5,6,7,8-Tetrahydro-1-Naphthylamine: A Deep Dive into Its Impact and Promise
Historical Development
Decades ago, curiosity about naphthalene’s transformations led researchers down the path to new amine derivatives. Chemists wanted amines that fit into complex molecules for dyes and pharmaceuticals. The emergence of 5,6,7,8-tetrahydro-1-naphthylamine came as synthetic organic chemistry matured. Thoughtful experimentation with catalytic hydrogenation in the post-war era made the selective tetrahydrogenation of naphthylamines possible—efforts that opened up unique intermediates for later use. Academic papers from the 1960s and 70s started mentioning this compound as a useful building block, though it never enjoyed blockbuster fame. Even as attention wandered, specialty chemical makers found ways to nurture small but steady demand. Companies with keen eyes for new pharmacophores or novel materials kept this amine on their shelves, appreciating its versatility long before broader commercial applications surfaced.
Product Overview
5,6,7,8-Tetrahydro-1-naphthylamine sits in a category of niche amines—organic compounds that often serve as the backbone for making bigger molecules. Its partially saturated naphthalene ring gives it certain unique advantages. The extra hydrogen atoms reduce aromaticity just enough to make downstream reactions easier, all while keeping the rigidity and electron distribution helpful for further synthesis. When labs want to make specific dyes, polymers, or drug candidates, this amine becomes more interesting than simpler anilines or cyclohexylamines. Rather than being just a starting material, it also plays a role as an intermediate for chemical modifications, small-scale R&D projects, or pilot-scale batches. Manufacturers with active R&D departments recognize its flexibility, using it both as a key raw material and as a model compound for new reaction methods.
Physical & Chemical Properties
In person, this compound usually appears as a colorless to pale yellow oily liquid. It doesn’t have the harsh, nauseating smell associated with aromatic amines, though it carries a distinct chemical note. Boiling points often line up around 250°C, depending on purity. It melts at low temperatures, which helps with handling and measuring, especially during prep work. This amine dissolves nicely in common organic solvents such as ether, chloroform, and benzene, but shows only moderate water solubility. Its molecular formula, C10H13N, reveals a compact structure, with a tetrahydro framework stabilizing the amine group. The compound reacts as expected for a secondary amine—ready for acylation or alkylation, but tough enough to hold up against mild bases or acids. Lab practitioners appreciate this stability. In air, it’ll darken if left exposed for too long, so smart storage is key.
Technical Specifications & Labeling
Suppliers who specialize in laboratory chemicals offer 5,6,7,8-tetrahydro-1-naphthylamine at purities above 98%. Labels give basic identifiers: chemical name, CAS number, molecular weight (147.22 g/mol), and lot-specific details about assay. Good suppliers include reports on trace impurities, as these can make or break reactions in sensitive syntheses. Bottles arrive sealed under inert gas when high purity matters—a practice borne out of lessons learned from careless oxidations. Buyers routinely request Safety Data Sheets, aware that regulatory agencies set high bars for exposure and risk. Even research-grade shipments come with batch certificates, reflecting a persistent culture of quality assurance rooted in both regulatory demand and the lived experience of avoiding failed experiments due to material inconsistencies.
Preparation Method
Producing 5,6,7,8-tetrahydro-1-naphthylamine reliably demands control over both hydrogenation and amination steps. Most routes start with 1-naphthylamine. Hydrogenate the molecule in the presence of a ruthenium or palladium catalyst under gentle pressure. This catches the four sites that need hydrogenation, leaving the core structure intact. Some labs skip direct amination of tetrahydronaphthalene, but the consensus holds that starting with the amine gives better selectivity. Catalysts need frequent regeneration, as heavy use produces fouling—something seasoned process chemists learn quickly. Reaction conditions matter as well: Too much temperature, and new byproducts show up; too little hydrogen, and yields crumble. Finished products get distilled under reduced pressure, eliminating high-boiling residues. Large-scale operators have invested in closed systems that minimize operator exposure and protect air quality, responding to both safety knowledge and evolving regulation.
