5-Difluoromethoxy-2-Mercapto-1H-Benzimidazole: Commentary and Insight
Historical Development
Chemistry has always found ways to tweak molecules for bigger impacts. 5-Difluoromethoxy-2-Mercapto-1H-Benzimidazole grew out of decades spent modifying benzimidazoles, a group that hit the scene in the early twentieth century and became go-to scaffolds in medicinal chemistry. The 1980s saw chemists pushing for greater selectivity and functional diversity, and adding difluoromethoxy groups brought a step-change in bioactivity and stability. Researchers saw early that sulfur atoms in the benzimidazole core delivered valuable biological tricks, boosting interest in this compound’s potential. Interest swelled as the pharmaceutical industry chased new gastroprotective drugs and kinase inhibitors, and bench chemists, including many in my own academic cohort, began building libraries of these compounds. Investment from industry came soon after as patent filings pointed to both process efficiency and new therapeutic applications.
Product Overview
5-Difluoromethoxy-2-Mercapto-1H-Benzimidazole landed in the spotlight thanks to its role as both a biological probe and a building block. On paper its formula suggests a pretty minor substitution — two fluorine atoms and a methoxy bridge attached to the benzimidazole ring, with a thiol group at the second position. That little change changes reactivity remarkably. Drug companies prize it for structure-activity relationship work. Agrochemical labs use it to block enzymes in fungi and worms. Academic teams, like the one I joined in graduate school, often relied on this backbone as a lead structure, adding further groups to push both biological activity and synthesis know-how.
Physical & Chemical Properties
In the solid state this benzimidazole derivative forms off-white crystals that handle moisture decently, with a melting point usually between 167°C and 172°C. The difluoromethoxy side group introduces some lipophilic bite so the material dissolves readily in most polar aprotic solvents — DMSO, DMF, and acetonitrile handle it well — and stays reasonably stable in basic and neutral conditions. Under acidic washes, the thiol group can oxidize and start changing the molecule’s profile. Its relatively low molecular weight compared to other benzimidazoles supports decent cell permeability, a fact often mentioned in early ADMET screens published in the late 2000s. Longevity on the bench and in formulation raises it above less stable sulfur analogs, and the presence of the “CF2O” group means NMR and mass spectrometry both provide clear, unambiguous signals for routine characterization.
Technical Specifications & Labeling
The best suppliers provide purity certificates showing levels above 98%, checked by HPLC and NMR for trace contaminants or analogs. Technical sheets list melting point range, water content (measured by Karl Fischer titration), and heavy metal screening. Labels must follow both GHS safety standards and regional compliance requirements, calling out thiol reactivity, hazard statements for skin and eye irritation, storage advice below 25°C, and the lot-specific batch number. Reliable transport requires packaging under inert gas or sealed vials to beat air-driven oxidation. Labs using the compound in regulated environments demand documentation for traceability and certification regarding animal- or allergen-free supply chains.
Preparation Method
Most routes rely on cyclization between a suitable o-phenylenediamine preloaded with a difluoromethoxy group on the benzene ring and a thiourea source. One-pot methods using acetic acid and sulfurizing reagents like Lawesson’s reagent have trimmed waste and improved yield. Industrial plants tend to use a two-step process for higher purity, first building the intermediate via nucleophilic aromatic substitution and then performing thiolation under mild conditions to protect the sensitive difluoromethoxy moiety. I’ve seen process chemists use H2S and basic aqueous workups for bulk synthesis, then finish by eschewing chromatography with successive recrystallizations — a practical fix for scaling up. The greener approach replaces solvents like dichloromethane with ethanol or even water, borrowing ideas from green chemistry groups who demand lower environmental footprints.
