4-Vinyl-m-Xylene: From History to Modern Application
Historical Development
Chemists discovered the value of aromatic hydrocarbons in the late nineteenth century, focusing on controlled substitutions on the benzene ring. The emergence of 4-vinyl-m-xylene didn't take shape in isolation; researchers in the 1950s grew interested in vinyl-substituted aromatic compounds because they helped bridge simple petrochemical feedstocks and more structurally intricate molecules. Laboratories in the US and Europe set their sights on methylated styrenes, searching for improved physical properties and easier incorporation into polymer chains. The process for obtaining 4-vinyl-m-xylene came as a logical step forward, borrowing methodology from toluene derivatization and harnessing rapid developments in catalytic chemistry in the decades after the Second World War. As synthetic chemistry evolved, so did the ability to consistently produce aryl-vinyl monomers like this one at both bench and industrial scales.
Product Overview
4-Vinyl-m-xylene acts as a vinyl-aryl monomer and starts as a clear to pale yellow liquid. It holds an intermediate position: not as widely adopted as simple styrene, yet picked for specialty polymers, particularly where improved rigidity or modified thermal performance takes priority. Lab catalogs and bulk suppliers list it along with other methyl styrenes, underscoring its place among specialty aromatic vinyl groups. Chemists often look to this compound when they want to introduce both methyl and vinyl groups to an aromatic ring—giving materials a unique edge in electronic or photoresist applications.
Physical & Chemical Properties
The chemical formula for 4-vinyl-m-xylene stands as C10H12, with a molecular weight close to 132.21 g/mol. It forms a liquid at room temperature, with a boiling point typically around 215–220°C, and a melting point well below ambient. Its density falls near 0.92–0.93 g/mL, less dense than water and most common laboratory solvents. This monomer evaporates slowly but still releases a noticeable aromatic odor if left exposed. Its solubility profile fits non-polar solvents much better than water, reflecting the aromatic core and methyl attachments. Reactivity centers around its vinyl group, which opens up easily under radical or anionic conditions, while the m-xylene backbone grants some resistance to oxidation and acid attack compared to unsubstituted vinyl benzenes.
Technical Specifications & Labeling
Labels on commercial bottles state purity (typically greater than 98%), presence of polymerization inhibitors (like tert-butylcatechol), and handling instructions that warn of skin and eye irritation risks. Safety Data Sheets cite the chemical’s UN number, hazard codes, and packaging recommendations—usually amber glass bottles under nitrogen to slow any accidental polymerization. Lots supplied for research commonly come with a batch analysis listing spectral confirmation (NMR and IR), GC purity, and occasionally, information about trace metal content, given its use in electronics and demanding polymer syntheses.
Preparation Method
The most common route for making 4-vinyl-m-xylene starts with methylation of m-xylene, generally by Friedel–Crafts alkylation. Alkylated xylene then undergoes halogenation—often bromination—on the desired ring position. Careful control of stoichiometry and reaction temperature directs the bromine to the para position relative to the methyl group. Subsequent dehydrohalogenation, often using strong base like potassium tert-butoxide, steals a proton and eliminates bromide, forming the vinyl group. Some companies utilize catalytic dehydrogenation processes, particularly for larger runs, leveraging palladium or copper catalysts and high throughput flow reactors to minimize side reactions. End-stage purification usually requires distillation under reduced pressure and passage through basic alumina to strip out inhibitors or colored impurities.
Chemical Reactions & Modifications
The defining reactivity lies in the vinyl group, which enters free radical, anionic, and cationic polymerizations, forming high molecular weight chains. Block copolymers or graft copolymers can incorporate 4-vinyl-m-xylene alongside other functional styrenes or acrylates, shifting glass transition temperatures and mechanical profiles. The methyl groups on the aromatic ring further bolster resistance to unwanted crosslinking, making polymers that survive harsher environments. Electrophilic substitution reactions take advantage of the electron-donating methyls, enabling additional functionalization if needed. The aromatic ring resists hydrogenation under mild conditions but transforms under catalysis—useful in fine-tuning polymer properties for custom applications in electronics or coatings.
