Rethinking 5-Amino-1,3,3-Trimethylcyclohexanemethylamine: Beyond the Basics
Historical Development: The Roots of a Versatile Chemical
People usually look to history for the stories behind well-known molecules, but compounds like 5-Amino-1,3,3-Trimethylcyclohexanemethylamine often fly under the radar. The tale of this amine traces back to the surge of synthetic organic chemistry after World War II, when industrial and academic laboratories across Europe and the United States scrambled to unlock new building blocks for plastics and specialty chemicals. Through the decades, researchers refined alkylation and amination strategies, driven by the need for versatile intermediates. This amine grew in popularity along with monomer synthesis and resin formulation, especially as fine chemical producers learned to streamline multi-step methods. As more focus landed on ring-modified and sterically hindered amines, chemists saw the value in its unique structure and functional profile—far from just an afterthought or laboratory footnote.
Product Overview: What Sets This Amine Apart
The appeal of 5-Amino-1,3,3-Trimethylcyclohexanemethylamine lies in its quirky setup: a cyclohexane core studded with three methyl groups and a primary amino group on a side chain. This configuration doesn’t just look good on paper. In practical settings, the molecular framework brings both rigidity and a bit of steric bulk. For folks working in industrial labs, this means building with a scaffold that resists easy breakdown, making it a popular pick when durability and well-defined reactivity matter. Its compatibility in aminolysis and alkylation shines when crafting custom additives, curing agents, or active pharmaceutical ingredient intermediates. While not as instantly recognizable as aniline or ethylenediamine, it’s carved out a loyal following among chemists seeking something tough, predictable, and more than a little off the beaten path.
Physical & Chemical Properties: Not Just Another Amine
Once you crack open a bottle, you notice its sharp, somewhat fishy odor—a calling card for primary amines. The molecule exists as a colorless or pale liquid, depending on purity and storage. It boasts a relatively high boiling point courtesy of its cyclohexane ring and methyl padding, and resists freezing well below zero Celsius. The compound dissolves readily in many polar organic solvents, holding its own in both bench-scale and pilot-scale settings. Its lone nitrogen atom, sitting comfortably on the side chain, craves protons and offers nucleophilic punch in synthesis. Yet the ring’s methyl groups shield it against overzealous reaction, balancing activity with selectivity. Its atmospheric stability makes storage less of a headache, provided the container seals up tight to avoid water uptake or slow air oxidation.
Technical Specifications & Labeling: Clarity for a Demanding World
Those of us who’ve browsed supplier catalogs know the confusion that comes with chemicals carrying a smattering of nearly identical names and synonyms. 5-Amino-1,3,3-Trimethylcyclohexanemethylamine’s labeling usually pinpoints high purity levels, minimal water content, and defined refractive index. Seasoned chemists check density, molecular weight, and assay by GC or NMR to dodge any surprises on reaction day. Proper handling calls for sturdy labeling—since more than one researcher has grabbed the wrong flask and ended up with an unexpected byproduct or worse, a scrapped experiment. Its status as an amine flags it for moderate regulatory scrutiny, nudging suppliers to add hazard codes for skin irritancy and toxicity research flags—issues every responsible lab needs to keep at the forefront.
Preparation Method: Crafting the Cyclohexanemethylamine Backbone
Synthesizing this molecule isn’t just a rote exercise. Old-school methods leaned heavily on alkylation of cyclohexanone derivatives followed by reductive amination, where hydrogen gas and a metal catalyst did the heavy lifting. Improvements in catalytic hydrogenation and selectivity have made the process both greener and more scalable. Some labs prefer a route through Grignard reagents coupled with amination steps, especially for custom or isotopically labeled compounds. The mismatched steric demands from the trimethyl groups mean not every preparation method will give clean yields without careful control of reaction conditions—pressure, temperature, and base selection. Good chemists keep the waste streams clean and recyclables sorted, since downstream purification chews up both time and resources.