Chemical Reactions & Modifications
The compound’s two main handles — the mercapto (-SH) group and the difluoromethoxy side chain — open doors to targeted chemistry. S-alkylation swaps the hydrogen on the thiol for aryl, alkyl, or even heterocyclic bulks, enhancing cell specificity or shifting metabolic fate. Some groups use the sulfur atom for gold or palladium-catalyzed couplings, pushing into materials chemistry beyond pharma. The difluoromethoxy wing brings resistance to oxidative metabolism, which helps in lead optimization for slow clearance drugs. Under UV or strong oxidants, the ring system resists breakdown, while aggressive nucleophiles chip at the thiol. Cross-couplings on the aromatic ring sometimes challenge even seasoned synthetic chemists, since the electron-withdrawing difluoromethoxy can kill reactivity at targeted positions — a real puzzle in late-stage diversification. Much of my lab time was spent troubleshooting these transformations and finding new ways to tap that unique reactivity.
Synonyms & Product Names
Aside from its full chemical name, the compound appears in chemical catalogs under names like DFMO-MBI, Difluoromethoxy-Mercaptobenzimidazole, and at times as 2-(Difluoromethoxy)-5-mercaptobenzimidazole. Regulatory documents also list it by CAS numbers specific to verified batch synthesis. Research groups may give it “MBI-DF2” shorthand or apply project code numbers, sometimes making literature searches difficult. Recognizing all these synonyms ensures nobody misses important safety data or cross-references in patent filings — a habit drilled into me while reviewing literature for grant applications.
Safety & Operational Standards
Lab workers handling this molecule need gloves, lab coats, and eye protection at minimum. The free thiol reacts with oxidizing agents and can trigger allergic responses in sensitive individuals, which shows up in MSDS documentation, though acute hazard ranks below harsher benzimidazoles. Chronic exposure data remain thin, so batch manufacturing facilities rely on closed systems and local exhaust. Containment and air monitoring matter most during weighing and transfer; users log spills and clean up with standard spill kits. Safety audits ask about the presence of peroxides — thiols never age gracefully — so disposal involves deactivation in alkaline hypochlorite, not straight landfill. Each drum or vial carries hazard pictograms flagged by transport code UN numbers and GHS statements, a legal requirement following international shipments.
Application Area
Work on this molecule started with bioactivity screens against bacteria and fungi, but recent years have seen applications sprawl. Pharmaceutical firms consider it as a lead compound for proton pump inhibition, anticancer hits, and molecular probes for kinase studies. Veterinary drug candidates are in the mix too. Crop protection teams review it for antifungal seed treatment because the difluoromethoxy group delays enzymatic breakdown in the soil. Material scientists eye it as a ligand for metal complex formation, dabbling in catalysis and organometallic frameworks. Academic labs still use it to train students in making derivatives and exploring benzimidazole-related SAR. I’ve sat in research meetings where every team seemed to find a new angle, proving the molecule’s wide appeal.
Research & Development
The playground for discovery work has not shrunk, either. Labs use 5-Difluoromethoxy-2-Mercapto-1H-Benzimidazole as a platform for drug optimization, targeting enzyme inhibition, anti-inflammatory activity, and even neuroprotection. Collaborations with biologists push the molecule into genetic and cell screening for understanding disease pathways. Medicinal chemists appreciate the robust stability and amenability to molecular modifications, which permits rapid analog synthesis for lead selection. Scale-up teams design cleaner, solvent-minimal syntheses as green standards get stricter. Industry partners invest in computational studies to anticipate metabolic liabilities before a full panel of in vivo tests. By pushing both AI-driven and hands-on experimental work, research teams bridge the old-school bench approach with new machine learning strategies, extracting the most information from each derivative. This hybrid workflow, which I have witnessed in several cross-industry projects, boosts efficiency and quickens the feedback loop between synthesis and biological testing.
Toxicity Research
Much of the current safety knowledge stems from periodical testing in zebrafish, rodent, and cell cultures. Acute oral exposures in rats suggest moderate toxicity with LD50 values higher than sulfur-rich analogs. Cell studies show low mutagenic potential, yet knowledge gaps remain on chronic outcomes and reproductive effects. Some metabolites formed under liver microsome reactions call for caution if considering long-term therapeutic dosage. For environmental profiles, the difluoromethoxy part drags out soil persistence, raising flags for run-off concerns in agrochemicals. Industry standards push for pre-market environmental fate and aquatic toxicity studies. Researchers in toxicology continue drilling into structure-toxicity relationships, often deploying CRISPR-edited cell lines to get finer details on off-target effects.
