4-Nitrobenzyl Cyanide: A Practical Review for Chemists
Historical Development
For over half a century, chemists have turned to 4-Nitrobenzyl cyanide as a building block in both academic research and commercial synthesis. 4-Nitrobenzyl cyanide emerged thanks to advancing nitration and cyanation techniques from the twentieth century, riding the wave of organonitro chemistry’s rapid expansion. The introduction of selective nitration on aromatic rings, followed by relatively straightforward cyanide incorporation, gave rise to this compound’s routine lab preparation and consistent use across pharmaceuticals and agrochemicals. As synthetic organic chemistry grew more sophisticated, this molecule held its ground as a versatile intermediate, largely due to how easily its reactive handle—the nitrile—could transform into amides, acids, and heterocycles. Often found referenced in research from the 1960s and 1970s, 4-Nitrobenzyl cyanide still appears in modern routes, demonstrating its staying power through decades filled with new reactions and reagents.
Product Overview
In the laboratory, 4-Nitrobenzyl cyanide appears as pale yellow crystals or powder, easy to spot on a shelf among other nitriles. Some chemists favor it for its solid form, which keeps handling simple and dosing accurate. The compound’s structure—one benzene ring with a nitrile at the para position vis-a-vis a nitro group—balances reactivity and stability. Suppliers offer it in various package sizes, but it remains affordable compared to many specialty chemicals, partly because its stepwise synthesis uses accessible starting materials. While many look for niche intermediates with elaborate scaffolds, 4-Nitrobenzyl cyanide provides a tried and tested starting point for custom modifications thanks to its clear chemical logic and accessible cost.
Physical & Chemical Properties
Many who measure solids in the lab appreciate 4-Nitrobenzyl cyanide’s melting point, consistently between 94–97°C. The pale yellow hue comes from the nitro group’s contribution to conjugation, a cue for identification in a mixed lot. Its moderate solubility in common organic solvents like ethanol, acetone, and ethyl acetate means purification often doesn’t require complex protocols—just classic recrystallization. The compound displays appreciable stability when stored under proper conditions away from light and moisture, resisting spontaneous hydrolysis or decomposition. While the nitro group acts as an electron-withdrawing handle, it also makes the aromatic ring slightly more susceptible to nucleophilic substitution and functional group interconversion at predictable positions.
Technical Specifications & Labeling
Manufacturers disclose high purity levels, typically above 97%, and reliable batch records stem from straightforward synthesis and purification. Labels should list the CAS number (5556-21-8), molecular weight (172.14 g/mol), and chemical formula (C8H6N2O2), alongside GHS pictograms calling for awareness of potential hazards. SDS documents clarify the need for gloves, goggles, and fume hood operation due to the compound’s moderate toxicity and potential for skin and inhalation irritation. Clear batch labeling by suppliers adds another layer of reassurance on traceability—a welcome detail when facing audits or process reviews.
Preparation Method
Most chemists prepare 4-Nitrobenzyl cyanide through a two-step route. The typical process starts with p-nitrotoluene, which undergoes benzylic bromination (often via NBS or elemental bromine). This intermediate then reacts with sodium or potassium cyanide in a polar aprotic solvent, yielding the nitrile through a classic nucleophilic substitution reaction. Filtration and recrystallization follow, removing unreacted materials and providing high-purity product. For scale-up, operators watch for toxic gas risks during bromination and ensure cyanide scavenging post-synthesis to protect the environment and personnel. More recent literature explores green chemistry alternatives—like microwave-assisted synthesis or flow chemistry adaptations—yet most academic and industrial settings stick to these classic steps due to reliability and yield.
