Pushing Limits for Mood Research
Scientists explore new antidepressants every year, and 5-MT often plays a part in that story. Because its structure closely resembles both melatonin and serotonin, it fits into the same biological 'locks' those molecules use. By studying its effects, researchers reveal fresh ideas for drugs that could help people struggling to find the right depression or anxiety treatment. There's still a lot to learn here, but every bit of data helps.
Biohacking the Sleep Cycle
Melatonin supplements get more supermarket shelf space every year. While melatonin works for some jet-lagged travelers or shift workers, it doesn't act alone in the body. 5-MT helps researchers look closer at what controls the body’s clock. If future therapies could tap into the same machinery, people with insomnia or circadian rhythm issues might get another tool in their kit. People often underestimate how much stable sleep improves life, both short-term and decades down the road.
Lab Work and Drug Development
Not every discovery hits pharmacy shelves right away. A compound like 5-MT might act as a building block in drug testing. Scientists use it to test new compounds and to map out which regions of the brain certain drugs actually target. In this sense, 5-MT doesn’t become a medicine, but it makes other drugs possible. This is behind-the-scenes science. What happens at the research bench eventually works its way toward better patient care in clinics and hospitals.
Responsible Investigation
No commentary about brain chemistry is complete without saying this: all scientific work with 5-MT follows careful procedure. Universities and pharmaceutical companies face strict oversight when dealing with these kinds of molecules. That keeps the focus where it belongs—on evidence, ethics, and safety. Responsible advances come from slow, steady science, not hype or shortcuts.
Looking Forward
As more stories highlight mental health and sleep, we can expect research chemicals like 5-Methoxytryptamine Hydrochloride to keep showing up in big studies. That may sound technical, but the end result touches daily life—from treatments for insomnia to breakthroughs in depression therapy. Real lives are at stake. Careful research now promises new tools tomorrow, and that keeps hope alive for people who need better solutions.
What is the recommended dosage for 5-Methoxytryptamine Hydrochloride?
Why Dosage Matters For Any Substance
Many people look at 5-Methoxytryptamine Hydrochloride, also known as mexamine, and wonder how much is safe or effective. Before diving in, it’s smart to remember: the right amount makes all the difference. Taking too little might not do much. Taking too much turns curiosity into risk. Anyone handling substances like this needs to take dose recommendations seriously, especially when research is limited.
The Science We Know So Far
Mexamine is a naturally occurring tryptamine in plants and animal tissues. Scientists started looking at it decades ago for its possible role in melatonin production and neurotransmitter activity. Today, you can find it in some research laboratories and chemistry supply companies. Research studies often look at rodents or isolated tissue samples. Human trials are rare, so clear, universally accepted dosing guidelines just don’t exist.
From experience in scientific labs, dosage for experimental compounds will always lean on caution. I’ve seen colleagues start at the lowest detectable level and slowly bump up the dose, sticking with protocols and safety data whenever available. For mexamine, animal studies sometimes use doses around 1-10 milligrams per kilogram of body weight for injections in rodents. Extrapolating those numbers to humans just doesn’t work straight across due to differences in metabolism, body chemistry, and routes of administration.
Responsible Use and Safety First
For anyone considering working with or studying this compound, the first place to turn is the safety data sheet, research papers, and expert toxicology input. If you ask a medical researcher or practicing toxicologist, most will say that without FDA approval or thorough clinical studies, no one should recommend or use mexamine outside of an approved research protocol. Even promising molecules require strict safety oversight before anyone talks about dosage for humans.
Accidents and overdoses with untested compounds can happen far more easily than people think. Even well-trained chemists keep their sample sizes tiny and their logs detailed. The scientific record holds too many examples of hopeful discoveries backfiring because someone cut corners, or assumed “natural” means “harmless.”
Supporting Evidence and Reliable Resources
A recent scan through journals like the Journal of Psychopharmacology or Neuropharmacology pulls up only animal dosing details, with no consensus for human use. Even databases trusted by hospitals or poison control centers point out the gap: Without proper studies, no one can say what’s “safe” or how mexamine interacts with other drugs. Too few facts, too much risk. This is one area where sticking with established clinical substances pays off, because dosages have been worked out by people whose job is to minimize harm.