Future Prospects
Looking ahead, both the pharmaceutical industry and agrochemical developers treat 5-Difluoromethoxy-2-Mercapto-1H-Benzimidazole as fertile ground for innovation. Chemical engineers design smarter workflows for larger batch synthesis with less waste. Biologists pursue selectivity in disease treatments, hunting for targeted delivery and resistance-busting derivatives. Drug discovery can evolve with smarter libraries drawing from this core, aiming for personalized therapies. Environmental scientists investigate breakdown pathways and design antidotes for accidental overdose or spill. The compound’s unique mix of stability, tunable reactivity, and biological compatibility provides a launchpad for next-generation applications in medicine, agriculture, and beyond. The pace of research ensures this benzimidazole will stay relevant, as new discoveries build on the foundation laid by decades of practical lab work and cross-discipline collaboration.
What is the chemical structure of 5-Difluoromethoxy-2-Mercapto-1H-Benzimidazole?
Diving into the Chemical Design
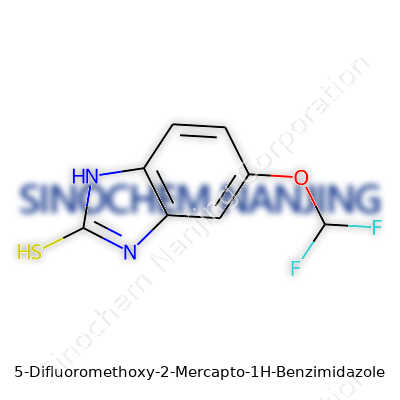
5-Difluoromethoxy-2-Mercapto-1H-Benzimidazole sounds technical, but its chemical structure spells out a clear story. Sitting on a benzimidazole core, the molecule features two standout groups: a difluoromethoxy group and a mercapto group. The benzimidazole core brings the familiar fused ring system seen in bioactive molecules like omeprazole or albendazole. With the difluoromethoxy group at the 5-position, two fluorine atoms hitch a ride on a methoxy group, pushing both electron density and size around. Sitting at position 2, the mercapto group — a sulfur carrying a hydrogen (–SH) — adds reactivity and opens doors for binding in biological systems.
What This Structure Does for Chemists
The layout of these substituents shapes both the molecule’s character and promise. Fluorine introduces electronegativity, changing how this molecule responds in a cell or test tube. I’ve seen plenty of times in a lab how simply swapping a hydrogen for a fluorine forces new physical and biological behavior. The difluoromethoxy group makes the compound tougher, slashes metabolic breakdown, and helps it last longer in the body. These kinds of changes can take a molecule from “interesting” to drug-worthy.
The mercapto group pulls in biochemists and synthetic chemists. That –SH handle links to metals, reacts with proteins, or tethers to a solid support in research. In my own work, these sulfur groups show up in enzyme inhibitors and chelators—not just on paper but in tubes and flasks. They bring both stickiness and reactivity, helping chemists build new scaffolds or probe biology in clever ways.
Importance in Medicine and Research
This family of benzimidazole compounds sits at the crossroads of medicinal chemistry. Many drugs succeed because medicinal chemists fine-tune the positions and choices of elements on a scaffold exactly like this one. Adding a difluoromethoxy presents patentable new properties—modified behavior in metabolic pathways, different targeting outcomes, or even shifting the balance between safety and potency.
In research circles, chemists chase these subtle changes. A sulfur here or a fluorine there sometimes draws the line between a new treatment for a neglected disease and another candidate left behind on the shelf. Actual cases abound: benzimidazoles fight parasites, block cancer cell growth, or control acid secretion in the stomach because someone explored just this kind of atomic switch. Documented studies in the past five years have reported modifications at similar positions on the benzimidazole ring, leading to improved antifungal, antiviral, and anti-inflammatory effects.
Challenges and Paths Forward
Working with sulfur, especially mercapto groups, isn’t straightforward. They oxidize and react with ease, creating hurdles for storage and purity—issues I know well from time spent coaxing unstable intermediates to their next destination. Solutions include using antioxidants or protecting groups during synthesis, though these strategies need time, money, and patience.
