4-Toluidine Hydrochloride: A Deep Dive
Historical Development
4-Toluidine hydrochloride has roots reaching back to the explosive growth of organic chemistry in the late 19th century. At that time, researchers started isolating and refining aniline derivatives to make synthetic dyes, and 4-toluidine soon became indispensable. Once chemists realized how its structure could help synthesize azo dyes and other colorants, the floodgates opened for its use in textile manufacturing and laboratory experiments. Unlike many compounds whose popularity faded, 4-toluidine hydrochloride stayed relevant, riding waves of innovation from the early days of industrial chemistry to present-day research labs.
Product Overview
Recognized for its off-white crystalline appearance, 4-toluidine hydrochloride belongs to the family of amines, packing both the punch of a reactive amine group and the influence of a methyl group on its benzene ring. Most catalog suppliers list it for chemical synthesis, dye production, and even niche pharmaceutical research. Its value goes beyond mere utility—it represents a bridge between first-generation synthetic compounds and the highly engineered molecules of today.
Physical & Chemical Properties
4-Toluidine hydrochloride comes as a stable solid at room temperature, with an unmistakable faint yellowish tone after exposure to air. It melts in the range around 200°C and dissolves smoothly in water. In my own work with aromatic amines, I’ve had to respect the pungent odor and volatility, which underscores its active nature as both a reagent and an intermediate. On the chemical front, it carries the C7H10ClN formula, featuring a para-oriented amino group and a methyl group—giving chemists a lot to play with in substitution and modification reactions.
Technical Specifications & Labeling
Reputable suppliers offer 4-toluidine hydrochloride at purities upwards of 98%, often confirming batch identity with GC-MS or NMR data. Labelling reflects both its regulatory status and safety concerns: the packaging typically displays GHS pictograms for acute toxicity and irritancy, while technical data sheets outline melting points, solubility, and spectral properties. For researchers new to aromatic amines, it’s essential to reference safety datasheets, since exposure thresholds run much lower than for many other lab solids.
Preparation Method
Most manufacturers rely on a simple, robust synthesis: starting with 4-toluidine, hydrochloric acid is added under controlled cooling to yield the crystalline hydrochloride salt. The choice of acid stoichiometry, cooling temperature, and rate of acid addition all affect yield and product quality. In practice, I’ve watched even minor changes—like temperature drift—lead to impurity formation or incomplete salt precipitation, so careful monitoring makes all the difference in batch consistency.
Chemical Reactions & Modifications
4-Toluidine hydrochloride excels as a substrate for diazotization—a hallmark reaction for synthesizing azo dyes. Its amine group allows for facile conversion to diazonium salts, opening pathways to linkages with phenols or other aromatic amines. Beyond diazotization, its methyl group opens the door for alkylation, oxidation, and sulfonation chemistry. In dye synthesis, it remains a backbone for generating vivid reds and yellows, which can be found in textiles and inks. Its versatility keeps it on the bench of any synthetic chemist with an eye for aromatic transformations.
Synonyms & Product Names
On the label, it might show up as p-Toluidine hydrochloride, 4-methylaniline hydrochloride, or para-toluidine hydrochloride. Some trade catalogs shorten it to 4-Tol HCl or just Toluidine HCl. These synonyms reflect both chemical tradition and marketing simplicity, but the underlying molecule remains unchanged—a testament to its resilience in chemical nomenclature.
Safety & Operational Standards
Working with 4-toluidine hydrochloride demands proper lab hygiene and protective gear. Both the amine and the hydrochloride salt trigger strong responses in mucous membranes, so gloves, eyewear, and lab coats are the minimum. Chronic exposure connects historically with toxicity concerns, especially in dye factories of the past, where workers showed elevated risks of carcinogenic effects. Rigorous training and fume hood operation help lower those risks in today's settings, turning what was once a dangerous routine into manageable laboratory work. In any chemical storage, incompatibilities with strong oxidizers and bases stand out, driving the need for clear segregation in chemical inventories.
