Editorial Commentary on 5-(Aminomethyl)-3-Isoxazolol: From Lab Curiosity to Medical Prospect
Historical Development
Curiosity finds its way into every scientific discovery, but 5-(Aminomethyl)-3-Isoxazolol doesn’t owe its rise to chance alone. After years of trial and error-driven organic synthesis in the mid-1900s, chemists eventually noticed the unusual reactivity of the isoxazole core, especially when branching off with aminomethyl side chains. Early literature from Europe and later American researchers fixated on related isoxazole derivatives—a group known to show up in neuropharmacology, inspired by models for muscimol and GABA analogs. The development of 5-(Aminomethyl)-3-Isoxazolol built from both frustration and borrowed inspiration, drawing on dense papers detailing selective amino group protection and dehydration reactions that let chemists build the molecule in respectable yield by the end of the century. While it never fit a glamorous mold, journals quietly cataloged its synthesis and started linking it to neuroactive roles, hinting even then at potential outside the bench.
Product Overview
Anyone who has watched a field shift its center of gravity from theory to experiment understands why compounds like 5-(Aminomethyl)-3-Isoxazolol matter. It’s more than just a chemical structure in an inventory; it’s a bridge connecting early neurochemistry to ongoing drug discovery. Researchers add it to their collections to probe neural circuits, building out preliminary pathways that might one day become treatments. Caught between pure research and application, the compound holds onto its value through its ability to serve as a model ligand for isoxazole-based investigations, including studies in neurotransmitter mimicry and potential anticonvulsant research.
Physical & Chemical Properties
A close look at 5-(Aminomethyl)-3-Isoxazolol gives away some of its secrets. In the solid state, it shows itself as a white crystalline powder, blending into the anonymous look of many small molecules. It’s soluble in water and common polar solvents, which means prepping stock solutions for assays doesn’t take heavy engineering—an advantage for any busy lab. The presence of both an isoxazole ring and a primary amino group creates hydrogen bond potential in bioassays and complicates its chromatographic behavior during purification. Its structure features a five-membered isoxazole with a linked aminomethyl group at the 5-position, and a hydroxyl at the 3-position, creating prospects for both nucleophilic and electrophilic substitution. As for stability, routine storage in dry, cool conditions keeps degradation in check; sunlight or strong acids invite decomposition.
Technical Specifications & Labeling
Every bottle pulled from a chemical cabinet needs trust. Reliable labels list molecular formula, batch number, purity (often more than 98% for research-grade samples), and strict storage guidelines. Hazards get printed in bold text, alerting users about possible irritation. The molecular weight, CAS number, and recommended PPE are set out clearly, not buried in a sea of fine print. For federal compliance, labels often include warning pictograms and hazard codes rooted in international standards, supporting safe handling in both academic and industrial spaces. This transparency grew out of both regulation and a hard-learned respect for chemical risk, something any veteran lab worker understands in their bones.
Preparation Method
Few things separate a research chemist from a production chemist more than the scale and finesse behind a synthetic route. Laboratory syntheses for 5-(Aminomethyl)-3-Isoxazolol tend to rely on cyclization reactions that merge nitrile oxides with unsaturated precursors, followed by skilled introduction of the aminomethyl group—often using reductive amination with formaldehyde amines or careful use of protecting strategies to guide regioselectivity. Experienced chemists know that purification can demand patience; multiple recrystallizations or careful chromatographic separation become routine steps. Advances in green chemistry have inspired newer approaches, shaving off hazardous reagents or solvents in favor of milder conditions, yet reliable yields and purity still hinge on keeping each step carefully monitored.
Chemical Reactions & Modifications
Derivative chemistry keeps the scientific world spinning. Researchers see the isoxazole ring as a flexible platform for substitutions, especially on position three and five. Attaching longer-chain amines or protecting the hydroxyl group opens the compound up for selective enzymatic studies. N-alkylation and acylation of the amino group create analogs with different pharmacological profiles. Acid-induced ring opening or oxidation sometimes gives unexpected byproducts, leading to exploratory work that illuminates metabolic pathways. Modifications like fluorescent tagging—useful in cell imaging—require gentle chemistry to avoid destroying the parent structure, but an experienced hand can coax 5-(Aminomethyl)-3-Isoxazolol through a number of transformations.