Fluorination also comes with environmental and safety baggage. Tuning reactions for precise fluorine placement eats up resources. Industry is pushing for greener, less hazardous fluorination techniques, like electrochemical or photochemical methods, to meet both regulatory and sustainability goals.
Rising demand for fresh chemical space—new patents, new functions, new treatments—means the world will keep tinkering with classic cores like benzimidazole. The structure of 5-difluoromethoxy-2-mercapto-1H-benzimidazole stands as both a technical puzzle and an open invitation: solve its quirks, and you might unlock safer, more reliable molecules for patients, researchers, and manufacturers alike.
What are the main applications of 5-Difluoromethoxy-2-Mercapto-1H-Benzimidazole?
Pharmaceutical Pathways and Drug Development
Fluorinated organic compounds hold a steady spot in medicinal chemistry, and 5-Difluoromethoxy-2-Mercapto-1H-Benzimidazole shows just how much influence two tiny fluorine atoms can have on drug design. This benzimidazole derivative earns attention in research labs for its scaffold, which shows strong promise in the search for new drugs. Benzimidazoles, in general, take part in treatments for infections, cancers, and inflammatory diseases. The integration of a difluoromethoxy group at the 5-position can raise chemical stability and tweak how the molecule interacts with its environment, giving it an edge during the screening phases for new therapeutics. A close friend who works at a biotech startup once told me that modifying small parts of a molecule like this can help researchers tune activity and lower side effects—a constant struggle in making better medicine.
Researchers investigating 5-Difluoromethoxy-2-Mercapto-1H-Benzimidazole often focus on its ability to serve as a core for kinase inhibitors or anti-viral candidates. Since resistance to current drugs remains a large problem in cancer or viral infections, adding a novel benzimidazole derivative to the mix gives hope for treatment options that break through the typical resistance patterns. Over the past five years, the scientific publishing world has seen dozens of articles mentioning new benzimidazole analogues with various fluorinated groups, pointing to an active and expanding field.
Crop Protection and Agrochemical Development
The agriculture industry relies on molecules that can protect crops from fungal diseases or pests without building up harmful residues in fields. Fluorinated benzimidazoles attract attention here, as they often show strong fungicidal properties. Large agrochemical producers have synthesized and tested dozens of analogues based on this skeleton, aiming for products that prevent crop loss and help farmers manage resistance in the field.
Adding a difluoromethoxy group can help boost overall potency against fungal targets. This chemical tweak also improves how the compound sticks around on plant surfaces, leading to longer-lasting protection. Friends who run small farms have pointed out that they depend on innovations from industry scientists who keep tweaking these molecules so that products work longer, reduce costs, and avoid harming pollinators. Regulatory agencies also require regular updates about toxicity and breakdown products, so the push for safer, more effective crop protection never lets up.
Material Science and Specialty Chemicals
Beyond medicines and pesticides, this molecule’s unique mix of functional groups brings new possibilities in material science. Chemists have explored using benzimidazol-thiol hybrids as building blocks for electronic materials, especially in projects requiring stable compounds that can conduct electricity or interact with metals. The sulfur atom allows binding to certain substrates or nanoparticles, while the difluoromethoxy group resists chemical degradation. These features matter when designing sensors, coatings, or catalysts. At chemistry conferences, I’ve seen posters describing new polymers made from benzimidazole derivatives, revealing a research area where small changes can lead to products with real-world uses, from batteries to protective films.
Challenges and Responsible Innovation
Work with specialized benzimidazoles calls for diligence, not just in the lab but also in how companies manage safety, environmental impact, and intellectual property. Concerns about toxicity and persistence often come up, especially as governments increase scrutiny on chemicals entering food chains or water systems. Manufacturers and research groups share responsibility here. They need strong data to back both the usefulness and the safety of these compounds.
Continuous conversation between chemists, regulators, and the people who use these chemicals daily helps target research efforts. Applying clean chemistry practices, investing in alternative testing methods, and supporting data sharing give these molecules the best shot at making a positive mark, whether in medicine, crops, or new materials.
