Digging Into 5-Bromo-2-(2-Methyl-2H-Tetrazol-5-Yl)-Pyridine: From Lab Bench to Future Possibilities
Historical Development
Chemistry often mirrors the changing demands of society, and the story of 5-Bromo-2-(2-methyl-2H-tetrazol-5-yl)-pyridine runs through both meticulous academic labs and industrial research programs. The drive behind researching halogenated heterocyclic compounds took off with the race to develop more selective pharmaceutical intermediates and innovative crop protection tools. Through the late 20th century and into the early 2000s, scientists trawled through all kinds of aromatic systems, searching for biological sweet spots. The brominated pyridines, with their potential for cross-coupling and fine-tuning, quickly picked up momentum. The tetrazole ring, known for mimicking carboxyl groups in bioisosteric drug design, found a niche in cardiovascular, antidiabetic, and anti-inflammatory research. Combining bromine and tetrazole on a single pyridine backbone didn’t come out of nowhere; it took years of trial and error, hundreds of failed syntheses, and quite a few clever analytical chemists with patience for NMR puzzles.
Product Overview
Many chemists meet this compound at junctures focused on molecule building. 5-Bromo-2-(2-methyl-2H-tetrazol-5-yl)-pyridine serves as a valuable intermediate. Adding a bromine atom gives plenty of room for further modifications, from Suzuki-Miyaura couplings to nucleophilic substitutions that transform this building block into richer, more biologically potent chemistries. The tetrazole moiety resists hydrolysis, stands up in a wide variety of pH conditions, and offers reliable mimicry for functional groups found in many enzyme substrates. Companies end up stocking this material because it serves as a springboard for more complex molecules—whether for the medicinal chemist looking for a new lead compound, or the agrochemical developer searching for the next breakthrough fungicide.
Physical & Chemical Properties
On the bench, this compound stands out for its off-white to light beige crystalline appearance. The melting point hovers between 115°C to 122°C, offering a solid under most ambient conditions but never so stubborn as to resist basic purification steps. Solubility features deserve respect: good enough in common organic solvents like DMSO, DMF, and acetonitrile, but predictably limited in pure water. You’d expect its stability under standard storage, though the bromine can raise eyebrows for sensitive reactions. Analytical chemists appreciate a clear NMR spectrum (aromatic shifts easy to spot), and mass spectrometry confirms a molecular ion at 226 g/mol. The compound’s logP value lands on the low side for a halopyridine, mainly due to the polar tetrazole, which changes the way it interacts with membranes or chromatography columns.
Technical Specifications & Labeling
Chemical suppliers often provide this material at a minimum purity of 97%, measured by HPLC or GC analysis, along with strict residual solvent tests. Labels on commercial bottles list key identifiers such as CAS number, molecular formula (C7H6BrN5), batch code, and suggested storage guidelines (cool, dry place, tightly capped). Material safety data sheets flag combustion hazards linked to organic bromides and recommend Class D fire extinguishers—an important detail for smaller research outfits with limited safety infrastructure. Each lot ships with both CoA and, where necessary, RoHS or REACH statements for proof of responsible sourcing and compliance with European regulations.
Preparation Method
Lab prep involves a two-phase approach that starts with 2-bromo-5-chloropyridine or occasionally a pre-halogenated pyridine. The tetrazole segment usually gets introduced using a [3+2] cycloaddition with sodium azide and a suitable methyl nitrile precursor, often catalyzed by a copper or zinc salt in polar aprotic solvent. After cyclization and neutralization, the product comes out through either crystallization or liquid-liquid extraction, followed by silica gel chromatography for purification. Avoiding metallic contaminants is a big deal, especially for pharma-grade intermediates. The entire process stands out for moderate efficiency, rarely dipping below 60% yield if the reagents behave.
