4-Vinylpyridine: A Closer Look Across Science and Industry
Historical Development
The story of 4-vinylpyridine takes shape in the laboratories of the early twentieth century, a time when chemists worldwide chased new building blocks for the growing polymer and pharmaceutical industries. Pyridines were already old friends, bustling with potential since the days of organic synthesis’ infancy. Substituted vinyl derivatives looked especially promising when researchers realized that they could tweak existing molecules into unique players for more tailored applications. By the mid-1900s, 4-vinylpyridine no longer belonged to the realm of strange lab curiosities. Its straightforward structure—vinyl attached to the pyridine ring at the para position—transformed it into a serious candidate for specialty polymers and advanced chemical synthesis. Of all the pyridine isomers, the 4-position’s distinct electronic environment offered improved reactivity and handling for folks searching for more control in both production and application.
Product Overview
In industry and research, 4-vinylpyridine shows up as a clear to pale yellow liquid. This isn’t some esoteric lab reagent that sits untouched on a shelf; production runs both small and large serve a global market that values 4-vinylpyridine for its role in resin and copolymer manufacturing, as well as its knack for boosting chemical diversity in synthesis work. Suppliers tend to offer the compound with a high stated purity—typically above 95%—for those who want predictable outcomes. Industrial clients often ask for larger drums sealed tightly, whereas researchers prefer smaller, more manageable bottles. The commodity status of 4-vinylpyridine stems from both its routine utility and the careful regulatory packing demanded for anything this reactive.
Physical & Chemical Properties
4-vinylpyridine behaves much like its vinyl and pyridine cousins. One whiff reveals its sharp, almond-like odor, a reminder to reach for the gloves and protect the nose. Sitting just below room temperature at around 60°C (140°F), its boiling point matches what chemists expect for light organic liquids. The compound mixes well with most organic solvents, but water only takes in a modest amount. Its molecular formula—C7H7N—places it at 105.14 g/mol. The vinyl group craves electron-rich partners for reactions, while the nitrogen atom brings an adaptable basic site, perfect for forming salts or grabbing protons on demand. There’s an edge to its reactivity, thanks to conjugation across the pyridine ring, that makes it invaluable in specialized modification steps.
Technical Specifications & Labeling
Manufacturers label bottles with all the details—CAS: 100-43-6, purity, batch number, manufacturing date, and storage notes like “Keep cool. Protect from light.” MSDS sheets walk users through recommended PPE, first aid, and fire protocols, and there’s zero patience for ambiguity. Some producers provide extra data for compliance with regional standards, since both the EU and North America agree on the importance of tracking hazardous ingredients. Certificates of analysis come with technical quality indicators like water content (often under 0.1%), color, and GC purity. This data helps researchers and process managers avoid unwanted variables in the final product. That forms the backbone of every serious order for a substance as tightly regulated and functionally important as 4-vinylpyridine.
Preparation Method
Making 4-vinylpyridine isn’t a DIY kitchen experiment. Commercial synthesis usually starts with 4-methylpyridine, a starting material produced in bulk for the vitamin B3 market. Chemists treat 4-methylpyridine with bromine to load a bromine atom onto the methyl group, often forming 4-(bromomethyl)pyridine. Next, a base helps knock off the bromine and create a reactive intermediate. Skilled chemists coax the molecule through a dehydrohalogenation step, pulling off both hydrogen and bromine to string out a double-bonded vinyl group. Each step is carefully watched—impurities can sneak in from skipped purification, and safety rules demand robust ventilation and poison controls due to exposure hazards at multiple points. What lands in the bottle: a vinylpyridine pure enough for polymerization, fine chemicals, and lab research.
Chemical Reactions & Modifications
The double bond shakes hands with a variety of reactants. For polymer chemists, that vinyl sits ready to dive into free-radical or ionic polymerizations, latching onto styrene or acrylonitrile for specialty copolymer chains. The pyridine ring, for its part, accepts functional groups at the nitrogen, making the compound interesting for quaternization and N-oxidation. Cross-linking possibilities widen further by using 4-vinylpyridine as a comonomer—ion-exchange resins, protein separation membranes, or advanced adhesives stem from these creative matchups. Synthetic chemists benefit from the nitrogen’s lone pair in catalysis or as a leaving group in tailored reactions. Every tweak builds in options for custom functionality, from selective reagent carriers to next-generation composite plastics.
