4-Nitrosophenol: An In-Depth Look at a Chemical with Many Faces
Historical Development
Organic chemistry has always thrived on curiosity and accident. 4-Nitrosophenol, an aromatic nitroso compound, reflects the kind of chemical that drew attention back in the days of dye research and synthetic experimentation. In the late 19th century, a handful of chemists probed the nitrosation of phenol derivatives, pushing reactions under acidic or basic conditions and taking note of vivid green and yellow hues. Early documentation linked nitrosophenols to color-changing reactions in analytic tests. The story of 4-Nitrosophenol spins out of those foundational studies. Labs searching for new dyes and intermediates documented methods for direct nitrosation, publishing data on yields, tints, and behaviors. From those early days, this compound found its way into both basic textbooks and applied research. Living through two world wars and the evolution of chemical industry standards, 4-Nitrosophenol stuck around because of its reactivity and its use as a stepping stone to other, more complicated molecules.
Product Overview
This compound, with the chemical formula C6H5NO2, belongs to the group of aromatic nitroso compounds. Structurally, it’s a phenol ring with a nitroso group hanging at the para (4) position opposite the hydroxyl group. What once sat in glass bottles as a curiosity now shows up in fine chemistry labs and industrial catalogs under a range of names. Producers typically supply it in crystalline form or in well-sealed vials to avoid air and light-induced changes. It has gained steady demand because it acts as a useful intermediate during the manufacture of dyes or as a test reagent.
Physical & Chemical Properties
4-Nitrosophenol presents itself as a pale yellow-green crystalline solid, sparking curiosity with its taste for color changes under different conditions. Its melting point ranges between 150 and 155 °C, depending on purity and sample handling. Solubility patterns in common solvents mirror most phenols: it dissolves readily in alcohols, slightly in water, and displays moderate behavior in ether. The nitroso group gives it a stronger electron-withdrawing effect compared to regular phenol, shifting its acidity and reactivity. It often undergoes slow changes when exposed to air, picking up oxygen and sometimes fading or darkening with time.
Technical Specifications & Labeling
Suppliers dealing in analytical-grade 4-Nitrosophenol will detail purity percentages, moisture content, and trace impurity profiles. Labels usually include batch numbers, molecular weight (123.11 g/mol), and hazard warnings. Certificates of analysis, checked through gas or liquid chromatography, are part of modern compliance. Product packaging, often in amber glass to block UV light, must carry the appropriate GHS/CLP hazard pictograms and first-aid instructions. Workers handling stocks in industrial settings quickly learn to follow manufacturer's MSDS sheets, which clarify operational limits and incompatibilities.
Preparation Method
Synthesis of 4-Nitrosophenol has carried the flavor of classical organic chemistry and process improvement over decades. The standard pathway runs through direct nitrosation of phenol, carried out by introducing sodium nitrite into an acidic aqueous solution of phenol. This in-situ release of nitrous acid helps limit side reactions. Reaction temperature never strays above the cool range, frequently held near 0–5 °C. Later, extraction pulls the product from the mixture using ether or another solvent. Crude solids get washed and recrystallized to yield product pure enough for most research or industrial uses. Process chemists have tinkered with minor improvements, such as phase transfer catalysts or organic bases, to nudge up yields and streamline purification steps.
Chemical Reactions & Modifications
The molecule stands out because both of its functional groups—the phenolic hydroxyl and the nitroso group—lend themselves to further transformations. In alkaline conditions, 4-Nitrosophenol can oxidize to quinone imines or rearrange, opening new synthetic routes. The electron withdrawal of the nitroso group tempers its reactivity in electrophilic aromatic substitution, leading researchers to use special techniques for additional modification. In the dye industry, it often couples with aromatic amines or acts as an intermediate in azo dye synthesis. Adding mild reducing agents converts it to the corresponding amine, while oxidizing agents can push it toward nitrophenol derivatives. Such reactivity keeps it front and center in synthetic strategy sessions.
Synonyms & Product Names
Anyone who has spent time in a chemistry stockroom recognizes how confusion sprouts from synonym lists. 4-Nitrosophenol also appears as para-nitrosophenol, 4-hydroxynitrosobenzene, and p-nitrosophenol. Chemical catalogs sometimes flip the numbering for European audiences (1-hydroxy-4-nitrosobenzene). Research journals sticking to IUPAC notation provide clarity, but shorthand names persist in practice. Such naming diversity means care is needed when ordering, documenting experiments, or cross-referencing safety data.