Chemical Reactions & Modifications
Chemists chasing diverse scaffolds lean heavily on the versatility of the nitrile group in 4-Nitrobenzyl cyanide. This functionality opens up hydrolysis routes to the corresponding carboxylic acid or amide, catalyzed by base or acid, with scalable protocols in batch or flow. The nitro functionality supports reduction chemistry, enabling formation of 4-aminobenzyl cyanide—a useful intermediate in drug design. Combining both functional group transformations, synthesis of heterocyclic frameworks becomes efficient and streamlined, especially in the context of pyridine, quinoline, or indole analog development. Nucleophilic substitutions, hydrogenation, and cross-coupling reactions all make appearances in the research literature, each finding new ways to elaborate the benzyl cyanide scaffold for specialized targets in bioactive molecules or material science.
Synonyms & Product Names
In catalogs and papers, 4-Nitrobenzyl cyanide takes on several names: p-Nitrobenzyl cyanide, 1-Cyano-4-nitrobenzene, and 4-Nitrophenylacetonitrile remain the most common. Trade literature sometimes abbreviates it as NBCN or uses the EINECS number (226-717-3) for regulatory submissions. This variety of naming occasionally causes confusion for those researching or ordering it for the first time—prompt identification in inventory systems or analytical reports means cross-referencing all known names, especially in global laboratory collaborations where language barriers can complicate communication.
Safety & Operational Standards
Lab safety teams insist on careful handling of 4-Nitrobenzyl cyanide, as its nitrile function carries moderate toxicity and risk for skin or lung irritation. Anyone working at the bench should wear protective gloves, goggles, and a lab coat, with most synthesis steps performed under a strong fume hood. Strict protocols exist for spills or accidental exposure, and storage requires flame-proof cabinets away from acids or strong bases. Waste containing residual cyanide or nitroaromatics needs separation followed by approved neutralization, aligning with hazardous waste management policies. In large-scale settings, operators install monitoring for air quality and double-check their processes with routine health and safety reviews. Annual refresher training on handling and disposal procedures remains critical, especially in academic environments where staff and student turnover challenges institutional memory.
Application Area
Medicinal chemists prize 4-Nitrobenzyl cyanide for its simplicity and modularity. Its two reactive groups put a wide swath of biologically relevant molecules within reach, especially where nitrile-containing scaffolds or nitro-to-amino conversions prove essential. The agrochemical industry incorporates it in paths to selective herbicides and insecticides. Material scientists put it to use building blocks for custom polymers and dye research, relying on its aromatic backbone to impart unique optical or thermal properties. In teaching labs, 4-Nitrobenzyl cyanide provides a clear lesson in aromatic substitution and functional group manipulation, its transformations forming the core of undergraduate synthesis projects and workshops.
Research & Development
Academic groups report a steady stream of new transformations built off the 4-Nitrobenzyl cyanide template. Recent studies showcase its use in expanding the range of carbon–carbon bond-forming strategies, including metal-catalyzed couplings and multicomponent reactions for library synthesis. Pharmaceutical innovation grows from the quick derivatization of the nitrile or nitro groups while maintaining strong yields and purity. Flow chemistry applications explore continuous production as a means to improve safety during cyanide introduction and manage scale-up economics. Patents reflect ongoing interest across crop sciences and specialty chemicals, where process intensification and green chemistry hit economic and regulatory targets. Open access journals and preprint archives catalog a diverse portfolio of synthetic exploits emerging from this unassuming aromatic nitrile.
Toxicity Research
Toxicologists classify 4-Nitrobenzyl cyanide as a compound requiring care due to the potential for acute toxicity. The presence of both nitro and nitrile groups raises red flags in risk assessment models, prompting ongoing research into possible mutagenicity and environmental impact. Animal studies show limited oral and dermal acute toxicity, but the compound’s breakdown products demand monitoring for possible cyanide release. Most regulatory bodies call for use of gloves and respiratory protection, and continued investigation into less hazardous analogs continues in the background. Environmental monitoring focuses on minimizing effluent of both parent compound and hydrolysis byproducts. Industry groups compile data in shared repositories, supporting regulatory compliance and enabling researchers to select safer alternatives where possible.