Chemical Reactions & Modifications: Flexibility on Demand
Amines act as the workhorses of many synthetic pathways, and this compound’s nucleophilicity opens plenty of doors. Reactivity with acid chlorides leads to tough amide bonds, ideal in resin chemistry or as linkers for drug candidates. Epoxide ring openings with this amine yield stable β-amino alcohols, adding value in the surfactant and coatings industries. Its sterically shielded core stymies unwanted side reactions, handing researchers a clean slate for selective functionalization. The molecule doesn’t just accept modifications—its architecture enables creation of libraries of analogs ripe for screening in pharmaceuticals, polymers, and beyond. I’ve seen it used as a backbone for functional dyes and even as a specialty curing accelerator for advanced composite materials.
Synonyms & Product Names: Language Shapes Discovery
Chemical communication falls flat if you can’t pin down exactly what you’re working with. This amine’s mouthful of a name sometimes appears in shorthands like AMCMA or as an isomer-specific cyclohexanemethylamine. The difference between a simple misreading and a lab disaster can come down to a single digit or misplaced methyl group. Commercial suppliers tag the product with inventory numbers and common alternates, making cross-referencing in global research easier but sometimes a headache. Synonyms abound, but accuracy ensures everyone, from the research assistant to the plant manager, stays on the same page and avoids costly or dangerous missteps.
Safety & Operational Standards: Lessons Learned From Practice
Lab safety hinges on far more than gloves and goggles. Handling 5-Amino-1,3,3-Trimethylcyclohexanemethylamine means respecting its irritant capacity—one splash on skin or in the eyes brings burning and redness. Vapors in high concentrations annoy mucous membranes, but careful users work under fume hoods and keep spill kits ready. It absorbs quickly through skin; I’ve seen even careful colleagues miss a drop and race for the wash station. Disposal protocols direct waste into proper organic containers, with clear logs recorded. Facilities with years of experience keep training up to date and rethink procedures when new uses or volumes emerge. Keeping hazards transparent and acting promptly on exposure keeps the compound a tool—not a risk.
Application Area: Where Science Meets Industry
Years back, the chief use I saw for this compound involved resin and polyurethane formulations. Today, researchers push its utility into specialty coatings, epoxy curing, and even pharmaceutical manufacturing. Its unique ring system and reactivity slot nicely into synthesis routines chasing new catalysts or surface-active agents. High-performance elastomers based on variants of this amine withstand tough thermal cycling and harsh chemical environments. The amine’s structure allows it to toughen adhesives or modify the solubility of drug candidates. Tailored analogs wind up as linkers in molecular imaging—bringing the compound from industry straight into patient care, a leap that highlights just how interdisciplinary chemistry has become.
Research & Development: Pushing the Boundaries
Innovation seldom moves in a straight line. I’ve seen academic collaborations trying to coax new coordination complexes from this backbone, hoping for catalytic activity or unique electronic effects. Researchers see its role not as a static feedstock but as a stepping stone toward engineered molecules—responsive adhesives, smart coatings, or biodegradable plasticizers. The global research community, driven by open data and rapid idea-sharing, explores how alkyl substitutions alter the amine's properties, banking on machine learning and predictive modeling to shorten the path from concept to commercial reality. There’s a competitive edge in unlocking greener, safer, or more cost-effective processing routes.
Toxicity Research: Walking the Careful Line
An honest discussion about specialty amines shouldn’t skip toxicity. Detailed studies track acute and chronic exposure risks, both for those handling the compound and for potential uses in consumer products. Primary amines like this often flag moderate toxicity profiles—skin sensitization tests and inhalation LC50 figures give researchers benchmarks for safe design. Long-term studies sometimes reveal organ system impacts in animal models, making documentation and regulatory compliance crucial. The promise of the molecule comes paired with accountability—thorough risk assessments, responsible manufacturing, and clear labeling. Teams weighing its use factor in downstream products’ environmental impact, end-of-life handling, and potential for bioaccumulation.
