4-Nitrophenetole: A Deep Dive into a Legacy Chemical
Historical Development
4-Nitrophenetole walked onto the chemistry scene over a century ago, drawing interest thanks to its unique structure and versatility. Chemists across Europe and North America experimented with nitro compounds and ethereal linkages during the late 19th century, seeking novel dyes and intermediates. The synthesis and practical application of 4-nitrophenetole flourished alongside the dye industry boom, especially as researchers learned how nitration and etherification could be combined in aromatic chemistry. Over the decades, industrialization brought sharper methods, and the chemical found its way into mainstream catalogs and technical handbooks. Today’s scholars still refer to early German and French publications for guidance when running classical preparations or mechanistic studies, a reminder that behind every modern bottle stands a long trail of lab work and collaboration.
Product Overview
4-Nitrophenetole, also known as p-nitrophenetole, brings together a nitro group and an ethoxy substituent on a phenyl ring. This combination balances polarity and organic solubility, opening doors to synthetic routes and complex applications. The substance appears as a light yellow liquid at room temperature, marking it out from those nitro aromatics stuck as solids or thick oils. Commercial suppliers offer this compound for researchers in organic synthesis, pharmaceuticals, and agrochemical development. The ability to tweak solubility and reactivity, thanks to its functional groups, gives this molecule an edge in custom synthesis projects that rely on fine-tuned aromatic systems.
Physical & Chemical Properties
The physical properties of 4-nitrophenetole support its broad utility. It melts at about 7-9°C and boils near 290°C, signaling notable thermal stability for an ether-nitro compound. Its characteristic odor and faint yellow tint set it apart from odorless, colorless phenetole counterparts. It dissolves well in common solvents like ethanol, diethyl ether, and chloroform, which offers practical handling in most lab processes. Chemically, the nitro group acts as a deactivator for electrophilic aromatic substitution—making further aromatic manipulation selective and controlled. Reductive conditions transform the nitro moiety into an amino group, which can funnel the molecule into dyes, pharmaceuticals, or complex ligands.
Technical Specifications & Labeling
Reputable suppliers provide 4-nitrophenetole with clear technical data: purity ranging from 97% up to 99.5%, GC and HPLC chromatograms, and certificates of analysis for every lot. Labels specify batch numbers, CAS number 100-17-4, and storage conditions typically calling for a tightly sealed container in a cool, ventilated space away from direct sunlight. Quality control reports check for moisture and residual solvents, vital for applications sensitive to trace contamination. Researchers and process engineers benefit from a combination of straightforward labeling and extensive documentation, including SDS sheets detailing first aid, disposal, and emergency measures.
Preparation Method
Traditional preparation of 4-nitrophenetole relies on the Williamson ether synthesis, using 4-nitrophenol and ethyl iodide (or bromide) in the presence of a base such as potassium carbonate. Reactants mix in anhydrous conditions to avoid byproducts, and product isolation uses simple distillation or recrystallization. Large-scale operations often tweak reaction times and temperatures to maximize yield, and some modern processes swap out classic bases for greener alternatives that cut down on waste and environmental footprint. Several patents reference continuous-flow or microwave-assisted protocols for time efficiency and thermal control. Real-world practice often demands process optimization, such as reducing hazardous reagent use, scaling up safely, and minimizing purification steps to meet industrial demand.
Chemical Reactions & Modifications
The chemical backbone of 4-nitrophenetole welcomes several transformations. Reduction with iron filings in acidic media or catalytic hydrogenation gives 4-aminophenetole, which serves as a key dye intermediate and pharmaceutical building block. Nucleophilic aromatic substitution swaps the ethoxy group for other substituents under strongly basic conditions, especially when paired with electron-withdrawing auxiliaries. Nitration or halogenation on the aromatic ring, although less common due to deactivation from the nitro group, still garners attention from method developers pushing reactivity limits. In multi-step syntheses, 4-nitrophenetole stands out as a practical linking unit that can survive harsh conditions and offer selective reactivity at later stages.
Synonyms & Product Names
4-Nitrophenetole goes by many names across literature and supply catalogs: p-nitrophenetole, 1-ethoxy-4-nitrobenzene, and para-nitrophenetole are among the top variants. Different regions and companies favor their own nomenclature, sometimes leaning toward systematic IUPAC names, other times preferring shorter trade names. In my own lab notes, I’ve always logged the CAS number as well as the product code from the specific supplier—a small yet crucial detail that avoids miscommunication with colleagues or regulatory bodies.