Chemical Reactions & Modifications
In the hands of an experienced chemist, the utility of this compound comes alive. The bromine atom doesn’t just sit there—it opens the door for palladium-catalyzed cross-coupling, making it possible to snap on biaryls, alkynes, or even amines. The tetrazole ring tolerates robust reaction conditions: you can alkylate, acylate, or even try N-oxide formation without much risk. Suzuki and Buchwald-Hartwig reactions run smoothly, helping medicinal teams rapidly sketch out structure-activity relationships. Post-functionalization of the tetrazole ring sometimes brings surprises—some modifications can tune solubility or tweak distribution coefficients, giving biochemists or formulation scientists a sharper tool for later studies.
Synonyms & Product Names
This compound hides under a few different names in catalogs worldwide. Chemical databases point to synonyms like 5-Bromo-2-(2-methyl-2H-tetrazol-5-yl)pyridine, 2-(2-Methyl-2H-tetrazol-5-yl)-5-bromopyridine, and a range of trivial names in different regions. In European and Asian markets, reference catalogs might label it as the “Bromo-MethylTetrazole Pyridine Intermediate.” While these synonyms aim to clear up confusion, they sometimes cause trouble for warehouse searches and ordering systems. Grabbing the correct CAS number usually solves the problem.
Safety & Operational Standards
Handling brings its own set of rules. While the brominated pyridine base isn't the most toxic in its class, it still calls for routine gloves and splash goggles. Open bottles only in a fume hood—volatile organic bromides don’t belong in the general air supply. Emergency procedures boil down to standard chemical spill kits and plenty of ventilation. Waste disposal goes through a halogenated organic stream, handed off to professional incineration. Chemists take safety training seriously, bringing new interns up to speed through routine drills—attention to detail on this front makes sure accidents stay rare.
Application Area
Plenty of actionable uses come attached to this intermediate. Pharmaceutical chemists keep it close at hand for rapid analog synthesis in lead optimization. The tetrazole mimics carboxylic acids, helping teams develop high-affinity angiotensin antagonists and sitting behind blockbuster drugs for hypertension and congestive heart failure. Agrochemical companies use this scaffold for fungicides and herbicides, especially tackling resistance in cereal crops. Material scientists have explored spin-off applications in developing new electronic materials—the electron-rich pyridine and the energetic tetrazole show up in some specialty polymers. Every lab I worked in kept a stash for “unplanned inspiration”—the kind of compound that doesn’t always make front-page news, but solves tough synthetic problems in big programs.
Research & Development
Innovation means always re-examining the basics. Chemical R&D teams summarize progress in two main lanes: synthetic methodology and biological activity profiling. The drive for greener chemistry shows up in approaches aimed at reducing heavy metal catalysis or switching to water-based solvents. Some labs even experiment with enzymatic conversion methods, looking to shave off hazardous side-products. On the screening front, new derivatives get plugged through enzyme binding assays, cell line panels, and environmental fate models. Each cycle brings stronger SAR (structure-activity relationship) maps and tighter control over selectivity or toxicity profiles. Collaboration stands out—a single intermediate often serves a dozen teams, with regular meetings to debate which modifications matter most for project objectives.
Toxicity Research
Risk assessment keeps pace with each new use. Dose-range finding studies in rodents show low acute toxicity at single exposures, but attention turns to chronic studies, especially with the tetrazole’s metabolic pathways. Researchers monitor for methylated metabolites, worried that certain transformations in the liver could hint at off-target effects. Cell profiling screens rule out mutagenic risk before compounds advance. The bromine atom sometimes raises regulatory questions, especially around disposal or environmental accumulation, so both research and compliance departments track ecotoxicity markers in run-off studies. The push for green chemistry translates to less persistent byproducts and careful documentation on every lot.
