4-Tert-Butylphenol: A Commentary on Its Journey and Impact
Historical Development
Years ago, the quest for phenolic compounds opened new doors in the world of synthetic chemistry. Among those discoveries, 4-Tert-Butylphenol made a mark thanks to its unique structure. Chemists first explored its use as they searched for ways to improve resin production and find agents that wouldn’t break down easily under harsh conditions. Lab notes from the early 20th century show researchers curious about simple substitutions on phenol rings, but the addition of a tert-butyl group at the para position offered something more—resistance to oxidation and thermal stress. Through decades of research, as more industries relied on advanced plastics and coatings, this compound found a home not only in factories but in the products seen every day.
Product Overview
This white crystalline substance pops up in everything from adhesives to polymer synthesis. In my own time working alongside coatings engineers, 4-Tert-Butylphenol stood out for its ability to modify resins and add both flexibility and durability. It’s more than just a chemical—it has become a workhorse for manufacturers who need reliability, somewhere between mass production and precision tweaking.
Physical & Chemical Properties
With a melting point around 98°C and a boiling point near 238°C, 4-Tert-Butylphenol holds up in environments that would destroy other phenols. Its low volatility at room temperature lets workers handle it with fewer worries about inhaling dangerous fumes. Chemically, it behaves just like a typical phenol when treated with strong bases, but the bulky tert-butyl group adds a stubbornness, making it less likely to break apart or react in unwanted side reactions. Its solubility in organic solvents like benzene and alcohol has helped it blend in so well with both polar and non-polar systems.
Technical Specifications & Labeling
Industries have pressed for clear labeling as regulations tightened. I’ve seen boxes marked “4-tert-Butylphenol, 99% pure, CAS 98-54-4” shipped to polymer labs. Manufacturers now include data on melting point, minimum assay, and the packaging’s compatibility with reactive chemicals. These labels aren’t just regulatory hurdles—they carry essential technical details for process engineers who have to hit narrow margins for quality. Even the type of packaging material shows up on spec sheets, something that matters more than folks outside the plant might think.
Preparation Method
Making this compound takes careful planning. One large-scale method starts with reacting phenol with isobutylene under acidic conditions. Strong acid catalysis encourages the tert-butyl group to attach at the para position rather than anywhere else on the ring. Controlling reaction temperature, pressure, and acid type makes all the difference in yield. For folks in smaller labs, milder routes exist, but upscaling always means keeping an eye out for runaway reactions—something I’ve seen colleagues sweat over while keeping glassware cool and reaction times short.
Chemical Reactions & Modifications
The chemistry behind 4-Tert-Butylphenol keeps drawing in curious researchers. It behaves as a typical phenol for esterification, etherification, or oxidative coupling. That tert-butyl branch resists attack from mild oxidizers, so the core remains functional through tougher processes. Modifications often target the hydroxyl group, forming novel resins or hardeners. In some specialty resin applications, derivatization helps boost resistance to UV or chemicals, a pretty useful trick for coatings used outdoors or in automotive parts.
Synonyms & Product Names
Ask around in a chemical warehouse, and you’ll hear names like “P-tert-Butylphenol,” “4-(1,1-dimethylethyl)phenol,” and sometimes “PTBP.” Extra trade names sprout up wherever specialty chemical suppliers want to lock in a brand. Regardless of how it’s labeled, the structure and consequences remain constant.
Safety & Operational Standards
Sitting in on trainings with plant operators, the warnings come thick and fast. Skin contact leads to irritation and redness, so gloves and goggles are standard. Inhalation risks exist in powder-handling operations, but well-ventilated spaces and dust collectors go a long way. Regulatory agencies like OSHA and REACH want up-to-date risk assessments and traceability. Over the years, I’ve watched safety data sheets expand from a page or two to multi-section dossiers that cover fire-fighting, spill procedures, and chronic exposure risks. Emergency showers and first-aid kits stay close by—not just for show, but as a core part of daily routines.