Safety & Operational Standards
Handling nitroaromatics means respecting both acute and chronic toxicity risks. 4-Nitrophenetole can cause skin and eye irritation, respiratory distress, and methemoglobinemia in severe cases due to its ability to disrupt oxygen transport in blood. Proper operation asks for gloves, goggles, and a well-ventilated hood during weighing or reaction setup. Waste streams must be segregated to avoid accidental release into water systems, reflecting both personal and environmental responsibility. Laboratories and manufacturing sites conduct regular risk assessments; many now run annual safety drills focused on spill response and emergency evacuation. Policy-wise, local regulations and global standards such as REACH or OSHA set out clear guidelines for storage, handling, and transport. As public awareness grows about legacy and emerging risks, training programs have moved from simple worksheet handouts to hands-on drills and video tutorials.
Application Area
Researchers and manufacturers use 4-nitrophenetole as an intermediate for fine chemicals, dyes, and certain pharmaceuticals. Its role in custom syntheses stands out, especially for aromatic amines and azo compounds that fuel textile and ink industries. Some specialty insecticides and herbicides draw on derivatives, thanks to the molecule’s reliability in complex synthetic routes. In academic labs, it often enters the syllabus as a demonstration substrate in undergraduate organic classes—teaching students how aromatic reactivity changes with nitro and ether groups. Over decades of bench work, there’s always been conversation about new applications in smart materials or niche electronics, though uptake there remains small compared to classic uses.
Research & Development
4-Nitrophenetole’s R&D journey stays lively, especially as sustainable chemistry and green synthesis take priority worldwide. Chemists report on new catalytic systems for etherification or reduction, aiming to boost atom economy and cut waste. Analytical chemists focus on developing better detection methods for trace impurities, while environmental scientists study its degradation profile in soils and water. Some groups design new analogues by tweaking ring substituents or replacing the ethoxy group; usually, this work goes hand-in-hand with computational predictions and high-throughput screening. Those invested in process engineering often revisit classic protocols to lower hazard ratings or introduce continuous processing. Friendlier scale-up—and safer, more cost-efficient routes—can make or break adoption in modern manufacturing.
Toxicity Research
Toxicologists pay close attention to nitro aromatics since many of them act as persistent environmental contaminants or pose direct health risks. Studies on 4-nitrophenetole show moderate acute and sub-chronic toxicity, mainly due to the nitro group’s metabolic reduction in living organisms. Animal tests have documented methemoglobinemia, liver stress, and minor neurotoxic effects at high doses. Regulatory bodies list it as a substance of concern, with occupational exposure limits enforced in major economies. More recent work explores biodegradation rates and byproduct formation to estimate persistence and buildup in ecosystems. Trend lines point toward stricter scrutiny and the ongoing search for less hazardous alternatives in key applications.
Future Prospects
Looking ahead, 4-nitrophenetole sits at the crossroads of tradition and innovation. Core uses in dyes and synthetic intermediates will likely continue, though environmental and health concerns push for reformulation and greener practice. The move toward continuous-flow chemistry and automated platforms could reduce operator risk and solvent use, echoing a broader sustainability trend. Digital twin tech and AI-driven reaction optimization promise to refine everything from raw material sourcing to waste management. Advances in elimination of nitroaromatic pollutants, inspired partly by compounds like this, may trigger new remediation technologies. Researchers in academia and industry keep a close eye on regulatory shifts and market demands—knowing that resilience and creativity often shape the way legacy chemicals evolve for a safer, cleaner future.
What is the chemical formula of 4-Nitrophenetole?
Understanding the Structure
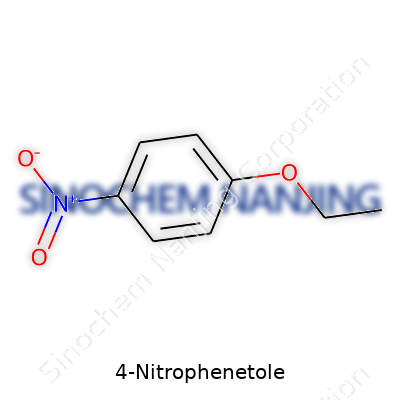
4-Nitrophenetole isn’t a name that rolls off most people’s tongues, but once you break it down, there’s something straightforward about it. The compound’s formula is C8H9NO3. Think of this molecule built from a benzene ring. On one side, you’ve got a nitro group at the fourth position, and across the ring, there’s an ethoxy group holding its ground. That setup matters not just for naming, but for everything from reactivity to safety in storage.