Application Area
The backbone role of 4-toluidine hydrochloride in dye synthesis continues in textile processing and pigment manufacture. As an intermediate, it extends its reach into rubber processing chemicals, plastic stabilizers, and selected pharmaceuticals. Some agricultural research outfits use it in the study of novel herbicides and growth regulators. In my experience, any lab working with custom or specialty dyes keeps this compound within easy reach, whether for color fastness studies or for molecular probes that rely on aromatic amine chemistry. Its reach extends beyond manufacturing—into forensics, chemical education, and as a reference standard for analytical labs.
Research & Development
Active research efforts aim to optimize yields, reduce byproducts, and improve safety profiles for both users and the environment. Green chemistry initiatives test ways to streamline synthesis, minimize hazardous waste, and find biodegradable alternatives to current dyeing agents. Some advanced materials projects repurpose aromatic amines like 4-toluidine hydrochloride as building blocks for functional polymers and novel electronic materials. In toxicology, ongoing studies target metabolites and breakdown products, pushing forward the debate about occupational limits and allowable concentrations in finished goods.
Toxicity Research
Studying the health impact of 4-toluidine hydrochloride has driven much of its regulatory oversight. Animal studies document impacts on the liver, kidneys, and blood cell production after sustained low-level exposure. Occupational exposures in the dye and pigment industries link to increased cancer risks, showing why industrial hygiene must stay front and center. Current research examines how metabolic enzymes break down aromatic amines and how genetic variability shapes individual susceptibility. Updating workplace standards depends on a constant flow of new data, emphasizing that safety is a moving target, not a box to check off once.
Future Prospects
Looking ahead, I see a compound caught between two worlds—one grounded in traditional industry and another reaching for sustainable innovation. As regulations tighten and public awareness grows, the pressure will keep rising for safer, greener alternatives. Research into less hazardous substitutes and improved handling techniques responds directly to that demand. Advances in computational chemistry point toward non-toxic analogs with similar performance, while process chemists experiment with recycling waste for circular manufacturing. The story of 4-toluidine hydrochloride isn’t finished; its future will depend on how researchers and regulators balance utility, safety, and environmental responsibility in a rapidly evolving world.
What is 4-Toluidine Hydrochloride used for?
Understanding 4-Toluidine Hydrochloride
4-Toluidine hydrochloride belongs to a class of chemicals called aromatic amines, which pop up often in the world of chemistry. In my experience, walking through chemical supply facilities, you see jars of this compound labeled as essential—not because the average person uses it at home, but because industries lean heavily on its unique properties.
Role in Dyes and Pigments
Dye manufacturing stands out as the biggest user of 4-toluidine hydrochloride. Factories feed this compound into production lines, especially for creating azo dyes—those vibrant reds, yellows, and oranges used for textiles, plastics, and even leather. Azo dyes account for a huge part of the world's colorants, especially in countries with booming textile industries. Researchers found the color-staying power of these dyes depends largely on molecules like 4-toluidine hydrochloride doing their job in the background.
Pharmaceutical Synthesis
Folks often overlook the behind-the-scenes chemicals that give us medicines. Years ago, working in a lab, I noticed how certain pharmaceutical intermediates rely on aromatic amines like 4-toluidine hydrochloride. Chemists use this compound to build more complex molecules, eventually creating drugs that treat everything from infections to chronic diseases. This step in pharmaceutical synthesis needs precision and clean raw materials, which keeps quality in check and ensures safe medicine for the end user.
Testing and Research
Laboratories keep small amounts of 4-toluidine hydrochloride around for research and testing. It serves as a reference compound in chemical analysis, helping scientists identify unknown substances or check purity. It shows up in processes like chromatography and spectroscopy. Thinking back to early days in analytical labs, I remember how a single slip in handling such chemicals could skew results or introduce errors that throw off entire experiments.
Health and Safety Concerns
Handling 4-toluidine hydrochloride brings its share of risk. Various studies, including those reviewed by the International Agency for Research on Cancer (IARC), flagged certain aromatic amines as potential carcinogens. In practical terms, this means any company or worker dealing with 4-toluidine hydrochloride has a responsibility to invest in proper protection—think gloves, fume hoods, and tight safety protocols. Poor ventilation and improper storage of aromatic amines like this one have, in some cases, caused accidents and health scares.