Chemical Reactions & Modifications
This amine offers a handy launching pad for all sorts of modifications. In the hands of a skilled chemist, it easily accepts alkyl groups onto the nitrogen. Acylation leads to key intermediates for specialty dyes and polymers. If you need to build out more complex pharmacophores—for example, prepping molecules that fit into neuroreceptor binding pockets—the tetrahydronaphthyl framework provides rigidity with a hint of flexibility. Reductive amination is smoother than with simple naphthylamines, since the saturated ring resists undesired overreactions. Formylation, sulfonation, and even selective halogenation all proceed in predictable fashion, delivering functional materials for ongoing research. These features help explain its gradual entrance into custom synthesis offerings by contract manufacturing organizations. Chemists who have spent time troubleshooting stubborn routes often return to this compound, appreciating the balance of reactivity and resilience.
Synonyms & Product Names
Literature and suppliers use several synonyms: 5,6,7,8-tetrahydro-1-naphthylamine, THNA, and in pharmaceutical contexts, sometimes “tetrahydronaphthylamine” without specifying position. Careful researchers double-check numbering, since mislabeling has tripped up more than one synthetic effort. Some catalogs use less precise terms like “hydronaphthylamine”; these usually reference the same compound, but confirm by structure. Chinese and European sources sometimes assign in-house product codes or trade names. Anyone buying or interpreting older journal data tends to review both structure and spectra, rather than trusting nomenclature alone.
Safety & Operational Standards
Workers planning to use this amine treat it with healthy respect. Skin and eye protection come standard, and good ventilation remains a must. Though not as acutely toxic as aniline, it can cause irritation or sensitization if handled poorly. Chemical hygiene plans flag it as a possible skin absorber. Those running larger scale preparations know how easily spills escape during transfers, so secondary containment and gloves—nitrile or butyl rubber—never get skipped. Fume hoods and glove boxes rule out airborne exposure. Training emphasizes careful waste management, with used solvents and residues collected for incineration or controlled disposal. Labeling must comply with national hazardous materials regulations: pictograms, hazard statements, and clear risk labeling as required by agencies like OSHA, REACH, and GHS. Audits of facilities handling this material have picked up fewer avoidable incidents where teams follow these playbooks.
Application Area
Organic synthesis remains the main game—especially for those preparing intermediates in pharmaceuticals, agrochemicals, or dyes. Medicinal chemists lean on the rigid yet modifiable backbone for candidate drugs affecting the central nervous system. Dye makers use it for color-fast intermediates, since the partially saturated naphthyl ring alters shade and binding behavior on fibers. Some additive manufacturers experiment with it in stabilizer blends for plastics or rubbers. Research groups interested in selective enzymatic transformations pick this amine as a substrate model—its structure gives insight into the role of ring saturation in enzyme binding and turnover. Chemical biology work capitalizes on its amine group for fluorescent tagging and building ligand libraries. Downstream products disperse into medicine, materials science, and specialty coatings.
Research & Development
Investigators in drug discovery and fine chemicals have spent decades exploring 5,6,7,8-tetrahydro-1-naphthylamine’s subtle features. Its structure pops up as a motif in patents for CNS-active agents, antimicrobials, and anti-cancer scaffolds. Medicinal chemists like its blend of aromaticity and conformational constraint—a rare mix that can increase receptor specificity. Some R&D teams in polymer chemistry probe how the saturated naphthyl ring influences polymer flexibility and stability. A handful of groups have published methods to attach PEG, sugars, or custom linkers, broadening its reach into biomedical materials. Graduate students tinkering with new coupling reactions often select this compound, since it reveals both successes and shortcomings more reliably than many "easier" substrates. Academic collaborations between industry and universities continue to drive new applications.
Toxicity Research
Animal- and cell-based studies from the last twenty years suggest this compound has moderate systemic toxicity, though nowhere near the risks posed by more volatile or reactive aromatic amines. Acute exposures in rodent models pointed to mild hepatic effects at high doses, while repeat-dose protocols flag mild nephrotoxicity after prolonged exposure—results consistent with most secondary aromatic amines. In vitro screens trigger mutagenicity alarms at concentrations higher than typical exposures; overall, findings fall in the caution-but-not-panic range. Modern risk management doesn’t wait for trouble, so research facilities and manufacturers monitor worker exposures closely and invest in engineering controls. Public and regulatory interest may still steer further toxicity studies, especially if new applications make human exposures more likely.