What is the purity specification for 5-Difluoromethoxy-2-Mercapto-1H-Benzimidazole?
The Real World of Chemical Purity
Purity standards for active pharmaceutical ingredients and fine chemicals aren't just any detail; they determine everything from safety to effectiveness. 5-Difluoromethoxy-2-Mercapto-1H-Benzimidazole, a compound with potential applications in research labs and specialty industries, comes under scrutiny for this very reason. Purity levels for such compounds often start at 98% and go higher for testing or production that demands extra rigor.
Why Purity Matters—Not Just a Number on Paper
People who work with chemicals understand that purity isn’t just a technicality. Impurities even in small traces can spark side reactions during synthesis, damage analytical equipment, or even cause harm in final formulations. Some labs use this compound in studies exploring enzyme inhibition or as intermediates for downstream products. At that stage, even trace amounts of unknowns can unravel months of work.
Standard laboratories stick to trusted suppliers who provide full certificates of analysis. A standard benchmark for purity usually sits at 98%. Analytical techniques like HPLC, NMR, or mass spectrometry verify this number, and suppliers get asked about residual solvents and moisture levels too. I've seen a project fall apart because a batch barely reached 95%, and it never performed reliably in a key reaction step.
Guardrails for Researchers and Manufacturers
Regulations in pharmaceutical research and manufacturing lean hard on traceability. The U.S. Pharmacopeia, among other watchdogs, asks for rigorous documentation and impurity profiling. Without strict purity requirements, end-users shoulder all the risk. It isn’t all about quality control labs doing their jobs—good results also come from rigid procurement policies. Consistent, high-purity raw materials set up the rest of the process for reliability.
Chemical suppliers use multiple purification and testing steps before anything leaves the warehouse. Infrared spectroscopy confirms compound identity, while chromatography sorts out impurities from the main lot. Water content and heavy metals get reported down to fractions of a percentage point on certificates. Some buyers seek 99% or higher for clinical or regulatory reasons, but 98% often balances performance and cost in real-world settings.
Challenges and Solutions
One roadblock is that not all batches survive transport or storage without picking up moisture or breakdown products. I’ve opened containers to find the slight odor or color shift that signals contamination; in such cases, retesting becomes the only way forward. Refrigeration and vacuum-sealed packaging help, but busy research cycles sometimes mean stocks linger longer than ideal.
For users who need peace of mind, working with suppliers who offer rapid retesting and open data helps. Scalability can also muddy the waters—small lab-scale batches may show great numbers, but ton-scale manufacturing brings on fresh hurdles with process residues and cross-contamination. Setting up protocols that demand fresh analysis for every lot minimizes risk.
Letting Science and Safety Drive Decisions
End-users depend on a transparent supply chain, robust analytical reporting, and honest communication when something’s amiss. That’s not just good science. It’s respect for the people and products downstream of every purchased gram. When it comes to 5-Difluoromethoxy-2-Mercapto-1H-Benzimidazole, a clear focus on purity creates value money can’t replace—reproducibility, safety, and trust in every flask.
How should 5-Difluoromethoxy-2-Mercapto-1H-Benzimidazole be stored?
The Importance of Proper Storage for Lab Chemicals
Chemistry labs fill up fast with strange names and stranger bottles. 5-Difluoromethoxy-2-Mercapto-1H-Benzimidazole might sound like a handful, but at its core, it’s a substance like many others: helpful for research, but dangerous if you treat it carelessly. Everyone knows stories where a forgotten vial or poorly capped bottle caused a mess—or worse, a health emergency. Safety isn’t only about rules—it's about avoiding trouble long before it rears its head.
Why This Chemical Demands Respect
Some compounds break down with moisture. Others react with air or light. This one falls under those that want a dry, cool, and dark space. Most benzimidazole derivatives handle poorly the heat and humidity common in crowded lab shelves. Inconsistent temperatures or stray sunlight can wreck your sample, making experiments useless or even dangerous.
I spent long nights helping a team run stability tests. Labeling the samples always meant more than writing neat numbers on tape. It meant thinking about whether the next person would know how to handle what they were holding. Lab experience taught me: the real work begins when the shipment arrives, not just during measurement or analysis.