Sustainable and Safe Practices
Some companies have started exploring greener alternatives that cut down on the need for such chemicals. Researchers now look for less harmful chemicals or tweak existing processes to use smaller quantities with the same results. Anyone in a position to design or approve industrial processes should encourage frequent safety reviews, better worker training, and a push toward sustainable chemistry. Regulatory agencies have begun to set tougher limits on exposure, but enforcement sometimes lags. Industry leaders could drive change by investing in new technology and ensuring every worker—from floor staff to management—knows the risks and best practices for handling these compounds.
Takeaway
4-Toluidine hydrochloride doesn’t make headlines, yet it plays a quiet but critical role in producing dyes, pharmaceuticals, and research tools. The choices made around its use—balancing industrial benefits with health and environmental safety—will shape not just products, but the welfare of workers and consumers alike.
What are the safety precautions when handling 4-Toluidine Hydrochloride?
What Makes 4-Toluidine Hydrochloride Risky?
Chemical safety is a topic that gets dusted off whenever a substance with a tough name—like 4-Toluidine Hydrochloride—shows up in the workplace. You’ll find this chemical in dyes, pharmaceuticals, and lab research. It carries some real health risks. Direct skin contact, breathing in dust, or even swallowing small amounts can harm you. People handling this chemical face cancer risks. A report from the International Agency for Research on Cancer (IARC) flagged similar aromatic amines as carcinogens. The stakes go up for folks who spend hours in the lab, on factory floors, or in dusty storage rooms.
Setting Up Your Workspace for Safety
From personal experience, sharp attention to preparation can prevent most accidents. Always work in a fume hood or a well-ventilated area. That might sound simple, but even short exposure in stuffy rooms can irritate your eyes and lungs. Even after years of daily lab work, it still surprises me how quickly vapors can spread.
Personal protective equipment (PPE) forms your front-line defense. Always wear chemical-resistant gloves. Nitrile usually works; latex sometimes breaks down, so check manufacturer guidance for compatibility. Don’t forget a lab coat and safety goggles. Chemical-resistant aprons should be thrown on for larger batches or accident-prone tasks. If you ever had a chemical splash on your arm, you’ll never skip that apron again.
Handling, Storage, and Spill Cleanup
Handling risky chemicals calls for focus. Avoid eating or drinking nearby. Before even opening the jar, make sure all bottles and pipettes get labeled clearly. This sounds dull until you watch someone grab the wrong jar by mistake. Air-tight containers help prevent moisture, since this chemical reacts with water over time. Keep containers off the edge of work surfaces; spills near edges waste time and cause panic.
Spills never happen on schedule. Have an absorbent spill kit within arm’s reach. Sand and clay-based powders work better than commercial towels for this compound. Always collect waste in clearly marked containers—accidental mixing with bleach or acids generates dangerous gases. Double-bag waste, then store it for a hazardous waste company. Regular trash bins never cut it for compounds with carcinogenic potential.
Health Checks, Training, and Emergency Plans
Keeping up with training matters, not just for new staff. Refresher courses on chemical handling remind everyone of what’s at stake. In busy facilities, people get rushed and cut corners, which just encourages trouble. If anyone feels dizzy or nauseous, head outside and call for help immediately. Quick drenching in eye-wash stations or safety showers can keep a small incident from becoming permanent damage.
Pushing for a Stronger Safety Culture
Safety doesn’t come down to checklists and catchphrases. It’s about clear habits, strong oversight, and accountability. Supervisors have to enforce rules every day, or people stop following them. Breakrooms, supply closets, and storage rooms all matter. Ventilation systems need regular checks, and gloves need swapping way more often than some budgets allow.
Smaller companies sometimes skip air-monitoring or waste tracking, but those shortcuts catch up fast. I once saw a small lab lose two weeks to cleanup and health checks after a simple mislabeling mistake. Annual health screenings can pick up symptoms before bigger problems show up: blood checks, allergy tests, and skin exams help keep teams safe and on task.