Real-Life Use: Why Bother?
Plenty of folks ask why anyone would bother memorizing or even talking about these chemical formulas. It's like the backbone of understanding reactions in organic synthesis. I've spent enough time unraveling the differences between similar compounds to know a missed methyl or nitro group changes outcomes quickly. 4-Nitrophenetole’s formula tells the story upfront, flagging the pieces that make it useful for making dyes, pharmaceuticals, and sometimes even as a reference standard in labs. Knowing that nitro group isn’t tucked anywhere random, but right at the para position, guides both how it behaves in reactions and what safety protocols make sense.
Health and Safety Considerations
With a formula like C8H9NO3, you’re dealing with a compound that’s not exactly kitchen-table safe. The nitro group makes it more reactive and, under certain conditions, hazardous. In my own college lab years, two lessons always stuck: label everything, and memorize the formulas for anything you even think about touching. The ethoxy and nitro pairing means things get energetic under heat or shock. This also means clear communication for safe chemical handling, especially for students new to organic chemistry. It’s not just a matter of theory—the real world reminds us quickly.
The Importance of Chemical Literacy
Chemical formulas look like a jumble to some, yet even a quick glance can reveal a lot. Public understanding of chemical formulas protects communities near factories and inspires smart questions about environmental risks. Folks with a baseline grasp of what those formulas mean can hold companies accountable, spot unsafe storage, and demand transparency. Accurate formulas lead to better safety data sheets, and better record-keeping when solutions spill or waste needs disposal.
Potential Solutions for Improved Safety
As a community, we could support chemistry education by encouraging hands-on molecule modeling in classrooms. Real models, even just ball-and-stick sets, allow students to see for themselves how nitro groups and ethoxy branches attach. I’ve watched high schoolers who barely glance at formulas grow engaged when they snap models together. Beyond schools, clear labelling and standardized formulas on chemical storage containers set a low bar that some companies still miss. This doesn’t have to stay wishful thinking. Industry regulations already demand clear labelling, but enforcement and public education put real teeth behind those lists of letters and numbers.
Final Thoughts
The chemical formula C8H9NO3 for 4-nitrophenetole carries more weight than just three types of atoms. It tells us how the molecule reacts, where it fits in broader supply chains, and how we should treat it to keep workplaces and the wider public safe. The more we invest in chemical literacy—from detailed safety protocols in labs to transparent information in local zoning—the safer everyone becomes and the more trust we build in scientific processes.
What are the common uses of 4-Nitrophenetole?
Understanding the Compound
4-Nitrophenetole does not make headlines, but its fingerprints show up in a lot of chemical processes. One of those everyday lab smells that lingers in your memory, this yellow liquid belongs to the nitroaromatic family. From pharmacy labs to large-scale chemical factories, it carves out a place because of how it bridges other molecules together.
The Pharma Backbone
Medicines start as chemical puzzles. You need building blocks that respond well to reactions and give you room to add whatever functional groups matter to your therapy. 4-Nitrophenetole gets used a lot as that kind of scaffold. Chemists reach for it when they want to build more complex aromatic compounds. You’ll find it popping up in research on painkillers, antiseptics, and other therapeutic agents. Its nitro group works as a launchpad for further transformations, including reductions that generate amino derivatives, which the pharmaceutical world practically runs on. If you work in medicinal chemistry, odds are you have seen its name on a reaction scheme during drug design cycles.
Dyes and Pigments Find a Friend
The colors in textiles, inks, and plastics often hide long histories of chemical innovation. It might seem odd to trace that bright blue or red back to a compound like 4-nitrophenetole, but in the dye industry, it’s valued for its ability to open a path toward azo and other nitro-based pigments. Whether it’s a chemical company scaling up batches for synthetic dyes or a research group trying to fine-tune a color profile, this molecule helps lay the groundwork. Once chemists reduce the nitro group, more aromatic amines come into play, giving vibrant colors stability against light and washing.
