4-Vinyl-1-Cyclohexene: A Deep Dive into Its Substance and Impact
Historical Development
Chemists in the early twentieth century began exploring cycloalkene derivatives as the world turned toward polymers and synthetic rubbers. 4-Vinyl-1-cyclohexene took shape during this period of wild experimentation, rising from lab notes to solid, real production around the boom of industrial chemistry in the 1950s and 60s. Production scaled quickly due to demands for low-cost intermediates in the plastics sector. Companies valued its structure and reactivity; researchers found its two double bonds opened doors for synthetic routes other cyclohexenes just didn’t offer. This chemical’s story is tied with the push for faster, more reliable rubber and plastic technologies in everything from car tires to hoses and adhesives.
Product Overview
Today, 4-Vinyl-1-cyclohexene stands out as a colorless liquid, often coming with a faintly sweet but noticeable odor. Industry leans on it as both an intermediate and end product, with annual global output counted in tens of thousands of metric tons. Producers favor it for its straightforward two-alkene structure, which slots into downstream manufacturing of specialty rubbers, coatings, and chemical modifiers. Its presence in commercial channels signals broad utility, touching countless consumer products indirectly through its use in process chemistry and materials innovation.
Physical & Chemical Properties
Density clocks in at roughly 0.86 g/cm³ at room temperature, keeping the compound lightweight and convenient for large-batch storage. Boiling occurs around 173°C, offering a sensible thermal range for both handling and chemical transformations. The molecular formula, C8H12, gives it a molecular weight near 108.18 g/mol. Its structure brings moderate reactivity, especially attractive for Diels-Alder and free-radical polymerizations due to those two double bonds—one exocyclic, one within the ring. Insoluble in water, it dissolves readily in ether and common organic solvents, presenting engineers with flexible mixing options during synthesis.
Technical Specifications & Labeling
Manufacturers guarantee minimum purity levels upwards of 98%, commonly checked by gas chromatography. Typical impurity limits cover residual cyclohexene and polymerization by-products, which, if unchecked, gum up both reactors and analytical results. Containers receive flammability and inhalation hazard warnings. Industry uses standardized hazard communication, referencing GHS codes and packing group II for transport. Labels plainly state its UN number (1993) and recommend personal protection and special storage outside direct sunlight or heat. Clear documentation smooths transport and purchasing, but ultimately, end-users need to backstop with their own process-specific quality assurance protocols.
Preparation Method
Most production routes rely on a dimerization process starting with butadiene. Under carefully controlled conditions—temperatures holding around 150°C and pressure above atmospheric—metal catalysts like nickel or titanium drive two molecules of butadiene together, producing a mix of 4-vinyl-1-cyclohexene and 1,5-cyclooctadiene. Refiners separate the products by fractional distillation, optimizing for yield with tweaks to temperature, catalyst loading, and reaction time. Recovered solvents and mother liquors circle back into the process loop, minimizing waste. Some research shops try out alternative feedstocks or greener catalysts, aiming for better atom economy and lower carbon footprints, but the butadiene route still rules heavy industry.
Chemical Reactions & Modifications
Those two double bonds open the door for plenty of chemistry. Direct polymerization under radical or cationic conditions produces elastomers with unique cyclic structures that standard linear acrylates just can’t match. Diels-Alder reactions tap the cyclic alkene to build larger polycyclic frameworks, paving paths for fine chemical syntheses and specialty intermediates. Hydrogenation knocks out both double bonds, turning the liquid into saturated cyclohexyl derivatives for further chemical building. Epoxidation, followed by ring opening, lets formulators introduce oxygen-containing functionality, tuning product performance for specialty plastics, adhesives, or coatings. Processes built around this molecule change up downstream performance in ways that chemists have learned to suit to evolving market needs.
Synonyms & Product Names
Suppliers and research chemists recognize the compound under several alternative titles—1-Cyclohexene, 4-ethenyl-; 4-Vinylcyclohex-1-ene; VCH. Commercial catalogues push brand names like VeoCyclo or Cyclohexene, 4-vinyl-, depending on region and marketing conventions. Legacy chemical literature references its structure-based IUPAC names, so cross-referencing becomes part of daily practice for anyone tracking down supplier sheets or comparing regulatory documentation across countries.