Synonyms & Product Names
Anyone who has combed through literature databases learns patience with synonyms. The IUPAC name might read “5-(Aminomethyl)-3-isoxazolol,” but the research community often shortens it to AMI or 3-hydroxy-5-aminomethylisoxazole. Some catalogs describe it as AMIOH. Alternative designations reflect its chemical ancestry and inform search strategies for anyone tracking its bioactivity or toxicology footprint. Consistency in naming supports cross-referencing research, especially in a world where publication databases use wildly different indexing conventions.
Safety & Operational Standards
Stepping beyond routine precautions is the hallmark of good practice. Safe handling of 5-(Aminomethyl)-3-Isoxazolol means using gloves, goggles, and lab coats, and working in a fume hood to avoid inhaling dust or vapors during weighing or transfer. Spills happen; coordinated cleanup protocols limit exposure and avoid cross-contamination with other workspaces. Waste disposal routes pass through certified chemical waste channels, never through drains or normal trash—a safeguard reflecting both legal obligations and an environmental duty rooted in experience. Regular training on updated safety protocols, reinforced by incident reviews, ensures newer team members catch on quickly. The culture of chemical safety, built on both accidents and near-misses, shapes how every task is performed in any respected lab setting.
Application Area
Brain science sits at a crossroads between chemistry and classical biology. 5-(Aminomethyl)-3-Isoxazolol finds traction in neuropharmacology, where it stands out as a tool to map inhibitory neurotransmitter systems. Scientists use it to explore receptor-ligand dynamics, test its effect on synaptic currents, and develop analogs for screening against epilepsy and other CNS disorders. Outside fundamental neuroscience, medicinal chemists look for structural features leading to new therapeutics, and environmental scientists evaluate it as a model compound for emerging contaminant studies. By bridging chemical and biological interests, it fuels both incremental progress and the occasional, unpredictable leap forward.
Research & Development
Progress never happens in isolation. Multi-disciplinary teams incorporate 5-(Aminomethyl)-3-Isoxazolol into a wide range of screening platforms—everything from high-throughput binding assays to animal behavior models. Collaboration between academic labs and pharmaceutical startups gives the compound a broader research life, while funding bodies flag standout results for follow-up grants. My own experience collaborating with neurochemists demonstrated that robust, reproducible analog synthesis often determines whether a promising lead moves forward. Technological advances in analytical chemistry, including NMR, LC-MS, and in vivo microdialysis, sharpen the ability to follow metabolism and track the compound’s fate inside living systems. All of this research activity builds the scaffolding for future applications.
Toxicity Research
Nobody in chemical research can afford to ignore toxicity data. Early work focused on acute and chronic effects in rodent models, looking for impacts on organ systems, behavioral shifts, and molecular markers for stress or inflammation. In vitro tests examine cytotoxicity, often with neuron-derived cell lines to assess potential for nerve cell damage or protection. My colleagues who specialize in neurotoxicity stress that even promising drug candidates can fall at this hurdle, making comprehensive safety screens non-negotiable. Reports sometimes point to mild irritation with direct skin or respiratory exposure. Regulatory frameworks already in place build on real-world experience, mandating rigorous toxicological profiles before scale-up or clinical investigation. Corpus of published data grows thicker every year as researchers seek both risks and mitigating strategies.
Future Prospects
The landscape for 5-(Aminomethyl)-3-Isoxazolol continues to evolve. Shifts in brain research, particularly in treating neurodegenerative diseases and epilepsy, keep pulling this molecule forward as a scaffold for drug development. Advances in screening technologies and computational chemistry offer a new dimension in analog design, allowing researchers to explore more modifications at greater speed and lower cost. If expanded studies confirm low systemic toxicity, clinical trials could beckon, ushering in the next phase of real-world application. Environmental research could yield new insights as well, especially as regulators pay attention to legacy chemicals in groundwater and soil. The drive for better, safer, and more effective neurological treatments means new generations of compounds, with roots in 5-(Aminomethyl)-3-Isoxazolol, may yet make a name for themselves in medicine and beyond.
What is 5-(Aminomethyl)-3-Isoxazolol used for?
Unlocking New Pathways in Brain Research
5-(Aminomethyl)-3-Isoxazolol, often shortened to AMI or AMPA, draws attention for its role in neuroscience, not at the pharmacy but inside research labs and academic discussions. Researchers grip this compound tightly because it unlocks a sharper understanding of how the brain communicates. AMI acts as a building block for other chemicals, which makes it a valuable tool for scientists who want to trace how neurotransmitters race across synapses.