Synonyms & Product Names
A few different names circulate in chemical catalogs: 4-vinylpyridine, para-vinylpyridine, 4-ethenylpyridine, and sometimes just abbreviated “4-VP.” Trade names depend on the supplier, with some using proprietary codes, but global commerce relies most on IUPAC’s clear structure: “1-ethenyl-4-pyridine.” Researchers and engineers spot these names on container labels, regulatory documents, and analytical reports, ensuring there’s no confusion across languages or technical levels. This clarity supports chemical tracking, inventory checks, and compliance efforts in both academic and commercial settings.
Safety & Operational Standards
Everyone working with 4-vinylpyridine hears the reminders about toxicity and flammability. Direct skin contact brings risks of burns, eye injuries, and allergic reactions. Safety goggles, nitrile gloves, and fume hoods make up non-negotiable equipment whenever handling the compound outside sealed systems. Inhalation hazards come from both liquid and vapor, and spills—no matter the size—trigger full cleanup protocols. Storage stays under lock, in ventilation-equipped chemical cabinets, with far distance from oxidizers or acids. Fire control teams look for dry chemical or CO2 extinguishers if things go wrong. Most regulatory authorities limit workplace exposure, and employers must provide hazard education, written procedures, and prompt medical responses when exposure takes place.
Application Area
4-vinylpyridine shows up everywhere people need new materials with select properties. Specialty polymers capture metals, remove contaminants, and act as medical carriers. Textile manufacturers look for coatings or fiber modifiers that can stand up to tough treatment or intense dyeing. Water treatment plants rely on ion-exchange beads prepared partly from 4-vinylpyridine copolymers—helping pull out heavy metals or unwanted ions from supply streams. Paint formulators add small amounts to anchor anti-corrosive layers onto iron and steel. Adhesive makers find value in the unique polarity of the pyridine ring, which sticks better to certain surfaces than non-polar alternatives. In pharma, 4-vinylpyridine acts as a synthon for more complex molecules, unlocking new medicinal scaffolds. Its versatility shapes countless finished products, both visible and behind the scenes.
Research & Development
Academic work keeps shining new light on 4-vinylpyridine, since its basic structure gives birth to so many creative possibilities. Polymer science groups test different monomer blends, observing how small tweaks change flexibility, thermal stability, or adhesion properties. Nanomaterials labs build custom surfaces by binding modified 4-vinylpyridine copolymers to carbon nanotubes or silica, searching for improved biosensors or environmental monitors. Bioconjugation research explores how pyridine sites interact with proteins or enzymes for next-gen drug delivery or diagnostics. Each publication or patent reveals another facet—a new angle for manufacturers or clinicians searching for reliable, customizable chemistry.
Toxicity Research
Health risk studies paint a careful picture of both acute and chronic hazards. Short-term exposure causes skin and eye irritation; even brief inhalation leads to nose and throat discomfort. Animal studies highlight liver and kidney changes at moderate doses, flagging potential for long-term organ impacts. Carcinogenicity results remain mixed, so regulations classify 4-vinylpyridine as a hazardous material needing strict workplace controls and medical surveillance in ongoing production settings. Environmental scientists look closely at breakdown products—since vinyl-containing aromatic compounds can persist in soil or water and affect aquatic species. Manufacturers work alongside regulatory bodies to monitor effluent, air releases, and worker health using regular testing and incident logs. Responsible chemical stewardship stays at the forefront for everyone involved.
Future Prospects
Directions for 4-vinylpyridine stretch from greener polymer synthesis to advanced functional materials. Demand for selective adsorbents in water cleanup, carbon capture, or rare metal recovery keeps growing, and manufacturers hunt for tailored resins that outperform today’s options. In medicine, research teams envision modified 4-vinylpyridine surfaces as platforms for smart drug release or as scaffold materials guiding tissue regrowth after injury. Electronics designers see opportunities for new conductive or semiconductive blends as industries reach for lighter, smaller, and more adaptable devices. Each leap forward borrows from lessons learned through decades of development, while regulatory landscapes push for safer, cleaner, and more sustainable chemistries. The compound’s journey stands far from finished—every new application inspires another round of study, debate, and reinvention within science and industry.
What is 4-Vinylpyridine used for?
What 4-Vinylpyridine Actually Does
4-Vinylpyridine might sound like something you’d only see on a chemist’s shelf, but this colorless liquid actually plays a starring role in a lot of things people rely on every day. Chemists use it mostly for its ability to build bigger, more complex molecules — it acts like a building block. If you’ve noticed how modern materials keep getting smarter and more versatile, there’s a good chance compounds like 4-vinylpyridine had a hand in that progress. For example, it helps make polymers stickier, tougher, or even better at grabbing onto specific molecules.
