4-Nitrophenol: From Discovery to Modern Application
Historical Development
People have experimented with aromatic nitro compounds since the days of early organic chemistry, chasing after dyes and medicines that would change industries. 4-Nitrophenol found its spot in textbooks after chemists isolated it through controlled nitration, watching a simple benzene ring reveal more layers. Decades back, before plastic beakers and fume hoods, glass tubes and Bunsen burners brought forth this yellow, crystalline compound, marking it as one of those chemicals that proves essential both in laboratories and on factory floors. During the World Wars, 4-Nitrophenol gained attention as intermediates in ammunition and dye production, but soon, it settled into wider roles: a reactant for pharmaceuticals, a substrate in enzyme assays, and even as a pesticide precursor. Its history isn’t just about science—it reflects shifting human needs and the drive to harness chemical reactions for new solutions.
Product Overview
4-Nitrophenol, commonly known as para-nitrophenol or p-nitrophenol, distinguishes itself with that unmistakable yellow color and a reputation for versatility. Sourcing pure material from suppliers often means securing an off-white to yellow solid, packed safely in sealed containers, each batch tracked for purity and date of manufacture. Laboratories rely on analytical grade 4-Nitrophenol for precise work—a choice that reflects confidence in stable, reproducible reactions. Out in the world, its uses span from creating herbicides that shape the world’s farmland yields, to acting as a pH indicator in biology classrooms. Teams working in pharmaceutical development look to it as a stepping stone, transforming the molecule into new drugs and research agents. Not many compounds serve both as classroom teaching aids and as keys to unlocking medical advances.
Physical & Chemical Properties
4-Nitrophenol’s bright yellow crystals don’t just catch the eye; they signal the presence of a strong chromophore, with an absorption maximum near 400 nm, making it easy to spot in spectrophotometric studies. It melts at around 113°C, a fact which helps chemists identify it when characterizing unknown samples. The slightly bitter, medicinal smell can remind anyone who’s handled organic compounds of labs and chemical stockrooms. This molecule dissolves well in hot water, alcohol, and ether, offering flexibility for synthesis and analysis. The phenolic hydroxyl group lends some acidity—more so than plain phenol—so it participates in acid-base reactions, aiding as an indicator. In terms of reactivity, the nitro group sitting at the para position limits further electrophilic attack, but opens up opportunities for reduction and substitution, providing a playground for experimental chemists. Handling this solid takes careful attention to avoid inhaling dust or letting it contaminate hands, because even a small amount can irritate skin or mucous membranes.
Technical Specifications & Labeling
Bottles of 4-Nitrophenol shouldn't arrive with ambiguous details. Labels lay out the CAS number (100-02-7), purity rating (usually above 99% for lab use), batch number, and recommended storage conditions—often cool, dark spaces away from incompatible reactive agents. Material Safety Data Sheets (MSDS) accompany every shipment, highlighting potential hazards, disposal instructions, and protective measures for users. In regulated markets, barcoding and QR tracking support accountability, letting institutions trace shipments from production to end-use. Chemists and technicians need specs they can trust: accurate melting point, molecular formula C6H5NO3, and clear warnings about toxicity. Lapses in labeling can cause mix-ups, which, in environments dealing with toxic and flammable goods, become more than just administrative headaches—they become genuine safety threats.
Preparation Method
Industrial production of 4-Nitrophenol taps into controlled nitration of phenol. Factories introduce a mixture of nitric and sulfuric acids to phenol, carefully cooling the vessel to prevent runaway heat and unwanted by-products. It’s a classic example of finding balance between speed and selectivity, as operators monitor temperature and flow rates. After reaction, workers isolate the product through extraction, followed by purification steps—sometimes crystallization, sometimes distillation—until achieving a batch that passes spectral and chromatographic purity checks. Small-scale synthesis in teaching labs follows similar steps, substituting elaborate controls for ice baths and cautious stirring. Environmental regulations push plants to recycle solvents and neutralize waste, a response to early mistakes when disposal was an afterthought, not a part of planning. Wastewater containing residual nitrophenols gets treated before release, closing a cycle that, if ignored, stains rivers yellow and damages ecosystems for decades.