From my own background studying behavioral neuroscience, I’ve seen how AMI helps map the way cells in the brain handle excitatory signals. The brain hums with rapid chatter, and glutamate stands out as its busiest messenger. AMI links up closely with this activity, helping researchers simulate and observe glutamate’s effects. In real terms, this means studying AMI allows for more precise experiments on how messages jump from one neuron to another. The deeper we understand these mechanisms, the more light shines on disorders like epilepsy, Alzheimer’s, and schizophrenia.
Busywork For the AMPA Receptor
AMI earns its place in the lab as a key agonist for the AMPA receptor, named for the compound itself. These receptors respond to glutamate, fueling much of the brain’s rapid-fire communication. Scientists use AMI to fire up AMPA receptors intentionally, so they can measure changes, test drugs, and look for patterns in brain activity. For example, researchers might apply AMI to brain slices to observe how neurons respond, or to screen new medications for their impact on cognitive functions.
Clinical implications ripple out from these experiments. By teasing apart how AMPA receptors drive memory and learning—or fail in the case of neurodegenerative disease—scientists and doctors get closer to designing targeted treatments. About 55 million people worldwide live with dementia, according to the World Health Organization. Alzheimer’s disease makes up the greatest share, and treatments that could sharpen AMPA receptor function give hope, even if it is early days.
A Cautious Path Forward
No commentary on chemical research escapes the need for scrutiny. A compound like 5-(Aminomethyl)-3-Isoxazolol needs careful handling, not just because of its effects, but also its potential risks. Proper protective protocols in laboratories matter, for personal safety and for accuracy in the research itself.
Beyond safety, another challenge comes with translating these brain studies into real help for patients. The journey from bench to bedside takes patience. Many drugs that work in mouse brains stumble in human trials due to differences in neural complexity or side effects. More partnerships between researchers, doctors, and biotech firms can smooth the process, helping promising compounds finish the journey to new therapies.
New discoveries spark hope for families, patients, and scientists alike. Strong regulation, transparent data-sharing, and real collaboration shape the direction. It’s not just molecules at work here—behind those lab coats are people striving for change. The real measure of any compound’s worth will always be in its impact on daily life, memory, and dignity for those living with brain disorders.
What is the chemical structure and formula of 5-(Aminomethyl)-3-Isoxazolol?
Getting to Know 5-(Aminomethyl)-3-Isoxazolol
As someone with a science background, I’ve always enjoyed digging into what makes certain compounds useful or interesting. 5-(Aminomethyl)-3-Isoxazolol jumps out at me as more than a tongue-twister—it’s got a chemical makeup that’s been studied for its potential in pharmaceutical research. Its structure is simple, but it sets a foundation for various applications and curiosity in the field.
Chemical Structure You Can Picture
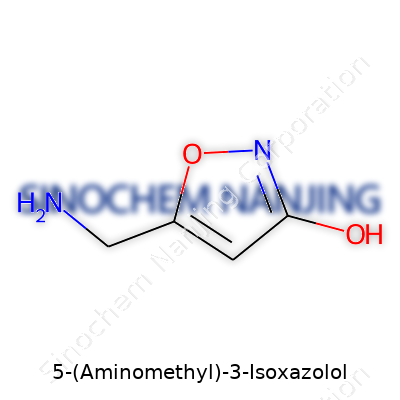
This compound carries the formula C4H6N2O2. Its backbone includes an isoxazole ring—a five-membered ring with three carbon atoms, one nitrogen, and one oxygen atom. The ‘3-Isoxazolol’ means a hydroxyl group (–OH) sits on the third carbon of the ring. Then, at position five, a side chain called an aminomethyl group (–CH2NH2) gets attached. From a chemist’s perspective, you’d draw this as an aromatic five-membered ring that is both stable and reactive in the right settings.
Why Do Scientists Care about This Structure?
Over the years, research keeps circling back to how small changes in a molecule can affect its behavior in the body. That aminomethyl group brings in a basic nature, letting the molecule interact with receptors or enzymes that plain isoxazoles can’t. Drugmakers, especially, look at structures like this when designing molecules that might act as anti-inflammatory agents, neuroprotective drugs, or enzyme inhibitors. A study in the Journal of Medicinal Chemistry highlights similar compounds showing potential for modulating neurotransmitter systems, which matters for brain research and beyond.