How Industry Puts It to Work
Big industries count on 4-vinylpyridine for more than just a science experiment. Take rubber, as an example. It’s easy to assume tires and hoses just come straight from latex, but the process ties in plenty of chemistry. Manufacturers add 4-vinylpyridine to the mix, so the rubber can adhere to metal or fabric reinforcements. Without it, tread might peel easier, and hoses could split quicker. It’s not about being fancy; it’s about making stuff last.
Another big use shows up in water treatment. A lot of purification systems rely on resins, which are specialized plastics that filter out impurities. 4-Vinylpyridine goes into these resins, giving them a kind of “magnetic” hand to grab stuff like heavy metals and even trace chemicals. Cities depend on clean water; having smarter ways to purify it benefits everyone.
Scientific Research Leans on It
Research labs have a soft spot for 4-vinylpyridine because it works as a crosslinker, helping researchers tie tiny molecules together for things like diagnostics. Labs also use it to tweak DNA or proteins for drug development and bioengineering. Getting these tweaks right supports better medical treatments. Precision matters here. Mistakes slow things down — careful chemistry makes labs more confident in their results.
Safety and Environmental Concerns
Like a lot of specialty chemicals, 4-vinylpyridine demands respect in the workplace. Inhaling its vapors causes eye and lung irritation, and direct contact with skin can be risky. I’ve seen lab teams put a lot of effort into airtight storage and proper ventilation. You don’t want to take shortcuts just because you’re familiar with something. Companies that handle this chemical follow strict safety rules, backed by decades of workplace research, and keep training new workers to do the same. Mistakes could have real costs, from accidents to environmental spills.
On the environmental side, there’s a push to make sure leftover material doesn’t end up in the wrong place. Labs and factories collect all their waste and process it using chemical treatments or incineration, following legal standards. These aren’t just hoops to jump through. Cleaner manufacturing means fewer headaches for neighbors, and it aligns with growing expectations for greener industry. In my career, I’ve seen regulators inspect labs for compliance, sometimes unannounced. Knowing someone’s watching keeps everyone sharper about waste management.
Looking Ahead
As research marches onward, there’s growing interest in designing new molecules that do what 4-vinylpyridine does — maybe even better and safer. Some folks hope for bio-based alternatives down the road. For now, though, this compound keeps showing up wherever toughness, stickiness, or selective filtering matters. Its contributions feel invisible only until you pause to ask how everyday products really come together.
What are the safety precautions when handling 4-Vinylpyridine?
Why Respecting 4-Vinylpyridine Matters
Anyone who’s worked around chemical labs knows you can’t treat every bottle the same. 4-Vinylpyridine brings pungent vapors and a reputation for skin and respiratory irritation. Mistakes aren’t fun here: Splash this on your arm, you feel it. Inhale its fumes, and your nose lets you know immediately. The experience really sticks with you, and not in a good way. It brings home a simple truth—safety training exists for a reason.
What Real Safety Looks Like
Goggles may steam while you’re handling flasks, but you don’t skip them. 4-Vinylpyridine can hurt your eyes badly if anything splashes, and a simple pair of chemical splash goggles stands between your vision and an ER visit. Nitrile gloves last longer than latex against this substance—nobody wants to change gloves every ten minutes, and thin gloves rip when things get messy.
I remember a grad student who tried moving fast, didn’t check the glove box—ended up with nasty red hands before he made it to the eyewash. The skin burning lasted hours. That happened because gloves were chosen for comfort, not resistance.
Ventilation Keeps the Room Bearable
Often in chemistry, the smell tells you more than the color or clarity. 4-Vinylpyridine stinks strong and sharp, way past “unpleasant.” Good labs keep this work in fume hoods; they aren’t fancy extras, they’re daily lifesavers. Without proper airflow, your lungs take a beating. The headaches and nausea prove this chemical means business. Respiratory exposure builds up over time, and chronic effects call for regular monitoring—the simple air badge on your coat never feels like overkill around this compound.
Spill Happens: Quick Response Counts
Even the neatest hands drip something sooner or later. The difference comes from how you clean it up. Paper towels just spread things. The right absorbent pads, maybe even some sand for larger splashes, soak up the mess fast. Contaminated gloves and used wipes go straight into a sealed container, not the regular trash. Nobody wants the custodian exposed without warning. That habit comes from learning the hard way. I once saw a trash bag turn into a chemistry set of its own; nobody wants another surprise fire in a garbage bin.