Safety & Operational Standards
Flammability characterizes its risk profile, with a flash point below 40°C that earns it a spot on most fire marshal watch lists. Vapors outpace air, collecting in low spots and potentially lighting up if leaks occur. Chronic inhalation creates notable central nervous system effects; dermal exposure dries out skin and can even cause mild chemical burns. Workers fit with proper gloves, goggles, and lab coats, using chemical fume hoods during sampling or blending. Facility managers enforce strict grounding and bonding to kill static ignition threats. International standards—like OSHA regulations in the U.S. or REACH directives in the EU—guide process design and training, with periodic drills to keep emergency response sharp.
Application Area
Industrial rubber production turns most of the world’s 4-vinyl-1-cyclohexene into a core comonomer in ethylene-propylene-diene monomer (EPDM) synthesis. These rubbers turn up in automotive weather seals, hoses, belts, and white goods gaskets. Its crosslinkable diene group ensures low-temperature flexibility and aging resistance in finished products. Paint and coating formulators grab it for crosslinking agents and adhesion promoters. Some specialty plastics developers experiment with it for new resins or reinforcement compounds. Even crop protection and pharmaceutical sectors investigate its cyclic scaffold as a starting material for active ingredient synthesis. Across these fields, it rarely ends up present in consumer hands directly, but its role in upstream transformation drives downstream performance.
Research & Development
R&D shops push boundaries by tuning catalytic dimerization—seeking both higher selectivity and fewer by-products. Academic groups dig into its potential as a renewable monomer, looking at engineered microorganisms or alternative bioroutes as the feedstock mix shifts away from petroleum base. Chemists test new polymerization initiators or crosslinkers that harness the dual unsaturation, broadening choices in elastomer design and specialty resins. Collaborations between universities and industry frequently chase lower-energy separations, greener catalysts, and new chemical transformations off the seven-carbon backbone. Publications map both the advances and the lingering hurdles, written with an eye toward both competitive edge and sustainability.
Toxicity Research
Toxicologists have published studies indicating moderate acute toxicity via inhalation or skin exposure, especially at higher concentrations inside poorly ventilated plants. Long-term animal bioassays reveal potential for liver and kidney effects; some links to possible carcinogenicity have led regulatory agencies to flag it as a chemical of concern. Chronic workplace exposure gets monitored carefully, with mandatory air monitoring and health checks in larger production operations. Safety data sheets warn against overfilling and call for quick spill containment, citing both inhalation risks and the threat of fire. Packaging and storage now follow stricter protocols to limit leaks or uncontrolled exposures during handling, shipping, and use.
Future Prospects
Demand for 4-vinyl-1-cyclohexene can shift with economic cycles and moves in synthetic rubber use, but ongoing infrastructure buildouts for vehicles, appliances, and building materials show little sign of slowing. Sustainability trends push researchers to seek renewable feedstocks, more selective catalysts, and safer reaction controls to limit exposure and environmental load. New developments in chemical recycling and advanced resins keep it in play, as teams seek products that resist aging, carry improved environmental performance, and meet tougher global safety standards. The push for green chemistry gives it another lease on relevance: not just for legacy plastics and rubbers, but as a flexible platform for tomorrow’s materials.
What are the main uses of 4-Vinyl-1-Cyclohexene?
A Backbone for Everyday Materials
Walk through any modern factory, and there’s a good chance you’ll find products that trace their roots back to chemicals like 4-vinyl-1-cyclohexene. This colorless liquid, with a sharp odor, might sound exotic, but it shows up in familiar places. The biggest role comes from its use as a building block to create specialty rubbers and plastics. Working in a materials research lab, I saw how key these ingredients are—without them, things we count on every day would disappear.
Most people never think about where car tires or shoe soles come from. The truth is, these items depend heavily on synthetic rubbers. 4-Vinyl-1-cyclohexene steps in as a starting material when making ethylene-propylene-diene monomer (EPDM) rubber. Rubber made from EPDM handles sun, water, and heat better than old-school types, and it doesn’t crack as quickly. Car makers choose EPDM for weather seals, hoses, belts, and insulation. Without this ingredient, cars and refrigerators would be a lot less reliable.
Innovations in Plastics and Polymers
Beyond rubber, 4-vinyl-1-cyclohexene is useful in plastic production. Polymer scientists appreciate the way it opens the door to tweaking properties like toughness and flexibility. Think about the packaging that keeps food fresher or the appliances that last for years—they both get a boost from advances in polymer chemistry.