Synonyms & Product Names
Listing synonyms helps avoid confusion: some suppliers use names like 1-Ethenyl-3,5-dimethylbenzene, 4-Ethenyl-m-xylene, or 4-Vinyl-1,3-dimethylbenzene. Chemical Abstracts and regulatory bodies may catalog it under the CAS number 8198-34-3. In European or Asian markets, local naming conventions can switch word order but reference the same compound. Academic studies, especially those exploring polymer science, sometimes label it as m-xylene derivative with vinyl substitution to emphasize structure over trivial names. These variations reflect not only regional preferences but also the history of aromatic chemistry nomenclature itself.
Safety & Operational Standards
Lab workers and plant operators must respect the health risks. 4-Vinyl-m-xylene irritates skin and respiratory tissues upon direct contact, and exposure controls call for gloves, goggles, and well-ventilated fume hoods. Inhalation of vapors over time produces headaches and dizziness, so storage containers stay tightly sealed and marked as hazardous, with spill protocols in place. National safety organizations put limits on allowable workplace concentrations. The chemical behaves as a moderate fire risk—flash point around 91–100°C—requiring grounding during transfer and exclusion of open flames. Waste involves incineration by licensed facilities, with extra caution to segregate it from oxidizing agents and acids. Handling guidelines emphasize slow, deliberate transfer to avoid static build-up and use of compatible materials for gaskets and seals.
Application Area
Specialty polymer developers value 4-vinyl-m-xylene as a comonomer in advanced resins that need stiffer backbone structures or altered solubility. Flexible electronics manufacturing employs derivatives in dielectric layers and encapsulation films, balancing insulation and heat resistance. High-resolution photoresists for semiconductor processing use it for its fine patterning capabilities, which stem from precise molecular architecture. The adhesive industry brings it into formulations where chemical resistance and flexibility matter under pressure or stress. Coating chemists tap into its unique aromatic-vinyl balance for paints and films that challenge UV degradation and chemical wear. Industrial researchers in automotive or aerospace look for more temperature-stable, less brittle polymers, finding that blends including this compound satisfy those constraints.
Research & Development
Materials scientists have tested countless vinyl aromatics to map how methyl substitution shifts polymer characteristics. Academic labs build libraries of copolymers blending 4-vinyl-m-xylene with acrylates, maleimides, or fluorinated styrenes, publishing comparative studies on impact resistance, clarity, and dielectric constants. Start-up companies push pilot projects that scale up new catalysis routes or greener production methods, sometimes replacing hazardous reagents or transitioning to continuous-flow systems. Researchers file patents on specialized uses in medical device coatings or next-generation lithography, believing that small tweaks to the vinyl-aromatic scaffold translate into measurable performance differences. Especially in the last decade, a push for sustainable manufacturing drove fresh interest into minimizing waste, optimizing catalysts, and recovering byproducts for circular production.
Toxicity Research
Toxicologists have run rodent studies to check for chronic and acute effects. High-dose inhalation prompts respiratory tract irritation and transient CNS effects, while repeated low-dose exposure doesn’t cause cancer or severe organ damage in standard tests, though comprehensive long-term data still lack. Skin absorption happens more slowly than for unsubstituted styrene but can sensitize allergic individuals over time. Wastewater studies show poor breakdown in municipal systems, calling attention to possible aquatic toxicity. Regulatory authorities include 4-vinyl-m-xylene on lists requiring registration, labeling, and use-specific exposure controls, largely because aromatic compounds share mechanistic toxicity pathways and environmental persistence.
Future Prospects
With rising demand for specialty polymers and consumer electronics, more companies examine monomers that tailor material performance at microscopic scales. Research circles buzz about 4-vinyl-m-xylene’s potential in recyclable thermosets or high-performance composites. New additive manufacturing techniques, especially 3D printing, create routes for its use in functional resins and smart coatings. Environmental pressures keep pushing for greener production, compelling chemical engineers to design processes that cut emissions and reclaim solvents. Some see opportunities in medical technology, where non-leaching, stable polymers can serve as implant coatings or drug delivery matrices. As regulatory landscapes shift and new scientific findings emerge, the role of thoughtfully designed aromatic vinyl monomers only grows, placing compounds like 4-vinyl-m-xylene squarely in the spotlight for years to come.
What is 4-Vinyl-M-Xylene used for?
Behind the Chemistry
4-Vinyl-m-xylene might sound like something only chemists talk about, but this compound moves through factories and research labs every day. Its real value comes from how it serves as a building block for many different things. Talking to folks in chemical manufacturing, it’s clear they view 4-vinyl-m-xylene as one of those raw materials that quietly link together so many products we rely on, especially in plastics and specialty coatings.