Labels, Storage, and Good Habits
Clear labels save lives, not just paperwork. 4-Vinylpyridine sits with other organics, far from acids or heat. Glass bottles with tight caps work better than anything loose or poorly sealed—vapor builds up and escapes fast. Small things, like handing off bottles with dry, clean gloves, make a difference. Old stains on labels invite confusion in emergencies. Fresh tape, a thick marker, and a date change your memory and your odds for the better.
Practical Knowledge, Stronger Teams
Veterans in the lab can spot a rookie fumbling with the bottle right away. Passing along real-world stories does more than any safety quiz. Labs should run training sessions with live demos, not just laminated sheets. Peer checks and regular discussions bottom out forgetfulness. If you see someone skipping out on protocol, a quiet comment helps sooner than later. By building a culture of speaking up, staff create a safer, friendlier place for everyone.
Learning from Mistakes Beats Overconfidence
Anyone handling 4-Vinylpyridine owes it to themselves and their colleagues to keep rules sharp and respect high. Keeping a first aid kit stocked, emergency showers working, and protocols visible keeps small mistakes small. The hard lesson? It only takes one slip for routine to become an emergency. That memory stays with you far longer than the faint odor lingering in your coat.
What is the chemical structure of 4-Vinylpyridine?
Understanding the Makeup
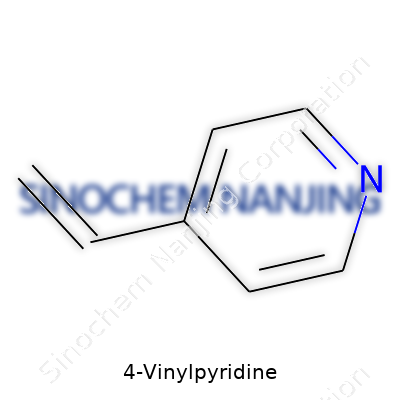
Walk into any lab where polymers or advanced chemicals are staples, and you’ll probably spot 4-vinylpyridine—often abbreviated as 4-VP. The name gives away its skeleton: a pyridine ring with a vinyl group attached at the fourth position. Pyridine is basically a benzene ring where one carbon swaps out for a nitrogen atom, changing both its reactivity and what it can connect to. In 4-vinylpyridine, the vinyl group (–CH=CH2) is stuck onto the ring, just opposite the nitrogen. In chemical shorthand, you'd draw that pyridine ring and put a vinyl group straight across from the nitrogen atom.
The backbone, that six-membered ring with five carbons and one nitrogen, acts as the foundation. Chemists might recognize the term aromatic; this sort of ring acts like a tiny stadium for electrons, letting them take the shortest route possible. That one nitrogen? It brings in a lone pair, making this molecule way more than just another ring—now you’re looking at something basic, something able to grab or donate protons under the right circumstances.
Where 4-Vinylpyridine Shows Up
I remember years back, getting my hands on 4-VP for the first time—it smelled sharp, a bit like fish or ammonia, which made sense after reading about its structure. The nitrogen gives it chemical teeth, boosting its ability to interact with acids and even metals. That vinyl group? It craves to polymerize. You throw in the right initiator and 4-VP will link together fast, building chains used in molecular imprinting or as a key player in copolymer blends.
This is where things get more interesting for industry and science. Pharmaceuticals can tap into the reactivity of that vinyl group, using it to anchor drug molecules onto specialty materials or to build selective binders for separating compounds. Environmental engineers benefit by catching toxic metals—4-VP-based polymers have the perfect spots for trapping heavy metals like mercury or lead, thanks to the ring’s nitrogen and the sticky nature of vinyl-derived polymers.
Importance Beyond the Lab
Start digging, and the influence of 4-vinylpyridine extends to water purification, adhesives, and even electronics. Synthetic chemists favor 4-VP because it acts as both a reactive partner and a building block with options. It stands as a bridge between classic aromatic compounds and newer vinyl chemistry, so anybody pushing the boundaries of material science or green tech should care.
There’s another angle worth thinking about: safety. 4-VP, like many pyridine derivatives, shouldn’t just be handled carelessly. The molecule can irritate skin and lungs. Its vapor warns you with that strong odor. Gloves, goggles, and a fume hood become must-haves. The structure connects right to these safety notes—without the nitrogen, you wouldn’t see the same volatility or solubility in water. Recognizing these traits keeps mistakes at bay.