Future Prospects
The world won’t run out of uses for halogenated pyridines or tetrazole-containing pharmaceutics anytime soon. Trends in precision medicine and sustainable crop protection give intermediates like this more weight than some realize. AI-driven drug design, mixing traditional organic chemistry with machine learning predictions, leans on combinatorial intermediates to feed their massive data engines. Advances in catalysis could lower the cost and speed up access, cutting out steps or using more robust, earth-friendly reagents. Environmental, health, and safety teams remain vigilant, working with synthetic chemists to keep one eye on regulatory shifts and societal expectations. For researchers, the lesson sticks: keep building, keep questioning, and stay ready for the next surprise discovery.
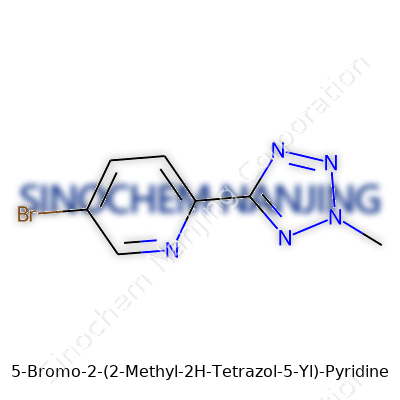
What is the chemical structure of 5-Bromo-2-(2-Methyl-2H-Tetrazol-5-Yl)-Pyridine?
A Walk Through the Chemical Name
Reading a chemical like “5-Bromo-2-(2-Methyl-2H-Tetrazol-5-Yl)-Pyridine” often feels like staring at alphabet soup for the first time. It isn’t just a jumble of letters. Each word carries a specific meaning that tells the story of atoms, rings, and substituents. Here we see a pyridine core kicking things off—a flat, six-sided ring with one nitrogen atom instead of carbon. Chemists treat this ring like a familiar friend; it pops up in medicines and metal catalysts alike.
Add a bromine atom to the fifth spot on that ring, and suddenly you’ve got changes in the molecule’s reactivity. Bromine is a heavy element, and that comes with real effects. Molecules with bromine usually grab attention in labs since brominated compounds can interact in unique ways compared to their non-brominated cousins. In pharmaceuticals and agrochemicals, swapping in bromine sometimes means a world of difference for how the substance’s behavior shakes out.
Tetrazole: Nitrogen’s Place in the Spotlight
Stuck off the second position of the pyridine sits what chemists call a 2-methyl-2H-tetrazol-5-yl group. This isn’t something you bump into at the grocery store. The tetrazole ring, made up almost entirely of nitrogen atoms, is a structural wildcard. It acts a bit like a Swiss Army knife in medicinal chemistry. Swapping a tetrazole group into a molecule can boost its ability to interact with certain enzymes and receptors—so drug designers use these structures to dial in biological effects.
With a methyl group riding shotgun on the tetrazole, this piece of the molecule brings a touch of bulk and changes the landscape for how it interacts with other chemicals or living tissue. That methyl group can help fine-tune the molecule’s ability to dissolve in fats and water, and even influence how long it sticks around inside living bodies.
Peering Inside the Molecular Blueprint
So what does this all look like in a structural sense? Drawing it out, you start with the aromatic pyridine ring. Bromine clings to the ring’s fifth carbon. On the opposite side, the tetrazole group reaches out from the second carbon, looking almost like a nitrogen-packed star. Add the methyl tail to the tetrazole, and the molecule starts to look crowded in just the right places for chemistry or biology to care.
Careers in chemistry are built around understanding how even tiny tweaks—like switching in a bromine atom or adding a methyl group—reshape a molecule’s character. This is how scientists uncover new treatments and cutting-edge materials. To do this work well, grounding every claim in tested evidence and clear, logical thinking stays important. Years of practical lab experience show that unexpected changes pop up once molecules reach the real world. Researchers tend to run thorough tests and weigh new findings against published data to stay firmly on track, from the initial sketch to the final analysis.
Using Chemical Structure to Open Doors
A structure like 5-Bromo-2-(2-Methyl-2H-Tetrazol-5-Yl)-Pyridine is more than an academic puzzle. It represents a set of choices rooted in deep knowledge about atoms and their neighborhoods. Chemical scientists, whether designing medicines or new materials, harness tools like X-ray crystallography, NMR, and computational modeling to confirm that molecules form just as intended. Getting the structure right opens doors: patent protection, regulatory approval, and ultimately, products that shape the way we live and treat illness.