A good example involves specialty plastics, where a touch of 4-vinyl-1-cyclohexene shifts the physical qualities just enough to suit a particular need. Chemical companies tune the material to handle impacts, or to stay flexible without breaking down. I remember early attempts at making rugged phone cases; once these newer polymers came into the mix, the cases finally lasted through drops and rough handling.
Industry Safety and Environmental Concerns
Jobs in chemical plants highlight something people outside the industry might not see: every material with industrial value carries a burden of responsibility. Handling 4-vinyl-1-cyclohexene means following strict worker safety standards. Factory teams use protective gear, monitor air quality, and respond fast to any signs of leaks.
A troubling fact stands out—4-vinyl-1-cyclohexene gets flagged as a possible carcinogen, and inhalation over long periods can create real risks. This pushes both manufacturers and health agencies like OSHA to track its use closely and reduce accidents. Safe chemical handling rules aren’t just paperwork; they keep factory workers healthy and local communities safe from toxic leaks.
Room for Smarter Progress
Decades in materials science have shown me how much can change with thoughtful innovation. Research groups keep working on catalysts that boost efficiency, hoping to make materials with fewer byproducts and less waste. Some projects look for gentler substitutes that keep performance high but shrink the health and safety footprint. For now, 4-vinyl-1-cyclohexene holds its spot because it works—producing tough, weather-resistant, and long-lived materials.
Smarter regulations, stronger monitoring, and honest sharing of data all play a part in protecting people while improving material quality. Safe, high-performance goods depend on more than the right chemical recipe. Real-world experience proves you need balance: demand for better rubber and plastic never stops, but progress only matters when it goes hand in hand with health, transparency, and environmental care.
What are the safety precautions for handling 4-Vinyl-1-Cyclohexene?
Recognizing the Risks
4-Vinyl-1-cyclohexene pops up in several chemical manufacturing processes. It finds use as an intermediate but brings with it a mix of hazards. The liquid form gives off a flammable vapor, and more than a few studies flag its toxic and possibly carcinogenic properties. I've spent time in labs handling challenging chemicals, and sitting around hoping for the best never paid off. You need clear protocols, reliable gear, and good habits.
Personal Protective Equipment and Mindset
PPE isn’t just a box to check. I’ve seen coworkers skip safety glasses because things seemed under control—until they weren’t. Goggles, chemical-resistant gloves (nitrile or neoprene), and a properly fitted lab coat stand between you and a trip to the emergency room. If splashing is a risk, a face shield steps up the protection. Short sleeves and open-toed shoes have no place near this stuff. One whiff or splash is enough to make you rethink a casual attitude.
Ventilation and Containment
Fume hoods make all the difference. The liquid releases vapors that not only irritate but also threaten long-term health. Running reactions in a well-maintained hood limits exposure. Labs without that luxury struggle to manage risk, and few engineering controls can substitute for properly moving air. I learned early to avoid working with volatile chemicals anywhere near an open flame or heating element. That advice earned its keep more than once.
Storage Tactics
Forget balancing jugs of reactive liquid on shaky shelves. Flammable liquid storage cabinets set the bar for safe storage of 4-vinyl-1-cyclohexene. The chemical belongs far away from oxidizers, acids, and ignition sources. I’ve watched containers crack after sitting near sunlight or heat—direct rays or warm rooms weaken seals, which leads to leaks and vapors seeping into shared air. Inspecting containers regularly doesn’t take long, and finding a leak early beats mopping up a spill later.
Spill and Exposure Response
If a spill happens, everyone benefits from clear roles and the right tools close at hand. Absorbent pads, sand, and specialized spill kits should sit in the workspace, not in a locked cabinet three doors down. Quick action cuts risk, but training sets people up to respond correctly. Washing skin thoroughly after contact with soap and water always mattered more than trying to tough it out. Eyes exposed to chemical splash need an emergency eyewash station right nearby, not a run to the nearest sink.
Waste and Decontamination
Disposing of waste isn’t glamorous, but it brings peace of mind. Sealing spent material in labeled containers made of compatible material and sending it off with a certified handler keeps the workspace safe. Water, soap, and scrubbing the work surface wrap up each session, clearing away any invisible residue. The workday ends better knowing sharp corners haven’t been left behind for someone else.