Room for Better Solutions
Challenges aren’t in short supply. Disposal of used 4-vinylpyridine-based polymers presents a headache, since the nitrogen ring doesn’t break down easily. Biodegradable alternatives and stronger recycling efforts are overdue. Plenty of research digs into greener synthesis, aiming for fewer byproducts and better reusability. Fresh approaches in catalysis could help get more yield out of every gram, shrinking the footprint of the overall process.
Using chemistry in sustainable ways depends on knowing each piece at a molecular level. 4-vinylpyridine isn’t just another obscure lab chemical; its structure tells a story—and understanding that story can spark smarter, safer, and cleaner uses down the line.
How should 4-Vinylpyridine be stored?
Why 4-Vinylpyridine Storage Matters
Too often, chemicals like 4-vinylpyridine just sit on a shelf until someone needs them. Nobody really thinks much about the risks, though the safety section of the datasheet always feels thick with warnings. My experience in a university lab taught me to respect these warnings. One wrong step—sudden warmth, loose cap, a little too much light—can set routine work back with spilled solvent, weird odors, or safety evacuations. Leaks turn nasty fast.
4-Vinylpyridine is not something I want loose in the air. The compound's pungency hits the nose hard, way rougher than paint thinner or gasoline. It irritates eyes, nose, and throat, and prolonged exposure raises the risk for more serious symptoms. Skin contact isn’t just annoying; it burns and sensitives quickly. People have wound up with lasting irritation because they took shortcuts or skipped gloves. There’s no good excuse for letting this stuff lie around uncapped or out of the fridge.
Critical Storage Steps That Prevent Problems
Temperature is a deal breaker. 4-Vinylpyridine polymerizes—meaning it clumps and changes properties—if it sits too warm, especially with air or light present. Room temperature won’t cut it. In my old lab, we always kept it in a deep, chemical-grade fridge, and we made sure the door got closed quickly. At lower temperatures, the risk of unwanted reactions plunges, so products stay usable and safe. The compound keeps best at around 2–8°C, but always look at the latest SDS for guidance.
Moisture and light play dangerous roles. Even a small amount of water can cause it to act up. Air causes slow polymerization, while light speeds things along. Sealing the bottle tight, keeping it dry, and sticking it in an opaque container away from windows works wonders. Those big brown glass bottles don't just look vintage—they’re protecting sensitive chemistry from going off. After a rush job where air exposure ruined our last supply, I learned to double-check that every container was not just closed but well-wrapped and paperwork updated.
Choosing and Labeling the Right Container
Never pour 4-vinylpyridine into some random bottle. The right container matters. Polyethylene or glass with tight-fitting lids blocks leaks and doesn’t react with contents. Labeling has to go beyond a scrawled name and date. Everyone needs to know what’s inside, hazards involved, and the storage requirements. No one should have to squint at faint handwriting and guess whether their gloves are enough.
Culture Shift: Training and Personal Accountability
Storage rules won’t help unless people actually follow them. New lab staff—students, interns, even veteran chemists—should see proper storage as a basic part of their jobs, not red tape. I learned early that quick walkthroughs of storage areas, routine label checks, and frequent small reminders go much further than emails packed with bullet points. If a container feels sticky, or the bottle’s color changes, somebody needs to pull it right away and report the issue. A strong safety culture stops near-misses from turning into the next headline-worthy accident. Storing 4-vinylpyridine well isn’t fancy—just wise. The health and safety of every lab and worksite starts here.
What is the purity specification for 4-Vinylpyridine?
Why Purity Standards Matter
4-Vinylpyridine doesn’t show up in casual conversation, but in the world of chemistry labs and polymer research, it’s a crucial player. Purity isn’t just a box on a checklist. Anything less than clean material, and experiments start to act weird. Side reactions creep in, polymer blocks turn out off-spec, and sometimes instruments clog or corrode. Even tiny amounts of leftover solvents or byproducts can throw months of careful work into doubt.
How Purity Gets Measured
Having tried to order and use 4-vinylpyridine in different labs, I’ve seen suppliers list typical grades right on their product sheets. Scientists rely on these numbers. Almost every research supply house grades their best 4-vinylpyridine at a purity of 97% or higher, and in some demanding processes, the number jumps beyond 99%. This figure isn’t picked out of thin air. Suppliers measure it using gas chromatography and liquid chromatography, backed by certificates of analysis. The remaining 1-3%? Those are usually minor impurities such as traces of pyridine, water, and stabilizers (to stop polymerization during storage).