Safety & Operational Standards
Handling a compound with both a hydroxyl and a nitroso group calls for respect. The nitroso functional group, historically overlooked, can liberate toxic species or engage with DNA. Irritation of skin, eyes, or respiratory tract remains a risk, especially in large-scale handling or poorly ventilated spaces. Workplace standards in Europe and North America require goggles, robust gloves, and fume extraction. Waste handling must avoid mixing this material with reducing agents or acids outside controlled conditions, since unpredictable decompositions or releases of nitrogen oxides can occur. Training focuses on storage in cool, dark, dry locations and immediate cleanup of spills. In academic labs, undergraduates learn these skills early, while industrial sites mandate regular safety reviews and emergency drills.
Application Area
The compound’s real-world use comes through its chemistry, which finds relevance in pigment synthesis and analytic testing. The vivid color shift that 4-nitrosophenol provides forms the bedrock of several colorimetric assays, especially for trace metals. It crops up in procedures that check for the presence of formaldehyde via chromogenic reactions. Those same color properties made it an early favorite in dye research, although newer, safer compounds have whittled down its direct use. In pharmacology, sometimes it shows up as a probe for studying enzyme activity, especially enzymes seeking out nitroso groups. In rare cases, engineers turn to it as a model substrate in photochemistry, giving it credit for helping to elucidate details about solar energy capture in organic molecules.
Research & Development
Graduate students and postdoctoral fellows who have explored nitrosoarene chemistry know that 4-nitrosophenol has pulled its weight in mechanistic and kinetic studies. The two functional groups offer useful contrast in transition-metal catalysis. As synthetic targets grow more involved, chemists lean into the compound’s ability to act as a foundation for new ligand structures or as a launching point for combinatorial libraries. Researchers in environmental science sometimes use it when testing methods for wastewater purification or radical-driven degradation. IT continues to hold its niche, not as a blockbusting chemical, but as a reliable tool for probing reactivity and tracking reactions through easy visual cues.
Toxicity Research
Decades of work probing aromatic nitroso compounds have flagged 4-nitrosophenol for careful handling. Animal studies and in vitro data hint at cytotoxicity, with mechanisms involving interaction with thiols and production of reactive nitrogen species. Repeated exposure can irritate mucous membranes or lead to contact dermatitis. Longer-term research flags concerns about mutagenicity, though conclusive links remain sparse. Regulatory attention picks up for nitroso compounds in general, leading to classifications as potential occupational hazards. Animal studies, often cited in safety documentation, show dose-dependent acute toxicity, but better epidemiological data are needed to firm up risk assessments for regular human exposure. Current advice focuses on minimizing contact and using the smallest amounts required for analytic or industrial aims.
Future Prospects
Anyone spending time in chemical research knows trends can shift quickly. 4-Nitrosophenol, though not a star commodity, will remain relevant as a reference and mechanistic probe. Environmental chemists may revisit it as nitroso compound fate in soils, or as markers for pollution events. Its role in teaching laboratories appears secure: few compounds showcase such a mix of recognizable structure, useful color changes, and informative reactivity. Medical research sometimes circles back to nitroso compounds during drug development, given their reactive oxygen and nitrogen profiles. The next decade may see its applications inch forward through catalysis or as a model system for green chemistry innovations.
What is 4-Nitrosophenol used for?
What We Know About 4-Nitrosophenol
Ask a group of chemists about 4-Nitrosophenol, and some might remember handling it in the lab, often with a bit of caution. This bright yellow compound comes up in a handful of chemical processes and research experiments. Though it’s not something you’ll spot in daily life, it plays a quieter but important role in the background of science and industry.
Why Chemists Reach for 4-Nitrosophenol
The spotlight lands on 4-Nitrosophenol in dye manufacturing. It forms a core raw material in creating certain azo dyes, some of which end up coloring textiles, inks, or food. These dyes rely on strong, stable base chemicals, and 4-Nitrosophenol brings those qualities. Color chemists need agents that deliver consistent shades and don’t wash out quickly—traits built on molecular stability. Using 4-Nitrosophenol challenges people to pay careful attention, because it can react fast and lead to a range of end products.