Future Prospects
The future of 4-Nitrobenzyl cyanide lies in fine-tuning preparation methods for greener, more cost-effective production, especially as regulatory scrutiny increases around nitriles and nitro aromatics. The expansion of continuous-flow synthesis promises greater control and less risk, possibly reducing chemical waste and energy use. New work in computational chemistry offers predictions for functional group compatibility, opening up an expanded toolbox of transformation possibilities. Combined with pressure across industries for safer and more sustainable intermediates, innovation continues in process chemistry to lower hazards and environmental footprint. Researchers consider biocatalytic alternatives and renewable feedstocks, gradually shifting traditional practices to ones aligned with today’s environmental and safety priorities.
What is 4-Nitrobenzyl Cyanide used for?
What Role Does 4-Nitrobenzyl Cyanide Play?
A chemical like 4-Nitrobenzyl cyanide sounds a bit intimidating at first glance. In practice, it has carved out a quiet but important role in both laboratory and industrial settings. During my time in a university research lab, stories often circled about how certain “niche” chemicals end up powering entire classes of medicines or new materials. 4-Nitrobenzyl cyanide fits that mold. This compound joins a long line of aromatic nitriles that researchers lean on as building blocks. They don’t make headlines, but they provide key skeletons for complex molecules that show up in the real world later.
Molecule Building with Purpose
Pharmaceutical chemists often turn to this nitrile as a starting point for life sciences research. Making custom molecules, especially anything in the world of new drugs, usually means stringing together smaller building blocks. 4-Nitrobenzyl cyanide offers a unique combination: the nitro group opens doors for further tweaks, and the cyanide functionality leads to more reactive chemistry. Scientists use these reactions to build out parts of antimicrobial drugs, cardiovascular treatments, and other therapeutic agents. That early step— choosing a small, well-behaved starting molecule— determines whether a project can deliver a new medicine in a matter of years instead of decades.
Applications Beyond Pharmaceuticals
Fine chemicals companies often stock 4-Nitrobenzyl cyanide for more than just academic projects. Organic chemists in the specialty materials field require versatile starting points for dyes, pigments, and optical materials. I once visited a factory responsible for coatings on electronics screens and found out several steps in their process relied on compounds that started their journey in bottles marked “toxic, aromatic nitriles.” Despite the risks, strict safety protocols let workers draw out real-world value from these chemicals.
We Owe Safety and Oversight to Ourselves
Experience in the chemical supply industry shows me how oversight cannot rely on trust alone. Cyanide groups and nitro aromatics carry legitimate safety concerns — accidental releases or poor handling lead to injury. Authorities like OSHA and the EPA demand best practices, including good ventilation, rigorous labeling, and specialized training for anyone near these chemicals. Responsible distribution matters even more, considering nitrile compounds carry a potential for misuse far outside the bounds of research or manufacturing. Monitoring sales, following up on end-user declarations, and educating buyers builds a culture of responsibility without choking innovation.
Room for Safer Chemistry
Innovation doesn’t slow down, even with old standards like 4-Nitrobenzyl cyanide at work. The green chemistry movement invests in processes that use less hazardous chemicals or swap out toxic reagents altogether. Companies and research labs experiment with milder reaction partners or green solvents, which reduces the risks for both people working in the lab and the environment outside. Supporting these developments— even if they start small— shows that progress means safer, more sustainable innovation built on real expertise rather than shortcuts.
Finding Balance in Chemical Manufacturing
Plenty of chemicals with tricky names earn their spot in the toolbox through years of trial and error. 4-Nitrobenzyl cyanide stands as one such case, quietly enabling breakthroughs. The drive to improve safety and encourage responsible use ensures its benefits reach society without sacrificing well-being or trust. That might not make for splashy headlines, but these kinds of choices steer chemistry toward a better future.
What is the chemical formula of 4-Nitrobenzyl Cyanide?