Future Prospects: Where This Chemical Could Go Next
The road ahead for 5-Amino-1,3,3-Trimethylcyclohexanemethylamine spirals out from specialty chemicals toward greener, smarter applications. Tighter environmental standards in Asia and Europe nudge companies to explore more benign synthesis routes, pursuing lower-emission or renewable pathways. Expansion into pharmaceutical precursors, advanced polymer design, and functional materials depends on smart R&D and a willingness to confront both technical and regulatory challenges. Sustainable manufacturing, collaboration between academia and industry, and a sharp focus on both worker safety and product stewardship drive progress. In hands that know both the science and the risks, this compound turns from obscure catalog entry into a springboard for new technology and a safer, cleaner chemical future.
What is 5-Amino-1,3,3-Trimethylcyclohexanemethylamine used for?
What’s Behind the Tongue-Twister Chemical Name?
Seeing a name like 5-Amino-1,3,3-Trimethylcyclohexanemethylamine can be intimidating. Underneath all that jargon sits a chemical compound that has carved out a role in modern industry and scientific research. Trends in manufacturing and innovation often depend on specialty chemicals like this one. An even mix of practicality and chemistry shapes how and why a compound becomes invaluable across certain sectors.
Diving Into Its Key Uses
Factories and labs prize this molecule for its role as a curing agent. In my early years working near a specialty epoxy plant, the phrase “curing agent” got thrown around a lot. These chemicals start or speed up hardening in plastics and coatings, especially epoxy resins. Epoxy can’t set up properly without amine curing agents, and this compound adds reliability and toughness to the final material. Automotive parts, electronics, aerospace components, wind turbine blades—more often than not, while handling or repairing these, you’re interacting with parts made possible by toughened epoxies.
Paints and coatings manufacturers also count on it for durability. Protective surfaces on equipment, metal structures, and bridges don’t just act as decoration. Paints laced with epoxy deliver hard-wearing outer layers, shrugging off water, salt, and corrosive air. Over the years, I’ve watched public works crews repaint aging bridges, sometimes getting two decades from a good epoxy-based coating thanks to curing agents like 5-Amino-1,3,3-Trimethylcyclohexanemethylamine.
Chemistry Behind Performance
What sets this compound apart from other amines comes down to its structure. The cyclohexane ring and the three methyl groups make for a bulkier amine, which can affect how quickly and evenly it reacts with epoxy resins. This can mean stronger bonding, better heat resistance, and sometimes more predictable results on the production line. Having reliable chemistry at your back helps prevent costly failures—from cracked windmill blades to delaminating electronics casings.
Worker Safety and Environmental Impact
With production comes concern for safety and the environment. In any chemical setting, proper personal protective equipment, safe storage, and responsible disposal remain the bare minimum. Amines like this one can irritate skin and eyes and shouldn’t get into water supplies. My mentor at the plant always said: “Respect what you handle.” Regulations keep evolving as science uncovers new details about health risks and environmental impact.
Green chemistry trends have picked up in recent years. Researchers keep hunting for ways to make tough, reliable resins without leaving a heavy chemical footprint behind. This includes developing safer modifications of old compounds or inventing entirely new ones. Companies rigorously test and document employee exposures, emissions, and accidental release data as part of the regulatory dance.
Where Progress Meets Responsibility
Specialty compounds—especially those with such a complex name—don’t gain traction in industry without bringing performance benefits to the table. Accountability shapes how these chemicals see continued use. Proper worker training, regular reviews of emerging research, and honest environmental reporting make a difference, both for workers and the communities where these plants operate.
Is 5-Amino-1,3,3-Trimethylcyclohexanemethylamine safe to handle?