Applications Where It Matters
Walk into any facility that produces specialty polymers, and there’s a good chance 4-vinyl-m-xylene plays a role. Companies use it in the making of resins and plastics to influence strength, durability, and flexibility. My time spent covering industrial chemistry confirmed that engineers count on its ability to help control molecular structure. What goes on under a microscope translates eventually to how long a plastic part will last in your car or home appliance.
Specialty coatings depend on this compound too. Think about protective finishes that sit on pipelines and heavy machinery—these coatings don’t just fight rust, they handle tough working conditions. Resin chemists mix 4-vinyl-m-xylene into coating formulas to target weather resistance and keep surfaces smoother. Once I visited a plant where big farm equipment got treated with layers of these coatings, and the bit of extra toughness came right from changes made at the chemical level with ingredients like this one.
Why the Small Ingredients Count
The world doesn’t see the drama inside a mixing vat where workers combine all these chemicals. Spending time around people who manage industrial safety makes it clear: even substances used in small amounts need careful handling. 4-vinyl-m-xylene, like many synthetic chemicals, raises health and environmental questions. Breathing in its vapors or spilling it in water near a plant could cause real problems. A few years back I saw firsthand how teams had to scramble to contain a leak and follow strong chemical hygiene rules just to keep everyone safe. The way forward involves better training, stricter storage, and investment in spill response, not shortcuts.
Room for Improvement and Safer Paths
Looking at industry changes over time, I notice more push to refine how chemicals like this one get used. Groups like OSHA and the EPA continue tightening regulations on handling and exposure. It’s good to see more manufacturers investing in fume hoods, smarter ventilation, and remote monitoring for chemical tanks. This doesn’t just protect folks at the plant—it makes a dent in long-term environmental risk. Most plant managers I’ve met want safer setups because chemical incidents carry costs, not just fines but real harm to reputation and the community.
Science never stops moving. Researchers keep hunting for replacement materials that offer similar performance with fewer hazards. I see some hope in recent development of bio-based alternatives, though they still need time to catch up in terms of performance and cost. Until those solutions scale up, safe and responsible use of ingredients like 4-vinyl-m-xylene stays essential. With diligent oversight and a culture of safety, workers and neighbors alike can breathe a little easier.
What is the chemical structure of 4-Vinyl-M-Xylene?
Breaking Down the Basics
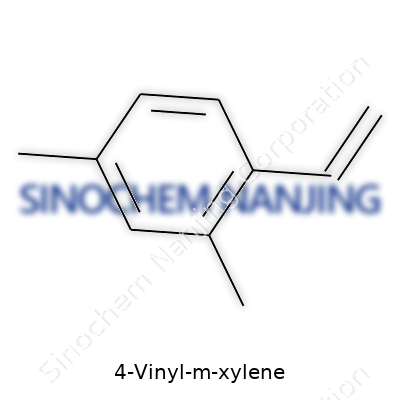
Staring at the name “4-vinyl-m-xylene,” you see a puzzle built from pieces familiar to chemists and industry insiders. To appreciate what makes it tick, start by splitting the name. There’s xylene, a benzene ring with two methyl groups squeezed onto it, and “4-vinyl,” which plants a vinyl group—two carbons and a double bond—at a specific spot on the ring.
Unlike simple xylene, which comes in three flavors (ortho, meta, and para), m-xylene’s methyl groups land on spots 1 and 3 of the benzene ring. Add a vinyl group to spot 4, and now you've constructed 4-vinyl-m-xylene, formally known by its IUPAC name as 1,3-dimethyl-4-ethenylbenzene.
Drawing the Skeleton: From Paper to Lab Bench
Lay out the carbon backbone. Benzene’s famous hexagonal ring sits at the core. From carbons one and three, you branch off methyl groups, short -CH3 chains that subtly change how the molecule behaves. Take the fourth spot and tether a vinyl group—a simple -CH=CH2—and you’ve got an unsaturated feature begging to take part in chemical reactions.
What does this structure look like in shorthand? Chemists might jot down its chemical formula as C10H12 or draw a six-carbon ring (the benzene) with two methyls and a vinyl flag on the edges. For the seasoned chemist, the SMILES code “CC1=CC(=CC=C1C)C=C” spells it out for software, confirming every atom’s place.