Knowing the Structure: C8H6N2O2
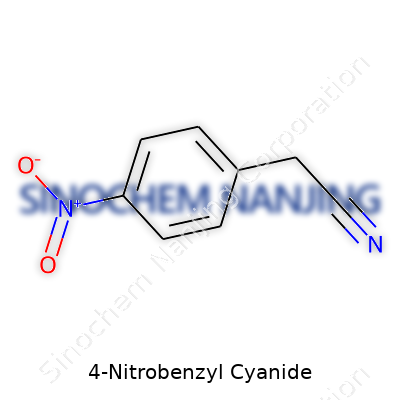
If you crack open any reliable chemistry reference, the formula of 4-Nitrobenzyl Cyanide appears as C8H6N2O2. This compound rarely shows up in everyday conversation, but in labs, it’s a familiar tool. At its core, the structure involves a benzene ring with a cyanide and a nitro group attached. The nitro group sits on the fourth position relative to the cyanide. Every atom matters—altering just one would turn this chemical into an entirely different tool.
Why 4-Nitrobenzyl Cyanide Matters
Back when I spent late nights in an undergraduate organic chemistry lab, I ran into this compound while mapping out synthetic routes for complex molecules. Unlike many textbook examples, 4-Nitrobenzyl Cyanide pops in real research. It’s a core building block for making pharmaceutical intermediates, dyes, and agricultural chemicals. Its structure allows for reactions like nucleophilic substitutions, giving chemists control over what comes out in the flask.
In the field of pharmaceuticals, small molecules like these can end up as key steps in making painkillers or even cancer drugs. Chemists choose it because nitro and cyano groups can transform through reduction or hydrolysis. The nitro group’s electron-withdrawing power tunes the reactivity of the adjacent groups. Useful outcomes follow—they can reduce the nitro to an amino group or turn the cyanide into a carboxylic acid. These reactions give access to new drug candidates, or sometimes, to research tools for understanding disease pathways.
Handling Chemicals with Care
Safety always surfaces as a major concern. My first close-up encounter with cyanides left me uneasy. Their toxic reputation stays engraved in the mind. Nitriles (that cyano part) deserve respect. Inhalation or skin contact brings risks, so gloves, goggles, and a working hood become non-negotiable in routine handling. Most university and industrial labs thrive on students and staff respecting the hazards—not just with cyanide, but especially when combined with reactive nitro groups.
In some small-scale companies, I noticed that staff lean on supplier-provided safety data sheets but don’t always transfer that caution into the lab. Real-world accidents often come from skipping steps or ignoring storage guidelines. A proactive safety culture matters far more than a locked cabinet. Promoting clear labeling, easy access to antidotes, and regular safety drills would go a long way to mitigating risk.
Making Chemistry Safer and Greener
Newer research shifts toward greener pathways for making 4-Nitrobenzyl Cyanide. In speaking with friends at a university green chemistry center, many highlighted low-waste synthesis and solvent-free approaches as driving current research. Catalysts that run on water instead of traditional organic solvents show promise. Technology lets researchers monitor reactions in real time and lower the energy toll of synthesis. Cutting down on hazardous byproducts not only saves on disposal, it makes life easier for the people actually running the reactions.
Supporting responsible chemical management in teaching labs and industry settings stays essential. Real accountability means more than checking regulatory boxes. Ethics in science come from training and leadership that prioritize safety, traceability, and transparency from start to finish.
Summary Table
| Property | Value |
|---|---|
| Chemical Name | 4-Nitrobenzyl Cyanide |
| Chemical Formula | C8H6N2O2 |
| Common Uses | Pharmaceuticals, Dye Synthesis, Research Intermediates |
| Hazards | Toxic, Irritant, Environmentally Harmful if Unchecked |
What are the safety and handling precautions for 4-Nitrobenzyl Cyanide?
What Makes 4-Nitrobenzyl Cyanide Tricky to Handle?