Chemical Reactions & Modifications
The real power of 4-Nitrophenol lies in how it opens up to new chemistries. Reducing the nitro group leads to 4-aminophenol, anchor of paracetamol production—a drug that fills medicine cabinets worldwide. Reacting it with alkali generates phenolate ions, shifting color and letting students observe acid-base chemistry in real time. Substitution at other ring positions remains tough, thanks to the electron-withdrawing nitro group, but skillful chemists manage selective modifications under careful conditions. For enzymology, 4-nitrophenyl esters serve as chromogenic substrates, giving researchers an easy visual readout of enzyme action. Smarter processes use catalysis to speed up reaction times and minimize waste, responding to the call for greener, more sustainable routes. The nitrophenol backbone shows surprising adaptability: it's the scaffold for dyes, photographic developers, and a handful of insecticides. This flexibility isn’t just theoretical—it shows up in patents for new derivatives and refinements in bulk chemical outputs.
Synonyms & Product Names
Anyone scanning chemical catalogs has seen 4-Nitrophenol go by several names. Para-nitrophenol serves as a technical alternative, highlighting the position of that nitro group. Some supplies shorten it to p-nitrophenol or simply PNP—easy on the tongue but easy to confuse if context slips. In the pharmaceutical world, it might show up as Mexen or Paroxol, a nod to brand history or specific uses. While the IUPAC name spells out the arrangement, quick codes form the real language in fast-paced labs. Synonyms grow from industry needs, sometimes tied to patents or marketing, sometimes echoing the language across generations of textbooks. Consistency in naming means fewer mistakes, especially in settings where similar compounds carry widely different risks.
Safety & Operational Standards
Every bottle or drum of 4-Nitrophenol demands respect. Skin contact causes redness, itching, and sometimes long-term sensitization. Breathing in dust or vapors leads to headaches, nausea, or worse, cyanosis from disrupted blood oxygen transport. Decades of handling cases in labs and plants shaped current safety standards: full gloves, goggles, and working fume hoods stand as non-negotiables. Facilities train staff for spills, using absorbent pads, waste bins labeled for hazardous materials, and neutralizers if acids mix in. Emergency protocols focus on prevention and speed: immediate eyewash and showers, rapid access to professional help, and ongoing monitoring for long-term health effects. Workers review labels and MSDS files before opening bottles, learning not just the hazard symbols but also the real dangers behind them. Regulations in the US and EU spell out storage limits and exposure controls, supported by inspections that catch lapses before they turn tragic. These lessons didn’t come cheap; hard-earned from generations of accidents and improvements.
Application Area
The way 4-Nitrophenol finds its way into so many fields impresses even seasoned chemists. Analytical labs use it as an enzyme substrate in spectrophotometric assays, where it reveals how biological catalysts work under different conditions. Agriculture taps into its chemistry to build pesticides that control pests across thousands of hectares; each molecule traces back to those first nitrations of phenol. In the pharmaceutical industry, it stands as a crucial intermediate, becoming painkillers or antiseptics with a few more steps. Water quality experts detect tiny amounts in rivers and lakes, knowing even low concentrations cause harm to aquatic life. Its color-shifting response to pH lets it act as an educational tool, teaching students about acids and bases in vivid demonstrations. Environmental agencies use it to check for contamination, forcing polluters to explain why such compounds end up in groundwater. Every new use carries echoes of the needs and risks that have shaped this product’s place in society.
Research & Development
Innovation doesn’t slow down, and 4-Nitrophenol remains a focus in both academic and industrial labs. Researchers design greener synthesis routes, shifting away from harsh acids toward milder catalysts and renewable feedstocks. Universities probe its reactivity with novel enzymes, mapping reaction pathways that could lead to new drug designs. Analytical chemists improve sensors for rapid field detection, shrinking the hardware needed for accurate readings in environmental surveys. Formulators look for ways to limit contamination in final products, using selective adsorbents and better purification steps. The drive toward sustainability fuels experiments with alternative solvents, reducing the carbon footprint for every kilogram produced. Pharmacologists explore new derivatives, seeking medicines that work better or avoid known side effects. Multidisciplinary teams build data models to predict reaction outcomes before a single jar opens, blending chemistry’s rich history with tomorrow’s digital tools. Research churns up new insights every year, proving that even old molecules have new stories to tell.