Synthetic chemistry leans on 4-Nitrosophenol for making compounds beyond dyes as well. The nitroso group grabs interest for its ability to join with other chemicals, opening doors to more complex molecules. Researchers turn to this compound when studying chemical reactions involving nitroso groups. Sometimes it sparks off tests of antioxidants or measurements of enzyme activity, especially in biochemistry labs.
Safety and Health in Handling
In my own lab shifts, nobody passed a bottle of 4-Nitrosophenol without a proper glove check. The compound poses health risks—skin contact, inhalation, or improper storage can spark trouble. Laboratories store it under tight protocols with good ventilation and secure containers. Research published by the National Institute for Occupational Safety and Health links similar nitrosocompounds to health hazards, so extra caution isn’t optional. This means anyone working with 4-Nitrosophenol often wears full PPE, and chemical hoods run at full suction.
Environmental Impact
Disposal and wastewater management raise bigger questions. 4-Nitrosophenol’s chemical family sometimes includes substances flagged for pollution concerns. If factories let effluent slip out untreated, communities downstream pay the price. Results from studies in environmental journals highlight nitrosophenol residues as persistent in soil and water. This problem needs real-world solutions—tighter discharge policies, better treatment processes, and regular monitoring. Limiting environmental damage starts with setting real accountability and investing in cleanup technology.
What Could Make a Difference
Science shouldn’t only serve product pipelines or patents but also public health and ecosystems. Manufacturers could hunt for safer alternatives or greener processes. Lab leaders and institutions might share best practices on training, labeling, and containment. Investing in analytical technology helps trace leaks before they spread. Fostering a safety culture takes more than rulebooks; it’s built on open conversation and shared responsibility. Regulators push for stricter standards, yet action at the ground level—inside labs, on factory floors—often drives real change.
Where Curiosity Leads
Young chemists, environmental advocates, and industrial decision-makers all touch the 4-Nitrosophenol story from different angles. Learning how such compounds shape, color, and sometimes harm our world puts power in our hands. Every step in handling, using, or replacing a chemical carries weight. When those choices get made carefully, with eyes on safety and sustainability, both science and society benefit.
What are the safety precautions when handling 4-Nitrosophenol?
Knowing the Risks Before You Start
4-Nitrosophenol doesn’t get used in everyday labs for no reason. It possesses a strong chemical punch, both in terms of its potential toxicity and its hazards to skin and airways. Its nitroso group signals harm pretty clearly; I’ve seen too many researchers rush into similar substances, underestimating them out of routine. Those days are over once you’re exposed to enough headaches, rashes, and ruined gloves.
Personal Protection in Practice
Forget bare hands. Nitrile or butyl rubber gloves offer the biggest barrier against 4-Nitrosophenol’s stubborn nature. Lab coats help, but tight cuffs and full-length sleeves save you from regret later. Splash-proof goggles keep your eyes from accidental sprays—even small pipetting mistakes can shoot droplets where they shouldn’t land.
A quality lab coat and gloves do more than impress safety auditors. Years ago, a postdoc I worked with tried swapping her gloves for latex—and ended up with a chemical burn. Nitrile lasted through the task, no traces left. Chemical resistance charts aren’t for show; they’re survival manuals.
Air Quality and Fume Safety
Open benches present too many risks. The fumes that 4-Nitrosophenol can produce won’t always have an obvious smell, but inhalation risks grow with every open container. Work inside a certified fume hood, not close to the door or near fans where eddies pull vapors out into the room. I watched a seasoned chemist skip this step; the resulting cough stuck for days and reminded the rest of us that even quick tasks deserve respect.
Spills: Preparation Beats Panic
Nearly every chemist I know has dropped a vial or watched a bottle tip. Spills with this chemical demand calm and fast action. Pouring kitty litter or commercial absorbent around the spill keeps it contained. Dam the area, scoop up solids using dedicated dustpans, and never sweep. Wet paper towels soak up small droplets, but wear gloves throughout. Bag everything as hazardous waste; labeling matters, especially when bins sit for days before pickup.
Storage: Smart Choices in the Stockroom
Never toss 4-Nitrosophenol on a shelf next to acids, bases, or oxidizers. Dedicated chemical storage cabinets, built for flammables and toxics, give peace of mind. Store the material in a sealed container with clear labeling—date, contents, hazard warnings. Climate control keeps it from degrading, which could spike risks even higher.