Extra effort in planning and specialist training pays off every time someone goes home healthy. Safety isn’t a slogan. It’s a daily responsibility, especially with chemicals as risky as 4-Toluidine Hydrochloride.
What is the chemical formula of 4-Toluidine Hydrochloride?
What’s 4-Toluidine Hydrochloride, Really?
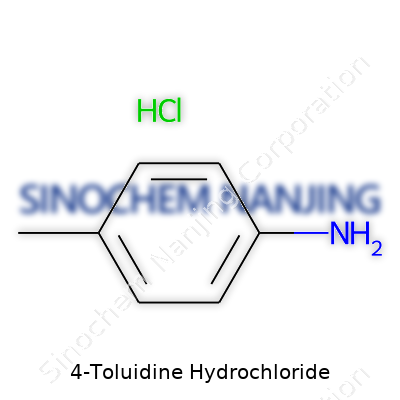
4-Toluidine Hydrochloride, with the chemical formula C7H10ClN, does more than just sit on a shelf in a laboratory. It earns its place in both chemical research and the world of manufacturing. The compound comes from 4-toluidine, a derivative of aniline, tweaked so the molecule packs a methyl group onto the fourth position of its benzene ring. Toss in hydrochloric acid, and a salt forms that many in chemistry call 4-Toluidine Hydrochloride or para-toluidine hydrochloride. The chemical name packs some punch, but the formula—C7H9N combined with HCl—keeps the composition straightforward and easy to grasp for those interested in its core essence.
How Is This Formula Useful in the Real World?
I remember the first time I handled 4-Toluidine Hydrochloride as a young lab tech. It didn’t jump out like some chemicals with wild colors or strong smells. Most people who come across it are usually working with dyes, pharmaceuticals, or certain types of polymers. The clear formula—C7H10ClN—means it fits neatly into reactions where a controlled amine is needed. Manufacturers rely on substances like this because they can be predictable and offer clean endpoints for processes, no guesswork involved.
Inside the factory, 4-Toluidine Hydrochloride often serves as an intermediate—meaning it steps into the spotlight for a short time and then passes the baton to something else. It plays a big part in making azo dyes, which end up coloring everything from fabrics to inks. Medical chemists also value the hydrochloride salt form, which they can handle more efficiently due to its increased stability and solubility compared to its free base form.
Stressing Safety with Everyday Chemicals
One thing I’ve learned through years spent surrounded by chemicals is the importance of safety. 4-Toluidine Hydrochloride is no exception. The Science Journal and regulatory data flag it as a substance that needs careful handling. Workers exposed to 4-toluidine derivatives have faced health issues; multiple toxicity studies have linked exposure to problems like skin and respiratory irritation, and chronic contact could even contribute to longer-term risks. Community and company protocols exist for a reason, and those who ignore gloves, goggles, and proper storage put themselves and coworkers at risk.
Pushing Forward with Responsibility
Responsible use starts with knowledge. Anyone working with chemicals like 4-Toluidine Hydrochloride needs to know not just the formula but also the potential impact on health and the environment. Following up-to-date safety data sheets, jumping on regular health monitoring, and keeping up with local and global guidelines can help lower workplace incidents.
On a larger scale, chemical producers can invest in closed systems to reduce leaks and spills, as well as better air filtration and waste reduction strategies. The science community keeps researching alternatives and green chemistry approaches—looking for ways to get the same performance with less risk. These solutions won’t wipe out risk overnight, but by giving attention to both formula and real-world application, the industry keeps edging closer to safer and smarter practices.
Final Thoughts on Knowing the Chemical Formula
Chemical formulas aren’t just memorization tasks for science class. They connect us to the nature of substances that make our world possible and remind us of the responsibility we hold. C7H10ClN—4-Toluidine Hydrochloride—is more than a string of elements. For anyone working in labs, factories, or environmental safety, knowing the formula sparks a deeper respect for both the molecule and the wider world it touches.
How should 4-Toluidine Hydrochloride be stored?