Toxicity Research
Awareness of 4-Nitrophenol’s toxicity runs deep in chemical safety circles. Chronic exposure damages blood and the nervous system, with animal studies showing mutagenic and teratogenic effects at higher doses. Short-term contact burns skin and eyes, while ingestion—through accident or contaminated water—brings more severe symptoms: dizziness, loss of coordination, organ impacts. Safety testing in labs and factories goes beyond legal minimums, using advanced sensors to catch leaks or airborne particulates before workers notice smells or headaches. Environmental biologists track its impact on fish and invertebrates; even at trace levels, it disrupts reproductive cycles and lowers oxygen in water bodies. Regulatory agencies in many countries classify it as a hazardous substance, controlling how much anybody lets out or disposes of. Reporting requirements tie back to real-world consequences—runoff or illegal dumping can wipe out fish populations and threaten human health. Long-term studies push for alternatives wherever possible, spurring development of less toxic processes or tighter containment systems. Every page in the MSDS tells part of this story, connecting risk to real protections for people and environment.
Future Prospects
The path ahead for 4-Nitrophenol responds to shifting pressures—tightening regulations, public concern about environmental impact, and the search for safer chemical processes. Researchers keep finding new applications in biotechnology, where selective detection and controlled modification of biomolecules call for reagents that provide clear, measurable changes. Modern green chemistry pushes for alternatives to legacy nitration methods, reducing both the amount of hazardous waste and the risk to operators handling strong acids. Advances in catalysis, continuous processing, and automation enable cleaner, more consistent outputs. Environmental scientists demand better measurement tools to track contamination, motivating improvements in sensor development and rapid testing. Health professionals and industrial users keep an eye on substitutes that rival 4-Nitrophenol’s effectiveness but lower the toll on people and the planet. The push for sustainability will keep reshaping its production and application, reminding everyone that chemistry’s benefits always come paired with a duty to minimize harm. Every advance draws on chemistry’s deep playbook, while asking tough questions about safety, efficiency, and the long-term consequences of each new reaction.
What is 4-Nitrophenol used for?
Digging Into 4-Nitrophenol’s Many Roles
Some chemicals pop up just about everywhere once you know what you’re looking for. 4-Nitrophenol lands on that list, making appearances in places that range from chemistry labs to big industrial plants. It's a yellow powder, probably best known to science students as a pH indicator, but that’s really just the tip of the iceberg. Every time I ran a simple color change experiment in college, I always saw 4-Nitrophenol go from colorless to bright yellow. What I didn’t realize was the number of different ways this stuff shapes daily life.
Tool in Pharmaceutical Production
It’s tempting to think of 4-Nitrophenol as just a science fair tool, but it matters much more outside the classroom. Drug manufacturers count on it to make medicines like acetaminophen (Tylenol). Here, 4-Nitrophenol acts as a chemical stepping-stone—a starting piece that gets tweaked and transformed by chemists. What stands out is the level of precision needed when using 4-Nitrophenol this way. Any contamination or misstep means the medicine wouldn't be safe. That’s why pharmaceutical companies run strict tests for impurities and check their work at every stage. Regulatory agencies make sure those standards don’t slip, and that's good—everyone who reaches for a pain reliever deserves to trust what’s on the label.
Pesticides and Environmental Monitoring
4-Nitrophenol's job doesn’t end in the medicine cabinet. It helps create pesticides, especially parathion and methyl parathion. These substances protect crops from insects, but they also leave behind traces in soil and water. I grew up surrounded by farmland, so I often saw people talk about pesticides in worried tones. 4-Nitrophenol serves as a marker for researchers: if it shows up downstream or in the groundwater, there’s a signal that the pesticides, or their breakdown products, stuck around longer than intended. For this reason, environmental labs have built sensitive tests just to spot it, making sure streams and drinking water stay safe. Government limits keep an eye on how much ends up in food and water, but monitoring and enforcement take real work.
