5-Hexen-2-One: Looking Beyond the Lab Bench
Tracing the Roots: How 5-Hexen-2-One Found Its Place
5-Hexen-2-one has a story that carries echoes of organic chemistry’s relentless search for diversity in molecules. Decades ago, research chemists in both the fragrance and pharmaceutical spheres began to notice how subtle changes in carbon skeletons led to unexpected shifts in both scent and bioactivity. Picture the world of the 1950s and 60s, when the expansion of synthetic organic toolkits made it possible to turn out chains and rings at a scale previously unimagined. Simple molecules like 5-hexen-2-one gained value not because they starred in major industrial syntheses, but because of their versatility—both as building blocks and as functional additives. Anyone who spent early years hunting for new flavors or niche medicinal leads has crossed paths with this compound in journals and catalogs, often bundled with other unsaturated ketones.
The Personality of 5-Hexen-2-One
This compound isn’t the friendliest to the nose, striking a balance between fruity-green notes and sharp, almost pungent edges. In terms of practical properties, 5-hexen-2-one walks a line familiar to ketones—moderate boiling point, low viscosity, clear appearance. A lone double bond in the chain catches attention, both chemically and spectroscopically, while the keto group at the second position drives reactivity. Pour some into a beaker and you spot the classic liquid, colorless if pure, with an odor that sticks around longer than many realize. Its molecular weight, formula, and the expected refractive index land right where you’d expect for a small unsaturated ketone.
Technical Details and What They Mean in Daily Use
Labels and specs can sometimes read like riddles to a newcomer, but old hands in organic laboratories catch on quickly. The CAS number for 5-hexen-2-one makes ordering clear in crowded storerooms. Purity standards set by flavor or pharma regulators generally hang over production, with water content, isomer control, and storage warnings printed clearly on bottles. Manufacturers must choose between glass and lined metal for container options, especially since the double bond can make storage tricky under heat or light. Companies pursuing “green” credentials might chase lower solvent residues or animal-testing-free guarantees, answering a market that has ethics on its mind.
How 5-Hexen-2-One Arrives: Paths from Idea to Molecule
There’s no shortage of tricks to build 5-hexen-2-one. Common routes draw from classic carbonyl chemistry—aldol condensations, catalytic isomerizations, and oxidation of corresponding alcohols. Those who learned synthetic methods before the modern era will remember the treatise on controlling double bond placement, juggling temperature, pH, and sometimes the wits of a skilled hand. Newer, greener methods emerge with catalysts that promise less waste or milder conditions, using palladium, nickel, or base metals when possible. These changes aren’t just good press—they cut down on disposal costs and workplace hazards, a factor that matters for chemists who still spend long hours on the bench.
Chemical Reactions and Creative Tweaks
This isn’t a molecule that likes to keep still. The combination of a double bond and a carbonyl sets it up for everything from simple hydrogenation to wild polymerizations under the wrong conditions. In flavor and fragrance labs, careful manipulation—reduction, acetylation, and epoxidation—can build libraries of analogs with similar backbones but altered sensory footprints. Drug discoverers look for conjugated ketones in search of biologically active leads, and tweaking the position or substitution on 5-hexen-2-one can swing properties wildly. That’s what makes it valuable to both tinkerers and those chasing new patents.
Name Game: Synonyms and Common Aliases
Chemists rarely stick to a single naming convention, and 5-hexen-2-one shows up under a handful of banners. Some call it “1-Penten-3-on”, using older Germanic influences, while others log it as “hex-5-en-2-one” in more IUPAC-flavored systems. Literature searches and supplier catalogs might list unsaturated methyl ketone as a generic pointer, though experienced folks know to double-check structures. Any chemist worth their salt has juggled enough synonyms to appreciate why clear communication still sits at the core of good science.
Playing It Safe: Rules and Best Practices
Handling 5-hexen-2-one means more than gloves and goggles, especially since liquid ketones can bite with both volatility and reactivity. Standard operating guidelines call for secure ventilation, tightly sealed storage, and quick cleanup of spills. This care isn’t just to spare the operator’s lungs or skin; many small ketones have routes of toxicity that demand respect, with both inhalation and dermal absorption in play. Updated safety data—from animal studies and high-throughput toxicity screens—drive revisions in handling protocols and shelf-life calculations. Practical safety takes experience and a respect for how quickly a small leak or spill can escalate in a warm lab or poorly ventilated classroom.