Pesticide Intermediates Matter on the Farm
Crop protection relies on getting the chemistry right. 4-Nitrophenetole serves as an intermediate for certain pesticides and herbicides. It’s the kind of substance that rarely ends up in the final product but makes that final step possible. Companies depend on nitro derivatives for the early stages of synthesis, and without them, a lot of the food on our tables would be more vulnerable to pests and disease. This role doesn’t get much attention outside lab circles, but it enables both food security and safer pest management by helping companies build molecules with targeted action.
Lab Training Wheels and Teaching
Any chemistry undergraduate who’s run a nitration or reduction experiment has probably worked with nitroaromatics like this one. It stands out as a classroom example for organic transformations. The visible changes in color, odor, and reactivity give trainees a hands-on lesson in classic organic chemistry reactions. People often remember the smell or the ease with which the product can be tracked on TLC plates. Educational labs help reinforce safety, purification skills, and reaction monitoring, and this compound shows up on a lot of those checklists.
Health and Environment Cautions
My own time swapping stories with colleagues highlights a running anxiety: toxicology. Nitro compounds carry an edge, healthwise, and demand good ventilation and gloves. Simple reminders—don’t breathe the vapors, keep it off your skin—get repeated because the risks are real. Waste disposal needs careful planning, too. Acting responsibly with these intermediates protects not only workers but also the downstream environment, especially when wastewater can carry traces into broader ecosystems.
Looking Forward: Cleaner Chemistry
Green chemistry pushes companies and academics to find alternatives, cut down on waste, and rethink every step. Catalytic hydrogenation, safer solvents, and closed-loop manufacturing start showing up in more process descriptions. 4-Nitrophenetole's future in industry depends on how well labs adapt their safety culture, monitor emissions, and invest in cleaner routes. In the quest for safer chemistry, it acts as both a stepping stone and a reminder of why progress still matters.
What are the safety precautions for handling 4-Nitrophenetole?
Why Respect for Chemicals Matters
Walking into a lab or small manufacturing setup, 4-Nitrophenetole catches more than one person off guard. Its uses might pop up in research chemistry or product development, but safety sits at the center of those projects. I once joined a team tasked with scaling up a reaction involving 4-Nitrophenetole. Folks from the health and safety committee made it clear: treat this compound with caution if you want to walk out healthy at the end of the day. Years later, those lessons keep ringing true across all sorts of chemical workspaces.
The Nature of 4-Nitrophenetole
This compound packs both health and environmental hazards. Direct skin contact can lead to irritation. Breathing in its vapors risks headaches, nausea, or worse. Swallowing even a small amount could trigger toxic effects. Above all, 4-Nitrophenetole stays stubborn in the water and soil, and that's why spill controls become non-negotiable. There’s memory on the shop floor of a missed glove change—one quick touch, and the redness and itching that followed felt like solid proof not to cut corners.
Everyday Protective Measures
Good habits start right at the door. Pull on chemical-resistant gloves—nitrile or butyl rubber work well. Safety glasses make eye accidents rare, and splash-proof goggles make them nearly impossible. A full-length lab coat blocks accidental drips; closed-toed shoes make sure spills never reach skin. If the operator plans to work in a spot with no fume hood, stop and move to one. Ventilation reduces airborne exposure, and wearing a mask rated for organic vapors adds an extra layer of defense if it gets hectic.
Safe Storage Choices
Forget stacking jars on high shelves or near windows. Darkness, dryness, and a cool climate preserve the integrity of 4-Nitrophenetole and lower the risk of runaway reactions. Containers made from high-density polyethylene or glass with tight, chemical-resistant lids help keep air and moisture out. Labels should stay clear and bold. One poorly labeled bottle risks the wrong reaction or an emergency cleanup, and that’s not a story anyone wants to repeat.
Taking Spills and Waste Seriously
Working with chemicals long enough, spills become a “when,” not an “if.” Even small releases need immediate cleanup. I used to keep spill kits nearby—absorbent pads, neutralizing agents, disposal bags. Sweep up solids. Mop up liquids with pads. Never flush 4-Nitrophenetole waste down the drain; trained waste handlers should take over at this point. I watched a team skip this advice once and got a front-row seat to a contaminated sink and furious environmental officers. Take those moments as warnings, not just stories.
Training and Response Readiness
No one benefits from skipping the basics. Training employees about emergency plans, exit routes, and first aid builds confidence and protects lives. Local fire codes and environmental laws usually require safety data sheets on hand. In my experience, reading through those sheets once helped me spot gaps in our own procedures. It only takes a single incident to realize that safety culture protects more than just the company; it safeguards everyone’s health and future.