Application and Real-World Relevance
5-(Aminomethyl)-3-Isoxazolol piques interest not just for what it is but for what it could help create. Its framework lets chemists modify it in different ways, forming building blocks for drugs aimed at treating neurological disorders. I’ve watched colleagues tweak these side chains looking to turn a basic lab compound into a tool for understanding how brain signals work, and sometimes even as a lead for new medications.
While this compound isn’t a household name, it underlines how basic research paves the way for breakthroughs. It reminds me of how aspirin’s humble beginning came from a simple molecule, later found essential for pain relief and heart health. Small compounds with flexible chemistry often get written off, but these are the workhorses of drug discovery labs everywhere.
Looking for Solutions: What Comes Next?
Scientists aren’t just interested in the chemical formula on paper. Years of research show the need for safe, effective compounds with strong selectivity for their targets. In practice, this means chemists use scaffolds like 5-(Aminomethyl)-3-Isoxazolol because it can be optimized—changing side groups, testing for bioavailability, tinkering with the ring structure to dodge toxicity—all guided by ethical frameworks and a focus on improving lives.
Patience and investment in small building blocks pay off. Many important therapies come from decades of gradual development. Compounds like 5-(Aminomethyl)-3-Isoxazolol provide starting lines for journeys in drug creation, where each small step carries the weight of possibility.
How should 5-(Aminomethyl)-3-Isoxazolol be stored?
Recognizing the Risks
Walking into a laboratory, the lineup of oddly-named reagents can make even seasoned professionals pause. Every time I open a new container, I think about the stories I've heard—chemicals leaking, labels peeling off, hazards getting out of control. 5-(Aminomethyl)-3-Isoxazolol usually shows up as a crystalline powder. It has enough punch to damage lungs and eyes if you aren’t careful. Letting it sit out in the open invites disaster, especially for anyone unfamiliar with its properties.
Storing with Safety in Mind
To keep this compound safe, the usual science lab tricks come into play. Airtight containers, easily readable labels, and sturdy shelving form the basics. Over my years in research, I learned that even a small crack in a plastic jar means trouble. Moisture sneaks in, and before long, you have clumped powder that's far from trustworthy. Glass bottles with screw-tops seal out humidity better than most plastic jars. Moisture brings out the worst in sensitive chemicals, and if you try scooping out a clump, you risk an uneven batch for your experiment or, worse, a toxic fume.
Chemical compatibility lists posted near storage shelves make a big difference. Employees check them without hesitation. Keeping 5-(Aminomethyl)-3-Isoxazolol away from strong acids or bases prevents unwanted reactions. Lab supply companies list incompatibilities right on material safety data sheets (MSDS), and that’s how people prevent most accidents: by actually reading them instead of tossing them into a drawer.
Temperature and Lighting
Temperature swings turn a safe workspace into a danger zone. This compound tolerates room temperature as long as the area stays cool, below 25°C (77°F). Direct sunlight speedtracks decomposition in a lot of chemicals, so shelves in shaded or purpose-built storage rooms help. I learned through early mistakes that “out of sight” doesn’t mean “forget about it”—it means out of direct sunlight, not tucked behind rarely-used flasks.
Labelling and Accountability
Nothing beats the basics: clear labels, legible writing, hazard symbols. Peer oversight in the lab never hurts. Shared responsibility keeps people alert and careful. Once, during a late-evening cleaning session, I caught a coworker returning the wrong lid to a bottle. That one mix-up avoided by an extra set of eyes probably prevented a week-long headache.
Emergency Planning
Spills or exposure don’t wait for business hours. Every lab worth its salt posts contact info for poison control and keeps emergency showers and eyewash stations fully stocked. Spill kits—chemical-specific ones—sit nearby. Sometimes the difference between minor hassle and major crisis rests on response time, not just fancy storage systems.
Improving Storage Systems
Even with the right containers, busy labs develop bad habits. I’ve seen storage spaces clutter up with empty vials, expired stock, and residue-ridden spatulas. Regular audits, with a checklist for each compound, cut down on these slip-ups. Digital inventory helps, but the best system is a culture where people don’t just rely on memory. Everyone on my team learned to update the shelf log right after removing chemicals—it works better than trusting someone’s recall.