Why Structure Matters for Use
Chemical structure goes beyond theory. It deeply shapes how 4-vinyl-m-xylene interacts with other molecules. That vinyl group triggers attention. In my research days, I watched how tiny tweaks—like adding a vinyl group—turned a lazy aromatic into a live wire ready for polymerization. That single double bond means the compound locks arms with others during industrial synthesis, so it becomes a link in everything from resin to specialty plastics.
Industries chasing new polymers crave monomers like this. Put it in a reactor and the vinyl site leaps into action, attaching itself to other chains. That means chemists can customize resins—targeting specific flexibilities, strengths, or heat resistance—by stacking 4-vinyl-m-xylene alongside other aromatic monomers.
Handling Risk in Laboratory and Industry
Anyone handling aromatic chemicals knows their power and their danger. Xylene derivatives raise red flags for toxicity and air quality. I’ve always respected the need for proper ventilation, gloves, and goggles in the lab. Reference sheets stress that this material can irritate skin and eyes, and breathing its vapor for long can stress your body. Facts back up those warnings—studies from regulatory groups underscore the effects of exposure. Safe storage demands sealed containers and a cool spot away from open flames, since volatility lurks in these aromatic hydrocarbons.
Solutions and Safer Practices
Industrial chemists and safety officers have stepped up efforts to control aromatic hydrocarbon exposure. I’ve seen workplaces switch to improved closed-system handling, install better air scrubbers, and conduct regular training sessions. Fact-based guidance from groups like OSHA and the European Chemicals Agency pressures producers to improve, not just comply. Green chemistry drives research into safer substitutes, but for now, strong safety culture stands as the line between discovery and disaster.
Is 4-Vinyl-M-Xylene hazardous or toxic?
Chemical Curiosity Meets Health Questions
Factories rely on all sorts of chemicals to keep industries moving, and 4-vinyl-m-xylene has carved out its place on production floors. Most people don’t stop to think about what’s behind that long chemical name. It's used for creating plastics, specialty rubbers, and even as an intermediate for dyes. The reality is, folks working in labs or near manufacturing lines come closer to this compound than they might like.
Hazards Live Beyond the Safety Sheet
Let’s talk real risks. Reports and scientific studies flag 4-vinyl-m-xylene as a chemical that can hurt your body if you breathe it or let it touch your skin. It can irritate your eyes, nose, or throat, possibly leaving you coughing or with a headache after even a short burst of exposure. The bigger worry grows with long hours on the job, handling this compound day in and day out. Chronic exposure is no small thing—liver problems, nervous system issues, and effects on blood count have all been linked to compounds in this chemical’s family.
Not Just a Lab Problem
Most folks expect factories to follow strict rules about handling risky substances, but rules written on paper sometimes don’t cut it. Training might miss the mark, or equipment could run past its prime. I’ve seen places run ventilation to keep workers safe, but all it takes is someone skipping gloves or leaving a mask on a bench. That’s when health problems move from being warnings in a manual to being part of someone’s life. It’s not just the folks directly using the substance. Spills or poor waste management can send fumes through a whole building. Families living near plants also deserve clean air, and if a chemical can linger in the environment, it turns a workplace hazard into a neighborhood problem.
Research Adds Clarity but Leaves Gaps
Regulators keep tabs on chemicals like 4-vinyl-m-xylene, and for good reason. Agencies such as the EPA and OSHA ask manufacturers to label dangers clearly and set exposure limits. Still, gaps stick around. Long-term effects can take years to show up or need more research. Data on cancer risk lacks full agreement. Industry sometimes waits for studies, but workers shouldn’t have to wait for complete research before taking precautions. The principle of “better safe than sorry” carries real weight.
Keeping Workplaces and Neighborhoods Safer
Change starts small. Any company using chemicals with tough-to-pronounce names should push for real safety—proper gear, good extraction systems, and open training sessions. Health checks at work can catch early signs of trouble, allowing people to act before symptoms grow. Factories choosing safer substitutes—where possible—show care not just for profit but for people. Regulators pressing for more thorough studies empower communities and build trust.
Everyday Choices Matter
Anyone with a connection to manufacturing, whether on the job or in the community, has a stake in chemical safety. Workers asking questions, employers keeping standards high, and agencies staying alert all play a part. A cleaner, safer future for everyone doesn’t rest on one person’s shoulders—it comes from steady, shared responsibility, grounded in science and daily vigilance.