Why Small Steps Make a Big Difference
Sticking with best safety practices doesn’t happen by accident—it comes from experience, shared stories, and the knowledge that accidents impact people first and careers next. Respecting hazardous materials like 4-Nitrophenetole turns routine tasks into safe ones and keeps workspace drama where it belongs—in the stories of what could have gone wrong, not in the headlines.
What is the physical appearance and odor of 4-Nitrophenetole?
Physical Traits Set 4-Nitrophenetole Apart
If you ever open up a vial or a flask with 4-Nitrophenetole, two things stand out: color and texture. Most samples look like yellow crystals or a yellowish powder rather than a clear liquid or a fine white dust. That color signals the presence of the nitro group—chemists recognize right away that bright or pale yellow often means something has been nitrated. Hold it up to the light, and the color stays pretty consistent: no shimmer or sparkle, just a mellow yellow. It clumps a bit between your fingers, almost gritty or grainy if it’s pure. Some commercial samples come a little sticky in warm air, since it’s known to soften up above 50 degrees Celsius.
You probably won’t see much variation in color or clarity unless impurities slip in. Out in the lab, a good portion walks right out of a reaction vessel looking like this, especially if you’re working with clean glassware and dry reagents. It doesn’t melt like wax or smear on your glove—solid at room temperature, with no drama.
Odor: Sharp and Slightly Sweet
Now, the smell. Open a container of 4-Nitrophenetole, and you’ll catch a distinct, sort of piercing scent. People often call it sweet—the way some solvents or simple ethers tend to be—but beneath that, there’s a sharpness that can catch in the throat. It isn’t as tear-inducing as nitrobenzene, but it’s still pretty noticeable, especially if you don’t have great air flow. Even low concentrations can fill up a small room, so fume hoods are a must. Anybody who’s spent time around nitro compounds learns to respect that tang—persistent and unmistakable.
For folks handling this material outside of research or production, that sharp odor might raise quick safety flags. It isn’t just unpleasant; inhalation shouldn’t ever become routine. Experience teaches that even trace exposure, day after day, adds up. This reinforces the need for strong awareness: managing risks keeps researchers safer in the long run.
Why Paying Attention Matters
There’s a good reason companies, universities, and chemical providers care about how 4-Nitrophenetole looks and smells. Clear identification supports safety and quality control. Spotting the right shade of yellow and the expected odor means the right stuff came off the line and into the bottle. A darker or off-tone sample could hint at leftover starting materials or foreign contaminants—things you don’t want to mix into further syntheses. My own lab runs quality checks because unexpected colors or odors can mean trouble. Consistency keeps experiments reliable and reduces the risk of surprise reactivity, which matters most when scaling up or switching batches.
Some places install sensors or carry out regular air tests to catch drifting vapors from open containers. Simple steps like these protect student chemists, seasoned researchers, and everyone else coming through the door. Better ventilation, sealed packaging, and formal odor monitoring keep people healthy and labs in business.
Practical Steps for Better Awareness
Placing safety data sheets where everyone can see them makes a difference—no one has to wonder about hazards or what to do if they spill something. Training sessions, sometimes overlooked, let even the most experienced team members brush up on best practices: how to recognize 4-Nitrophenetole, proper PPE, and why working in a hood trumps “just being careful.”
If something doesn’t look or smell right, it’s worth pausing for a second opinion. Surface stains, faded labels, or weird odors tell a story you shouldn’t ignore. The more you see and sniff, the better your nose becomes at picking out the outliers. In chemical work, that kind of attention could mean the difference between a smooth project and a hazardous mistake.
How should 4-Nitrophenetole be stored and disposed of?
Why Storage Choices Matter
Storage for chemicals like 4-Nitrophenetole can’t be an afterthought. I’ve seen forgotten bottles on crowded shelves turn into real hazards, especially if they’re not handled well. 4-Nitrophenetole sits in a category of organic compounds known for causing long-term issues if a leak or spill goes unnoticed. It’s also an irritant, both for the skin and airways, so you don’t want a forgotten broken cap at the back of a supply closet.
Instead of just finding a spot on a shelf, bring the focus to containment and labeling. Glass bottles with tight caps work best to prevent evaporation and accidental exposure. Don’t store it with acids or reducing agents. Some compounds interact in ways that catch you off guard. I’ve seen cluttered labs where incompatibles ended up together, and cleaning up those messes wastes a lot of time and causes unnecessary risk. Keep 4-Nitrophenetole in a cabinet labeled for organics—ideally lockable, away from heat or sunlight. Excess heat or UV can start slow decomposition.