What’s Next For Curious Minds
Anyone interested in learning more can read publications in PubMed or reach out to professional societies like the American Chemical Society or the Society for Neuroscience. At the end of the day, education saves lives. Ask questions, look for real answers, and don’t accept crowd-sourced guesses about chemicals that deserve respect and careful handling. For now, the only responsible answer about 5-Methoxytryptamine Hydrochloride dosage is: Treat with care, research responsibly, and leave human dosing to the experts.
Are there any side effects associated with 5-Methoxytryptamine Hydrochloride?
Getting Closer to the Molecule
5-Methoxytryptamine Hydrochloride might not set off alarms in the average person's mind, but this compound quietly grabs attention among scientists and those interested in neurology and biochemistry. The molecule comes from the same family as melatonin and serotonin, so it plays into the body's natural rhythm. Many think of it as a research chemical and not a supplement you can pick up from the corner shop. Still, mentions of its potential use for sleep, mood, or even cancer research have made their rounds.
Learning from Other Tryptamines
Looking at its family tree, 5-Methoxytryptamine shares features with compounds that affect serotonin receptors in the brain. Similar tryptamines sometimes cause changes in sleep, body temperature, or mood. While tracing its effects, some see an overlap with melatonin, which signals sleep, and serotonin, which plays into emotional balance.
Known Side Effects—Science and Stories
Actual clinical data on 5-Methoxytryptamine Hydrochloride in humans comes in short supply. Published studies mostly stick to animals or test tubes. So, what can be said with honesty? Given what we know about related tryptamines, it makes sense to suspect this compound could bring sleepiness, shifts in mood, or changes in appetite. Sometimes, similar chemicals cause dizziness, headaches, nausea, or digestive issues. Melatonin, for example, sometimes triggers vivid dreams, next-day grogginess, or a dip in body temperature. Serotonin-acting compounds sometimes raise blood pressure or heart rate, though most data don't show this with 5-methoxytryptamine.
Lab tests link 5-Methoxytryptamine to possible changes in circadian rhythms, which may leave some people feeling out of sync after taking it at the wrong time of day. Animal studies flag high doses as possibly toxic to certain cells and organs. While no human poisoning cases get reported in published literature, no one should mistake lack of reports with proof of safety.
Risks and the Value of Caution
Some risk comes from not knowing enough. Even well-known melatonin causes surprises for many people—things like hormone changes, weird dreams, or morning grogginess. With 5-Methoxytryptamine, the gaps in studies make it harder to predict who's most at risk. Kids, pregnant women, or adults with mood disorders stand out as groups needing special caution. Mixing it with other serotonin boosters could mean more serious complications, like serotonin syndrome.
The quality and purity of chemicals from unregulated sources can't be trusted. I've known people who ordered so-called "pure research compounds" online, only to develop headaches or rashes, later realizing the batch was tainted.
Toward Safer Use—What Makes Sense
Trust in a chemical grows when scientists and doctors get years to track its effects. This simply hasn't happened yet with 5-Methoxytryptamine Hydrochloride. Anyone considering it for research or personal use should talk with medical professionals, check legal status locally, and stick to legitimate suppliers with strong reputations.
If future studies prove benefit over risk, clearer guidance will come. Until then, walking carefully, staying informed, and looking beyond anecdote to solid science protects both the curious and the hopeful.
How should 5-Methoxytryptamine Hydrochloride be stored?
Knowing What You’re Handling
5-Methoxytryptamine hydrochloride has captured the attention of researchers for its role in neurotransmitter studies and possible links to circadian rhythm regulation. In a lab or clinical environment, this compound brings value only when its properties remain stable and safe for use. Mishandling can lead to wasted resources, questionable data, and safety risks nobody should brush off.
The Real Risks of Careless Storage
Live long enough working in laboratories, and a few horror stories pile up—chemicals stashed next to a heat vent or left loosely capped, and months of work unravel in an afternoon. Chemical compounds like 5-Methoxytryptamine hydrochloride break down under heat, humidity, or exposure to light. A little moisture creeping in, or sun streaming across the bench, accelerates decomposition. Research by chemical suppliers has shown that tryptamine derivatives stored at room temperature under bright conditions lose potency and develop unknown byproducts.