Future Prospects
This amine's simple starting point belies the diversity of pathways it opens. Innovations in catalytic hydrogenation, continuous-flow synthesis, and greener solvents make its production more attractive each year. With fragment-based drug discovery on the rise, more research teams want naphthyl-based motifs with controlled saturation levels to fine-tune biological activity. Analytical advances provide clearer pictures of reaction intermediates, knowledge that will push fine chemical scale-up strategies forward. Materials science keeps expanding its appetite for robust amines that can survive demanding thermal or oxidative environments, especially in new polymer and sensor materials. As regulations evolve and consumer safety demands intensify, chemists who can deliver pure, well-documented 5,6,7,8-tetrahydro-1-naphthylamine with a minimum of ecological impact will earn steady demand—cementing its role as a go-to specialty intermediate for years to come.
What is 5,6,7,8-Tetrahydro-1-Naphthylamine used for?
A Closer Look at a Quiet Workhorse
5,6,7,8-Tetrahydro-1-Naphthylamine doesn’t show up in everyday headlines. To most people, it sounds more like a password than something connecting to real-world products. From years spent working with fine chemicals and talking to engineers who source intermediates for manufacturing, I know stories like this often stay hidden behind layers of supply chains. But even chemicals that rarely make it past the warehouse door have a big say in the things we trust and use.
Where It Shows Up
This amine rides under the radar because its life happens away from the spotlight. Most buyers pick up this compound not for end-consumer goods, but for what happens several steps earlier. 5,6,7,8-Tetrahydro-1-Naphthylamine serves as a building block for dyes, agrochemicals, and specialty pharmaceuticals. Its rigid naphthalene backbone lets chemists stick on all kinds of useful groups, turning it into a tool for making advanced materials or drug candidates. One conversation with a textile chemist a few years back reminded me how much these molecules matter for dye stability; the right precursor holds color fast under harsh washing, which means less waste and longer-lasting clothes.
Some specialty rubber products come out more durable by using ingredients made from this amine, according to technical papers from industry consortiums. Plants use derivatives of naphthylamine compounds to generate herbicides and pesticides with tailored performance, when the old generic ones can’t do the job anymore. With regulators getting stricter on chemical residues, reliable molecules like this one let formulators design products with specific breakdown rates and safety profiles, protecting both farmers and consumers.
Facts to Know
The world of fine chemical manufacturing tends to emphasize reliability and traceability. Compounds like 5,6,7,8-Tetrahydro-1-Naphthylamine get tracked with batch records, and their purity shapes the fate of dozens of downstream reactions. Years ago, an API project I joined for a cardiovascular drug nearly stalled due to an impurity in a supplier’s lot of an amine just like this. Quality control teams often go through days of batches to trace the source, reminding everyone how one weak link affects safety and cost.
Large-volume players focus on non-toxic alternatives and greener synthesis, too. Groups like the American Chemical Society publish case studies urging manufacturers to swap old routes for new ones with less hazardous waste or better atom efficiency. These shifts trickle down—if a precursor like 5,6,7,8-Tetrahydro-1-Naphthylamine gets made in a cleaner way, finished products gain in sustainability and companies meet tighter ESG targets. Even investors now care about how their portfolios line up with greener processes.
Improving the Industry
Investing in transparent sourcing lets buyers verify where sensitive chemicals come from and how they're handled. I’ve seen project managers ask for site visits and third-party audits before signing contracts for intermediates. This scrutiny grows every year, shutting out fly-by-night brokers and encouraging real accountability. Digital tracking, such as blockchain-backed certificates, is no longer an experiment. These technologies are making it harder for poor-quality or counterfeit precursors to slip in.
Some labs team up with universities to streamline reactions using amines like this—one R&D group I worked with shaved hours off their production step and cut raw material waste by nearly 20%. These small victories, multiplied across thousands of processors, cut down pollution and save meaningful money.