Taking on Industrial Chemistry
Big factories rely on 4-Nitrophenol too. It acts as an intermediate in dye manufacturing—think yellow and orange dyes used to brighten everything from clothes to paper. Power plants and refineries sometimes release it by accident, so that's a problem worth tracking. Years back, I worked near a paper mill, and we’d get updates on which chemicals showed up in the river. Local communities pushed hard for cleaner waste processes, and 4-Nitrophenol featured on those lists. It reminds you that just because something serves a purpose doesn’t mean it can slip through the cracks without attention.
Human Safety and Better Solutions
Exposure matters with 4-Nitrophenol. It can irritate skin and eyes, and breathing in the dust isn’t safe. Factory workers and lab staff wear protective gear for good reason. Businesses follow tight guidelines for storage and disposal. Regulatory agencies—like the EPA and OSHA—update safety rules as new research comes in. Some companies now look for greener production methods, exploring alternatives with fewer side effects for people and nature. Bio-remediation techniques, which use bacteria to break down 4-Nitrophenol, see increased testing as part of this shift.
Why Attention Counts
Living with 4-Nitrophenol means taking responsibility seriously. It delivers benefits in health care, agriculture, and industry, but it doesn't disappear on its own if left unmonitored. Keeping water and air safe, protecting workers, and updating industrial practices all make a difference. Paying close attention, applying strict safety checks, and researching better options shape how this chemical fits responsibly into life—now and in the future.
What are the safety precautions when handling 4-Nitrophenol?
Understanding What You’re Dealing With
4-Nitrophenol pops up in more research work than most folks realize. It’s a pale-yellow solid, mainly used for making dyes, pesticides, and medications. Sure, it doesn’t look dangerous at first glance, but problems sneak in pretty quickly. This compound gets absorbed right through the skin and can do real harm if people breathe it in or swallow it. It can irritate the eyes or skin, but the truly worrying part is what happens inside the body. Once inside, 4-nitrophenol can mess with red blood cells, making it tough for them to carry oxygen—a problem you really don’t want in any lab, classroom, or factory.
Why Gloves Aren’t Optional
I’ve learned not to cut corners when it comes to gloves. Even a tiny drop slipping through can leave skin red and sore, or even trigger headaches and dizziness if you’re unlucky. The usual latex gloves might not cut it—going for a thicker, chemical-resistant material such as nitrile or neoprene delivers way more peace of mind. Long sleeves help, too. Gone are the days of rolled-up lab coats and bare arms—one small splash could mean trouble.
The Simple Power of Good Ventilation
I used to underestimate good airflow, but taking shortcuts with this stuff lands you in hot water fast. 4-Nitrophenol gives off fumes that you can’t always smell, and they still irritate your airways if you work in a cramped space. Fume hoods give you protection you can count on. Regular fans or open windows just don’t cut it in a lab environment. Running the hood before you start and keeping your work inside the sash helps avoid any guesswork.
Eye Protection: Not Just a Suggestion
Getting anything in your eyes is brutal, but 4-nitrophenol stings fiercely and does lasting damage. Safety goggles aren’t negotiable. The wraparound kind gives better coverage—no project should move forward without them. For anyone handling a lot of powder or preparing solutions, a face shield ups the protection level, especially when you’re mixing or dispensing.
Cleaning Up After Yourself
Leaving behind even a few crumbs of 4-nitrophenol on the bench puts the next person at risk. Dedicated waste containers and properly labeled bottles are a big help. I remember seeing someone dump leftover solution down the regular sink outside a fume hood—one stern warning later, nobody in that lab made the same mistake. Chemical waste belongs in the right spot so it doesn’t circulate back into water supplies or air. Surface spills should go straight onto absorbent pads, not just paper towels, followed by a wipe-down using a cleaning agent fit for chemicals (not just soap and water).