Waste Disposal: No Shortcut Worth Taking
Down the drain isn’t an option. I’ve seen students tempted, pressured by deadlines. Regulations cover disposal of any nitroso compounds. Always use containers designed for hazardous waste, and keep a waste log. Many universities have formal pickup programs; that one phone call keeps the chemical stream safe for everyone downstream—from custodian to landfill worker.
Step Up Training and Culture
Routine safety drills, signage, and frequent reminders save more than just skin or lungs—they keep whole teams working. Encouraging every person in the lab to speak up about near misses and unknown risks changes how we all see hazardous work. Leadership doesn’t rest with the supervisor—it belongs with anyone holding the bottle.
Conclusion
Handling 4-Nitrosophenol demands a higher level of respect and preparation. Assign safety routines the same patience as experimental design; in a field where a single mistake can change lives, experience counts for everything.
What is the chemical structure and formula of 4-Nitrosophenol?
Looking Closer at the Structure
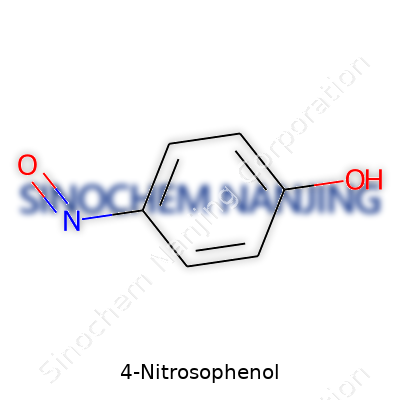
Many of us have passed through chemistry lessons without thinking too hard about the real-world significance of chemical structures. Take 4-Nitrosophenol, for instance. Right away, the name points to what’s going on: a phenol ring with a nitroso group sitting at position four. Simple on paper, maybe, but the arrangement of functional groups on this six-carbon ring shapes behavior and reactivity in the lab.
The skeletal formula for 4-Nitrosophenol is C6H5NO2. The molecule builds on a benzene ring, which brings a sort of chemical resilience and resonance. This ring features an -OH group (the phenol part) at one spot, and sporting a -NO group (the nitroso group) right across from it—hence the “4” in the name. Chemists call this the para position. The nitroso group carries a nitrogen double-bonded to an oxygen, so that brings a reactive flair, not just an extra atom here and there.
Bringing It Into Practice
Chemistry isn’t only about drawing lines and counting atoms. In the real world, these structures make a difference. Phenol rings turn up everywhere, in everything from aspirin to antiseptics. The nitroso group, by itself, gets noticed for interacting with biological molecules, for mediating color changes, and for shaping how compounds behave under light. With 4-Nitrosophenol, these two features combine on the same platform.
Experience in the lab has taught me to respect nitroso compounds. They demand careful handling, since such groups tend to react with ease. Work with phenols and you’ll memorize their sharp, medicinal odor, and how they shed protons, behaving a bit like acids. Mix those two tendencies—reactive and acidic—and the result is a chemical with personality.
Significance and Issues in Practice
Few people find themselves buying jars of 4-Nitrosophenol, but the molecule leads to broader issues—especially where safety and environmental stability matter. Nitroso compounds have ties to toxicology since they sometimes serve as stepping stones to nitrosamines, which researchers flagged for cancer risks decades ago. The story of these chemicals keeps evolving as science uncovers new evidence.
Handling phenols, especially derivatives like 4-Nitrosophenol, prompts concern for skin contact and inhalation. The chemistry community already recognizes this and sets clear safety standards. Those working around lab benches know the importance of gloves, fume hoods, and proper storage. Studies on biodegradability and breakdown under sunlight help green chemists find safer disposal options or alternative pathways.
Working Toward Better Practices
Safer design and handling rest on understanding these basics. When designing new molecules for use in dyes, medicines, or pigments, chemists look for ways to cut down on unwanted hazards. Using computational tools, it’s possible to predict some toxic activities linked to nitroso groups long before a new compound is synthesized. Creating less hazardous derivatives by shifting positions of functional groups or changing the base ring is often more than a theoretical exercise.