Application Area
Look around any shop using epoxy or phenolic resins, and 4-Tert-Butylphenol probably plays a behind-the-scenes role. Its reaction with formaldehyde or epoxy resins allows for strong adhesives, laminates, and insulation materials. In paints and varnishes, it helps plastics and coatings stay tough even in sunlight or variable weather. During my time looking at consumer safety, it came up in discussions around stabilizers for thermal papers and certain plastic housings. The compound even finds a niche in pharmaceuticals and agricultural chemicals, demonstrating just how widely it reaches.
Research & Development
Driven by demands for better and safer materials, R&D teams pour over new uses and derivative molecules. Research papers continue to track ways to tweak the tert-butyl group or add functional groups without losing the core phenolic activity. In collaboration with academic labs, I learned that computational chemists now model reaction pathways to minimize waste and energy use in synthesis. Investigating biodegradable alternatives has gained speed, but so far, 4-Tert-Butylphenol holds a unique spot thanks to its performance, especially where durability cannot be traded for green appeal.
Toxicity Research
Over years of safety meetings and literature reviews, one theme keeps coming up: about the impact on workers and the environment. Animal studies show acute toxicity can appear at high doses, leading to liver and kidney damage, but occupational exposures stay low with proper handling. Chronic effects tend to be minor at regulated levels, yet water solubility brings concerns about runoff. Researchers keep mapping out metabolic breakdown and environmental degradation, highlighting the need for tighter effluent control at production plants. Transparency in toxicology testing reassures regulators, but it also gives companies the knowledge to act on near-miss scenarios and manage risks before incidents happen.
Future Prospects
The push to keep products safe and high-performing drives the focus onto alternatives, but nothing delivers quite like 4-Tert-Butylphenol in tough resin or coating jobs. With stricter environmental regulations on the horizon and sustainability topping corporate agendas, demand for safer production methods and greener derivatives grows. Investment in closed-loop synthesis, improved recovery of spent chemicals, and real-time monitoring of environmental impacts stand out as practical solutions. Collaborations between academia and industry hold promise in designing next-generation resins that either degrade cleanly or recycle more easily without giving up on the qualities that 4-Tert-Butylphenol brings to the table. The compound’s future isn't just about churning out more product—it’s about responding to evolving standards, tighter rules, and customer demand for both performance and safety.
What is 4-Tert-Butylphenol used for?
Chemistry in Daily Life
4-Tert-Butylphenol often goes unnoticed in conversations, but plenty of us use products that depend on it. This compound, shaped by a distinct structure with a bulky tert-butyl group attached to a phenol ring, sits at the crossroads of industry, home, and the environment. In my own experience working on product development for coatings and adhesives, I’ve seen first-hand how crucial its role is. People may not ask for this name at the store, yet what it helps create surrounds us every day.
Resins, Adhesives, and Once-Unstickable Things
Try building a wooden chair that never wobbles, repairing a car interior, or simply handling glossy receipt paper—4-Tert-Butylphenol plays a quiet role in making these tasks possible. In phenolic resins, it boosts both heat stability and mechanical strength. Cabinetmakers, car repair shops, and even folks like me fixing squeaky doors often reach for adhesives where this chemical quietly does its job. Without it, many synthetic resins lose their resistance to heat and chemicals, so the results start to disappoint in a matter of months.
Electrical Goods and Safety
Anyone who’s opened up old electronics has seen brownish panels and circuit boards. That neat, solid plastic laminate owes a lot to 4-Tert-Butylphenol. Its ability to toughen insulation and help coatings resist harsh conditions makes it valuable in electrical insulators. During college, I spent hours in labs testing different plastic insulations—those containing this compound consistently outperformed others under heat and pressure. When you’re talking about things plugged into walls or living outdoors in all seasons, every bit of toughness counts.