Safe Storage Method: Step by Step
Keep it sealed. Air brings moisture, and even slow leaks can make a bottle lose purity. Screw the lid back on tight after every scoop. For long delays between uses, a layer of inert gas—argon or nitrogen—can push out that moist air.
Find it a cool home. Some research groups vote for the fridge, but a chemical storage cabinet out of direct sunlight often does the trick. Put it away high up and out of reach from heat sources like radiators or sunny windows. Temperatures under 25°C (77°F) tend to preserve sensitive benzimidazoles—the lower, the better, as long as the place isn’t too damp.
Avoid the water. Never open the bottle near sinks or where splashes can land. Benches should stay dry, and hands even drier. A little care keeps spoilage away and prevents ruined experiments.
Label everything. Handwriting fades. Old tape peels, especially from frosty fridge air. Mark bottles with chemical name, date opened, and your name, so there’s no guessing weeks or months later. Stick a reminder with best-by dates if you know them. Store with a group of similar stability—don’t mix with acids or bases, and avoid flammable bins if you can.
The Risks of Getting Storage Wrong
Impurities sneak in from sloppy habits. A little water can start decomposition, leaving you with a darkened, useless sludge. Mishandling sometimes means inhaling or touching compounds that should stay inside glass. Lab accidents aren’t just stories for safety meetings; they come out of skipped steps and misplaced trust in shortcuts.
Long-term exposure risks show up in health records: chemical dermatitis, allergies, sometimes more. NIOSH and OSHA both lay out what to avoid. Factories and labs both see what can go wrong when safety culture slips.
Smarter Solutions, Safer Labs
Smart sensors help; digital temperature and humidity loggers catch problems early. Routine audits—peer walk-throughs, not just paperwork—spot expired or leaking items. Training isn’t just for new hires; refreshers matter as chemicals or staff change.
Investing time up front—the careful storage, clean labels, dry bench—is a lot cheaper than losing a whole batch of research, or worse, risking health. Years in the lab taught me the only real shortcut is doing it right, right away.
Is there any safety or handling information available for 5-Difluoromethoxy-2-Mercapto-1H-Benzimidazole?
The Importance of Respecting Lab Chemicals
Every time I hear about a new or obscure compound like 5-Difluoromethoxy-2-Mercapto-1H-Benzimidazole, my mind goes straight to safety basics. Lab work can tempt anyone to get comfortable, but complex molecules deserve real respect—especially ones that don’t have a prominent safety history in public databases. When chemists run into compounds without a thorough hazard profile, it makes sense to treat them with the kind of caution reserved for the more notorious benzotriazoles, benzimidazoles, and mercapto-substituted aromatics.
Structure Tells a Story
The structure of 5-Difluoromethoxy-2-Mercapto-1H-Benzimidazole alone raises eyebrows for any synthetic chemist. A benzimidazole core often means biological activity—sometimes the kind that’s helpful for medicine, other times the kind that interferes with hormones or cell processes in unexpected ways. The difluoromethoxy group can introduce volatility or increase potential for bioaccumulation. Old hands know from sulfur-containing groups like mercapto, you often get unpredictable reactions with oxidizing agents and the unmistakable smell—sometimes followed by skin sensitivity.
Known Risks with Similar Chemicals
There isn't a lot of public toxicological information directly on 5-Difluoromethoxy-2-Mercapto-1H-Benzimidazole, but we can learn from its relatives. Many benzimidazole derivatives develop problems with acute toxicity, organ irritation, or allergic response. The mercapto group in particular relates to thiols, which have a reputation for being both sensitizing and irritating. Anyone with experience handling thiol reagents remembers the burning sensation and skin rashes after even brief, unprotected exposure. The difluoromethoxy side likely introduces volatility and possibly inhalation risk. There’s a persistent lesson in synthesis labs—treat anything with new fluorine groups with an extra layer of caution; fluorinated organics often have poorly characterized hazards.