How 5-Hexen-2-One Gets Used
If there’s a corner of the chemical industry where small, reactive ketones aren’t welcome, it’s hard to find. Perfumers and flavorists value 5-hexen-2-one for its ability to add crisp, fresh top notes or serve as a synthetic bridge between other, more costly raw materials. Agrochemical developers once toyed with functionalized analogs while chasing novel crop protection leads. In academic research, 5-hexen-2-one earns points as both a model for reaction mechanisms and a component in combinatorial syntheses. Lab instructors use it to teach selectivity and the hazards that can come with unsaturation.
The Pulse of Research and Development
The last decade has seen a slow boost in interest for efficient synthesis and “green” chemistry updates related to unsaturated ketones like 5-hexen-2-one. Academic groups chase new catalysts while industry looks for biobased feedstock integration. Analytical chemists have developed improved detection methods, using NMR, GC-MS, and IR, to track purity and trace contaminants down to fractions of parts per million. Universities pitch flavor and fragrance research as part of broader sensory science programs, often using 5-hexen-2-one derivatives as teaching models or as part of metabolomics screens in plants. The intersection of bench research and regulatory pressure isn’t always comfortable, but it does create opportunities for safer, cleaner synthesis and analysis.
Toxicity and the Questions Still Open
Every solvent and additive carries a backstory of hazard, and the toxicity profile of 5-hexen-2-one won’t surprise anyone familiar with unsaturated, low-molecular weight ketones. Acute exposure can cause mild irritation by inhalation or skin contact. Metabolism studies in animals hint at both rapid breakdown pathways and possible concerns over repeated exposure, particularly in higher concentrations. Regulatory agencies keep a close eye on new data, especially for compounds touching food, clinical trials, or everyday fragrance products. Consumer safety watchdogs and responsible manufacturers need clear reporting and risk assessments rooted in current animal and cellular data, not outdated case studies. This calls for constant vigilance, shared data across borders, and a willingness to reformulate or relabel as new toxicity discoveries emerge.
What’s to Come: Where 5-Hexen-2-One Could Go
Innovation always gravitates toward simple, modifiable molecules, and the chemistry community hasn’t run out of ideas for 5-hexen-2-one. Research angles cover sustainable production, safer process design, and the search for new functions in digital olfaction and synthetic biology. Some see promise in harnessing microbes or plant-based systems to convert renewable feedstocks, reducing reliance on petroleum. Others dig deep into structure–activity relationships, unpicking how slight modifications could yield new flavors, materials, or therapeutic candidates. Regulatory winds keep turning, nudged along by consumer and environmental advocacy, and future growth depends on transparency—not just about hazards, but about innovation, stewardship, and the ongoing dance between risk, reward, and real-world impact.
What is 5-Hexen-2-One used for?
The Real Value Behind 5-Hexen-2-One
5-Hexen-2-one slips under the radar for most people, but it’s a big deal in a few key industries—mostly because of its molecular trickery. On the surface, it’s a clear, sharp-smelling chemical not unlike many you’d run into in classic organic chemistry labs. Start looking into its real job, and you’ll see why chemical manufacturers and researchers keep it close at hand.
Creating Flavors and Fragrances
People instinctively respond to scent and taste, and that’s where 5-hexen-2-one shows up. Used by specialists working with flavors and fragrances, this compound acts as a building block. By bringing out subtle green, fresh, fruity, and vegetable notes, it helps “round out” the smell in items like perfumes, soaps, and even those fresh, grassy vibes you sometimes get in premium personal care products. Its scent profile has earned it a role as a specialty background note in artificial flavors, too—think imitation apple, grape or tomato, the hint that makes foods or drinks taste more “real.”