What To Do in Case of Accidents
Panic wastes time. Anyone handling 4-nitrophenol should know the drill ahead of time. Eyes get rinsed at the station for at least 15 minutes. Contaminated skin calls for immediate water—lots of it, and no scrubbing. Tell a supervisor or safety officer at once; don’t just sneak off and hope for the best. Medical teams need to know right away if someone’s exposed, since the symptoms kick in quickly and treatments depend on fast action.
Building a Culture of Respect for Chemicals
Handling 4-nitrophenol isn’t about fancy gear or hoping for good luck. Experience shows that respecting protocols—having the right protective clothing, working in a solid airflow, proper disposal, and clear emergency procedures—keeps people safe. Relying on memory or half-read labels usually backfires. Regular training, easy access to safety data sheets, and a habit of double-checking storage cabinets are simple steps that make a huge difference. No shortcut justifies a gamble with this compound—especially when the health costs might stick around far longer than any experiment.
What is the chemical formula of 4-Nitrophenol?
What 4-Nitrophenol Is and Where You Find It
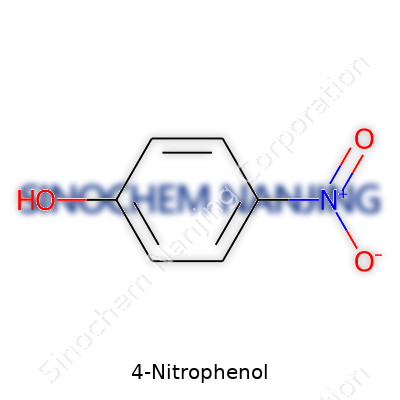
4-Nitrophenol, known for its pale yellow crystals, goes by the formula C6H5NO3. This compound shows up a lot in chemistry labs, industrial production, and sometimes even in environmental testing. I remember my college years, seeing those yellowish stains on lab coats, thanks to careless handling during organic synthesis labs. The nitro group, attached to the benzene ring at the para position, gives 4-nitrophenol that recognizably vibrant shade and distinguishes it from its relatives.
Why Chemical Structure Matters in the Real World
Chemical formulas are not just for quizzes and equations. The nitro group at the 4-position determines practically all of 4-nitrophenol’s reactivity. This compound often pops up as a breakdown product of pesticides and some pharmaceuticals. Environmental scientists monitor it because runoff from agricultural fields and some factories can send traces into local water sources. Toxins like these tend to grab my attention because a seemingly small detail—like one group on a ring—makes a big difference. The biochemical interactions depend heavily on how these atoms line up, making formulas like C6H5NO3 more than trivia—they point to risk and behavior in nature.
Impacts on Health and Environment
Chemicals like 4-nitrophenol are not silent bystanders. It can irritate the skin, eyes, and respiratory tract. Workers at facilities with weak ventilation or poor safety culture can develop symptoms unless they gear up properly. On the environmental side, I have seen reports of river samples turning up measurable amounts after pesticide application. These traces can affect aquatic ecosystems because the compound persists, breaking down slowly under sunlight and microbial activity. That yellowish water can look innocent but might signal bigger concerns for fish life and water safety down the line. Children and those using groundwater nearby often deal with consequences first.
How Labs and Industry Handle It
I once toured a specialty chemicals plant and saw how tightly controlled storage and spill prevention routines had to be with any nitrophenol derivative. Proper labeling, personal protective equipment, and engineered storage make the difference between safe practice and risky shortcuts. Analytical chemists often test samples by measuring the intense absorbance of 4-nitrophenol’s characteristic yellow color. The formula tells them what reactions to run—whether to reduce it to aminophenol or oxidize it further. Consistency in quality checks comes down to knowing what molecules you’re handling.
Moving Toward Safer Practices
Safer handling practices, stronger wastewater treatment, and timely environmental monitoring give some hope for managing risk from 4-nitrophenol. Some companies have invested in green chemistry approaches, seeking less persistent byproducts and more recyclable catalysts. It can be tempting to cut corners, but downstream health costs and regulatory headaches outweigh short-term savings. From my own work in lab management, updating training protocols and regular safety audits kept incidents low and peace of mind high for the team.
