4-Nitrobenzyl Chloride: Past Developments and Modern Realities
Historical Development
Stories about 4-Nitrobenzyl Chloride go back to a century of change in organic chemistry. This compound emerged as scientists looked for more direct ways to prepare benzyl chlorides with specialty nitro substitutions. Researchers tinkered with nitration and chlorination methods, sometimes in cluttered university labs with nothing but glassware and stubborn intent. As analytical techniques advanced, 4-Nitrobenzyl Chloride earned its spot as a practical benzylating agent. Publications before the digital era mention its use in photographic processing, pigments, and fragrance components, hinting at its versatility.
Product Overview
4-Nitrobenzyl Chloride stands out as a solid at room temperature, yellow and pungent. Its structure, a benzene ring sporting both a nitro and a chloromethyl group, keeps it tough enough for demanding synthetic routes. Chemists pick it for nucleophilic aromatic substitutions, protecting groups, and intermediate steps. Industries producing dyes, advanced polymers, pesticides, pharmaceuticals, or even optical brighteners see benefit from its reactivity. Its reliability grew as batch syntheses became standardized, and now global suppliers offer samples or drums depending on need.
Physical & Chemical Properties
This compound’s melting point makes handling predictable; it usually sits around 74-77°C. Its high density, about 1.38 g/cm³, lines up with expectations for nitro aromatics. Its moderate solubility in polar organic solvents lets labs and plants dissolve or extract it with little fuss. Its reactivity centers around the electrophilic chloromethyl group, which allows selective alkylation and other substitutions. The nitro group, a strong deactivator, influences its chemical fate—making certain side reactions less likely and providing chemists a specific range of transformations.
Technical Specifications & Labeling
Labels on 4-Nitrobenzyl Chloride packages tell a story of global standards. Industrial reference sheets flag purity levels, often above 98% for most lab work. Customers check for known impurities such as isomeric benzyl chlorides or leftover starting materials. CAS numbers, UN shipping handles, and GHS pictograms appear on every drum or bottle, as regulations steadily thicken. Labels also point to shelf-life studies and proper storage, keeping users honest and safe. Documentation reaches into every region, linking testing labs, customs authorities, and quality assurance teams.
Preparation Method
I’ve seen the most common route start from 4-nitrotoluene, which undergoes chlorination. This straightforward pathway has lingered in textbooks for decades because it delivers decent yields and clear byproducts. Certain techs harshly scrutinized reaction purity, switching from batch to continuous processes to meet modern environmental needs. Extraction, washing, and distillation steps get tweaked, aiming to cut waste and cut costs. Still, handling chlorine and controlling heat release define much of the risk. Purification almost always demands vacuum distillation or careful recrystallization.
Chemical Reactions & Modifications
Once inside a flask, 4-Nitrobenzyl Chloride rewards clever design. It acts as an alkylating agent, forming protective groups on phenols, amines, and nucleosides—giving synthetic chemists valuable tools to mask functional groups temporarily. In nucleophilic displacement reactions, the chloride leaves easily, replaced by oxygen, nitrogen, or sulfur atoms. Reduction of the nitro group opens up new product lines, including amines for dyes and pharmaceutical bases. Its derivatives extend the chemistry even further and spark innovation in supramolecular chemistry and catalysis.
Synonyms & Product Names
While lab sheets often list it as 4-nitrobenzyl chloride, you’ll also see p-nitrobenzyl chloride, para-nitrobenzyl chloride, and 1-chloromethyl-4-nitrobenzene. Older catalogs show alternate spellings and translations from German or French suppliers. Some manufacturers sell it under abbreviated codes for internal tracking. The chemical supply chains and customs routes demand a unified naming map to avoid confusion and shipping delays, especially as global regulations shift and expand.