Specialty Coatings and Papers
Thermal paper, used for receipts and tickets, relies on coatings that contain 4-Tert-Butylphenol. The paper reacts and develops sharp images when exposed to heat. As someone who’s worked in the printing supplies sector, I saw how reliable results demand chemical stability—faded receipts hurt both cashiers and customers. Water-resistant coatings for outdoor gear and floor varnishes also benefit from its presence, helping finishes last longer under scrapes, spills, and sun.
What About Safety and the Environment?
The chemical industry pays close attention to health. Prolonged exposure to 4-Tert-Butylphenol can cause irritation and shouldn’t be taken lightly by workers. Factories implement strict protocols—proper gloves, ventilation, safe handling—to protect workers, following rules laid out by experts and regulators. Many companies also track how much gets released into water supplies or air. This reflects a growing sense of responsibility throughout manufacturing. As a father, I understand the push to make products both sturdy and safe. Looking at data from environmental monitoring, most risks get controlled with smart design: recycling efforts, correct waste treatment, and sustainable sourcing of raw materials.
Innovation and Smarter Solutions
As science expands, demand grows for safer alternatives. Research centers keep searching for phenol replacements, especially in sensitive products like food packaging or toys. Developing alternatives takes time and patience; I’ve seen teams testing new combinations that might one day match the chemical’s unique properties without downsides. Good stewardship means pushing for less waste, safer substitutes, and clearer labelling so buyers can make informed decisions.
Beyond the Lab Bench
4-Tert-Butylphenol has shaped our household gadgets, construction, and industrial processes. Whenever people talk about chemical risks, it pays to remember the logic that drives the search for balance. Understanding why these ingredients matter makes it easier to support safer products and better choices in the long run. Real progress needs science, straight talk, and a willingness to look beyond the label toward real-life impact.
What are the safety precautions when handling 4-Tert-Butylphenol?
Rolling Up Sleeves: Dealing with a Serious Chemical
Seeing 4-Tert-Butylphenol pop up in the workplace isn’t rare, especially in paint, rubber, and plastic plants. This white, flaky substance may not look dangerous, but looks can fool anyone. Workers and lab techs face risks—from skin burns to breathing problems—without proper preparation.
Why Skin Matters Here
Anyone who’s ever had a run-in with chemical burns knows the pain doesn’t just go away when you rinse off. 4-Tert-Butylphenol is notorious for causing irritation or full-on burns after skin contact. I once watched a coworker deal with red, blistered patches after a single accidental splash. Gloves alone aren’t enough. You want to use chemical-resistant gloves—nitrile or neoprene, not just a cheap disposable type. Sleeves, aprons, and goggles round out this set of protection. Splatters bounce, and even a drop or two can mean a visit to the medic.
Air Quality: Not Just a Luxury
Noses and lungs take a beating when dust or fumes fill the air. Breathing in 4-Tert-Butylphenol dust stings the respiratory system and can trigger headaches or dizziness. Relying on natural airflow isn’t smart; you need exhaust ventilation and reliable respirators. In one shop, the difference between working with and without proper ventilation was obvious: fewer coughs, headaches, and panicked runs to the eye-wash station.
Mistakes Multiply in the Storage Room
Inside a busy plant, containers of this material often get stacked next to acids or bases. That’s an accident waiting to happen. 4-Tert-Butylphenol needs a cool, dry spot away from anything reactive. No metal shelves, since it can corrode them over time. Every container deserves a clear label, and everyone nearby should know where the spill kit sits. I’ve watched minor leaks turn chaotic when nobody knew how to react; practice spill drills like fire drills.
Spill Response: Speed Saves Skin
No matter how careful you are, spills happen. I once dropped a beaker thanks to sweaty gloves. Fast action matters. Throw on full gear: gloves, goggles, long sleeves. Scoop or sweep away the flakes—never sweep dry dust with bare hands since the powder goes airborne. Dispose of waste in a dedicated, sealed bin. Afterwards, wash every bit of skin and rinse eyes if there was even a chance of exposure.