The Stakes for Industry and Research
Purity might seem dry on paper, but it controls success in real-world uses. Chemical engineers in plastics manufacturing, for example, crave high-purity 4-vinylpyridine because it inserts into polymer chains during synthesis. Impurities disrupt the molecular structure, meaning you can end up with brittle batches or materials that don’t bond the way they should. In my days working with analytical instruments, a little bit of contaminant made the difference between clean data and a muddy, useless spectrum.
Specifying the Standard
Looking across different catalogs and published references, such as the Sigma-Aldrich and Alfa Aesar listings, the standard minimum for laboratory-grade 4-vinylpyridine clocks in at 97%. For really high-precision work—like pharmaceutical research or critical coatings—scientists reach for 99% or better. Every shipment comes with a certificate spelling out the exact content and the methods used to check. Quality control labs might even double-check purity themselves to confirm the supplier’s numbers before using a new batch.
Taking Control of Quality
The risk of handling impure or inconsistent chemicals feels personal after seeing a poorly characterized reagent ruin a run of expensive experiments. Pre-screening every lot using your own trusted techniques—sometimes just a quick GC or NMR scan—catches hidden surprises. Many teams share their best suppliers by word of mouth, building trust through experience instead of just glossy marketing claims. One chemical company gaining a solid reputation for reliable, high-purity 4-vinylpyridine can save headache down the road.
Solutions and Steps Forward
Labs keep tighter records than ever, documenting not just the batch number but also the precise purity and storage conditions. Setting clear guidelines helps everyone use the right grade for each project. Some researchers go straight to the source and ask suppliers for the full impurity profile, not just the headline number, to spot potential reactivity problems in sensitive applications. Better transparency across the supply chain means fewer surprises and more reproducibility in results.
Simply put, purity in chemicals like 4-vinylpyridine influences everything from consistency in manufacturing to trustworthy scientific results. A clear, verified standard keeps research on track and products reliable.
| Names | |
| Preferred IUPAC name | 4-Ethenylpyridine |
| Other names |
4-Ethenylpyridine 1-Vinyl-4-pyridine 4-Vinyl pyridine p-Vinylpyridine 4-Pyridinyl ethene |
| Pronunciation | /ˈvaɪ.nəl.pɪˈrɪ.din/ |
| Identifiers | |
| CAS Number | 100-43-6 |
| Beilstein Reference | 1818732 |
| ChEBI | CHEBI:34970 |
| ChEMBL | CHEMBL143876 |
| ChemSpider | 5171 |
| DrugBank | DB03813 |
| ECHA InfoCard | 100.019.178 |
| EC Number | 202-163-5 |
| Gmelin Reference | Gmelin Reference: 153780 |
| KEGG | C01717 |
| MeSH | D014805 |
| PubChem CID | 7905 |
| RTECS number | UY5600000 |
| UNII | Q74YD1276M |
| UN number | UN2810 |
| Properties | |
| Chemical formula | C7H7N |
| Molar mass | 105.14 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | amine-like |
| Density | D:0.976 g/mL at 25 °C(lit.) |
| Solubility in water | Soluble |
| log P | 1.51 |
| Vapor pressure | 0.6 mmHg (20 °C) |
| Acidity (pKa) | 5.58 |
| Basicity (pKb) | 8.90 |
| Magnetic susceptibility (χ) | -44.6×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.5069 |
| Viscosity | 4.24 mPa·s (20 °C) |
| Dipole moment | 2.32 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 259.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 22.6 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -3049 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H302, H312, H315, H319, H332, H335 |
| Precautionary statements | P210, P261, P273, P280, P305+P351+P338, P309+P311 |
| NFPA 704 (fire diamond) | 3-3-2-W |
| Flash point | 86°C |
| Autoignition temperature | 490 °C |
| Explosive limits | 1.2–8.6% |
| Lethal dose or concentration | LD50 oral rat 200 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 210 mg/kg |
| NIOSH | WN8575000 |
| PEL (Permissible) | PEL: 1 ppm |
| REL (Recommended) | REL: 1 mg/m³ |
| IDLH (Immediate danger) | 60 ppm |
| Related compounds | |
| Related compounds |
2-vinylpyridine 3-vinylpyridine pyridine 4-ethylpyridine |