5,6,7,8-Tetrahydro-1-Naphthylamine might sound like background noise to most people, but it’s a key character for anyone making durable goods, safer chemicals, or next-generation therapeutics. Watching how companies source it and handle it tells a bigger story about where modern chemistry—responsible chemistry—is heading.
What is the chemical structure of 5,6,7,8-Tetrahydro-1-Naphthylamine?
Getting to Know the Structure
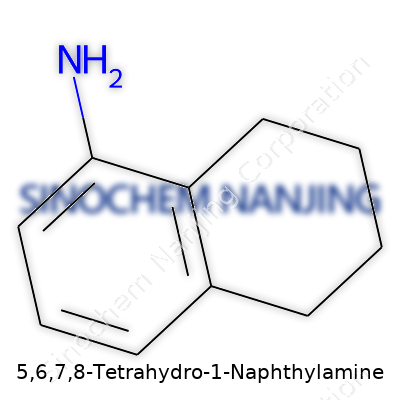
Building molecules often feels like a puzzle. 5,6,7,8-Tetrahydro-1-naphthylamine carves out its own space in the world of chemistry. The base, naphthalene, comes from two fused benzene rings. Take the first ring and add amine (-NH2) at position one. Hydrogenate carbons at 5, 6, 7, and 8. That’s where the “tetrahydro” part lands. It means those carbons swap their double bonds for single ones, fill in with hydrogen, and make the structure less rigid.
Drawing it looks like this: a classic naphthalene backbone, which chemists are quick to spot. Modifying this backbone changes the molecule’s properties. The amine function changes its behavior and makes it useful in synthesis. I’ve spent time in the lab prepping similar aromatic amines. They often wind up in pharmaceuticals and dye manufacturing. The tetrahydro addition opens new doors for the ring’s reactivity and shape, allowing for niche use in labs and industry.
Why Structure Matters
I’ve seen chemical structures make or break a project. This compound’s partially reduced ring stands apart from plain naphthylamines, offering less aromaticity and a more flexible skeleton. Lab tests show these tweaks influence solubility and the way enzymes interact during metabolism. Chemists rely on these differences for designing experiments, especially when scouting for drug precursors.
Think about how adding hydrogens affects the whole platform. Increased hydrogenation can dial back toxic tendencies—something aromatic amines get dinged for. Regular naphthalene derivatives carry mutagenic risks; tweak the core, and you often see a drop in that red flag. Still, the amine group needs careful handling. My time in the lab taught me to trust, but verify, with safety data.
Everyday Impact and Uses
This type of molecule attracts researchers seeking new treatments for neurological disorders. The flexible ring sometimes fits better into biological targets than stiffer, planar structures. One area gaining steam is the development of monoamine oxidase inhibitors. A compound with this structure could mimic or block neurotransmitter breakdown, which has implications for Parkinson’s therapy or mood disorder medications.
Aside from medicine, these building blocks inspire new organic materials. Early in my career, we explored polyaromatic amines for specialty polymers. Shape and electronic properties decide which sector—pharma, materials, or dyes—takes the biggest interest. Flexible naphthylamines are like Swiss Army knives in the hands of a creative chemist.
Addressing Risk and Next Steps
No conversation about aromatic amines finishes without talking environmental and safety impacts. Tetrahydro-naphthylamines earn cautious optimism for being less persistent and less toxic compared to their fully aromatic siblings. Still, I’ve learned that responsible sourcing, vigilant waste management, and close study during scale-up protect workers and the planet alike.
It pays to combine tradition and innovation. Green chemistry tools now let us synthesize compounds like this with fewer byproducts and lower energy. Universities and startups eye these strategies for both economic and ecological reasons. Future breakthroughs will likely depend on deeper profiling—metabolism studies, safer reaction conditions, and roundtable talks between researchers and regulators.
Understanding a structure like 5,6,7,8-Tetrahydro-1-naphthylamine shows what’s possible with a mix of curiosity, rigor, and practical chemistry. Each tweak to the core spurs new ideas, and the conversation keeps moving forward.
Is 5,6,7,8-Tetrahydro-1-Naphthylamine hazardous or toxic?