Final Thoughts
4-Nitrophenol’s formula may look simple: C6H5NO3. Underneath, it drives important decisions about safety, environmental impact, and regulatory action. Recognizing both its promise and challenge helps anyone who handles it tread carefully, for their own sake and for the world around them.
How should 4-Nitrophenol be stored?
4-Nitrophenol Isn’t Just Another Lab Chemical
4-Nitrophenol shows up in a lot of labs, but handling it always gives me pause. The way this yellow compound reacts to light, heat, and air humidity proves it can't just sit in any storeroom corner. Some researchers, even those with plenty of experience, can get comfortable with everyday risks—but anything that's toxic, potentially explosive, and easily absorbed through skin deserves more attention than most glass bottles on a shelf.
Keep It Cool, Dry, and Out of Sight
You can tell a lot about responsible chemical storage by how folks treat 4-Nitrophenol. Labs that value safety keep it in tightly sealed containers, always clearly labeled. I learned from safety officers and long-timers that amber glass works best. The reason? Sunlight will break 4-Nitrophenol down, and that can turn a safe substance into something unpredictable. Someone on my team once stored it near a window—we caught the mistake in time, but it made the lesson stick.
People tend to overlook moisture, but high humidity and water contact cause real headaches here. 4-Nitrophenol will decompose, leading to pressure build-up or nasty byproducts. Dry cabinets with desiccants go a long way. I always make sure desiccator beads go in with high-risk solids—some think it’s overkill, but no one wants to clean up an avoidable spill.
Don’t Mix with the Wrong Crowd
In shared labs, storage cabinets fill up fast. Convenience can tempt people to group chemicals by size instead of by compatibility. Pairing 4-Nitrophenol with alkalis, strong reducing agents, or organic matter increases the odds of an accident. More than once, haphazard organization led to minor scares—nothing grabs your attention like a container sweating or hissing. I always recommend keeping incompatible materials apart, storing oxidizers and reducers as far away from each other as possible.
Legal and Industry Standards Matter for a Reason
Legislation and industry standards shaped how labs treat chemicals like 4-Nitrophenol. Globally, the Globally Harmonized System (GHS) and local workplace safety laws give clear-cut rules. My own compliance checks often reveal gaps: old containers lacking hazard symbols, storage at the wrong temperature, questionable labels. I don’t shrug these off, and neither should anyone else. Keeping detailed logs, using secondary containment, and sticking to recognized best practices aren’t just about avoiding fines—they tell every newcomer in the lab that mistakes have real consequences.
Training, Reminders, and Respect for Chemicals
People forget storage rules over time, especially those who have only read about chemical dangers in textbooks. Regular reminders help. I like short safety drills: find the most dangerous item in your cabinet, check its condition, and review its storage instruction. For 4-Nitrophenol, this almost always leads to double-checking seals or moving it farther from incompatible substances. One direct experience beats a hundred warning stickers.
Tight storage protocols for 4-Nitrophenol protect not just the chemical, but also the people working alongside it. Mistakes in handling never stay small. Storing it right signals a commitment to safety, knowledge, and respect—qualities that carry more weight in a lab than the fanciest piece of equipment.
Is 4-Nitrophenol toxic or hazardous to health?
Toxic Chemicals Don’t Just Stay in the Lab
A world filled with chemicals like 4-nitrophenol can make anyone nervous, especially if you work in an industry that relies on dyes, pesticides, or manufacturing processes where it’s common. Most people see chemical names and think of white lab coats and science fair experiments, but the real impact often lands on folks who don’t have much say — factory workers, farmers, or people living near chemical plants. Based on what research shows, the risks attached to 4-nitrophenol go beyond academic discussions; we’re talking real health concerns for real people.
4-Nitrophenol Is Not Just a Yellow Powder
Seeing a yellow powder in a jar might not feel alarming, but 4-nitrophenol’s hazards stack up quickly. If you inhale it, swallow it, or let it touch your skin, there’s trouble. The body absorbs it easily. Many studies point to acute symptoms: headaches, nausea, dizziness, and even blood issues like methemoglobinemia. This problem reduces the blood’s ability to carry oxygen. The difference can mean the world for a worker breathing fumes in a confined space, versus someone reading a label in a classroom.