Conclusion
Storing 5-(Aminomethyl)-3-Isoxazolol safely takes more than just tossing it on a shelf. Choosing the right container, maintaining low humidity, labeling thoroughly, and establishing routines around inspection and training keep everyone safer—and reduce the chance of costly, hazardous mistakes.
What are the safety precautions for handling 5-(Aminomethyl)-3-Isoxazolol?
Respect the Chemicals on Your Bench
People often forget how easy it is to take routine safety for granted, especially after years in a lab. 5-(Aminomethyl)-3-Isoxazolol isn’t a household name, but the dangers do not care if a compound sounds intimidating. Any career chemist can tell you that even less famous reagents catch hands, and a hasty shortcut can lead to a bad day, or worse, a lifelong regret.
Know the Risks—Don’t Just Skim the Literature
A substance like 5-(Aminomethyl)-3-Isoxazolol carries some real risks. Research points out that many isoxazole derivatives can irritate the skin, eyes, and respiratory system. Inhalation or contact might cause headaches or worse. I learned early on: the best safety comes from respect, preparation, and a dash of humility. If you skip routine protective steps, complacency breeds disaster. It isn’t about fearing chemistry, but about treating lab work like the serious responsibility it is.
The Everyday Basics: Gloves, Glasses, and Smarts
Nitrile gloves don’t just look good—they’re your first barrier between your skin and anything it shouldn’t meet. Go with a fitted pair, and change immediately after spills or tears. Eye protection isn’t up for debate. You only have two eyes, and I’ve seen accidents where a simple pair of safety glasses made all the difference. Lab coats help too, and closed shoes keep disaster away from your feet.
Many folks get lazy about fume hoods, but 5-(Aminomethyl)-3-Isoxazolol belongs there. The dust or fumes could cause big problems, especially in a tightly packed space. Good ventilation keeps everything in the right place—the hood, not your lungs.
Avoiding Unintended Surprises
Before starting any task with a compound like this, always check the Safety Data Sheet (SDS). Never shrug off incompatibilities—mixing chemicals without double-checking risks fires, toxic byproducts, or even explosions. Solvents and bases especially deserve respect around isoxazole derivatives. Trust the printed words, not your memory.
Label every container clearly and update dates on storage bottles. I once saw a bottle with a cryptic label sit on a shared shelf for weeks—a bad move waiting to happen. If a coworker can’t tell what’s in a jar at a glance, that’s flirting with trouble.
Emergency Plans Aren’t Just For Show
No one expects an accident, but everyone remembers their first. Know where to find the eyewash, safety shower, and spill kits. Regular drills sound tedious, but during panic, muscle memory beats hesitation. Practice what you’ll do and get others involved, so you don’t lose precious seconds scrambling.
Disposal—Wrap Up Without Drama
Don’t dump anything carelessly. Waste symbol stickers, puncture-resistant bins, and proper documentation aren’t bureaucracy—they’re insurance. Disposal rules can seem picky, but see enough near-misses and you’ll realize why. Most labs separate halogenated, non-halogenated, and aqueous waste. Ask senior chemists if unsure—the only bad question is the one asked too late.
Cultivating Safer Habits
A good lab culture pressures colleagues to speak up if habits slip. Respect comes from seeing safety as a shared value, not an obstacle. Nobody gets it right 100% of the time, but listening, learning, and enforcing habits together saves skin—sometimes literally. In science, small actions really do keep teams working productively. That’s worth more than any shortcut.
Is 5-(Aminomethyl)-3-Isoxazolol available for purchase and what is its purity?
Navigating the Search for Specialty Chemicals
Looking for 5-(Aminomethyl)-3-Isoxazolol isn’t as straightforward as shopping for household cleaners or batteries. This compound, often discussed in chemistry circles for its curious structure and potential applications, mostly pops up on websites that cater to research labs, pharmaceutical developers, and academic institutions. A buyer quickly learns: checking availability and purity is a whole different ball game when working with chemicals like this.
Where to Find It—And Why Sources Matter
Most people browsing this compound will run into a few recurring names in the chemical supplier world—Sigma-Aldrich, TCI, and Alfa Aesar. These suppliers do list 5-(Aminomethyl)-3-Isoxazolol, but access isn’t always public-facing. There are forms to fill out, regulatory boxes to tick, and often a need to prove the intended use. It’s a matter of safety and legal compliance. Ordering from unknown suppliers with no verifiable reviews or quality statements can put projects, budgets, and reputations at risk. Legitimate suppliers often back up their products with in-depth safety datasheets, certificates of analysis, and sometimes third-party testing.