Worse, a degraded substance can behave in unpredictable ways. No one wants to run a cell viability assay only to get false negatives or toxic surprises because a powder didn’t get proper care. Keeping chemicals reliable is a foundation of responsible research.
Straightforward Steps for Proper Storage
People sometimes overthink chemical storage. For 5-Methoxytryptamine hydrochloride, a few practical rules serve well. Seal it tightly in a clean, inert container—amber glass jars or HDPE bottles with secure screw caps work best. These containers keep out air and moisture that can destroy the sample over time.
Store the container in a refrigerator around 2°C to 8°C. At that temperature range, organic compounds decay much slower, and the cool environment helps prevent reactions with trace atmospheric oxygen or water vapor. Avoid placing this chemical in the freezer unless the manufacturer or pharmacist recommends it, since freeze-thaw cycles can introduce condensation inside the packaging.
Keep the chemical in a spot shielded from direct sunlight or strong ambient light. UV rays strike sensitive molecules, cause photodegradation, and leave researchers with less or a mistaken product. Cabinets designed for chemical storage or even standard fridge shelving away from the door light work well.
Always label the container clearly with the name, concentration, and opening or preparation date. If you work in an environment where many people come and go, add hazard information and locks where required to protect less experienced colleagues. Fumbling with anonymous powders or mixing unlabeled samples has caused more than one lab mishap.
Learning from Everyday Experience
Everyone in the sciences has opened an old chemical jar only to find clumps or discoloration—signals that what was once useful has now turned useless, or worse, hazardous. Years ago, I managed a storeroom and saw budgets blown on replacement orders for ruined stocks. Labs with strong habits for sealing, labeling, and refrigeration wasted less and produced more reliable, reproducible work.
Mistakes happen, but most are preventable through simple awareness. Training everyone, including new hires and students, in storage routines protects people and investments. Manufacturers publish storage guidelines because they want you, the end user, to trust results and minimize liability.
Looking Forward: Consistency Brings Results
Watching out for lead times, expiration dates, and unusual temperature swings in the lab saves time and money. Document any storage quirks on shared log sheets, replace worn seals, and keep an eye out for the telltale signs of spoilage. If everyone invests a few minutes in storage care, the entire team benefits and your research stands on firmer ground.
Is a prescription required to purchase 5-Methoxytryptamine Hydrochloride?
What Is 5-Methoxytryptamine Hydrochloride?
5-Methoxytryptamine Hydrochloride, often called 5-MT or Mexamine, came up during my graduate research. Anyone who has spent time in a lab knows how often new chemical names float through, each with its own set of rules. 5-MT belongs to a family of tryptamines which, in some variations, have shown up in scientific discussion about brain chemistry, sleep, and mood regulation. As with other tryptamine compounds, this one draws attention from both researchers and, at times, folks just curious about neurotransmitters.
Legal Status and Prescription Rules
In most countries, a clear difference stands between chemicals for research and those used in medicine. In the United States, the Food and Drug Administration (FDA) controls which compounds a pharmacist can hand out. 5-Methoxytryptamine Hydrochloride never got approval from the FDA for any medical treatment. No pharmaceutical company has asked to market it. As a result, nobody fills a prescription for it at the corner drugstore. Instead, chemical suppliers sell it to research labs, not to patients or people looking for supplements.
Europe follows similar guidelines. The European Medicines Agency treats all novel compounds with caution. Researchers buy it through institutional accounts, not through doctors or pharmacies for personal use. The story repeats almost everywhere else: only accredited labs can order this chemical, and sellers ask for proper paperwork before shipping anything.
Why Regulations Matter
I’ve seen what happens when regulations get ignored. Novel research chemicals can carry real risks. 5-MT shares some structural traits with neurotransmitter serotonin, but the body doesn’t always handle synthetic analogues the way people expect. Scientific studies barely address its effects in living humans, except in a few narrow lab setups. Without long-term safety data, letting it loose on the open market invites an accident.