Continuous Learning and Prevention
Hazards evolve as work changes. Regular training fits real-world needs. An effective safety culture means people speak up when they spot a risk and treat every round with toxic materials as a test of skill, not a routine chore. I’ve seen teams that built the habit of reviewing procedures spot small slips before they snowball. 4-vinyl-1-cyclohexene isn’t forgiving, and neither should daily routines be when health is on the line.
What is the chemical formula and structure of 4-Vinyl-1-Cyclohexene?
The Chemical Blueprint: Formula and Structure
4-Vinyl-1-cyclohexene isn’t a chemical that most people bump into during daily routines, but it earns its space in the world of polymer science and industrial chemistry. The chemical formula for 4-vinyl-1-cyclohexene is C8H12. It features a six-membered cyclohexene ring with a vinyl group attached to the fourth carbon. If you draw it out, you’ll spot one double bond inside the cyclohexene ring, plus another in the side-chain, forming the vinyl group. The structure gives this molecule unique properties, influencing both reactivity and stability. You get two regions capable of certain chemical reactions, not just one—this matters a lot for making specialty plastics and rubbers.
Why the Structure Matters
Back in college organic chemistry, ring structures always brought out strong opinions. Some folks dreaded the aromatic stuff, while others gravitated toward these cyclohexene types. The single double bond in the ring makes it less reactive compared to aromatic rings, like benzene, but the presence of a vinyl group attached means it can polymerize and link up in bigger chains. That’s a big deal. That split personality gives chemists ways to fine-tune reactions, something not every compound offers. Vinyl cyclohexene pops up as a building block in making high-performance plastics and rubber, where you want flexibility but need some strength, too. From car parts to specialized pipes, it lends toughness to final materials.
Industry and Health Considerations
Factories handling compounds like 4-vinyl-1-cyclohexene invest heavily in safety measures for a reason. OSHA lists it as a hazardous material. People working around it need protective equipment and good ventilation, since chronic exposure or spills can cause harm. Studies have linked related compounds to possible cancer risks. This heightens the need for strong oversight, not only for worker health but for communities near production sites. Legislative controls like the REACH regulation in the European Union and firm guidelines from the EPA in the United States are in place for reasons grounded in real science and history.
Challenges and Better Paths Forward
Factories and labs both have a lot riding on careful handling and disposal routines. I saw how even small lapses could create headaches downstream—fines, production stops, or worse, contamination. Building in regular safety drills, using real-time air monitoring, and keeping up with research on less toxic alternatives help reduce risk long-term. Chemistry doesn’t stand still. Research around “green chemistry” invites new possibilities: molecules that do the job of 4-vinyl-1-cyclohexene but with a gentler footprint. Some labs experiment with biobased cycloalkenes or tweak the traditional process to cut down on hazardous byproducts. These efforts go beyond regulatory compliance. They represent a larger shift toward safer production and a cleaner environment. Facts show that such changes not only help people and ecosystems but also lead to stronger business profiles over time.
Looking Ahead with Responsible Innovation
Understanding how a structure like 4-vinyl-1-cyclohexene’s works means looking at both its advantages and the responsibilities that come with it. Getting every link in the chain right—formula, use, disposal—protects workers, strengthens the bottom line, and keeps communities safer. That’s the kind of value real expertise brings, whether you’re on the production floor or reading a safety sheet in a lab.
How should 4-Vinyl-1-Cyclohexene be stored?
Understanding What’s at Stake
4-Vinyl-1-Cyclohexene serves a crucial role in chemical manufacturing. Many folks working in labs or plants run into this compound, and its properties call for careful handling from the start to the end of its use. I’ve seen how small mistakes here can turn into headaches or even hazards, so being smart about storage means avoiding bigger problems down the line.
Real Risks Call for Real Storage Solutions
This compound comes with a flammability rating that can’t be ignored. So, storing it near heat sources, open flames, or places where sparks happen doesn’t cut it. Warehouse managers, lab techs, and even shipping staff all bear some responsibility for keeping it separate from ignition sources and out of direct sunlight. I remember a case where poor storage let vapor build up, and the cleanup cost everyone time, money, and nerves.
Temperature control also matters. At higher temps, vapors can build and pressure increases; this can push the limits of what storage drums or bottles were designed to handle. Keeping it cool—in a designated flammables cabinet, not just a regular closet—reduces this risk.