Safety & Operational Standards
4-Nitrobenzyl Chloride demands respect, and from my experience, gloves, goggles, and well-ventilated fume hoods stand as essentials. Its irritant and possible sensitizing properties carry real risk in field and factory settings. Safety Data Sheets list recommendations for spill management, accidental exposure, and waste disposal. Many labs reinforce practices through ongoing worker training and clear labeling. Proper containment and scrupulous cleanliness limit risks of skin contact and inhalation. Regulations keep growing as more epidemiological data emerges, and companies face strict reporting under REACH and OSHA guidelines.
Application Area
Uses for 4-Nitrobenzyl Chloride cut across industries. It shows up in DNA and RNA research as a key protecting group strategy. Pharmaceutical pipelines rely on it for intermediate synthesis, and agricultural firms use it in fine-tuning crop protection agents. Polymer chemists have adopted it for functionalizing specialty plastics. Optical materials and dyes draw on its persistent stability and unique color properties. Academic research tracks each new modification, with recent studies diving into light-activated molecules and targeted drug design. Demand rises and falls with cycles in R&D innovation, but interest keeps growing as new uses emerge.
Research & Development
In recent years, R&D spent real money and time investigating 4-Nitrobenzyl Chloride’s potential in advanced organic transformations. Research groups pushed its use in photolabile protecting groups, where controlled light exposure triggers precise molecular changes. Projects in chemical biology focus on enzyme labeling and time-resolved studies using this compound. Startups experiment with green chemistry, modifying process parameters to shrink waste and energy footprint. Patents occasionally surface for new methodologies, showing innovation remains alive in both high-tech and mainstream applications. Some universities have opened courses or projects exploring next-generation nitro aromatics inspired by its successes and limitations.
Toxicity Research
Talk to anyone in safety compliance, and discussions of 4-Nitrobenzyl Chloride’s toxicity often echo past lessons. Lab tests show it can cause irritation to eyes, skin, and lungs. Animal data suggests it holds moderate acute toxicity, reinforcing why handling protocols matter. Long-term exposure risks continue to draw research, especially after some nitro aromatics showed carcinogenic potential. Industries monitor effluent streams to keep environmental releases in check. Newer analytical tools identify exposure biomarkers, helping identify safer workplace limits. Regulatory agencies routinely review toxicological data, adjusting legal thresholds as scientific knowledge grows. Industries and labs bear responsibility to stay ahead of these changes to protect both workers and local environments.
Future Prospects
The future for 4-Nitrobenzyl Chloride looks shaped by stricter environmental standards, smarter manufacturing, and digital tracking of chemicals. More players lean on automation and process analytics to cut hazards and improve efficiency. Sustainability factors now drive R&D, from greener starting materials to closed-loop waste handling. Partnerships between industry, academia, and regulatory bodies stand out as the way forward, especially when anticipating changes to REACH or GHS rules. Research teams worldwide test new applications ranging from biodegradable plastics to smart drug delivery based on nitrobenzyl chemistry. As chemical supply chains integrate blockchain and IoT sensors, traceability, authenticity, and safety should only improve, building trust from the maker’s bench to the user’s shelf.
What is 4-Nitrobenzyl Chloride used for?
Chemistry, Not Some Distant Concept
Chemical names like 4-nitrobenzyl chloride usually mean very little outside a lab coat world. For chemists, though, this compound isn’t just a tongue-twister. It’s a workhorse. In lab notebooks, jars of yellowish powder, and fume hoods, 4-nitrobenzyl chloride pops up everywhere because its structure packs both a nitro group and a reactive chlorine atom. That combination matters, especially in making molecules that don’t just sit on a shelf.
Sparking Transformations in Research
Scientists value 4-nitrobenzyl chloride for its use as a protecting group in organic synthesis. It helps mask alcohol and amine groups during multi-step reactions, allowing chemists to build complex molecules step by step without everything falling apart. For every pharmaceutical, agricultural chemical, or dye that goes from idea to actual product, there’s a lot of trial and error. Protecting groups like this one make that possible. Skip that tool, and reactions turn chaotic—yields drop, and projects grind to a halt.