Learning Together
Every workplace should talk openly about safety lapses. There’s no shame in sharing a story about getting burned or leaving a lid loose—those stories keep others from repeating mistakes. Regular training connects new hires and old hands, and updates on gear or protocols reach everyone. Posting emergency contacts and procedure checklists in plain sight helps when nerves fray.
Building a Safety Culture
Careful preparation builds trust and reduces injuries. Even when routines get boring, fatigue or rushing leads to accidents nobody wants. Regular gear checks, clear labels, and honest talk make the difference between a safe day and a trip to the ER. Living with less drama in the lab or warehouse means everyone gets to go home in one piece.
What is the chemical formula and structure of 4-Tert-Butylphenol?
Why 4-Tert-Butylphenol Comes Up Often in Chemistry Circles
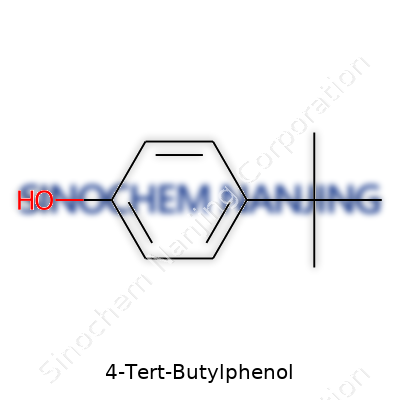
The name 4-tert-butylphenol pops up in plenty of labs, workshops, and chemical catalogs. It stands out as a building block for materials and specialty resins. Interest keeps growing around substances that mix durability with unique property sets, and this compound ticks many boxes. Its chemical formula, C10H14O, might look simple, yet a closer look at its structure shows just how much impact small molecular tweaks can have.
Breaking Down the Structure
Picture a benzene ring—six carbons joined in a hexagon. Now, if you attach a tert-butyl group at the fourth carbon, and a hydroxyl group (-OH) at the first, you get 4-tert-butylphenol. The tert-butyl group brings three methyl branches tied to a single central carbon. This arrangement bulks up the molecule, raising its melting point and changing how it dissolves in different solvents.
Here’s a closer look: The hydroxyl group sits attached to carbon number one on the ring, showing off its classic phenol nature. The tert-butyl group, located at position four, gives the molecule its name and distinct chunky structure:
- Benzene ring as the foundation
- Hydroxyl group at the para (1-) position
- Tert-butyl group at the para (4-) position
This formula, C10H14O, might not turn heads at first. Still, the positioning of these groups flips the script on the compound's reactivity and physical properties. As someone who’s handled 4-tert-butylphenol in the lab, I noticed the substance acts much less volatile than regular phenol. The tert-butyl tail makes it less willing to vaporize or react just for the sake of it, which saves headaches both for storage and safety.
Where the Real-World Impact Shows Up
In practice, this molecule makes waves in resin synthesis, adhesives, and even UV stabilizers used for plastics. The tert-butyl's size means it fits into polymer chains without letting them tangle too closely, which helps control brittleness and solubility. Chemists often lean on 4-tert-butylphenol to beef up epoxy resins that need to last through heat and weather.
This touch of stability isn’t just nice from a performance standpoint—it cuts down the risk during manufacturing too. Small changes in the structure influence the way resins cure and how much effort needs to go into cleaning up after a reaction. Less runaway reaction risk means safer workflow for folks on the production floor.
What Stands in the Way
Handling phenolic compounds, bulky or not, often raises questions about skin sensitivity and environmental safety. While the tert-butyl addition makes 4-tert-butylphenol less hazardous compared to raw phenol, you still need gloves and solid ventilation when working with it. Waste management rules keep tightening up, pushing manufacturers to upgrade handling and disposal.
Change is possible through better engineering controls and upstream substitutions. Some labs look for biosynthetic pathways or greener substitutes with lower toxicity, but 4-tert-butylphenol still holds its ground thanks to versatility and performance. The right mix: tighter controls, smarter safety routines, and continuing research into even safer molecular cousins.