Working in research for years, I’ve dealt with a range of chemicals that ask for more than just a pair of gloves. 4-Nitrobenzyl cyanide takes its place high up that list. It carries both a nitro group and a cyanide group. Toxicity sneaks in from that cyanide moiety, and the nitro boost increases the risk. One quick spill or lapse in protocols can bring lasting consequences. So, nobody in the lab can afford to get casual with a compound like this.
Breathing and Touching: Two Major Routes of Exposure
Fact: Cyanide compounds spell out trouble for your cells, especially for the parts that need oxygen the most. Inhalation or skin contact can bring on anything from headaches and dizziness to full-blown respiratory distress. Accidentally getting 4-nitrobenzyl cyanide on my hand once, even with gloves, drilled that lesson home. I washed it off within seconds, but my nerves were shot for the rest of the day. Chemical-resistant gloves, safety goggles, and lab coats aren’t props—they’re necessities. Splash goggles, not just eyeglasses. Nitrile gloves, not latex.
Avoiding Accidents: Good Habits Mean Fewer Emergencies
It’s easy to leave safety talks at the door and wade straight into work. But with chemicals like 4-nitrobenzyl cyanide, every shortcut chips away at luck. Fume hoods don’t simply clear up the air—they keep toxic vapors out of your lungs and away from anyone close by. Not every space offers proper ventilation, but pushing ahead without a fume hood turns an everyday mistake into an emergency call.
People often forget about secondary contamination. Spill a bit on a bench or pipette bulb, and it lingers. Dedicated glassware, immediate clean-up with spill kits, and readiness with eyewash and showers are critical. It’s no coincidence that labs using these standards see fewer accidents.
Knowing What to Do If Things Go Wrong
Early in my career, one colleague spilled a small amount on the floor. Instead of freezing, she knew where the cyanide antidote kit was stashed and got medical attention. That preparation turned an incident into a story, not a tragedy. Simple planning—labeling the compound, having emergency numbers on the wall, proper training—makes the difference. You don’t leave an antidote locked up in a distant cupboard. Keep it accessible, and make sure everyone knows how to use it.
Storage and Waste: Avoiding Unnecessary Risks
4-Nitrobenzyl cyanide won’t forgive lazy storage. Heat and sunlight can break it down and turn a bottle into a hazard. Segregate it from acids and bases—cyanide and acid fumes react to release hydrogen cyanide gas, and that turns a minor leak into a full-scale evacuation. Store it in a locked, ventilated cabinet, with a clear label stating its toxicity and incompatibilities. For waste, pour it in the right hazardous chemical container, not down the sink. Waste handlers need to know exactly what's in that bottle before they haul it away. Clear documentation protects everyone.
Stronger Culture, Fewer Incidents
4-Nitrobenzyl cyanide doesn't care if you're tired or in a hurry. Careful handling, regular training, and a willingness to speak up about unsafe conditions make the lab safer for everyone. Sharing stories, not just rules, helps drive the point home—one misstep can change a life. That’s reason enough to keep these best practices front and center every single day.
What is the molecular weight of 4-Nitrobenzyl Cyanide?
A Close Look at 4-Nitrobenzyl Cyanide
Most of us don’t stroll through life thinking about chemical formulas. Yet, 4-Nitrobenzyl Cyanide with the formula C8H6N2O2 has practical implications in labs and industries. This compound shows up in organic synthesis, where accuracy down to the decimal can mean success or failure. A molecule like this holds a story that stretches from a bench in an undergraduate lab all the way to pharmaceutical manufacturing, where even the tiniest miscalculation might derail a process. Getting the molecular weight right matters.
Breaking Down the Numbers
When dealing with chemical reactions, the math isn’t just hypothetical—it's the backbone of every mixture. The molecular weight of 4-Nitrobenzyl Cyanide stands at 160.15 g/mol. That number comes from the atomic weights: Carbon weighs 12.01 g/mol, Hydrogen 1.01, Nitrogen 14.01, and Oxygen 16. You add them according to the formula: eight Carbons, six Hydrogens, two Nitrogens, and two Oxygens. I’ve stood at a lab scale watching those milligrams pile up, knowing each one needs to be precise. Off measurements can lead to wasted resources or unsafe experiments.