Looking at the chemistry of 4-Nitrosophenol from all these angles—structural, practical, safety-related—delivers a deeper appreciation for why structural knowledge matters. The lessons learned from basic organic molecules have shaped lab safety and the kind of molecules chosen for further development.
How should 4-Nitrosophenol be stored?
Handling a Chemical That Demands Respect
4-Nitrosophenol isn’t something most folks will find in their garage. This compound gets its use in specialized labs and industry, but everyone who works with it knows the risks. A yellow-green solid by look, 4-Nitrosophenol sits among chemicals with an unpredictable edge. I’ve worked in a lab that stocked pounds of tricky reagents, and I learned early on that playing it safe means treating each bottle like it matters.
Why 4-Nitrosophenol Needs Discretion
Breathing in the dust or letting it land on your skin—those are sure invitations for irritation or worse. 4-Nitrosophenol isn’t the worst actor in the world of organics, but it still can cause harm. Some folks might shrug at a dusty jar, but I watched a careless moment snowball into a minor accident once. That stuck with me. It’s not fear, it’s respect.
Storage That Makes Sense
The basics for 4-Nitrosophenol slot right into what every lab hand learns: cool, dry, shaded, and secure. That’s not just a checklist tossed off in orientation—it’s about real consequences. This stuff doesn’t play well with sunlight; exposure nudges it to break down. Moisture can set off slow reactions, turning tidy shelves into a mess. I always pick amber bottles for photolabile compounds like this. It keeps out the light, and in my experience, helps everyone spot the risky cargo by color alone.
A shelf in a quiet cabinet, far from any sink or water source, works best. Good air circulation nearby helps stop vapors from building up. In poorly ventilated rooms, vapors can hang around and threaten people’s health. Secure caps on bottles matter—a loose-fitting lid invited trouble for us once, and nobody repeated that blunder.
Keeping It Separate From Mischief-Makers
Stacking chemicals that react can fuel a fire or, at best, ruin precious research. 4-Nitrosophenol shouldn’t share space with oxidizing agents or strong bases. Mixing those by accident doesn’t end in mild inconvenience. One time, I saw a young tech absentmindedly store bottles too close, and that near-miss sour smell in the air was warning enough. Good labeling and mindful sorting make a difference, even for the seasoned vets in the lab.
Personal Protection and Spills
Accidents don’t send invitations—they just show up. Nitrile gloves, goggles, and a trusty lab coat block most emergencies from becoming serious. Every responsible lab stocks proper spill kits—think sand, absorbent pads, and something to neutralize organic compounds. I’ve jumped in to tidy up a minor 4-Nitrosophenol spill with help from a well-stocked kit and saw how a minute’s preparation meant no harm done.
Solutions for Smarter Storage
Training stands as the first solution. Walking folks through safe storage gives them more confidence, not just rules to remember. Regular checks—opening cabinets, checking expiry dates, testing seals—beat any set-and-forget approach. Online databases help track chemical hazards, and digital inventory systems can flag items close to expiry or at risk. Sharing stories of real mistakes and fixes builds a stronger safety culture than warnings on paper.
4-Nitrosophenol deserves caution in how it’s stored, just like any chemical that has a sting hidden behind its label. Respect, not paranoia, is what keeps people and property safe.
What are the physical and chemical properties of 4-Nitrosophenol?
A Closer Look at Appearance and Physical Form
Crystals tell a lot about a substance. 4-Nitrosophenol stands out with its yellow-green color, signaling its presence quickly compared to countless pale or colorless organic compounds. Its structure brings a nitroso group (–NO) onto the fourth carbon of the phenol ring, steering both how it behaves and looks. With a melting point around 113–114°C, this powder doesn't need extreme temperatures before changing state, which might prompt precautions for storage both in academic labs and industry shelves.
Solubility and Interaction With Water
One thing that always sticks from years of lab work is the headache of dealing with unpredictable solubility. This compound isn’t one of those tricky cases. Water can dissolve it reasonably well, a contrast with many nitro or phenolic compounds that struggle to mix in. Ethanol and ether both welcome 4-Nitrosophenol too, so those seeking to use this molecule aren’t boxed into a corner by tough solvent limitations.