Every chemist remembers the day a molecule danced from a chalkboard sketch to a purified solid flake in the palm of their hand. These structures, packed with possibility, drive progress in medicine and industry. Stories like this one remind us that every atom counts.
What is the primary use or application of 5-Bromo-2-(2-Methyl-2H-Tetrazol-5-Yl)-Pyridine?
Discovery Fuels Medical Progress
Step inside any modern pharmaceutical lab and you’ll find shelves lined with countless small bottles. If you look closer, you might come across a label reading “5-Bromo-2-(2-Methyl-2H-Tetrazol-5-Yl)-Pyridine." That's a mouthful, but buried in those letters and numbers lies the hope for new, more effective medicines. Chemists use this compound as a building block, a chemical foundation for researching and creating active pharmaceutical ingredients (APIs) designed to tackle everything from infections to cancer.
Why Scientists Focus on This Compound
Drug discovery often begins with a core chemical structure that researchers can modify, testing new versions for treatable effects. This molecule, with its bromine and tetrazole groups, is full of potential. The bromine offers a site for further tweaks by introducing or swapping parts of the molecule, and the tetrazole group can imitate the properties of natural biological molecules, which matters a lot for how the body processes drugs.
Researchers love to talk about “medicinal chemistry.” It sounds technical, but really it means looking for creative ways to make families of new drug candidates that work better, last longer, and have fewer side effects. This compound fits right into that search. Its chemical shape lines up with targets in the human body, making it a top choice when designing molecules that might stop infections in their tracks or block pathways that lead to chronic diseases.
Innovation and Real-World Impact
Sometimes, medical breakthroughs seem overnight, but in reality, they rest on thousands of these smaller discoveries. Small changes at the molecular level can mean quicker action in the body or drugs that remain stable on pharmacy shelves. In fact, the tetrazole group has shown up in several FDA-approved medicines, including some blood pressure drugs, since it helps molecules stick around in the body just long enough to do their job without lingering too long.
The pharmaceutical industry values versatility. This chemical slips easily into test tubes for what chemists call “cross-coupling reactions”—basically sticking new pieces together like Legos. The more ways a molecule can be adjusted, the broader the possibilities for drug development. Over the past decade, scientists have taken inspiration from compounds like this to spark new work in antibiotics, antivirals, and anticancer agents.
Balancing Progress and Responsibility
Synthetic molecules hold a lot of promise, but safety always comes first. Testing never stops at just how well a new drug works. Chemists and pharmacists watch for stability, toxicity, and the way the body processes every candidate. Global agencies require deep evidence before any new medicine leaves the lab. Years of research help ensure that promising new candidates don’t bring unexpected risks to patients. Safety checks and quality controls drive the process, with lessons passed from scientist to scientist over decades.
Looking Ahead: Opportunity and Caution
New molecular starting points—like 5-Bromo-2-(2-Methyl-2H-Tetrazol-5-Yl)-Pyridine—represent the gutsy ambition behind every effective pill on the shelf. They won’t solve every problem overnight, but the families of medicine created from such innovative scaffolding keep hospitals better equipped to treat serious illness. Scientists continue to refine their methods, turn lessons from failed attempts into better outcomes, and add to the range of lifesaving options available to doctors and patients facing tough diagnoses.
What are the storage and handling requirements for 5-Bromo-2-(2-Methyl-2H-Tetrazol-5-Yl)-Pyridine?