Why Proper Storage Matters
Working with chemicals comes with responsibility. My experience in small laboratories taught me early that people easily overlook the basics, like storage conditions. 4-Toluidine Hydrochloride is not some benign compound. It’s toxic and can cause harm through skin contact, inhalation, or accidental ingestion. Proper storage protects the health of people working around it, and it keeps the compound stable. If you just toss the container onto a shelf, you invite trouble—spilled chemicals, ruined experiments, unplanned exposure.
Main Storage Practices to Stick With
Keep this material in a tightly sealed container that’s clearly labeled. Labels should never smudge or tear—no one wants to be guessing after a few weeks. Use only containers made from materials that do not react with 4-Toluidine Hydrochloride. Make space in a dry, cool, and well-ventilated spot, away from heat sources, direct sunshine, or moisture. Humidity causes degradation in many chemicals, and 4-Toluidine Hydrochloride is no exception.
I have seen what careless handling does. Dried-out crusts on a bottle’s rim, mysterious colors in powder, and crusty caps can all signal trouble. A dry environment and away from strong oxidizers keep this from happening. Safety Data Sheets highlight that mixing this compound with oxidizing agents risks dangerous reactions—fire, even explosions in rare cases.
Health and Safety Above Convenience
People sometimes leave toxins in open workspaces for convenience. It feels easier to grab what’s needed quickly, rather than walking across the room to a chemical cabinet. This shortcut ignores potent health hazards. The National Institute for Occupational Safety and Health (NIOSH) describes 4-Toluidine Hydrochloride as a possible carcinogen. Researchers studying benzidine-derived chemicals (like toluidine compounds) found links to bladder cancer. Long-term storage out in the open might look harmless, but vapors build up and surface contact risk grows day by day.
Best practice is to use a ventilated chemical-storage cabinet, lockable and separated from chemicals that react dangerously. Workers should always have easy access to gloves and safety goggles nearby and a way to quickly rinse eyes and skin in case of exposure.
Disaster Preparedness
It’s too late to think about emergency response once an accident happens. An updated spill response plan, with absorbents ready to go and a clear protocol posted where people work, makes a big difference. I’ve seen teams scramble, unsure how to clean a powder spill, wasting time searching for materials and instructions. Local regulations often require a register of hazardous chemicals and routine checks on expiry dates and container integrity. Relying on regular self-inspection and involving the whole staff can catch cracks, leaks, and drying-out of stored chemicals before they reach a dangerous point.
Moving Toward a Culture of Safety
Ignoring storage protocols isn’t an option for anyone who values health or reliable research. Keeping 4-Toluidine Hydrochloride in the right place lowers exposure, protects investment in materials, and supports compliance with workplace safety laws. It's not just about ticking boxes. Building habits around careful handling protects everyone, from seasoned chemists to newcomers just learning the ropes.
Is 4-Toluidine Hydrochloride hazardous to health?
Understanding 4-Toluidine Hydrochloride
Anyone working in the world of chemicals knows certain names carry a reputation. 4-Toluidine Hydrochloride fits that mold. It shows up in the lab, in research, and in the dye industry. Without much attention, people could underestimate what it brings to the table, especially for health and safety.
What Makes It a Concern?
4-Toluidine Hydrochloride doesn’t just cause a mess on the lab bench. Touching it or breathing it in comes with real risks. Evidence collected by agencies like the U.S. National Toxicology Program shows clear links to cancer in lab animals. The International Agency for Research on Cancer lists the parent compound of toluidines as a possible human carcinogen. On top of that, there’s a track record of irritation—think burning eyes, cough, or red and itchy skin after exposure. Some researchers also point out that chronic exposure potentially damages the liver and kidneys, though more investigation helps fill in the gaps.
A Glimpse Into Reality at Work
Researchers and factory employees come in contact with 4-Toluidine Hydrochloride far more than most people ever do. Protective equipment matters in these settings. I remembered my first year around these labs—one slip with gloves or a half-tied mask, and dizziness or headaches followed. These aren’t one-off problems. Years of studies back up what you feel after breathing unfiltered powder: headaches, nausea, and poor sleep that comes from worrying about long-term effects.