Looking at the Science Behind the Name
Chemical names often sound intimidating, especially unknown ones like 5,6,7,8-Tetrahydro-1-Naphthylamine. In basic terms, this is an organic compound, a derivative of naphthalene—a structure found in mothballs and some pesticide products. Experience teaches caution around chemicals linked to naphthalene because research shows repeated or high-level exposure to these compounds can mess with human health, especially the blood and the liver.
Why Safety Matters
Handling new or rarely discussed chemicals isn’t just for chemists and lab techs. Many workplaces and academic labs use and store compounds with long names but limited public data. If someone encounters 5,6,7,8-Tetrahydro-1-Naphthylamine in a lab, factory, or storage room, questions about toxicity need clear answers. Trust and safety both rely on honest information.
What the Literature Says
Studies on similar structures—naphthylamines—uncover some real red flags. For example, beta-naphthylamine has official recognition as a human carcinogen, especially linked to bladder cancer. The closer a chemical’s structure is to a known hazardous substance, the more cautious people should get. Most safety data sheets on these sorts of compounds, if available, place lots of emphasis on wearing gloves, eye protection, and working in a well-ventilated space, making it clear inhaling dust or vapors is a real risk.
Direct research on 5,6,7,8-Tetrahydro-1-Naphthylamine turns up a pretty thin record, which should raise eyebrows. If a chemical isn’t widely tested, nobody knows how it affects the nervous system, causes irritation, or travels around in the body. Some safety resources even flag it as a “suspected” irritant to skin and eyes, and potential issue for aquatic life. That sets off alarm bells for lab managers, safety officers, and students alike.
Experience and Risk Management in the Real World
Lab accidents can feel preventable in hindsight—one lax moment and trouble finds you. I've seen colleagues handle odd chemicals with the same gloves they wore outside the lab, forgetting how easily certain substances seep into the bloodstream through skin contact. Even compounds that haven’t been flagged as dangerous burn, sting, or irritate lungs after a spill. The best lesson: treat the unfamiliar with the same respect as the proven hazard.
Where Solutions Start
Anybody storing or using 5,6,7,8-Tetrahydro-1-Naphthylamine should push for a Safety Data Sheet (SDS). If a supplier can’t or won’t provide one, that’s a sign to rethink the purchase or ask for help from a chemical safety officer. Companies like Sigma-Aldrich, Fisher Scientific, and government agencies like OSHA invest years creating sheets packed with instructions for storage, disposal, and first-aid steps in plain English.
Training and ventilation form the backbone of chemical safety. Schools and workplaces need to invest in regular training sessions that go beyond the required minimum. A fume hood or strong airflow system can make the difference between a safe experiment and a trip to the emergency room. Wearing proper gloves and goggles needs to become second nature, not an afterthought. Responsible management includes proper disposal—nobody benefits from flushing chemicals down the drain or throwing them in office trash.
Respect the Unknown
New or little-known chemicals like 5,6,7,8-Tetrahydro-1-Naphthylamine remind us not to trust just absence of evidence. If health and safety data haven’t caught up with the lab supply chain, play it safe, ask questions, and keep people protected. Health is hard to replace, but simple steps and curiosity about every substance make all the difference.
What are the storage conditions for 5,6,7,8-Tetrahydro-1-Naphthylamine?
Everyday Chemistry, Real Problems
Anyone who’s worked in a lab for long enough knows that chemical storage matters as much as the projects themselves. Some compounds spoil with a little heat. Others need a blast of nitrogen just to keep them honest overnight. With 5,6,7,8-Tetrahydro-1-Naphthylamine, a few core rules stand the test of time. Skipping these steps can mean a spoil of the batch, a lab evacuation, or worse. The stakes aren’t just about product loss, but the safety of anyone nearby.
The Real Reason for Tightly Sealed Containers
I’ve seen amines break down sitting on a crowded shelf. They suck up moisture from the gulliest humidity, then brown or sour almost overnight. With this compound, air-tight storage pays dividends. Glass containers with PTFE-lined caps hold up better than most substitutes, especially if the bottle gets handled often. The last thing anyone wants is a surprise reaction with oxygen. Nitrogen or argon flushes buy peace and reduce fire risk. You’re not doing chemistry if you’re dodging explosions in the storeroom.