Getting Real About Handling Chemicals
I’ve spent years around laboratories and chemical plants, and there’s always one rule: never treat a new compound as harmless just because the name isn’t familiar. With 5-Amino-1,3,3-Trimethylcyclohexanemethylamine—let’s just call it ATMCA for short—the biggest risk starts with not knowing enough. Stories of safety slip-ups keep surfacing, where people underestimated a compound because it wasn’t a classic “hazard.” Sometimes it turns out they wish they had paid closer attention to the material's properties.
Basic Properties and Concerns
ATMCA, like other cyclohexanemethylamine derivatives, brings strong amine odors and can irritate the skin, eyes, or lungs. Amine vapors sometimes cause headaches or breathing problems in confined spaces. Gloves and goggles never feel like overkill, especially if your skin is sensitive. Even outside the lab, chemical supply shops warn about proper ventilation when handling strong amines. Anyone who has gotten a splash from an amine knows that burning sensation means you don’t want to take chances.
Looking at chemical safety data sheets, this compound falls into a group described as corrosive and potentially harmful through contact or inhalation. Data from recent industrial toxicology surveys mention eye and mucous membrane irritation as a recurring problem. Some closely related compounds produced allergic responses or skin burns. There’s no long-term research published specifically on ATMCA in humans, but chemical relatives show effects that make extra precautions wise.
Regulatory and Lab Practices
Good Manufacturing Practice (GMP) guidelines encourage limiting direct exposure to any intermediate like ATMCA, especially when its full effects aren’t completely mapped out. Regulatory agencies push for closed systems and point-of-use handling to cut leaks and exposure. The European Chemicals Agency recommends local exhaust ventilation, protective clothing, and training not just for formaldehyde, but also for lower-profile amines. American Chemical Society documentation suggests the same, supporting the idea that any new amine should get the full protective treatment.
Lessons from Hands-On Use
The biggest lesson I’ve learned is never to let routine make you lazy with PPE (personal protective equipment). Even if the packaging shows nothing dramatic and the lab manager shrugs, one accident with a chemical like ATMCA can mean a nasty day. In one plant I visited, routine exposure to low-level amines gave the whole place a chemical haze; after a worker developed serious skin irritation, management doubled down on gloves and fume hoods. People complained about extra steps, but skin complaints dropped.
Finding Safer Ways
Most solution comes down to common sense. Engineering controls like closed bottles, splash guards, and designated workspaces keep accidents contained. Proper labeling and easy-to-read safety sheets stick around in both universities and factories, letting people spot hazards quickly. Pushing for substitution—using less hazardous reagents for the same synthesis—gains traction too. For now, ATMCA sits somewhere between well-known and risky, and anybody working with it makes the process much safer just by staying alert and following proven protocols.
What are the storage requirements for 5-Amino-1,3,3-Trimethylcyclohexanemethylamine?
Understanding the Nature of the Chemical
5-Amino-1,3,3-Trimethylcyclohexanemethylamine stands out as a specialty amine with uses ranging from industrial chemical synthesis to niche research applications. Its amine structure points to reactivity—especially toward water and acids—but also a level of volatility and potential hazard. People responsible for handling this substance need more than a label and a number; they need a clear, practical roadmap for keeping it safe, stable, and effective.
Why Proper Storage Matters
A failure in storage can turn a routine workday into an emergency room visit or bring regulatory trouble no business wants. I’ve witnessed facilities deal with damaged containers, not due to carelessness but through misunderstanding of how amines behave in storage. Chemicals like 5-Amino-1,3,3-Trimethylcyclohexanemethylamine can release vapors that not only irritate the respiratory tract but also help corrode storage area metals. Poorly secured lids or exposure to humidity ramps up decomposition rates, making the compound less useful or even hazardous.
Temperature and Climate Control
Keeping the temperature steady sits at the core of preserving 5-Amino-1,3,3-Trimethylcyclohexanemethylamine. Heat accelerates chemical changes, sometimes generating pressure in the container and causing leaks or bursts. Storage at room temperature, out of direct sunlight, avoids most issues. Cold environments prevent excessive evaporation and reduce vapor risks. I’ve seen labs use dedicated climate-controlled closets with alarms: the peace of mind is worth the higher up-front setup costs, especially for more sensitive stockpiles.