What Safety in Storage Actually Looks Like
Years of handling specialty chemicals in the lab show that without clear routines, storage mishaps follow close behind. 5-Bromo-2-(2-Methyl-2H-Tetrazol-5-Yl)-Pyridine falls into that “not particularly forgiving” category. Exposure to air, moisture, or the wrong neighbors on the shelf can kick off trouble far beyond a lost afternoon at the bench. Glass containers with sturdy, airtight lids stand up best to both accidental spills and the creeping humidity that seems to find its way into every storeroom. Labels fade or fall off, leading to confusion, so chemical-safe, legible labels go a long way. Proper storage cuts down confusion, accidents, and, ultimately, the real risk to people working with it.
Stable Doesn’t Mean Carefree
A flashy molecular structure often signals unpredictability, and this compound doesn’t buck the trend. Shelf life changes based on how well you control temperature and light. From bitter experience on hot summer days, even “room temperature” fluctuates enough to degrade sensitive reagents. No one wants to discover decomposition just as an assay hits a time crunch. While refrigeration can prevent breakdown, it risks condensation when you pull it out for use, so always warm to room temperature inside a secondary container before opening. Flammable solvents or combustibles nearby only boost danger when someone works hurriedly or skips proper training.
Personal Responsibility and Group Accountability
Handling any pyridine derivative brings real hazards. Accidental skin contact, inhalation, or spills make for frantic moments, not just for the person involved but for everyone nearby. Relying on goggles and gloves gets drilled heavily in training, but regular check-ins and honest talks help keep safety from slipping with routine. Designated fume hoods protected more than one colleague from unnecessary headaches, and knowing where emergency showers or eyewash stations are saved a situation from getting worse. Communication matters. I remember a distracted undergrad forgetting to cap a reagent bottle, leading to overnight evaporation and a lingering odor for days – small mistakes become big problems quickly.
Planning for Unexpected Emergencies
Storage planning doesn’t stop when the bottle’s on the shelf. Chemical spill kits, clear labeling of cleaning supplies, and a standing order not to eat or drink anywhere near the chemicals saved a lot of folks from close calls. Disposal grows tricky—few local waste stations are equipped for compounds with halogenated groups and tetrazole rings, so working with reputable hazardous waste contractors takes the guesswork out of the equation. I’ve seen labs downgraded for cutting corners with disposal to avoid paperwork. Those shortcuts never end well; compliance isn’t just bureaucracy, it’s good science and sound ethics.
Finding Real Solutions
Ensuring safety isn’t just about meeting a checklist. Every person using or storing 5-Bromo-2-(2-Methyl-2H-Tetrazol-5-Yl)-Pyridine shares duty for reviewing storage guidelines, updating safety data sheets, and watching out for their labmates. Real solutions grow out of a culture where questions are welcome, training stays current, and researchers see themselves as stewards of both chemical and human well-being. Investing in high-quality storage containers, prioritizing proper ventilation, and taking seriously every spill or exposure builds trust and keeps science on sturdy footing. That trust lets the whole group work confidently, knowing a mistake won’t spiral into an emergency.
Is 5-Bromo-2-(2-Methyl-2H-Tetrazol-5-Yl)-Pyridine hazardous or toxic?
Chemicals in the Lab: A Personal Look at Hazards
Spending several years handling chemicals in research facilities leaves a person cautious about what goes into a beaker or onto a balance. 5-Bromo-2-(2-methyl-2H-tetrazol-5-yl)-pyridine, a compound you’ll spot on lists for advanced chemical synthesis, isn’t one most people recognize at a glance. Its structure places it firmly in the realm of specialty chemicals, and this is where risk and care meet science.
How Toxic Is This Stuff, Really?
Let’s break it down based on what’s known. There are no glowing safety reviews for this compound. It falls between the cracks of industrial databases—the sort of spot that makes chemists dig deeper before popping a lid. Close relatives of this molecule, like other bromopyridines or tetrazoles, bring their own hazards: skin and respiratory tract irritation, lasting environmental impacts, and in some cases, acute health risks after inhalation or direct contact.
Looking at the published literature, you won’t find animal testing data lining up for this molecule, and safety data sheets usually play it safe: wear gloves, use a fume hood, keep it sealed. This is not overkill—tetrazole rings have a reputation for being unpredictable, especially when functionalized with other reactive groups like bromides. I remember colleagues swapping stories about similar compounds giving off odd odors and causing headaches in poorly ventilated rooms.