Pharmaceuticals: More Than Just Pills
Pharma chemists lean on 4-nitrobenzyl chloride to build molecular frameworks for new drugs. They attach and remove this group to shield sensitive parts of a drug molecule through harsh reaction conditions. In practice, that means the difference between a lifesaving treatment making it out of clinical trials or getting stuck due to unstable chemistry. The moment a new drug works because a protecting group did its job is the moment this chemical proves its value, even if no patient sees its name on a label.
Molecular Biology: Lighting Up Discoveries
In the biotech field, 4-nitrobenzyl chloride plays a role in crafting sensitive chemical probes and linkers. Fluorescent tags that help researchers spot changes in proteins, DNA, or living cells often rely on this compound to block, then release, active sites at just the right moment. Precision matters. Miss a step, and the experiment goes dark. That’s why nobody shrugs off the role a single chemical like this can play, even in the age of AI-powered science.
Synthetic Dyes and Beyond
Beyond medicine and molecular biology, the textile and electronics industries tap into the reactivity of 4-nitrobenzyl chloride for crafting specialty dyes and intermediates for colorants. In printed electronics, those same chemical bonds show up in photolithographic processes. There, engineers lay down precise patterns for circuits and screens. Some of those processes would stall without such a reliable starting point.
Safety Must Come First
Handling 4-nitrobenzyl chloride isn’t like handling baking soda. The compound can irritate skin, eyes, and lungs. Labs keep it in tightly sealed bottles and only open them inside fume hoods. Everyone wears gloves, goggles, and lab coats. Manufacturers provide clear safety data, and regulators check shipments to keep workers and the environment safe—not out of some vague obligation, but because the health risks are real. Responsible handling stands at the center of chemical progress.
Future and Responsibility
Chemistry keeps moving forward. Green chemistry initiatives are changing how scientists work with reactive compounds, including 4-nitrobenzyl chloride. Labs are rethinking waste management, exposure limits, and finding alternatives when risk outweighs reward. Industry and regulators both have a stake. Everyone shares a goal—smart innovation without trading away safety. So, chemicals like 4-nitrobenzyl chloride stick around not for their name, but for the problems they help solve and the responsibility with which they’re handled.
What are the safety precautions when handling 4-Nitrobenzyl Chloride?
Real Dangers in the Bottle
Someone grabbing a bottle of 4-nitrobenzyl chloride for the first time might just see a pale yellow powder and not think twice. The reality hits harder. This stuff releases biting fumes that sting your nose and send you into a coughing fit. Even a small spill can leave you with burning eyes and skin. You want to keep your hands, face, and lungs out of harm’s way.
Protecting Yourself and Your Colleagues
Step one is personal gear. Nitrile gloves work best rather than basic latex. Laboratory coats that cover your arms matter, so roll those sleeves down. Proper goggles feel like a hassle until you realize a single splash can leave a nasty chemical burn on your cornea. The chemical moves quickly through skin and eyes, so rinsing for at least fifteen minutes in a safety shower or eye wash saves more than just your afternoon—it can save your vision.
Nobody ever wishes they spent less time in a fume hood. You want the draft on full blast to suck away vapors. It’s easy to think a quick scale-up doesn’t need it, but that’s when inhalation hits hardest. Once, I watched a friend ignore this and the entire lab cleared out for an hour, hacking and wheezing, until the acrid cloud dispersed.
Ventilation and Labeling Matter
Good airflow saves your nerves and your lungs. Basic aprons and gloves don’t cover inhalation risk. Even if the fume hood seems okay, don’t skip opening the sash before uncapping the flask. I’ve seen even experienced chemists overestimate their luck, only to regret it after a few minutes of watery eyes.
Label everything. Nobody wants to find an unmarked flask and sniff their way to identification. I once caught a tech reaching for what he thought was simple benzyl chloride—his hands came away tingling. Quick labeling makes the whole team safer and cuts down on mix-ups.