Not Just a Personal Story: The Facts Back It Up
Look at the science: inhalation data from the U.S. Environmental Protection Agency shows even small exposures to toluidines produce blood cell changes in animals. Chronic exposure increases cancer risks in tests with rodents. Occupational safety agencies—OSHA and NIOSH—recommend tight exposure limits, proof enough that these dangers deserve real attention.
Why This Matters Beyond the Lab
Most people don’t handle 4-Toluidine Hydrochloride at home. It creeps up the supply chain in dyes, plastics, and other industries. Workers without proper training or safety gear face higher odds of harmful effects. Sometimes, older factories or underfunded setups still use clunky protocols. When coworkers feel unsafe, they think twice about speaking up—nobody wants to lose a paycheck for asking about clean air.
How Can We Fix It?
Safety culture makes the real difference. Proper gloves, fitted respirators, fume hoods—these should never sound like luxuries. Training needs frequent updates, not a once-a-year seminar where half the staff zones out. Clear labels and quick-access safety data sheets help everyone make smart choices in the moment. Strong policies work best when supervisors take them seriously and when workers see managers lead by example. Speaking up for safer workplaces—whether by testing air quality more often or insisting on better exhaust fans—carries real weight.
Folks dealing with 4-Toluidine Hydrochloride every week deserve strong protections based on facts and real-world stories. Hard evidence, worker experience, and a commitment to safety all shape a workplace that keeps health risks as low as possible. That’s where trust gets built, on the shop floor and in every lab.
| Names | |
| Preferred IUPAC name | 4-Methylaniline hydrochloride |
| Other names |
p-Toluidine hydrochloride 4-Aminotoluene hydrochloride 4-Methylaniline hydrochloride p-Methylaniline hydrochloride |
| Pronunciation | /ˈfɔːr təˈluːɪdiːn ˌhaɪdrəˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | 636-21-5 |
| 3D model (JSmol) | `3D model (JSmol)` string for **4-Toluidine Hydrochloride**: ``` NC1=CC=C(C=C1)C.Cl ``` |
| Beilstein Reference | 87876 |
| ChEBI | CHEBI:86341 |
| ChEMBL | CHEMBL507360 |
| ChemSpider | 82906 |
| DrugBank | DB12775 |
| ECHA InfoCard | The ECHA InfoCard of product '4-Toluidine Hydrochloride' is **"100.013.933"**. |
| EC Number | 212-012-4 |
| Gmelin Reference | 7717 |
| KEGG | C14382 |
| MeSH | D016072 |
| PubChem CID | 8655 |
| RTECS number | XU6425000 |
| UNII | 4P4KUP399P |
| UN number | 2811 |
| CompTox Dashboard (EPA) | DTXSID6013865 |
| Properties | |
| Chemical formula | C7H10ClN |
| Molar mass | 127.60 g/mol |
| Appearance | White to light yellow crystalline powder |
| Odor | ammonia-like |
| Density | 1.259 g/cm3 |
| Solubility in water | Soluble |
| log P | -0.1 |
| Acidity (pKa) | 9.8 |
| Basicity (pKb) | 5.08 |
| Magnetic susceptibility (χ) | -59.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.633 |
| Viscosity | Viscous liquid |
| Dipole moment | 3.63 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 176.7 J mol⁻¹ K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -80.0 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, in contact with skin or if inhaled; causes skin irritation; causes serious eye irritation; may cause an allergic skin reaction; may cause cancer; suspected of causing genetic defects. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H302 + H312 + H332: Harmful if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P264, P280, P301+P312, P302+P352, P305+P351+P338, P330, P337+P313, P332+P313, P362+P364 |
| Autoignition temperature | 193°C |
| Lethal dose or concentration | LD50 oral rat 2410 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 2650 mg/kg |
| NIOSH | WW3150000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 4-Toluidine Hydrochloride: **0.5 mg/m³** |
| REL (Recommended) | 0.5 mg/m³ |