Lab Handling—Practical Steps Truly Matter
A well-fitted lab coat, splash goggles, and heavy-duty gloves become non-negotiable. Ventilation isn't just a bonus—fume hoods cut inhalation risks before you even notice vapors wafting up. Using closed systems limits direct contact. Nearly every researcher who takes shortcuts discovers trouble the hard way. A friend once underestimated a new benzimidazole derivative and wound up with a nasty dermatitis that took weeks to fade.
Personal knowledge blends with evidence from safety data sheets on similar chemicals: avoid skin and eye contact as much as possible; don’t let this sort of compound sit open on a benchtop. Expect it to be harmful if swallowed or absorbed, and don’t ignore the power of a good mask in case powders or aerosols form during transfers.
Where We Go from Here
In-house safety teams and materials scientists need to push for full hazard characterizations on specialty chemicals like this. Getting a professional toxicologist involved before scale-up saves a lot of trouble. Most companies rely on GHS labeling and the reaction profiles of similar molecules—sometimes that isn’t enough. Research with new benzimidazole derivatives calls for supporting safety data sheets, targeted risk assessments, and, where possible, in vitro studies on toxicity.
It never hurts to keep a fresh attitude about chemical risk. The unknowns with compounds like 5-Difluoromethoxy-2-Mercapto-1H-Benzimidazole demand that kind of rigor. Scientists with experience under their belts pass this mindset on to those just entering the lab. “Better safe than sorry” isn’t just a catchphrase; it really means something in a real-world research environment.
| Names | |
| Preferred IUPAC name | 5-(Difluoromethoxy)-2-sulfanyl-1H-benzimidazole |
| Other names |
5-(Difluoromethoxy)-2-mercaptobenzimidazole 5-(Difluoromethoxy)-2-sulfanyl-1H-benzimidazole 2-Mercapto-5-(difluoromethoxy)benzimidazole |
| Pronunciation | /faɪdɪˌflʊəroʊˈmɛθɒksi tuː mərˈkæptoʊ wʌn eɪtʃ bɛnzˈɪmɪdæzəl/ |
| Identifiers | |
| CAS Number | 1218937-66-4 |
| Beilstein Reference | 3728832 |
| ChEBI | CHEBI:61090 |
| ChEMBL | CHEMBL406059 |
| ChemSpider | 120606 |
| DrugBank | DB13520 |
| ECHA InfoCard | 05f081ea-eb73-484c-9aae-42ee811b0dff |
| EC Number | NA |
| Gmelin Reference | Gmelin 831978 |
| KEGG | C14894 |
| MeSH | D000072646 |
| PubChem CID | 159809 |
| RTECS number | KW6460000 |
| UNII | BO1P10KX8J |
| UN number | UN3276 |
| Properties | |
| Chemical formula | C8H6F2N2OS |
| Molar mass | 258.24 g/mol |
| Appearance | Light yellow crystalline powder |
| Density | 1.62 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 1.98 |
| Vapor pressure | 1.61E-06 hPa at 25°C |
| Acidity (pKa) | pKa = 7.42 |
| Basicity (pKb) | 13.06 |
| Magnetic susceptibility (χ) | -0.0005407 |
| Refractive index (nD) | 1.622 |
| Dipole moment | 4.02 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 253.6 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | A02BC05 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P264, P272, P273, P280, P302+P352, P305+P351+P338, P362+P364, P501 |
| Flash point | > 173.6°C |
| Lethal dose or concentration | LD50 oral rat 1580 mg/kg |
| LD50 (median dose) | LD50 (median dose): "1797 mg/kg (rat, oral) |
| NIOSH | Not listed |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 5-Difluoromethoxy-2-Mercapto-1H-Benzimidazole is not specifically established by OSHA. |
| REL (Recommended) | 0.1 mg/m3 |
| Related compounds | |
| Related compounds |
5-Methoxy-2-mercapto-1H-benzimidazole 5-Trifluoromethoxy-2-mercapto-1H-benzimidazole 2-Mercapto-1H-benzimidazole 5-Chloro-2-mercapto-1H-benzimidazole 5-Difluoromethoxy-1H-benzimidazole 5-Difluoromethoxy-2-methyl-1H-benzimidazole |