Chemical Manufacturing: Why Synthesis Matters
Chemists and process engineers often reach for 5-hexen-2-one as an intermediate—a stepping stone, really. Lining up the right building blocks means people can create more complex molecules that end up in things like agricultural products, pharmaceuticals, and specialty coatings. This compound contains both a double bond and a ketone functional group, allowing chemists to perform many types of transformations. For example, with some clever work, it feeds into the synthesis of vitamin E analogs, pharmaceuticals candidates, and antioxidants. These applications help produce ingredients that handle everything from preservation to human health on a large scale.
Special Use: Polymer and Material Science
Makers of new plastics and advanced materials sometimes use 5-hexen-2-one. Its reactive double bond activates polymer chains, supporting the development of tough, flexible materials or adhesives that don’t break down so easily. The result? Longer shelf life and better performance in everyday goods, from electronics to textiles. Not every consumer notices these changes, but the people in labs and on factory floors know how crucial this ingredient can be in the mix.
Concerns and Solutions for Worker Safety
Chemicals like 5-hexen-2-one bring risks. Anyone who has worked in industrial labs knows that proper handling and real training matter. Exposure can irritate skin, eyes, or lungs. The solution sits mostly in strong safety routines—ventilation, gloves, goggles, and full awareness about what you’re working with. Researchers keep detailed hazard sheets and enforce good clean-up practices for spills. The industry does not shrug away from tough regulatory standards: compliance with REACH in Europe, or the US Environmental Protection Agency’s guidance, keeps people and supply chains accountable.
Looking Ahead: Responsible Chemistry
Companies continue to refine how they use and dispose of 5-hexen-2-one. Old habits change as people develop biodegradable derivatives or safer alternatives where possible. As labs move toward greener chemistry, every choice about intermediate chemicals gets reviewed for both effectiveness and safety. So, while 5-hexen-2-one may not make headlines, its uses still reflect a constant push for innovation and responsibility in science-driven businesses. Decades in the lab have taught me: the little details in these specialty chemicals can drive huge changes in the products we all use—if they’re handled with skill and respect.
What are the safety precautions when handling 5-Hexen-2-One?
Knowing What You’re Dealing With
5-Hexen-2-One might fly under the radar in broader conversations, but people in labs and industry know it’s a reactive organic compound you don’t mess around with. Its sharp, almost fruity odor sends a signal: this is not something to sniff directly. Living with chemical safety every day, you learn to respect substances like this, even if you only work with small amounts.
Why Proper PPE Gets Personal
Personal protective equipment isn’t a box-ticking exercise. Anyone who’s watched acetone burn or felt an unexpected splash will remember: gloves, goggles, a fitted lab coat, and closed shoes are your real friends in chemical handling. Nitrile gloves keep this compound off your skin, and safety goggles block the irritation that comes from even the hint of vapor. I always reach for a face shield if the procedure could splash. It’s about not gambling on accidents.
Understanding Ventilation’s Role
Handling 5-Hexen-2-One in an ordinary room puts you at real risk. A lab with a fume hood pulls vapors away fast, keeping them out of your breathing zone. I’ve run afternoon syntheses with this compound, and ventilation made the difference between a clear head and a lingering headache. Open windows just can’t compete with a dedicated exhaust system.
Skin Contact and Spills Happen—React Fast
Even the most careful chemist deals with minor spills. If any lands on your skin, running water for fifteen minutes is not negotiable—no shortcuts here. Remove contaminated clothes before they can trap the liquid against your skin. Training for spills teaches you to clean small splashes with absorbent pads, but if a big container goes down, you leave, raise the alarm, and let the team with respirators handle it.
Safe Storage Makes Life Easier
Experience tells you that containers with a tight seal and a cool, dry place give less trouble over the long run. Label everything, date it, and keep volatile chemicals separate from acids and oxidizers. I’ve lost count of near-misses avoided just by checking labels twice. Don’t store organics above eye level—one slip can put a bottle in your lap.
Fire Hazards: Don’t Give Flames a Chance
5-Hexen-2-One catches fire easily. No open flames, no hotplates running longer than needed, and proper grounding for static. Keep a CO2 or dry chemical fire extinguisher within arm’s reach. Your emergency exit plan isn’t overkill; it’s what you fall back on when seconds count.