Moisture and Air Exclusion
This amine’s chemistry doesn’t get along with water or high humidity. Water can spark unwanted side reactions, creating new compounds or compromising purity. Airtight containers, preferably made of HDPE or stainless steel, block ambient air and moisture from creeping in. Routine checks on lid seals or gaskets matter here—I recall a bulk storage drum once picked up unexpected moisture after a deteriorated seal, creating a sludgy, unusable mess. Teams swapped to newer, gasketed drums and put a daily check on the inspection schedule. That quick fix ended the problem for good.
Segregation and Compatibility
Mixing strong acids, oxidizers, or even some metal salts with amines creates a recipe for dangerous reactions. Forgetting the necessary distance between storage shelves runs a real risk, especially in crowded stockrooms. Segregation isn’t just a lab safety principle—it saves livelihoods. Separate zones for acids, bases, oxidizers, and organics keep cross-contamination off the table. Physical barriers such as dikes or trays catch spills before they migrate.
Labeling, Record Keeping, and Regulation
Clean, precise labeling on every container isn’t busywork. In the rush of a busy afternoon, a missing label is all it takes to dump an incompatible material inside a drum. Documentation not only satisfies hazard communication standards but makes inventory efficient and less prone to dangerous mix-ups. Pairing storage with a digital tracking system has helped several operations I know reduce both waste and compliance headaches. Regulations require safety data sheets (SDS) on site. Those sheets need regular reviews since best practices evolve with new science and incidents.
Final Thoughts on Everyday Practice
Investing in the right storage for 5-Amino-1,3,3-Trimethylcyclohexanemethylamine pays dividends in productivity, worker safety, and operational continuity. Training everyone to recognize the risks and follow simple but strict routines keeps disasters at bay. From practical experience, small daily habits—tightening lids, recording batch movements, and checking shelf conditions—do more than expensive technology ever will. Safe storage isn’t a paperwork exercise. It’s a careful balance of respect for chemical reactivity and respect for your team’s health.
What is the chemical structure of 5-Amino-1,3,3-Trimethylcyclohexanemethylamine?
Diving into the Structure
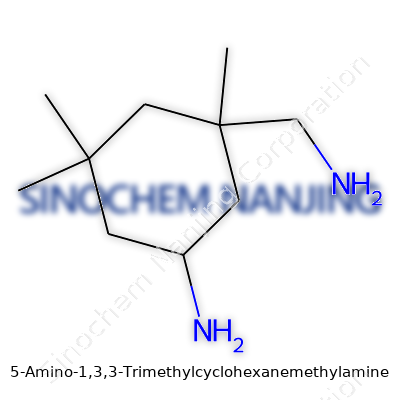
Chemistry often comes down to the shape of molecules and the kinds of atoms that stick together. For 5-Amino-1,3,3-Trimethylcyclohexanemethylamine, the name might spin heads at first glance, yet it reveals a lot about this compound. Picture a cyclohexane ring—six carbon atoms forming a closed loop, like a stop sign if you squint. Now, tack on three methyl groups at the first and third carbons, load an amino group at the fifth carbon, and bolt a methylamine group at the first carbon. This specific architecture gives the molecule both its mouthful of a name and its distinctive properties.
The backbone—the cyclohexane ring—helps this compound stand up to both acidic and basic environments. The three methyl groups, tightly clustered, dial up the hydrophobic character of the molecule. These methyls actually bend the cyclohexane ring out of its usual chair conformation, which can affect how the compound packs or dissolves in various solvents or within biological systems.
Those two amine groups, one attached directly to the ring and the other danging off as methylamine, play key roles in how the molecule interacts with other chemicals. Amines tend to grab onto protons, act as nucleophiles, and get into all sorts of reactions ranging from industrial synthesis to biological signaling. The basicity from these two nitrogens makes the compound reactive, and also marks potential spots for hydrogen bonding—this can make it a player for medicinal chemistry or specialty material creation.