Environmental and Laboratory Risks
Hazard doesn’t stop at the human body. Tetrazoles, by design, are energetic; the military uses some as propellants. Add a bromine atom, and you’re holding something that could persist if released, affecting water ecosystems and accumulating in soil or living tissue. Lab disposal requires special containers and chemical deactivation, since pouring this stuff down the drain could mean trouble.
My own rule of thumb: if a pyridine or tetrazole smells harsh or causes a tingle on the skin, ventilation and barriers come before curiosity. Accidental spills make a mess, and some of these chemicals stain or eat into plastic surfaces.
Straightforward Safety Measures
Facts speak loudly—precaution wins every time. Anyone tasked with working around this compound should take the same approach as with recognized hazards: work behind protective screens, don coats and gloves, keep emergency showers and eyewash stations nearby, and avoid working alone. Supplier-provided safety data sheets are a gold mine; manufacturers include them for good reason, even if the print runs long and dense.
There is room for improvement in risk education. Many academic labs still treat specialty organic compounds as if they all behave the same, even though just a few atoms can change the danger profile. Proper signage, chemical storage with good labeling, and up-to-date training keep accidents rare.
Better Solutions for Preventing Harm
It makes sense to push for green chemistry approaches, swapping out hazardous building blocks for safer alternatives where possible. More research into the human and ecological effects of emerging lab compounds, even those not produced at industrial scales, closes knowledge gaps before they become safety headlines. Insurance won’t cover shortcuts, so funding and policy should require research operations to do the basics—risk assessment and planning for unexpected exposure or spills.
At the end of the day, respect for specialty chemicals like 5-bromo-2-(2-methyl-2H-tetrazol-5-yl)-pyridine goes a lot further than bravado. Treating every unknown bottle as if it could hurt you is not paranoia—it’s just responsible science.
What is the molecular weight and CAS number of 5-Bromo-2-(2-Methyl-2H-Tetrazol-5-Yl)-Pyridine?
Numbers Tell a Story: Molecular Weight and CAS
Each chemical compound carries two ID cards. One is the molecular weight, which tells chemists how much a single molecule weighs. The other is the CAS (Chemical Abstracts Service) number, a unique code used to catalog and track chemicals. For 5-Bromo-2-(2-Methyl-2H-Tetrazol-5-Yl)-Pyridine, the molecular weight lands at 254.07 g/mol, and the CAS number to keep on file is 1025556-65-5.
Precision Matters in the Lab
In the world of research, numbers like these don't just float around for fun. Molecular weight plays into every calculation, whether you're mixing reagents in a pharmaceutical lab, checking the purity of a compound, or designing the next step in a synthesis pathway. I remember measuring out samples and needing to trust that every decimal was correct so the experiment didn’t spiral out of control thanks to something small, like one wrong digit.
Taking shortcuts around chemical nomenclature or registry numbers usually brings trouble later. When I searched for rare compounds after a late-night batch reaction, having the right CAS number shaved hours off the hunt. Tracking safety sheets, ordering samples, or looking up hazards—all get trickier when you don’t start with precise identification.
The Value of Reliable Identification
CAS numbers aren’t just bureaucratic paperwork—they protect labs from deadly mix-ups. Cross-contaminating reagents, especially with something like a pyridine derivative containing bromine and a tetrazole ring, brings real risks. Misplacing one element can introduce toxicity or destabilize reactions. In industrial settings, safety teams lean on these numbers to link a substance immediately to its hazard profile, storage requirements, and emergency protocols.
In drug development, regulatory filings demand bulletproof accuracy. Making a mistake with the molecular weight, even by a single dalton, could lead to the wrong compound passing through toxicology tests or, worse, winding up in a clinical trial. The stakes don’t get higher than that.