How should 4-Tert-Butylphenol be stored?
Challenges of Storing 4-Tert-Butylphenol
4-Tert-Butylphenol sits among the more stubborn chemicals in any workplace. Take it from anyone who has spent time around labs or warehouses: fumes and spills from this compound do more than frustrate—they carry real risks. Anyone who’s caught a whiff after an uncapped drum understands the irritation that follows. There’s more at stake than just an unpleasant odor. Skin, eyes, and lungs can get inflamed fast if basic precautions get ignored.
Why the Right Conditions Make a Difference
This chemical has no business near heat or sunlight. High temperatures push up vapor release and increase the odds of a fire. Even a modest spark finds more fuel if the environment stays hot or if vapors collect. Storing it in a cool spot, with good airflow, is not just about ticking safety boxes. It means fewer headaches—literally and legally.
Some folks might underestimate the damage moisture can do. Water creeping in leads to slow breakdown and messes up the container’s integrity. Leaks show up sooner than you'd think. A dry space with secure shelving and spill trays gives workers and property a fighting chance. Some older buildings let humidity creep inside; a humidity monitor and regular checks keep surprises at bay.
Container Choices and Labeling
Trust canisters made from materials proven to keep out air and light. Metal drums with lined interiors or thick HDPE containers work well, but only if they close up tight. Labels should never lose their text. Wear-and-tear, chemical drips, or fading markers have caused more confusion than many want to admit. Keeping safety symbols clear helps new employees avoid rookie mistakes and gives first responders the info they need during an emergency.
Segregation and Inventory Strategies
Some of the riskiest situations pop up when this chemical sits next to strong acids or bases. I’ve seen cross-contamination trigger odors sharp enough to chase people out of a room. That’s why shelves should separate such chemicals with a real barrier. Regular inventory checks do more than stop pilferage—they show if a container has started bulging or sweating, hinting at a bigger problem inside.
Extra Steps to Lower Risk
Think about the folks moving these containers. Training helps, but a simple floor plan with clear walkways and labeled emergency exits steps up real safety. I remember one warehouse with weekly drills—no one forgot what to do, and minor leaks got spotted before turning serious.
Absorbent mats near the storage area and easy-to-reach eyewash stations turn a spill from disaster into a quick fix. Eye protection, gloves, and long sleeves remain must-haves every time.
Staying Proactive Matters
Storing 4-Tert-Butylphenol safely means getting ahead of problems. Smart planning, regular maintenance, and teamwork stop issues from ever reaching crisis mode. Real safety grows from shared experience and honest attention to the basics, not just rules on paper.
Is 4-Tert-Butylphenol hazardous to health or the environment?
Real Risks—Not Just Laboratory Talk
People sometimes trust that chemicals with complicated names belong strictly in a lab. The truth feels different once you notice those same chemicals everywhere: plastics, paints, adhesives, even circuit boards. 4-Tert-Butylphenol stands as one of those compounds with a complicated reputation. Its uses stretch across manufacturing, but the question sticks around—what does it really do to people and the world outside those factories?
Human Health: Why Worry?
Anyone who’s handled cleaning supplies knows skin burns aren’t just a hypothetical risk. 4-Tert-Butylphenol packs a serious punch. The U.S. National Library of Medicine tracks documented cases of skin and eye irritation, and the EU describes it as corrosive to eyes. People exposed at work talk about rashes, itching, headaches, coughing, and nausea. Sometimes the symptoms show up before you notice a smell. Long-term exposure hasn't made headlines the way classic carcinogens like asbestos have, but chronic low-level contact raises big questions.
Inside the body, phenols often linger. 4-Tert-Butylphenol can get through the skin. Chronic exposure deserves attention in industries using this compound for coatings or resins. Companies owe it to their workers to provide solid gloves, face protection, and good ventilation, not just a quick slap on the wrist after an accident.