Storage and Chemical Reactions
4-Nitrobenzyl chloride doesn’t play nice with water or bases. It can decompose and throw off noxious gases. Keep it in sealed glass containers, tucked away from the main traffic in chemical storage rooms. Cool, dry spots away from sunlight do more to keep everyone healthy than any rushed clean-up after something goes wrong.
In my own experience, someone forgot to double-check if a storage cap was on tight. By morning, the room hummed with the sharp tang of chlorinated fumes. Venting the space and cleaning took most of the day—and the memory lasted a lot longer.
Dealing with Spills and Waste
Cleanup teams need respirators and chemical splash suits if things go south. Get a chemical spill kit ready before anything leaves the bottle. Work with absorbent pads and pour neutralizing agents only as directed—improvising often makes things worse. Knotted trash bags shouldn’t hold contaminated gear. Make sure hazardous waste bins are clearly marked.
Nobody wants a trip to the ER as part of their chemical routine. Treating 4-nitrobenzyl chloride with the respect it deserves keeps the science moving forward and keeps people from disaster.
What is the chemical formula and molecular weight of 4-Nitrobenzyl Chloride?
Chemical Formula and Structure
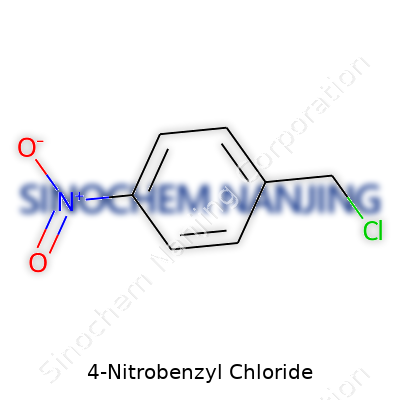
4-Nitrobenzyl chloride goes by the formula C7H6ClNO2. You get a benzene ring, four nitro at one end, plus a chloromethyl group replacing a hydrogen atom on the other. Chemists know the nitro group lands at the para position—directly across from the benzyl chloride. Its molecular weight clocks in at 171.58 g/mol, a detail that means something every time someone plans out a reaction. This isn’t just trivia. Having the right numbers helps avoid mistakes and wasted material in the lab.
Why Structure Matters
At first glance, chemical formulas might not mean much outside of a textbook, but they hold the key to understanding chemical behavior. Take 4-nitrobenzyl chloride as an example. The presence of both the nitro and chloride groups changes how the compound interacts with nucleophiles, bases, or the environment. This compound often enters the conversation in organic synthesis, especially among chemists who use it to build more complex molecules like pharmaceuticals or dyes. Each atom placement counts. The para-nitro group draws electrons away from the ring, increasing reactivity at the benzylic position. For synthetic chemists, small shifts in structure have huge consequences.
Guiding Safe Use
4-Nitrobenzyl chloride hasn’t earned a household name, but anyone who’s spent hours prepping reagents in a lab has probably seen it tucked away on a shelf. It’s reactive and somewhat volatile, so safety gear isn’t optional. Handling this compound with care isn’t just about protecting yourself from burns or fumes. Manufacturers and users must follow clear safety protocols, considering both toxicity and the risk of forming potentially harmful by-products. In a lab where this chemical is stored, consistent use of gloves, goggles, and fume hoods makes practical sense—not just to avoid short-term harm, but also to keep labs running smoothly.
Environmental Implications
Chemicals like this often end up raising questions about waste and disposal. If it finds its way into groundwater or soil, it doesn’t break down quickly. In my experience working with academic research labs, managing the leftover solutions that contain 4-nitrobenzyl chloride demands vigilance. You can’t just pour them down a drain and walk away. Proper procedures become part of daily routine, and every lab user gets briefed, sometimes more than once, to keep mishaps at bay.