Health and Environmental Stakes
You don’t want to breathe in 4-Nitrophenetole vapors, and you sure don’t want it in the water supply. The compound’s nitro group raises red flags, not just for toxicity but for persistence in the environment. Once a solvent or waste gets out of containment, cleaning up costs time, money, and effort. Agencies like the EPA track nitro compounds closely for a reason.
A 2018 report from the National Institute for Occupational Safety and Health lists nitroaromatic ethers like this one as materials of concern, especially in research and manufacturing settings. Spills in workspaces won’t just stop a project—they can trigger inspections and potential shutdowns.
Disposal: Skip the Sink, Call a Pro
Pouring chemicals down the drain damages trust, the environment, and sometimes your career. I’ve seen well-meaning folks try to “flush out” lab glassware, only to learn later that the chemical stuck around, affecting local water quality and causing real regulatory problems for their facility. 4-Nitrophenetole doesn’t break down easily in water or soil, so even small amounts build up over time.
Hazardous waste contractors know what to do here. Use clearly labeled containers, collecting waste in compatible solvent bottles—leave room for vapors. If your operation runs on a smaller budget, city or university hazardous waste programs often accept such materials. I’ve worked on campuses that arrange quarterly pickups for this reason. No one wants their chemistry experiments making ripples in wildlife or showing up downstream.
Staff training plays a part too. After one surprise inspection in a university lab, training sessions focused on simple steps: double-bagging waste, keeping logs, avoiding cross-contamination. Small habits save big headaches. Know where your closest eyewash and spill kit sit. Chemical hygiene isn’t just a checklist—it’s what keeps everyone safe and the environment clean.
Improving Everyday Practices
Working with 4-Nitrophenetole reminds me how constant vigilance keeps labs running. Regular checks on storage areas, clear guidance, and good communication between researchers and waste handlers close most of the gaps. Labeling supplies, sticking with scheduled clean-outs, and reporting questionable containers all help cut down on accidents. Chemistry runs better with these habits, and everyone downstream benefits.
| Names | |
| Preferred IUPAC name | 1-Ethoxy-4-nitrobenzene |
| Other names |
p-Nitroanisole 4-Nitroanisole 1-Methoxy-4-nitrobenzene |
| Pronunciation | /ˌfɔːrˌnaɪ.trəʊ.fəˈniː.təʊl/ |
| Identifiers | |
| CAS Number | '613-90-1' |
| Beilstein Reference | 1093354 |
| ChEBI | CHEBI:34636 |
| ChEMBL | CHEMBL186165 |
| ChemSpider | 16490 |
| DrugBank | DB08294 |
| ECHA InfoCard | ECHA InfoCard: 100.010.844 |
| EC Number | 210-599-4 |
| Gmelin Reference | 859388 |
| KEGG | C08343 |
| MeSH | D009602 |
| PubChem CID | 7418 |
| RTECS number | SJ8575000 |
| UNII | DR51K6S7FP |
| UN number | UN1662 |
| Properties | |
| Chemical formula | C8H9NO3 |
| Molar mass | 153.16 g/mol |
| Appearance | Light yellow liquid |
| Odor | Odorless |
| Density | 1.155 g/cm3 |
| Solubility in water | slightly soluble |
| log P | 1.98 |
| Vapor pressure | 0.012 mmHg (25°C) |
| Acidity (pKa) | 7.1 |
| Basicity (pKb) | 12.52 |
| Magnetic susceptibility (χ) | -57.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.602 |
| Viscosity | 1.041 mPa·s (25 °C) |
| Dipole moment | 4.73 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 178.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -42.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | −4324.0 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | Not assigned |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H302 + H312 + H332: Harmful if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P264, P270, P273, P301+P312, P305+P351+P338, P330, P501 |
| NFPA 704 (fire diamond) | 2-3-1 |
| Flash point | 105 °C |
| Autoignition temperature | 538 °C |
| Lethal dose or concentration | LD50 oral rat 2850 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 617 mg/kg |
| NIOSH | SN37800 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 2-8°C |
| IDLH (Immediate danger) | IDLH: 100 mg/m3 |
| Related compounds | |
| Related compounds |
Anethole 4-Nitroanisole |