The Value of Precision
Once, during my grad school days, I underestimated how fast small errors add up. I prepped a solution, thinking decimal places didn’t matter, and watched as a reaction fizzled because I got the maths wrong. The molecular weight is more than trivia; it decides solubility, how much reagent you weigh, and the outcome of your work. Labs around the world rely on these numbers for safety data sheets, risk assessments, and patent filings. Incorrect values don’t just mean botched experiments—they risk health, environmental safety, and in rare cases, compliance headaches with regulators.
Building Trust with Solid Data
There’s a reason scientists spend so much energy double-checking molecular weights. Industry giants and small-scale researchers both need confidence in what they’re working with. In drug synthesis, for example, the wrong calculation on a reagent like 4-Nitrobenzyl Cyanide introduces impurities that may not get caught until it’s too late. Dozens of recall incidents link back to compound misidentification or weighing errors. Molecular weights anchor the whole process, from inventory to synthesis to final QC.
Solutions to Keep Labs on Track
I’ve learned over time that a couple of simple habits go a long way. Triple-check the molecular formula before starting. Use reputable sources—trusted chemical suppliers, peer-reviewed databases, or dedicated resources like PubChem and ChemSpider. Many lab mishaps vanish with better training; teaching students and early-career chemists the critical role of molecular weights makes safer workplaces. Electronic lab notebooks automate part of this calculation now, catching mistakes before they turn costly. As green chemistry principles spread, precise measurements also cut waste and emissions.
Final Thoughts on the Numbers
Behind every round-bottom flask or industrial reactor lies a mountain of calculations. 4-Nitrobenzyl Cyanide’s molecular weight—160.15 g/mol—anchors decisions made in those spaces. Get the math right, and research moves forward, products stay safe, and resources don’t go to waste. These numbers bring chemical theory down to the real world, shaping results and careers alike.
How should 4-Nitrobenzyl Cyanide be stored?
Understanding the Risks
I’ve handled a fair share of chemicals in labs, and every time I see 4-nitrobenzyl cyanide on a shelf, experience reminds me how much respect these materials demand. This compound brings both toxicity and flammability. Skin contact, inhalation, even vapors – all pose real health risks. Not just for the people using the material, but for cleaning staff, delivery drivers, and anyone in the same building. Proper storage links directly to everyone’s well-being.
Physical Storage Strategies
You want glass containers with tight seals – the kind that prevent vapor from creeping out. Metal lids should resist corrosion. Plastic gives a false sense of security since this stuff can degrade many polymers. Kept away from direct sunlight, stored cool and dry, the bottle belongs in a cabinet marked for toxic chemicals. High humidity wrecks labels and screws up weight measurements. I’ve seen labels slide off in bad storage areas, no one needs that mystery in their lab.
Flammable storage cabinets stand out for a reason. The fire rating actually matters. These cabinets stay closed in emergencies, even as sprinklers go off and people evacuate. Firefighters recognize the yellow cabinet for what it holds and adjust their response – that could save a life. Shelves shouldn’t stack bottles two or three deep; every bottle should stay visible and easy to reach. This stops someone from knocking one over when searching blindly.
Limiting Access Matters
In my first year around hazardous chemicals, I realized the room’s lock wasn’t just about theft. High schoolers on a tour, visiting contractors, and even janitors have walked into storage rooms by accident. Chemicals like 4-nitrobenzyl cyanide belong behind at least one secure door, with access limited to trained folks. Written logs help track who opens that door. This record points out problems early, like repeated unsupervised access or bottles vanishing.
Labeling Beats Memory Every Time
It’s tempting to skip relabeling when a manufacturer’s sticker peels. People think their memory will carry the day. Wrong move. Good chemical hygiene means every bottle gets clear, standardized labeling right away. That label lists the full name, the date received, emergency numbers, and critical hazard words—something like TOXIC and FLAMMABLE, large enough that sweaty hands don’t have to fumble for a safety sheet.