Container Choices Make a Difference
Not every container is fit for handling strong organics. 4-Vinyl-1-Cyclohexene reacts badly with some plastics and unlined metals. Glass and high-grade, corrosion-resistant steel work better because they keep the chemical stable and avoid container breakdown or leaks.
If you handle multiple chemicals, color coding or clear labeling helps catch mistakes before they snowball. The wrong container on the wrong shelf just increases the odds of a spill or worse.
Ventilation Doesn’t Get Enough Attention
Good ventilation makes storage much safer and reduces danger from vapors. Companies sometimes overlook this, but the right fans, air exchanges, and sensors keep concentrations below trouble levels and protect both people and assets. One poorly ventilated storeroom once led to a minor health scare for a coworker of mine—she learned quickly the value of fresh air and monitoring.
Regulations and Practice Go Hand in Hand
OSHA, NFPA, and local fire codes give solid rules for anyone storing this material. Familiarity with those makes inspections easier and reduces fines. Workers need regular training and retraining so that everyone’s muscle memory lines up with safety protocols, not shortcuts. You can have fancy cabinets and sensors, but if workers don’t know what to do, you’ve gained nothing.
Spill kits and fire suppression tools in arm’s reach can turn a near-miss into just a lesson learned—not a disaster. Regular drills and real talk about accidents and what caused them help more than a stack of unread manuals.
Common-Sense Steps for a Tough Job
Time and effort matter. Keep only what you need on hand, rotate stock, and check containers for signs of age or weakness. It's also worth talking to staff regularly about what works and what doesn’t, because nobody knows the quirks of your site like the folks who use it every day.
Safe storage of 4-Vinyl-1-Cyclohexene isn’t just about checking off boxes. It’s about blending practical habits with respect for what the compound can do if ignored. A solid plan, the right tools, and honest experience from people on the floor help keep work safe and on track.
What is the purity specification for commercially available 4-Vinyl-1-Cyclohexene?
Looking at Purity Standards
Purchasing chemicals isn’t just a matter of handing over a purchase order and waiting for a drum to arrive. Anyone who’s spent time handling 4-vinyl-1-cyclohexene in a research lab or on a plant floor knows the details matter. Purity sits at the core of performance, safety, and regulatory compliance. Most suppliers offer this particular cycloolefin with a purity in the range of 95% to 99%. It’s not a one-size-fits-all standard though—small differences can create sharp changes in how a process does or doesn’t work.
Why That Few Extra Percent Matters
From experience, projects that stall unexpectedly often trace back to an underappreciated trace impurity. Small molecules lurking in that 1-5% leftover slice (often methylcyclohexene, cyclohexene, or 1,4-divinylcyclohexane) set off chain reactions. Polymerization steps, for example, skew toward unpredictable yields when the feedstock contains surprise extras. Every lab tech has seen the consequence—a failed batch, a frantic phone call to the supplier, and a scramble to confirm the real purity.
Supplier Declarations: More Than Just Numbers
Checking the certificate of analysis reveals far more than a marketing figure. Most major suppliers, including Sigma-Aldrich and TCI, list at least 97% purity by GC for their standard 4-vinyl-1-cyclohexene. Some push to 99% for specialized demands. That two percent gap might not catch every eye, but in silicone elastomer synthesis or advanced material manufacturing, it determines the color, clarity, or longevity of the final product. A memory sticks—being in the middle of a large polymer batch, realizing the haze came from a tiny contaminant flagged on the spec sheet.
The Safety Angle
Chemists working around 4-vinyl-1-cyclohexene know it has a low flash point and a reputation for being a skin and respiratory irritant. Unexpected impurities can push up workplace risks. Distillation residues or cross-contamination between bulk shipments change how the chemical reacts in a fume hood or on a plant scale. Reviewing purity specs—often buried in Section 9 or 10 of an SDS—turns into an everyday habit for anyone with responsibility over process safety.
What Actually Drives the Purity You Get
Some buyers think a catalog listing is law. In practice, the grade depends on the intended use. Manufacturers producing tire elastomers or automotive parts might ask for the lower end—95% or 96%—balancing process cost against end-use tolerances. Labs racing to develop new specialty plastics pay the premium for 99% purity since even a sniff of cross-reactivity can force months of retesting.