The Ripple Effect on the Environment
A lot of chemicals vanish down the drain or out smokestacks. 4-Tert-Butylphenol ends up in rivers and groundwater. Now, fish and small aquatic animals don’t have chemical-resistant gloves. This compound disrupts their reproduction and development, with studies linking it to hormonal interference. Those tiny disruptions ripple out: food webs spiral, populations drop, and water quality tanks. Scientists from Japan’s National Institute for Environmental Studies reported that water with just a whisper of this chemical lowered growth rates in fish larvae.
Plant life doesn’t dodge the problem. Soil contamination affects roots and growth, making it tougher for local plants to get their nutrients. Factories aren’t the only guilty party. Household runoff brings its own share—scrubbing paint trays or washing out plastic containers releases traces bit by bit.
The Search for Solutions: Responsibility and Action
Breakthroughs never arrive overnight, and chemical safety relies on both rules and real habits. Always check the safety data sheet before handling anything unfamiliar. Companies should update processes with safer alternatives, invest in closed-loop systems, and double-check waste disposal routines to catch leaks. Municipal water treatment, when it includes activated carbon filters, can catch some phenolic compounds before they hit the watershed.
Governments set the tone through laws and oversight, but regular folks can push for greener choices. Supporting companies that use less-toxic chemicals or recycled materials makes a difference. Each piece of the puzzle—whether it’s a careful factory manager or a homeowner recycling plastics—plays into the solution. Personal experience with chemical exposure reminds me that real progress feels slow but adds up. Small steps save skin, lungs, and rivers. Staying curious and informed about what surrounds us helps spot danger long before it turns into disaster.
| Names | |
| Preferred IUPAC name | 2-(tert-Butyl)phenol |
| Other names |
p-tert-Butylphenol 4-(1,1-Dimethylethyl)phenol 4-tert-Butylphenol 4-t-Bu-Phenol |
| Pronunciation | /ˌfɔːr tɜːrt ˈbɜːtəlˌfiːnɒl/ |
| Identifiers | |
| CAS Number | 98-54-4 |
| Beilstein Reference | 1209227 |
| ChEBI | CHEBI:34762 |
| ChEMBL | CHEMBL1522 |
| ChemSpider | 6922 |
| DrugBank | DB02240 |
| ECHA InfoCard | ECHA InfoCard: 100.011.042 |
| EC Number | 202-679-0 |
| Gmelin Reference | 6137 |
| KEGG | C06508 |
| MeSH | D016696 |
| PubChem CID | 7908 |
| RTECS number | OG5425000 |
| UNII | 82IUA43H7E |
| UN number | 2430 |
| CompTox Dashboard (EPA) | DTXSID9020814 |
| Properties | |
| Chemical formula | C10H14O |
| Molar mass | 150.22 g/mol |
| Appearance | White crystalline solid |
| Odor | Characteristic phenolic odor |
| Density | 0.943 g/mL |
| Solubility in water | slightly soluble |
| log P | 2.9 |
| Vapor pressure | 0.002 mmHg (25°C) |
| Acidity (pKa) | 11.3 |
| Basicity (pKb) | 13.2 |
| Magnetic susceptibility (χ) | -61.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.532 |
| Viscosity | 3.9 cP (20°C) |
| Dipole moment | 2.79 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 319.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -389.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3327.8 kJ/mol |
| Pharmacology | |
| ATC code | D11AX10 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H411 |
| Precautionary statements | P261, P280, P305+P351+P338, P310, P333+P313, P362+P364 |
| Flash point | 113 °C |
| Autoignition temperature | 545°C |
| Explosive limits | Explosive limits: 1.1–6.9% |
| Lethal dose or concentration | LD50 oral rat 3,600 mg/kg |
| LD50 (median dose) | 'LD50 (median dose): Rat oral 4000 mg/kg' |
| NIOSH | UR8750000 |
| PEL (Permissible) | PEL: 5 mg/m3 |
| REL (Recommended) | 8 mg/m³ |