Temp Check: Not Optional
Ambient warehouse temperatures jump around the world. Working summers in small university labs, the air in storerooms got stifling unless you kept chemicals cool. Most literature calls for 5,6,7,8-Tetrahydro-1-Naphthylamine to stay in the 2–8°C range. Standard refrigerators do a fine job. Flammable-proof fridges always bring a second level of safety, especially if the material sits next to anything volatile. Avoiding the sun and sudden swings keeps this compound stable and easy to work with later.
Drama with Water and Light
I’ve spilled water on an open bench before. It runs quick, unexpected, and can create big headaches. Amines hate moisture. One cracked seal invites water, and the breakdown begins—nasty smells, off-color residues, and a ruined sample. Lighting rarely helps either. Extended sun tanning doesn’t just fade paint; it wrecks many organic compounds. A dark or amber container, parked on a shelf away from direct sunlight, protects both the molecule and people working with it.
Labeling and Lab Culture
Label everything, every time. Years back, I found an unlabeled vial at the back of the fridge. Sure, some folks track every bottle with a notebook, but for others, those scrawled initials and “THN” scribbles mean nothing in a week. Precise labels stop confusion and prevent mix-ups. Keep a log of who opened what and when—some labs install RFID tags now. It’s not just bureaucracy. If an accident occurs, these records matter more than most realize.
Disposal: Beyond the Lab Manual
Accidents rise when folks treat leftover chemicals like old dishwater. Dumping unused amines into the sink pushes trouble downstream. Institutions post detailed disposal guidelines, and for good reason. You send hazardous waste to a licensed handler, not down the drain or to a landfill. Training refreshers remind everyone what goes where and how to handle a spill, so the risk stays low and fines stay away.
Room for Better Practices
Many labs coast on “good enough” until something goes sideways. Investing in better cooling, locking cabinets, and regular training always pays for itself. Reporting near-misses helps spot weak points early, and sharing advice across teams builds a safer routine. Storage isn’t busywork—it’s the backbone of good science and healthy workplaces.
Where can I purchase 5,6,7,8-Tetrahydro-1-Naphthylamine?
The Search for 5,6,7,8-Tetrahydro-1-Naphthylamine
Finding specialty chemicals like 5,6,7,8-Tetrahydro-1-Naphthylamine isn’t as simple as dropping into a store or browsing regular ecommerce sites. This isn’t a household cleaning product. I’ve spent years diving into research labs and have seen how chemicals like this raise questions not just about sourcing, but also about safety, regulations, and responsibility.
What Experience Tells Me About the Chemical Marketplace
Anyone who works in research, pharmaceuticals, or advanced manufacturing knows the reality of procurement. Suppliers for compounds like 5,6,7,8-Tetrahydro-1-Naphthylamine stick to established protocols. Only companies or institutions with real credentials get past the front door. It’s not because suppliers want to make life difficult; rules protect everyone – workers, neighbors, the public at large, and the reputation of those buying and selling these chemicals.
Why Regulation Exists
Tighter controls have nothing to do with red tape for the sake of it. Specialty chemicals carry risks: personal safety, environmental health, even potential misuse. A single mishap with a toxic or reactive compound can ruin a lab, harm the natural environment, or even impact local economies if something gets released. Many times, I’ve seen requests for chemicals get bounced because the end-use wasn’t clear, or the buyer lacked documented training.
What the Sourcing Process Looks Like
Getting 5,6,7,8-Tetrahydro-1-Naphthylamine usually starts by consulting catalogs from reputable chemical suppliers. Names like Sigma-Aldrich, TCI America, Alfa Aesar, and Fisher Scientific appear in many labs. Responsibility falls on buyers to check product listings, send queries, and supply paperwork proving research purpose and technical capability. More than filling out forms, accountability for safe storage, transportation, and disposal forms the backbone of the chemical supply chain. Any supplier worth its salt will ask for company credentials, details on intended use, and adherence to national and international laws.