How should 4-Vinyl-M-Xylene be stored and handled?
What Workers See on the Barrel
Every time I’m near a drum labeled “4-Vinyl-M-Xylene,” those red diamonds and warnings command respect. It’s clear—this compound isn’t your everyday worksite ingredient. It brings a flammable symbol, a risk for irritation, and sometimes the kind of smell that stays on clothes long after a shift ends. I’ve seen a lot of products over the years, but this one keeps me on my toes for a reason.
Why Safe Storage Matters
Chemicals like 4-Vinyl-M-Xylene don’t take kindly to careless storage. The stuff evaporates quickly, and nobody wants to risk breathing in those vapors or seeing what happens if it leaks. I’ve learned that a cool, well-ventilated spot goes a long way. Metal drums, kept away from heat, those have saved us more than once from a nasty incident. I remember a friend who stored a couple of cans too close to a steam pipe—he won’t make that mistake again after the fumes set off the detector. Keeping the lids tight, using the right seals, and giving each barrel its own spot—that’s job security in my book.
The Human Side of Personal Protection
Colleagues often ask if gloves really help. The answer: yes, and not just for your hands. I’ve seen 4-Vinyl-M-Xylene give someone a bad rash after a single spill. Nitrile gloves do the trick, along with goggles and a simple lab coat. But more than gear, routine makes a difference. If someone cuts corners on safety, the risk doesn’t vanish; it just waits for the next slip-up. On some days, protective gear feels excessive, especially in the heat, but those habits pay off. The first signs of trouble—dizziness, headaches—remind me to double-check my routine. Ventilation hoods and masks snag those fumes, so I’m not breathing them all shift.
Fire Safety is Non-Negotiable
Flammable means keep away from sparks, open lights, or anything that gets too hot. Too many workers ignore this part, thinking nothing will happen. A spilled cup is all it takes for vapors to turn a stray spark into trouble. That’s why extinguishers rated for chemical fires aren’t optional. I keep my routes clear so there’s no panic if we ever need to grab one fast. Even static electricity from dragging containers across a floor could start something, so it’s important to ground those containers properly.
Spill Management: Act Fast, Stay Safe
A small leak grows bigger if ignored. Spill kits with absorbent pads sit ready on our shelves, and everyone knows where to find them. Once, someone hesitated, letting a puddle spread near another solvent. We locked down the area and put on respirators, but it reminded us all of how easily things can spiral. Quick action and the right cleanup gear turn an emergency into a routine drill. Waste gets bagged and marked, nobody tosses it in the regular trash, and the disposal company picks it up every week.
It’s Everyone’s Job
No single person can handle safety alone. Training sessions, the right signage, and open communication keep everyone on the same page. If you’re not sure, you ask. That’s what keeps people healthy, accidents rare, and the boss off our backs. Chemicals bring risks, but careful habits turn risk into routine safety.
What are the safety measures for using 4-Vinyl-M-Xylene?
The Risks Many Ignore
Many people who spend time in industrial labs or manufacturing plants have run across 4-Vinyl-M-Xylene. Looking at it on a shelf, the chemical might not appear remarkable. It doesn’t shout danger the way something like hydrofluoric acid might. Still, its risks deserve just as much respect. Inhaling fumes or getting it on your skin causes real problems—nausea, breathing trouble, skin irritation, and even long-term damage if exposure becomes routine. Workers sometimes get overconfident if an accident hasn't happened in a while, but that’s when slips happen. I remember hearing about someone who developed awful dermatitis after one careless afternoon without gloves. Once you get a chemical burn or lung irritation, the lesson sticks.
Personal Protection: Don’t Cut Corners
It only takes a few seconds to put on proper gear, but plenty of people take shortcuts. PPE for 4-Vinyl-M-Xylene means more than just tossing on any old gloves or a mask. You want a chemical-resistant apron, goggles that leave no gaps, and sturdy nitrile gloves. Ventilation matters, too. Turning on a fume hood or working outside isn’t overkill—it’s common sense. Just opening a door or window doesn’t cut it if you’re handling more than a drop. For anyone who wears glasses, splash shields add another layer of confidence. Those small details often stop a small spill from turning into a bad afternoon or a trip to urgent care.