Stability and Chemical Reactivity
Stability matters, especially for those storing chemicals for months or shipping them across continents. 4-Nitrosophenol keeps its structure under ordinary lab conditions, but it's another story in the presence of light and air. The nitroso group sitting on the aromatic ring gets eager to react. Exposure to ultraviolet light nudges the compound toward oxidation or reduction reactions. It likes to play with reducing agents, converting to p-aminophenol fairly easily, and strong oxidizers, which convert it further. In solution, slight acidity or alkalinity throws a wrench into the mix, affecting stability and shifting its color.
Toxicity and Handling
Plenty of chemicals present risks, but 4-Nitrosophenol deserves a little more respect. Both the nitroso and phenol components bring health hazards, from skin irritation to wider systemic effects if inhaled or absorbed. My experience working in research teams underlines the point: gloves, goggles, and fume hoods shouldn’t feel optional. Accidental exposure, even just to low levels, can build up. The World Health Organization and European regulations flag this sort of organic nitroso compound as potentially hazardous. Their advice doubles as solid best practice—limit exposure, keep quantities small, and always use personal protective equipment.
Use in Research and Industry
Most research work relies on substances like 4-Nitrosophenol for their ability to transfer single electrons, acting as mild oxidizing or reducing agents. Scientists use it to follow redox reactions in solution and probe biochemical transformations. Its color also helps with visualizing changes, serving as an indicator in titrations or enzyme studies. Drug companies and dye makers sometimes use derivatives of nitrosophenol for their vivid colors and reactivity, though safer alternatives often edge it out.
Challenges and Safer Practices
The real issue for both scientists and industry isn’t so much the chemistry but the balance between usefulness and health concerns. Substituting less risky chemicals where possible brings down hazards in the workplace. Engineering controls—like better ventilation, splash guards, and sealed handling systems—reduce accidental exposure. Education goes a long way, too: knowing a chemical’s reactivity, toxicity, and optimal storage makes work safer and more efficient.
Toward Greener Options
Chemical safety isn’t just about individual responsibility but keeping up with alternatives. Organic chemistry continues to evolve, developing milder reagents and more selective catalysts that match or beat old standards like 4-Nitrosophenol. Pushing for better environmental profiles—less toxicity, easier disposal, greener synthesis—matters as much as the work done at the bench.
| Names | |
| Preferred IUPAC name | 4-nitrobenzenol |
| Other names |
p-Nitrosophenol 4-Hydroxynitrosobenzene p-Hydroxynitrosobenzene 4-Nitrosophenol |
| Pronunciation | /ˈfɔːr naɪˈtrəʊsəˌfiːˌnɒl/ |
| Identifiers | |
| CAS Number | 104-91-6 |
| Beilstein Reference | 878542 |
| ChEBI | CHEBI:78069 |
| ChEMBL | CHEMBL205115 |
| ChemSpider | 78480 |
| DrugBank | DB04111 |
| ECHA InfoCard | 100.012.683 |
| EC Number | 207-739-5 |
| Gmelin Reference | 7827 |
| KEGG | C02568 |
| MeSH | D009632 |
| PubChem CID | 74904 |
| RTECS number | SM8575000 |
| UNII | M5K2XV4F2O |
| UN number | 2811 |
| Properties | |
| Chemical formula | C6H5NO2 |
| Molar mass | 123.11 g/mol |
| Appearance | yellow crystals |
| Odor | phenol-like |
| Density | 1.34 g/cm3 |
| Solubility in water | slightly soluble |
| log P | 1.42 |
| Vapor pressure | 0.0228 mmHg (25 °C) |
| Acidity (pKa) | 7.23 |
| Basicity (pKb) | 10.40 |
| Magnetic susceptibility (χ) | -74.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.673 |
| Dipole moment | 2.98 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 129.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -36.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -59.5 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause allergic skin reaction. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06, GHS08 |
| Signal word | Danger |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | Store in a well-ventilated place. Keep container tightly closed. Store locked up. Dispose of contents/container in accordance with local/regional/national/international regulations. |
| NFPA 704 (fire diamond) | 3-2-2-详情 |
| Flash point | 79°C |
| Autoignition temperature | Autoignition temperature: 430°C |
| Lethal dose or concentration | LD50 oral rat 220 mg/kg |
| LD50 (median dose) | LD50 (median dose) of 4-Nitrosophenol: 620 mg/kg (rat, oral) |
| NIOSH | NIOSH SN 1055 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.07 mg/L |