Towards Responsible Chemistry
Looking forward, green chemistry offers tools to reduce reliance on potentially hazardous reagents like 4-nitrobenzyl chloride. Substitution—using less reactive or less toxic alternatives—continues to gain ground. Researchers now consider the full lifecycle of each chemical: creation, use, and disposal. Universities and industry labs now work together more closely around waste management and alternative synthesis strategies. Sharing these practices openly helps others avoid common pitfalls and supports safe, responsible innovation in the field.
How should 4-Nitrobenzyl Chloride be stored?
Why Careful Storage Matters
Working in labs, the safety tips packed into every protocol stick for a reason—one mistake with chemicals and lives change fast. 4-Nitrobenzyl chloride is no exception. I’ve seen bottles arrive, glass sweating, labels peeling off from the cold, and you start thinking, this isn’t just a shelf item. It doesn’t care if you’re sloppy or not paying attention. It reacts all the same.
4-Nitrobenzyl chloride brings together an aromatic ring, chlorine, and a nitro group. That sounds technical, yet what matters is how unstable that combination can get with moisture or heat. Sniffing a whiff of its pungent odor makes you realize this stuff isn’t forgiving. So, people ask if it just goes up on a shelf with other bottles. That’s a shortcut to disaster.
Best Storage Practices from Real World Experience
The best lab managers treat 4-nitrobenzyl chloride like a lit fuse. Dark, cool, and dry storage areas offer some protection. I keep it in sealed containers, away from sunlight or any source of warmth. Cabinets made for corrosive or hazardous chemicals work well because they stay sealed tight and handle accidental leaks or spills. At my last job, anything in that cabinet had double containment—another barrier for leaks.
Every bottle deserves a thick, clear label with big writing. I see too many colleagues making do with faded ink or only the chemical name, forgetting the date received or the hazard warnings. I’ve been guilty of this myself, but one close call taught me to always put the date and hazard codes where you can’t miss them.
The Science of Why Storage Rules Matter
4-Nitrobenzyl chloride reacts strongly with water. Leave a little moisture in the bottle and you can end up with hydrochloric acid escaping. That’s bad for your lungs, bad for your skin, and bad for whatever equipment stands nearby. Keeping humidity out is more than just a guideline; it’s non-negotiable. Silica gel packs become your best friend—pop a fresh one in any open bottle’s secondary container, and you’re instantly reducing risk.
Plastic doesn’t always cut it. Some plastics soften or even dissolve; glass usually wins here, but it’s still worth asking the supplier about compatibility. My own rule is never to guess—call the chemical safety office or check the latest documentation, even if it delays a project.
People Make the Difference
No storage plan works unless everyone in the space knows the rules. In my lab, we set quarterly reviews. Someone reads the Material Safety Data Sheet, checks inventory, and quizzes the team on emergency response steps. Laxity creeps in when routines lose urgency. Stories help keep everyone awake—a spill story told at break time sticks longer than any memo.
Access control also matters. Not every intern or visitor should reach for the hazardous shelves. Locking storage may sound strict, yet after seeing a novice pull the wrong bottle twice in one week, I started locking up anything that could harm with a single misstep.
Solutions Through Training and Accountability
Regular refresher sessions, mock drills for spills, and clearly posted emergency contacts at each storage location lift safety from the page into real habits. Good storage does not happen by chance; it comes from routines, shared stories, and strict habits built over years.
Treating 4-nitrobenzyl chloride with respect means thinking two steps ahead. If your workspace grows, invest in better cabinets and keep up with new storage standards as they change. Chemical safety isn’t a one-time choice. It’s a personal commitment every day you walk past that dangerous little bottle.
Is 4-Nitrobenzyl Chloride soluble in water or organic solvents?
Getting Real About Solubility
Anybody dabbling in lab work long enough knows that solubility isn’t some background detail—it shapes everything, from setup to cleanup. So, looking at 4-nitrobenzyl chloride, you won’t find it dissolving in water. Trying to mix it in with water leads to a cloudy mess, nothing more. Its molecular makeup—those aromatic benzene rings, the nitro group, the chloride anchored right on the ring—does not line up well with water’s tastes. Aromatic rings and water don’t exactly bond over coffee.