Disposal: Don’t Cut Corners
Pouring leftover chemicals down the drain leads to headaches for the whole community. Use the designated waste bottles. If you’re unsure, I learned the hard way that asking your safety officer or environmental team saves a lot of grief later. Regulations demand it, but so does common decency.
Continuous Learning Matters
Safety data sheets aren’t just paperwork. Get familiar with the section on first aid, exposure limits, and incompatibilities. I watch out for changes in recommended practices—science moves, and so do safety protocols. Refresher training with colleagues builds trust, and that can make all the difference during a real emergency.
What is the chemical formula and structure of 5-Hexen-2-One?
What’s Behind the Name
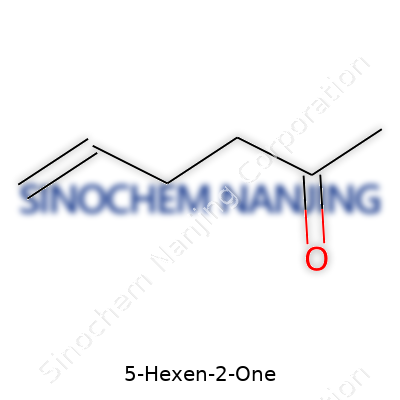
5-Hexen-2-one, often found in labs and flavor research, stands out because of its unique makeup. The chemical formula is C6H10O. This formula points to a six-carbon chain with one double bond and a ketone group.
Breaking Down Its Structure
A closer look at the skeleton of 5-hexen-2-one reveals a straight, six-carbon chain. One end of the chain holds a double bond between the fifth and sixth carbon, not in a ring but in a linear fashion. A ketone group (a carbon double-bonded to an oxygen) attaches to the second carbon. So you have the double bond at the far end, and the ketone nestled closer to the middle.
If you drew it, the structure reads CH2=CH-CH2-CH2-CO-CH3, counting from left to right. That double bond is what gives the “hexen” part of the name, and the ketone on the second carbon gives the “2-one” label.
Learning about this structure isn’t just a chemistry class exercise. It explains a lot about how this molecule acts out in the real world. Those double bonds and carbonyl groups create an environment for chemical reactivity, flavor, and aroma profiles that make this compound valuable.
Why 5-Hexen-2-One Catches Interest
A compound like this makes its mark in several industries. It acts as a starting material for fragrances, flavors, and specialty chemicals. Chemists notice that its structure lets it serve as a building block for more complex molecules. Manufacturers have used derivatives in creating green, fruity, or floral notes in perfumes and flavorings.
5-Hexen-2-one isn’t some exotic lab rarity. It appears naturally in some fruit and vegetable aromas, even if only in trace amounts. This connection ties chemistry and agriculture together in the world of food science and flavor design, with researchers often chasing molecules like these to mimic or modify authentic tastes.
Handling and Safety Concerns
People who’ve worked with this substance know the importance of proper handling. Its liquid form, mild scent, and flammability put safety front and center. Even small molecules like this can cause irritation if spilled or touched (facts supported by industry-standard safety sheets). Ventilation, gloves, and goggles represent more than just protocol—they become habits for anyone working in research or manufacturing settings.
If large-scale production comes into play, engineers often design systems to limit leaks and control vapor for good reason. Factory workers and researchers rely on documented chemical properties—boiling points, vapor pressure, and flash points. These properties influence every stage: from shipment to storage and disposal.
Potential Problems and Fixes
Improper storage or accidental release can pose environmental threats. Small molecules escape containers and can affect air quality. Plant managers and safety experts use tight-sealing storage and real-time monitoring technology. Trained staff who know what an odd odor means frequently catch problems early.
Continued research into safer alternatives can reduce hazards. Organizations building on strong safety habits and real chemical knowledge keep people out of trouble. Regulations developed over years reflect lessons learned through both success and error.
How should 5-Hexen-2-One be stored?
Why Proper Storage Matters
I've spent years working in labs and talking shop with chemists, and one thing everyone learns early is how storage can change everything. 5-Hexen-2-One, a colorless liquid with a sharp odor, isn’t some harmless compound you park on just any shelf. It's flammable, volatile, and doesn't play nice with many common chemicals. Safe storage isn't just a good idea—it's a duty to yourself, your team, and the planet.