What Makes Finding Specialty Chemicals a Challenge
The harder part isn’t just placing an order – it’s navigating import/export controls, hazardous substance regulations, and local licensure for possession. I’ve seen teams lose weeks to compliance paperwork, especially when shipping cross-border. That frustration is real, but there’s an important reason for those delays. Even routine chemicals became tools for harm in the wrong hands, so oversight matters.
Building Safer, Smarter Procurement
Better pathways exist. Some research consortia and universities now pool requests and use shared licensing for sensitive compounds. This reduces the exposure of amateur buyers and keeps untrained hands away from risky materials. Digital records and transparent reporting strengthen accountability. Training updates help staff spot red flags or weaknesses in chemical handling systems. Investing in a thorough vetting process—no shortcuts—saves trouble later and reflects respect for science and public good.
Key Facts and What to Watch Out For
Without verified credentials, direct access to 5,6,7,8-Tetrahydro-1-Naphthylamine simply won’t happen. Proper vetting stems from hard lessons learned worldwide, often the hard way. Scams and grey-market websites make big promises, but behind the glossy pages often lies risk: impure products, fake documentation, or legal trouble for both buyers and sellers. Protecting yourself means sticking with recognized suppliers and insisting on clear quality control, certifications, batch analysis results, and safety data sheets—every single time.
| Names | |
| Preferred IUPAC name | 5,6,7,8-tetrahydro-1-naphthalenamine |
| Other names |
5,6,7,8-Tetrahydro-1-naphthylamine Tetrahydro-1-naphthylamine 1-Amino-5,6,7,8-tetrahydronaphthalene 1-Naphthylamine, tetrahydro |
| Pronunciation | /ˌfaɪv.sɪks.sev.ən.eɪt ˌtɛt.rəˌhaɪ.drə wʌn næfˈθɪl.əˌmiːn/ |
| Identifiers | |
| CAS Number | 1005-80-9 |
| 3D model (JSmol) | `JSmol.loadInline("data/mol; 5,6,7,8-Tetrahydro-1-Naphthylamine; CC1CCCc2cccc(N)c12")` |
| Beilstein Reference | 1721347 |
| ChEBI | CHEBI:35498 |
| ChEMBL | CHEMBL2107736 |
| ChemSpider | 141214 |
| DrugBank | DB08357 |
| ECHA InfoCard | 05a3a5fe-6a1c-4e02-be4a-038b7763b3c5 |
| EC Number | 2123-70-2 |
| Gmelin Reference | 135193 |
| KEGG | C08391 |
| MeSH | D016692 |
| PubChem CID | 70046 |
| RTECS number | QJ8925000 |
| UNII | P7189I65N5 |
| UN number | UN2810 |
| CompTox Dashboard (EPA) | DTXSID4020845 |
| Properties | |
| Chemical formula | C10H13N |
| Molar mass | 149.24 g/mol |
| Appearance | white to light brown solid |
| Odor | Fishy |
| Density | 1.02 g/cm3 |
| Solubility in water | slightly soluble |
| log P | 1.89 |
| Vapor pressure | 0.17 mmHg (25°C) |
| Acidity (pKa) | 10.03 |
| Basicity (pKb) | 4.21 |
| Magnetic susceptibility (χ) | -6.26×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.6130 |
| Viscosity | 1.18 cP (20°C, lit.) |
| Dipole moment | 2.35 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 296.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -12.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3943 kJ mol-1 |
| Pharmacology | |
| ATC code | N06BX10 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H312, H332, H412 |
| Precautionary statements | P261, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | > 110 °C |
| Autoignition temperature | 630 °C |
| Lethal dose or concentration | LD50 oral rat 307 mg/kg |
| LD50 (median dose) | LD50 (median dose): 312 mg/kg (oral, rat) |
| NIOSH | NA9586 |
| PEL (Permissible) | PEL (Permissible): Not established |
| REL (Recommended) | 100 mg/L |
| Related compounds | |
| Related compounds |
1-Naphthylamine 2-Naphthylamine Naphthalene Tetralin (1,2,3,4-Tetrahydronaphthalene) 1,2,3,4-Tetrahydro-2-naphthylamine |