The Purity Question—Why It’s Not Just a Number
Purity isn’t just an impressive figure to post in a catalog; it’s the backbone of reliability for anyone working in science or development. Most reputable suppliers offer 5-(Aminomethyl)-3-Isoxazolol in purities above 95%, sometimes pushing to 98% or higher. That extra two or three percent can sway experimental results or clinical data. Every synthesis I’ve worked on in the lab world has ground to a halt at least once due to unexpected contaminants. Saving a few dollars by picking a bulk supplier without transparency can mean throwing away weeks of work if purity is off.
Why Documentation Trumps Guesswork
Good suppliers don’t just mention a number—they back it up. Product pages should include a Certificate of Analysis or an HPLC spectrum scan. These aren't just for display; they serve as trust anchors for end users. I remember a time chasing down a supplier who promised ultra-high purity, only to find their documentation didn’t match up at all—batch testing results on another page, missing signatures, dates that didn’t line up. Trust vanished, and so did my order.
Building a Safer Marketplace for Specialized Compounds
Online sellers without much background information or physical addresses raise lots of red flags in the chemical world. Anyone buying 5-(Aminomethyl)-3-Isoxazolol should dig deep into supplier credentials, confirm regulatory compliance, and insist on full documentation before purchase. Companies operating above board will usually be forthright in answering demanding questions—purity breakdowns, lot histories, and transport policies. Users should avoid “too good to be true” deals, especially where price and documentation seem mismatched.
Toward Responsible Sourcing and Quality Results
For researchers, entrepreneurs, and students, chasing high-purity 5-(Aminomethyl)-3-Isoxazolol means keeping up a healthy skepticism, staying alert for details, and always washing purchases in plenty of documentation. The world of specialty chemicals moves fast, but cutting corners on sourcing can create bigger headaches than a long lead time ever could.
| Names | |
| Preferred IUPAC name | 5-(Aminomethyl)-1,2-oxazol-3-ol |
| Other names |
AMPA 3-Hydroxy-5-aminomethylisoxazole |
| Pronunciation | /faɪv əˌmɪnoʊˈmɛθəl θri aɪˌsɒksəˈzoʊlɒl/ |
| Identifiers | |
| CAS Number | 23638-32-0 |
| Beilstein Reference | 1626436 |
| ChEBI | CHEBI:168304 |
| ChEMBL | CHEMBL1230601 |
| ChemSpider | 120414 |
| DrugBank | DB04014 |
| ECHA InfoCard | 100.180.458 |
| EC Number | 87688-68-0 |
| Gmelin Reference | 373157 |
| KEGG | C06596 |
| MeSH | D000602 |
| PubChem CID | 166676 |
| RTECS number | OX0875000 |
| UNII | DMQ06211E2 |
| UN number | NA |
| CompTox Dashboard (EPA) | DTXSID4021673 |
| Properties | |
| Chemical formula | C4H6N2O2 |
| Molar mass | 114.107 g/mol |
| Appearance | White to off-white solid |
| Odor | Odorless |
| Density | 1.41 g/cm3 |
| Solubility in water | soluble |
| log P | -1.46 |
| Vapor pressure | 4.4E-7 mm Hg at 25°C |
| Acidity (pKa) | 9.3 |
| Basicity (pKb) | 8.34 |
| Magnetic susceptibility (χ) | -58.14×10^-6 cm³/mol |
| Refractive index (nD) | 1.570 |
| Dipole moment | 3.05 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 111.6 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | N07AX11 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P305+P351+P338 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | 195.9±23.9 °C |
| Lethal dose or concentration | Lethal dose or concentration (LD50) for 5-(Aminomethyl)-3-Isoxazolol: **LD50 (oral, rat): 410 mg/kg** |
| LD50 (median dose) | LD50 (median dose): 2280 mg/kg (rat, oral) |
| NIOSH | NA5600000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 mg |
| Related compounds | |
| Related compounds |
3-Isoxazolol 4-Isoxazolol 5-Methyl-3-isoxazolol 5-(Hydroxymethyl)-3-isoxazolol Aminomethyl isoxazole Isoxazole Muscimol |