The Hazards Before Your Eyes (and Nose)
Anyone who has opened a bottle of 5-Hexen-2-One knows the fumes mean business. These vapors can catch fire at room temperature. Inhaling them can cause dizziness, eye and throat irritation, or worse if you're exposed long enough. The flashpoint sits around 32°C (90°F). The last thing you want is a spark or leaky container turning your workspace into a hazard zone. The National Institute for Occupational Safety and Health (NIOSH) recommends handling it inside a chemical fume hood, never in open air.
How Storage Can Save Lives
For years, I’ve seen what happens when people cut corners: corroded caps, sticky shelves, sticky lungs. Simple changes make the place safer. A cool, dry, well-ventilated storeroom keeps vapors from pooling. Metal cabinets ground away from sunlight, heat, and open flames reduce fire risk. Keeping 5-Hexen-2-One below its flashpoint slows down evaporation and tames its volatility.
What Works in Practice
In practice, the best approach starts with airtight, chemical-resistant containers. Glass bottles with Teflon-lined screw caps prevent leaks and help trap vapors. Every bottle deserves a clear, up-to-date label—you don’t want someone mistaking this for something else. I once saw a tech absent-mindedly grab a similar bottle, and the cleanup took hours.
Secondary containment acts as insurance. Plastic trays or spill-proof bins contain drips and leaks. Once, our lab’s secondary tray caught a slow leak overnight—a simple catch that stopped a terrible mess. Chemical storage cabinets designed for flammable liquids offer extra peace of mind. Metal shelving, not wood, works best.
Separate and Shield
Mixing volatile chemicals never ends well. Segregate 5-Hexen-2-One from acids, oxidizers, and bases—these can trigger violent reactions. The CDC and Material Safety Data Sheets back this up with clear lists of “keep away” substances. Close storage with these reactive chemicals creates a recipe for disaster. A separate cupboard, clearly marked, keeps one mistake from turning into an emergency.
Training, Checks, and Real Responsibility
People, not machines, make the biggest difference. Everyone in the lab or workshop must know what’s inside every bottle, what could go wrong, and how to respond if it does. Regular checks weed out corroded lids or expired chemicals. No one wants a forgotten compound brewing trouble in the shadows. I keep a simple logbook for routine inspections—small habit, big results.
Steps Toward Safer Workplaces
Fire extinguishers and spill kits need to stand ready wherever volatile organics sit. Good habits matter even more—closure after use, zero tolerance for mystery containers, constant reminders about the risks. Sick days and near misses rarely get the headlines, but prevention keeps everyone healthier and happier. Taking storage seriously means showing respect for your crew and for future workers who come after you.
Looking Forward
Progress shows up when storage goes from afterthought to daily practice. Every dry cap and safe shelf proves that careful handling pays off. In my circle, stories about accidents make the rounds as cautionary tales. Better to learn from those stories than to star in one. The peace of mind gained from well-stored 5-Hexen-2-One lasts far beyond the workday.
Is 5-Hexen-2-One hazardous to health or the environment?
The Substance in Everyday Industry
Factories reach for 5-Hexen-2-One due to its chemical structure, making it useful in labs and manufacturing. Flavors and fragrance sectors sometimes use it to synthesize compounds with a fruity aroma. Its molecular design, though, brings certain risks that tend to get downplayed outside chemistry circles.
Impact on Human Health
People who handle 5-Hexen-2-One face real dangers if they ignore proper safeguards. The liquid emits a sharp smell, signaling volatility. Inhaling its vapors irritates airways and the lungs. I remember helping out at a university chemistry demo where a tiny spill sent slick lemon-like fumes across the room — noses burned, a couple of students coughed hard. That moment drove home the sting of even low exposure.
Direct skin contact may leave skin red or cause an itchy rash. Eyes exposed to splashes get red quickly, creating strong discomfort and watery irritation. Longer exposure ramps up the risks, causing headaches, dizziness, and in extreme cases, damage to nerves. The U.S. National Library of Medicine links repeated inhalation to symptoms that mimic mild intoxication and even loss of coordination. Few folks recognize these symptoms as chemical exposure; problems often get brushed off as fatigue.