A handful of websites promise the world, selling unregulated chemicals to anyone with a credit card. This sidesteps safety checks, with no pharmacist or doctor to screen for interactions or toxicity. A short-term thrill turns into a long-term gamble. People can’t know for sure what’s in each package, and nobody tracks problems that might show up months later.
Tracking And Solutions
My own work in academic labs trained me to fill out chemical inventories, report unusual effects, and manage storage. This chain of custody builds trust. Requiring research-only sales, instead of over-the-counter access, helps keep risky compounds out of the hands of people looking for a shortcut. Public resources like poison control centers offer help for accidental exposures, but they rely on transparency and regulation to do their job.
Practical solutions often start with more education. People should understand that “research chemical” isn’t just a harmless label. Proper lab protocols and medical oversight protect both researchers and the broader public. New compounds deserve careful handling, especially before much is known about their risks. I’ve watched too many trends promise easy answers, only to backfire when rules get ignored. Respecting these safeguards keeps both science and individuals safer.
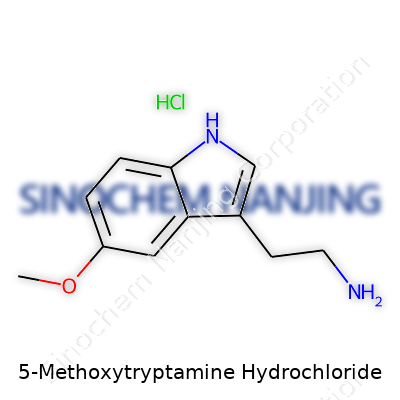
| Names | |
| Preferred IUPAC name | 2-(5-methoxy-1H-indol-3-yl)ethan-1-amine hydrochloride |
| Other names |
5-Methoxytryptamine HCl 5-MT HCl Mexamine hydrochloride |
| Pronunciation | /ˈfaɪˌmɛθ.ɒk.siˌtrɪpˈteɪ.miːn haɪˌdrɒx.iˈklɔː.raɪd/ |
| Identifiers | |
| CAS Number | 55486-13-6 |
| 3D model (JSmol) | `3D JSmol model string` for **5-Methoxytryptamine Hydrochloride** (C11H16ClN2O): ``` CNCCc1c[nH]c2ccc(OC)cc12.Cl ``` |
| Beilstein Reference | 72419 |
| ChEBI | CHEBI:47749 |
| ChEMBL | CHEMBL16211 |
| ChemSpider | 14533 |
| DrugBank | DB01850 |
| ECHA InfoCard | 100.235.914 |
| EC Number | 223-739-6 |
| Gmelin Reference | 1200937 |
| KEGG | C05584 |
| MeSH | D018470 |
| PubChem CID | 6137 |
| RTECS number | VL7520000 |
| UNII | 7T51O37U1D |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DTXSID80896459 |
| Properties | |
| Chemical formula | C11H16ClN2O |
| Molar mass | 250.74 g/mol |
| Appearance | White to off-white crystalline powder |
| Odor | Odorless |
| Density | 0.98 g/cm3 |
| Solubility in water | Soluble in water |
| log P | 0.5 |
| Acidity (pKa) | 9.34 |
| Basicity (pKb) | 6.75 |
| Magnetic susceptibility (χ) | -65.6×10⁻⁶ cm³/mol |
| Dipole moment | 2.42 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 234.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | ΔfH⦵298 = – |
| Std enthalpy of combustion (ΔcH⦵298) | -4885 kJ/mol |
| Pharmacology | |
| ATC code | N06AX15 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | Precautionary statements: P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P332+P313, P337+P313, P362+P364 |
| Flash point | 149.5 °C |
| Autoignition temperature | Autoignition temperature: 410 °C |
| LD50 (median dose) | LD50 (median dose): Mouse, intraperitoneal: 200 mg/kg |
| NIOSH | NLV1806000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10-50 mg daily |
| Related compounds | |
| Related compounds |
Serotonin Melatonin 5-Methoxy-N,N-dimethyltryptamine (5-MeO-DMT) Tryptamine Bufotenin |