Storage and Spills: Prevention Beats Cleanup
Lining up drums or bottles next to the workspace creates tempting access, but the smarter move keeps small amounts near the bench and the rest locked away. Label everything in bold text. There’s nothing worse than playing “what’s in that beaker?” in a stressful moment. Store 4-Vinyl-M-Xylene in a cool, ventilated place far from sparks. It’s flammable—one rogue spark from a misbehaving outlet can cause chaos. Good storage habits also limit evaporation into the air, which lowers risk for everyone in the space. If a spill happens, skip the paper towels. Spill kits with absorbent pads and proper bins help contain messes fast. Never send chemicals down the drain or toss them into regular trash.
Coworkers Watch Each Other’s Back
Experience in busy labs shows folks get lax if no one’s watching. My best memories involve teams who spoke up when someone almost skipped a safety step. Cultures that reward reminders save skin and lungs, literally. Training once a year isn’t enough—casual conversations about safety keep it alive. I’ve seen departments post “near miss” stories in the break room. Reading about a close call feels more real than re-reading manuals.
Regulations and Accountability
OSHA sets real standards around chemicals like 4-Vinyl-M-Xylene, from limits on airborne concentration to rules for disposal. Regular audits make a real difference. Updates to safety datasheets and refresher trainings lower the odds of disaster. Sometimes the best move is refusing to use dangerous chemicals if gear isn’t ready or if ventilation failed that day. Calling a halt slows work, but people’s health comes first. Management’s willingness to listen keeps workers honest about their concerns. Nobody should face hidden risks to keep a job.
Safer Alternatives and Commitment to Health
Some industries investigate alternative materials with lower toxicity. It’s worth checking if processes truly depend on 4-Vinyl-M-Xylene or if a less toxic substitute works. Lab safety never stands still. Careful design, honest feedback, and real accountability do more than check legal boxes—they keep families from worrying about who’s coming home sick. In any job involving strong solvents, treating every exposure as a big deal never gets old.
| Names | |
| Preferred IUPAC name | 1-Ethenyl-3,5-dimethylbenzene |
| Other names |
2,4-Dimethystyrene 1-Ethenyl-3,5-dimethylbenzene |
| Pronunciation | /ˈfɔːr vaɪˈnɪl ɛm ˈzaɪliːn/ |
| Identifiers | |
| CAS Number | 10039-89-9 |
| 3D model (JSmol) | `3D/4j/sJSmol/4VX?structure=4-vinyl-m-xylene` |
| Beilstein Reference | 1462107 |
| ChEBI | CHEBI:52045 |
| ChEMBL | CHEMBL1629449 |
| ChemSpider | 18688 |
| DrugBank | DB08795 |
| ECHA InfoCard | 100.126.236 |
| EC Number | 211-205-7 |
| Gmelin Reference | 809709 |
| KEGG | C11273 |
| MeSH | D014822 |
| PubChem CID | 136644 |
| RTECS number | ZE2625000 |
| UNII | IQD1L4F43G |
| UN number | UN2301 |
| Properties | |
| Chemical formula | C10H12 |
| Molar mass | 134.22 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | Aromatic |
| Density | 0.943 g/mL |
| Solubility in water | Insoluble |
| log P | 2.9 |
| Vapor pressure | 0.0698 mmHg (25°C) |
| Acidity (pKa) | pKa = 43.08 |
| Basicity (pKb) | 12.61 |
| Magnetic susceptibility (χ) | -70.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.539 |
| Viscosity | 1.27 mPa·s (25 °C) |
| Dipole moment | 0.68 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 354.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 67.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4817.9 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H319, H335 |
| Precautionary statements | P210, P261, P264, P271, P280, P301+P312, P304+P340, P305+P351+P338, P312, P330, P337+P313, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-2-Ʇ |
| Flash point | 64°C |
| Autoignition temperature | 550 °C |
| Explosive limits | Explosive limits: 0.9–6.1% |
| Lethal dose or concentration | LD50 oral rat 1970 mg/kg |
| LD50 (median dose) | LD50 (median dose): 3730 mg/kg (oral, rat) |
| NIOSH | NIOSH: GY2450000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 4-Vinyl-m-xylene is not established. |
| REL (Recommended) | 200 ppm |
| IDLH (Immediate danger) | IDLH: 140 ppm |
| Related compounds | |
| Related compounds |
m-Xylene Styrene 4-Ethyl-m-xylene 4-Vinyl-o-xylene 4-Vinyl-p-xylene |