Regulations urge workers to wear gloves, goggles, and respirators around this substance. OSHA outlines short-term exposure limits, but not every small manufacturer meets those standards. In some small soap and scent workshops, I’ve seen workers forgo masks or rely on open windows instead of extraction fans. Many learn the hard way about the cumulative effects.
Environmental Consequences
Spills don’t just endanger people in the room. 5-Hexen-2-One quickly evaporates and escapes into the air. These vapors break down under sunlight, but not before they can react with nitrogen oxides and other pollutants. Such reactions form ground-level ozone — a key part of urban smog which aggravates asthma and heart issues for nearby residents.
Waterways near manufacturing plants seem especially vulnerable. Should the liquid reach streams or soil, aquatic life bears the brunt. Research highlights its toxicity to fish and small invertebrates, disrupting local food chains and making recovery harder. Even microbes in the soil suffer, as residues slow down natural breakdown processes.
Mitigation and Safer Practices
In my experience, clear labeling and access to real-time air monitors reduce accidental exposure. Proper ventilation, spill kits, and secondary containment for storage should come standard, not just for big chemical plants but for every small shop or academic lab.
Switching to less hazardous substitutes, when possible, gives the best shot at curbing both health and environmental threats. For those who must work with 5-Hexen-2-One, regular training and easy-to-follow emergency plans make all the difference. I’ve watched as teams who drill for spills and cleanups respond calmly and effectively, stopping incidents from spreading further.
Bringing these practices into daily routine shows respect for both workers and the world around us. No one benefits if we ignore or hide risks. Addressing the hazards of 5-Hexen-2-One asks not only for technical solutions, but also a change in attitude and priorities — so that safety shapes every step, from ordering chemicals to discarding waste.
| Names | |
| Preferred IUPAC name | hex-5-en-2-one |
| Other names |
Hex-5-en-2-one 1-Pentenyl methyl ketone Pent-4-enyl methyl ketone Methyl 4-pentenyl ketone |
| Pronunciation | /ˈfaɪˈhɛksən tuː oʊn/ |
| Identifiers | |
| CAS Number | 109-49-9 |
| Beilstein Reference | 1209242 |
| ChEBI | CHEBI:89240 |
| ChEMBL | CHEMBL14368 |
| ChemSpider | 62689 |
| DrugBank | DB03816 |
| EC Number | 211-442-2 |
| Gmelin Reference | 8759 |
| KEGG | C09744 |
| MeSH | D006518 |
| PubChem CID | 12412 |
| RTECS number | HN9625000 |
| UNII | CHX6I4VS4H |
| UN number | UN1224 |
| Properties | |
| Chemical formula | C6H10O |
| Molar mass | 98.143 |
| Appearance | Colorless to yellow liquid |
| Odor | sweet, fruity, green, apple |
| Density | 0.847 g/mL at 25 °C |
| Solubility in water | Slightly soluble |
| log P | 0.97 |
| Vapor pressure | 0.923 mmHg (25 °C) |
| Acidity (pKa) | 13.1 |
| Basicity (pKb) | 5.05 |
| Magnetic susceptibility (χ) | -6.41 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | n20/D 1.439 |
| Viscosity | 0.759 cP (20°C) |
| Dipole moment | 2.79 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 332.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –209.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3326.6 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | H315: Causes skin irritation. H319: Causes serious eye irritation. H335: May cause respiratory irritation. |
| NFPA 704 (fire diamond) | 2-3-2 |
| Flash point | 41 °C |
| Autoignition temperature | 179 °C |
| Explosive limits | Explosive limits of 5-Hexen-2-One: 1.2% - 8.6% |
| Lethal dose or concentration | LD50 (oral, rat): 740 mg/kg |
| LD50 (median dose) | LD50 (median dose) of 5-Hexen-2-One: "426 mg/kg (rat, oral) |
| NIOSH | BV0800000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 5-Hexen-2-One: "OSHA has not established a specific PEL for 5-Hexen-2-One. |
| REL (Recommended) | 1 ppm |
| IDLH (Immediate danger) | 300 ppm |