Why This Structure Deserves Attention
It’s easy to overlook how much a chemical’s shape and composition can change what it does. Years of lab experience show that a slight tweak, like adding a methyl group or moving an amine, can flip a compound from being useless to incredibly useful. In the case of 5-Amino-1,3,3-Trimethylcyclohexanemethylamine, its specific arrangement makes it an interesting starting block for many applications. Cyclohexylamines as a class show up in corrosion inhibitors, fuel additives, and even pharmaceuticals. The positioning of groups on the ring sets the stage for everything that follows, making this molecule more than just a chemical curiosity.
Research published over the past decade shows that unique cyclohexylamines can customize polymers or serve as chiral building blocks for new drugs. Demand for new performance chemicals keeps rising, especially as industry pushes for higher selectivity or new kinds of sustainable materials. A molecule like this, with its easy-to-modify ring and basic nitrogens, can lead to pathways for new synthesis work.
Opportunities and Challenges
Not every molecule with a fancy structure gets mass produced. Some fizzle out in the early research stages because they’re too tricky to make, too expensive, or simply don’t deliver in real-world use. Scale-up remains a sticking point: reactions that go smoothly in a flask sometimes fail miserably in an industrial reactor. Cost and safety both matter. Amine compounds often give off sharp odors and can act as skin or respiratory irritants, which means careful handling.
By investing in smart, scalable synthetic methods—such as greener catalysts, fewer solvents, and milder conditions—labs and companies could unlock new ways to produce this kind of amine efficiently. Formal education focuses on mechanistic understanding and practical skills, and I’ve seen firsthand that chemistry coursework often skips over real-life challenges of scale-up, waste, and regulatory hurdles. Bridging those gaps with more hands-on training and tighter industry-academic collaboration could help push these promising molecules from benchtop curiosity to tangible innovation.
Where can I purchase 5-Amino-1,3,3-Trimethylcyclohexanemethylamine?
Understanding the Demand Behind Uncommon Chemicals
Some chemicals hold niche value, yet their role can mean the difference between research progress and hitting a brick wall. 5-Amino-1,3,3-Trimethylcyclohexanemethylamine might look like a string of tongue-twisters, but every chemist or industry insider recognizes it functions as a vital intermediate within specialty synthesis settings. Before even thinking about tracking down a seller, it makes sense to get a handle on why sourcing this compound differs from grabbing everyday lab stock.
Not every supplier deals in the rare. Distributors gravitate toward high-rotation chemicals, and obscure amines often stay off their regular catalogues. The compound might show up in medicinal chemistry routes, custom polymers, or the creation of specific catalysts—places where bulk chemicals never step foot. Labs working on small-batch experimentation or custom syntheses often stumble over finding a source willing to break out of “standard inventory” mode. The roadblocks get real: local distributors ask for large minimum orders, online marketplaces hesitate over exact product identity, and custom synthesis comes with both wait and price tags.
Sourcing Safely: Credible Vendors Only
In this situation, a quick Google search hardly cuts it. Authentic suppliers build their reputation over decades, not clicks. Once, a colleague needed a comparable secondary amine for a biotech prototype; she spent weeks filtering out resellers from global platforms playing fast and loose with chemical names just to locate a respected German distributor. That search taught us all a lesson in patience and vetting. Industrial chemical companies, certified lab suppliers like Sigma-Aldrich, Alfa Aesar, or TCI, and specialists like Fisher Scientific, often set the bar for quality control. These companies operate under strict verification processes and adhere to regulations set down by the US EPA, EU REACH, and other international standards. Sourcing through these channels costs more, but the product data, certificates of analysis, and handling advice come baked right into the experience.