Improvement and Solutions
Chemists and industry pros could see fewer errors if digital systems got smarter about linking names, structures, weights, and CAS numbers through robust databases. Some platforms do this right, pulling from trusted sources and flagging discrepancies. Training goes a long way, too—I always suggest that every person in the lab, from tech to postdoc, gets hands-on practice with chemical search engines and standard references. Overreliance on copy-pasting from PDFs or ambiguous trade names has led plenty of labs into trouble.
Opening up access to curated, community-vetted data reduces the risk of using outdated or misattributed molecular weights. Regulatory bodies might work better with industry by creating open platforms for new compound registrations. As more novel compounds pop up in research, collaboration between publishers, database curators, and researchers will keep the system robust.
The Human Factor
In a busy lab, details get lost. I’ve seen simple mistakes escalate, sometimes because people skip double-checking CAS numbers or scribble molecular weights from memory. Building a culture that values small details and taking time for verification saves money, reputations, and occasionally, lives. So while the difference between 254.07 and 254.70 might just look like numbers, they carry a story that goes beyond math—they speak to safety, accuracy, and trust.
| Names | |
| Preferred IUPAC name | 5-bromo-2-(2-methyltetrazol-5-yl)pyridine |
| Other names |
5-Bromo-2-(2-methyltetrazol-5-yl)pyridine 5-Bromo-2-(2-methyl-2H-tetrazol-5-yl)pyridine 5-Bromo-2-[2-methyl-2H-tetrazol-5-yl]pyridine |
| Pronunciation | /ˈfaɪˈbrəʊməʊˈtuːˈmɛθɪlˈtuːˈeɪtɛtrəˌzɒlˈfaɪlˈpɪrɪdiːn/ |
| Identifiers | |
| CAS Number | 1116743-24-6 |
| 3D model (JSmol) | `3D64F6BYE1EFLF-WJOKGBTGSA-N` |
| Beilstein Reference | 1813886 |
| ChEBI | CHEBI:139322 |
| ChEMBL | CHEMBL3584206 |
| ChemSpider | 14487004 |
| DrugBank | DB07722 |
| ECHA InfoCard | 03d93d95-d6c4-4293-bc2a-caba6c97c7c2 |
| EC Number | Not assigned |
| Gmelin Reference | 1361987 |
| KEGG | C19753 |
| MeSH | D000068406 |
| PubChem CID | 10037303 |
| RTECS number | TZ1970000 |
| UNII | Y912YZ8L6Z |
| UN number | UN3276 |
| CompTox Dashboard (EPA) | DTXSID3060188 |
| Properties | |
| Chemical formula | C7H6BrN5 |
| Molar mass | 266.08 g/mol |
| Appearance | Light yellow solid |
| Odor | Odorless |
| Density | 1.70 g/cm3 |
| Solubility in water | Slightly soluble |
| log P | 1.6 |
| Acidity (pKa) | 6.41 |
| Basicity (pKb) | 5.22 |
| Magnetic susceptibility (χ) | -87.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.669 |
| Dipole moment | 2.7343 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 369.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | The standard enthalpy of formation (ΔfH⦵298) for 5-Bromo-2-(2-Methyl-2H-Tetrazol-5-Yl)-Pyridine is: "-7.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2202 kJ/mol |
| Pharmacology | |
| ATC code | N03AX17 |
| Hazards | |
| Main hazards | H302 + H315 + H319 + H335 |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H315, H319, H335 |
| Precautionary statements | P261, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-2-1-ox |
| Flash point | Flash point: 230.2 °C |
| LD50 (median dose) | LD50: >5000 mg/kg (rat, oral) |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10-20°C |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
5-Bromo-2-(2H-tetrazol-5-yl)pyridine 5-Bromo-2-pyridyl tetrazole 2-(2-Methyl-2H-tetrazol-5-yl)pyridine 5-Bromo-2-pyridinecarboxamide 5-Bromopyridine-2-carbonitrile |