Preparing for Spills and Disasters
No matter how carefully bottles get shelved, spills happen. Absorbent pads, specific for organics, should sit close to the cabinet, not just under a sink across the room. People who work with the chemical need gloves and eyewash stations nearby. Fire extinguishers rated for chemical fires make the difference; old water extinguishers only spread the fire. Run drills every quarter so muscle memory kicks in when alarms sound.
Minimizing On-Site Quantities
Order only what you plan to use in a month or so. Old stock sits, degrades, and serves no one. Audits catch forgotten bottles sitting in the back for years, sometimes leaking before anyone realizes. Return or dispose of unused portions quickly, following proper regulations. Local hazardous waste programs can provide drop-off days – it beats waiting for a full storage room to become a real hazard.
Training Builds Habit, Habit Builds Safety
In every place I worked, the teams focusing on regular training avoided near-misses and accidents. Policies go up on a wall, but real safety happens when people drill routines, quiz each other, and know why every step matters. The strongest labs I’ve seen build redundancy into their storage plans, double-check each other’s work, and never brush off a question about storage as too basic.
The Takeaway
Storing 4-nitrobenzyl cyanide well doesn’t look flashy. It looks like an organized cabinet, matching bottles, clear labels, sharp logs, and a team of people who check on each other. The details protect everyone who steps into the space, giving confidence that what sits on the shelf now will remain stable, tightly contained, and ready to use—never to become the source of tomorrow’s emergency.
| Names | |
| Preferred IUPAC name | (4-nitrophenyl)acetonitrile |
| Other names |
p-Nitrobenzyl cyanide 4-Nitrobenzenecarbonitrile 1-Cyano-4-nitrobenzene 4-Nitrobenzenemethanenitrile |
| Pronunciation | /ˈfɔːr ˈnaɪtrəʊˌbɛn.zɪl saɪˈænaɪd/ |
| Identifiers | |
| CAS Number | 619-84-1 |
| Beilstein Reference | 1073880 |
| ChEBI | CHEBI:27841 |
| ChEMBL | CHEMBL234485 |
| ChemSpider | 14233 |
| DrugBank | DB08183 |
| ECHA InfoCard | 040000016367 |
| EC Number | 210-265-4 |
| Gmelin Reference | Gmelin Reference: "Gmelin 802299 |
| KEGG | C14306 |
| MeSH | D017923 |
| PubChem CID | 74929 |
| RTECS number | GV7875000 |
| UNII | WIZ38Z3682 |
| UN number | UN3439 |
| Properties | |
| Chemical formula | C8H6N2O2 |
| Molar mass | 148.14 g/mol |
| Appearance | White to pale yellow crystalline powder |
| Odor | Odorless |
| Density | 1.21 g/cm3 |
| Solubility in water | Slightly soluble |
| log P | 1.72 |
| Vapor pressure | 0.00017 mmHg (25°C) |
| Acidity (pKa) | 11.5 |
| Basicity (pKb) | pKb = 9.02 |
| Magnetic susceptibility (χ) | -8.45 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.5790 |
| Viscosity | 1.246 mPa·s (25 °C) |
| Dipole moment | 4.53 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 322.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -49.9 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS07, GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H312, H315, H319, H332, H335 |
| Precautionary statements | P261, P264, P270, P271, P273, P301+P312, P304+P340, P305+P351+P338, P312, P330, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-1-☠️ |
| Flash point | Flash point: 160°C |
| Autoignition temperature | Autoignition temperature: 540°C |
| Lethal dose or concentration | LD50 (oral, rat): 640 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 640 mg/kg |
| NIOSH | Not listed |
| PEL (Permissible) | Not established |
| REL (Recommended) | 25g, 100g |
| Related compounds | |
| Related compounds |
Benzyl cyanide 4-Nitrobenzaldehyde 4-Nitrobenzonitrile |