Some buyers wander into sketchier corners of the market searching for deals. This path rarely pays off. Fake products, mislabeled chemicals, dangerous impurities—each comes with heavy consequences for health, data integrity, and legal compliance. Responsible labs keep their receipts and certificates on file for a reason: traceability covers you. No reputable research institution or startup wants to risk project grants and safety just to shave a few dollars off the bill.
Legal and Safety Hurdles: Not Worth Skipping
Chemicals like 5-Amino-1,3,3-Trimethylcyclohexanemethylamine frequently show up on “watched” lists. Purchasers must confirm compliance with hazardous materials laws and licensing obligations before cash ever changes hands. Labs that miss a beat on these regulations may get stuck in customs delays or worse, face investigation. I’ve seen well-meaning researchers fall into gray areas simply out of ignorance. If in doubt, a call to your institution’s chemical safety officer or procurement department clears confusion up early.
Practical Solutions: Building the Right Networks
Making connections makes future sourcing easier. Larger institutions often share supplier reviews or organize bulk purchases for specialized reagents. Online chemistry forums and professional groups sometimes recommend sources—not just for the sale, but for technical know-how. Specialty suppliers occasionally provide small research quantities upon request, particularly for universities and accredited labs. Building a relationship with these vendors gives access not just to product, but also to troubleshooting—some even suggest alternatives if they know you’re facing a tight deadline or a patent roadblock.
Few shortcuts emerge when science leans on transparency and reliability. Chasing rare chemicals means asking tough questions, verifying every line of paperwork, and looking beyond tempting lowball offers. Trust comes with a price tag, but it consistently pays off for labs determined to keep their work above board, safe, and academically credible.
| Names | |
| Preferred IUPAC name | N-methyl-5-amino-1,3,3-trimethylcyclohexan-1-amine |
| Other names |
ISR 533 NSC 38643 |
| Pronunciation | /ˈfaɪ əˈmiːnoʊ waɪ ən ˈθri ˈθri traɪˈmɛθəl ˌsaɪkloʊˈhɛksən ˈmɛθəl əˈmiːn/ |
| Identifiers | |
| CAS Number | 30092-37-0 |
| 3D model (JSmol) | `3Dmol.js:load=JSmol,pdb:CC1(CC(CC(C1)(C)N)N)C` |
| Beilstein Reference | 1598734 |
| ChEBI | CHEBI:189017 |
| ChEMBL | CHEMBL1523635 |
| ChemSpider | 22938306 |
| DrugBank | DB16625 |
| ECHA InfoCard | 100.197.768 |
| EC Number | 211-996-3 |
| Gmelin Reference | 107211 |
| KEGG | C06735 |
| MeSH | D08.811.277.040.360.024.500 |
| PubChem CID | 11665012 |
| RTECS number | UY3150000 |
| UNII | 7U0J8F5A6Y |
| UN number | UN2735 |
| Properties | |
| Chemical formula | C10H22N2 |
| Molar mass | 170.31 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Ammonia-like |
| Density | 0.896 g/mL |
| Solubility in water | Soluble in water |
| log P | 0.51 |
| Vapor pressure | 0.18 mm Hg (25°C) |
| Acidity (pKa) | 10.70 |
| Basicity (pKb) | pKb = 3.27 |
| Magnetic susceptibility (χ) | -7.36×10^-6 cm³/mol |
| Refractive index (nD) | 1.482 |
| Viscosity | Slightly viscous liquid |
| Dipole moment | 3.05 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 322.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -120.8 kJ/mol |
| Pharmacology | |
| ATC code | N06DX01 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes severe skin burns and eye damage. Causes serious eye damage. Harmful to aquatic life. |
| GHS labelling | GHS07, GHS05 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H314, H317, H412 |
| Precautionary statements | P261, P280, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | > 101 °C |
| Autoignition temperature | 270 °C |
| Lethal dose or concentration | LD50 Oral Rat 910 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 > 2000 mg/kg |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.03 ppm |
| IDLH (Immediate danger) | Not listed |