Both approaches remain valid, but transparency keeps everyone honest. I worked with a supplier once who flagged a batch that missed its target by half a percent. The honesty saved weeks of troubleshooting, and we found a use for the material in a less demanding application. Such honest reporting lines up with what regulators and auditing bodies expect.
Solutions for Consistency
Trust always grows from experience and records. Regular quality checks with gas chromatography, clear supplier relationships, and verifying every drum’s lot specific numbers keep surprises at bay. Anyone with a background in chemical manufacturing can share stories about shortcuts backfiring—underscoring that paperwork isn’t just paperwork. Asking for complete impurity profiles and robust traceability pays off in reduced waste and safer labs.
4-vinyl-1-cyclohexene may sound esoteric, but keeping its purity on spec ripples into safety, reliability, and product lifetime—from factory to finished good.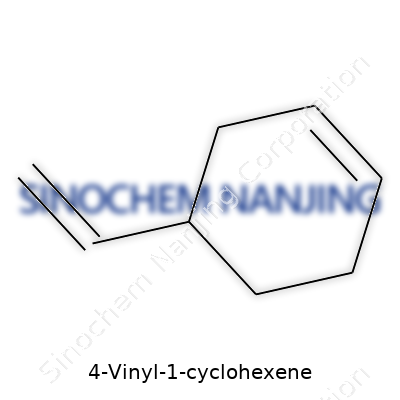
| Names | |
| Preferred IUPAC name | 4-ethenylcyclohex-1-ene |
| Other names |
Vinylcyclohexene 4-Ethenylcyclohexene 1-Cyclohexen-4-ylethylene 4-Vinylcyclohex-1-ene 1,2,3,4-Tetrahydro-4-vinylbenzene |
| Pronunciation | /ˈfɔːr ˈvɪnɪl wʌn ˈsaɪkloʊˌhɛksiːn/ |
| Identifiers | |
| CAS Number | 100-40-3 |
| 3D model (JSmol) | `3d:CCCC1=CCCCC1` |
| Beilstein Reference | 635873 |
| ChEBI | CHEBI:51435 |
| ChEMBL | CHEMBL51370 |
| ChemSpider | 6927 |
| DrugBank | DB14055 |
| ECHA InfoCard | ECHA InfoCard: 100.005.963 |
| EC Number | 202-898-1 |
| Gmelin Reference | 145251 |
| KEGG | C06746 |
| MeSH | D014823 |
| PubChem CID | 7501 |
| RTECS number | GV7875000 |
| UNII | 5T4VXJ8E09 |
| UN number | UN1302 |
| CompTox Dashboard (EPA) | `DTXSID6020725` |
| Properties | |
| Chemical formula | C8H12 |
| Molar mass | 108.18 g/mol |
| Appearance | Colorless liquid |
| Odor | mild sweet odor |
| Density | 0.841 g/mL at 25 °C (lit.) |
| Solubility in water | Insoluble |
| log P | 2.8 |
| Vapor pressure | 1.73 mmHg (25°C) |
| Acidity (pKa) | ca. 44 (for the most acidic hydrogen, typical of alkenes) |
| Basicity (pKb) | pKb = 10.63 |
| Magnetic susceptibility (χ) | -7.98 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.463 |
| Viscosity | 5.52 mPa·s (20°C) |
| Dipole moment | 0.51 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 322.04 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -26.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3586.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Precautionary statements | P201, P210, P261, P273, P280, P301+P310, P303+P361+P353, P304+P340, P308+P311, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 3-2-2 |
| Flash point | 33 °C (91 °F; 306 K) |
| Autoignition temperature | 244 °C (471 °F; 517 K) |
| Explosive limits | 1.1% - 6.7% |
| Lethal dose or concentration | LD50 oral rat 4810 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 2500 mg/kg |
| NIOSH | GV3325000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 4-Vinyl-1-Cyclohexene: **50 ppm (220 mg/m³)** |
| REL (Recommended) | REL (Recommended Exposure Limit) for 4-Vinyl-1-Cyclohexene is 0.1 ppm (0.42 mg/m³) as a time-weighted average (TWA) |
| IDLH (Immediate danger) | 50 ppm |
| Related compounds | |
| Related compounds |
Styrene Divinylbenzene Cyclohexene 1,3-Butadiene Vinylcyclohexane |