Long-Term Exposure Deserves More Attention
Short-term problems are just the start. Repeated contact with 4-nitrophenol, even in small amounts, has raised alarms about kidney and liver damage. Studies in animals sometimes prompt people to shrug it off, but experience tells me it’s unwise to wait for clear evidence in humans when lives are at stake. Some reports even hint at connections with cancers. Enough uncertainty exists that treating it as a serious threat keeps people safe.
The Trouble Goes Down the Drain
People often worry most about workplace exposure, but water contamination is a bigger story. Effluent from factories or pesticide runoff finds its way into rivers and streams. Small towns with limited resources to test drinking water might be at greater risk. Quite a few case studies show trace amounts of 4-nitrophenol sticking around in water, soil, and even in foods. The threat doesn’t disappear once the waste leaves a factory gate.
Laws and Training Matter, but Gaps Remain
Agencies like OSHA and the EPA have stepped in with regulations and workplace limits, which helps, but enforcement varies. Many workers don’t have enough training in how to handle or dispose of toxic chemicals. Sometimes, it just takes one leak, spill, or ignored warning sign to set off a health scare. The solution calls for better monitoring and stronger incentives for safe disposal, not just paperwork or periodic inspections.
Clear Steps Can Cut the Risks
Safer practices—good ventilation, protective gear, and updated procedures—make a big difference for workers. For communities, independent water testing can spot problems early. Factories and farms need more than just compliance; taking chemical hazards seriously should be part of everyday business. Prioritizing education keeps everyone—workers, families, and neighbors—a step ahead of the risks that 4-nitrophenol brings to the table.
| Names | |
| Preferred IUPAC name | 4-Nitrophenol |
| Other names |
p-Nitrophenol 4-Hydroxynitrobenzene p-Hydroxynitrobenzene PNP |
| Pronunciation | /ˌfɔːrˌnaɪtrəʊˈfiːnɒl/ |
| Identifiers | |
| CAS Number | 100-02-7 |
| Beilstein Reference | 1209223 |
| ChEBI | CHEBI:17698 |
| ChEMBL | CHEMBL1409 |
| ChemSpider | 1289 |
| DrugBank | DB03766 |
| ECHA InfoCard | 100.006.034 |
| EC Number | 4.1.3.9 |
| Gmelin Reference | 822 |
| KEGG | C01598 |
| MeSH | D009601 |
| PubChem CID | 980 |
| RTECS number | SM8575000 |
| UNII | M0J401G23K |
| UN number | UN1663 |
| Properties | |
| Chemical formula | C6H5NO3 |
| Molar mass | 139.11 g/mol |
| Appearance | Yellow crystalline solid |
| Odor | Odorless |
| Density | 1.48 g/cm3 |
| Solubility in water | 16.6 g/L (20 °C) |
| log P | 1.91 |
| Vapor pressure | 0.000016 hPa (25 °C) |
| Acidity (pKa) | 7.15 |
| Basicity (pKb) | 7.15 |
| Magnetic susceptibility (χ) | -76.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.628 |
| Viscosity | 0.0043 Pa·s (25 °C) |
| Dipole moment | 2.87 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 115.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -59.0 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -2063 kJ mol^-1 |
| Hazards | |
| Main hazards | Toxic if swallowed, causes skin and eye irritation, harmful if inhaled, suspected of causing genetic defects. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H315, H318, H400 |
| Precautionary statements | P261, P264, P270, P271, P301+P312, P302+P352, P304+P340, P312, P330, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-1-OX |
| Flash point | 79°C (174°F) |
| Autoignition temperature | 540 °C |
| Lethal dose or concentration | LD50 Oral Rat 282 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 282 mg/kg |
| NIOSH | SN 0875 |
| PEL (Permissible) | PEL: 5 mg/m³ |
| REL (Recommended) | REL: 0.1 mg/m3 |
| IDLH (Immediate danger) | IDLH: 200 mg/m³ |
| Related compounds | |
| Related compounds |
Phenol 4-Nitroaniline 4-Nitroanisole 4-Aminophenol 2-Nitrophenol |