On the flip side, hit the bottle of organic solvents and it’s a different story. Chloroform, ether, dichloromethane—the stuff common in organic chemistry labs—cut right through a sample of 4-nitrobenzyl chloride. This comes down to the “like dissolves like” rule, which sounds basic until you realize how much it simplifies life in the lab. Most organic chemists go with dichloromethane or ether because these solvents grab onto aromatic molecules and hold them steady, letting reactions move forward without much fuss.
Why Solubility Rules Matter
Some might ask why anybody should sweat the details about which solvent handles a compound best. The answer jumps out the moment you try to purify or react a stubborn chemical. Take synthetic pathways using 4-nitrobenzyl chloride. These usually show up when building intermediates in pharmaceutical projects or crafting specialty materials. If the compound clumps up in the wrong solvent, everything slows down: poor yields, annoying side reactions, or layers that never separate cleanly.
A friend of mine struggled with this exact problem. The project called for a benzylation step, and they tried to short-cut with water as the work-up solvent. Clouds started forming, crystals fell out, and the column chromatography drag went on for hours. Only after switching to organic solvents did the pathway run as planned. Actual experience teaches you that the textbook advice—pick the right solvent, don’t cut corners—saves time and money.
Balancing Lab Safety and Functionality
Organic solvents make chemistry run, but labs wrestle with their own rules. Chloroform, ether, and even dichloromethane come with health and fire hazards. In my own work, glove-box sessions with these chemicals leave me thinking about airflow and waste traps as much as reaction yields. Chemistry teachers love telling students to respect the fume hood, and for good reason.
Switching to “greener” organic solvents like ethyl acetate or using less solvent overall offers part of a solution. These alternatives won’t dissolve everything, but more chemists try swapping them in, searching for a middle ground between lab safety and reliable results. At big companies, sustainability teams have started tracking solvent use, pushing for clear guidelines on which ones to use safely and where water might step in for specific steps.
Keeping Curious, Staying Responsible
Compounds like 4-nitrobenzyl chloride underscore the importance of matching solubility to workflow. Scientists who respect solvent choice don’t just get better yield—they also build in more time for troubleshooting problems where health and safety share the stage. If curiosity drives the discovery, common sense has to keep it safe.
| Names | |
| Preferred IUPAC name | (4-nitrophenyl)methyl chloride |
| Other names |
p-Nitrobenzyl chloride 1-(Chloromethyl)-4-nitrobenzene 4-Nitrobenzylchloride p-Nitrobenzylchloride Paranitrobenzyl chloride |
| Pronunciation | /ˈfɔːr ˌnaɪ.trəʊ ˈbɛn.zɪl ˈklɔːr.aɪd/ |
| Identifiers | |
| CAS Number | 100-14-1 |
| Beilstein Reference | 1635924 |
| ChEBI | CHEBI:83017 |
| ChEMBL | CHEMBL141661 |
| ChemSpider | 5043 |
| DrugBank | DB14170 |
| ECHA InfoCard | 03be63c8-5146-43d8-97b7-cf2aba9ee3ea |
| EC Number | 202-204-7 |
| Gmelin Reference | Gmelin 80854 |
| KEGG | C01763 |
| MeSH | D017927 |
| PubChem CID | 9986 |
| RTECS number | CY8575000 |
| UNII | R31A9P8M4B |
| UN number | UN1664 |
| Properties | |
| Chemical formula | C7H6ClNO2 |
| Molar mass | 169.57 g/mol |
| Appearance | White to light yellow crystalline powder |
| Odor | Aromatic |
| Density | 1.41 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 1.83 |
| Vapor pressure | 0.0092 mmHg (25°C) |
| Acidity (pKa) | pKa = 9.15 |
| Basicity (pKb) | 10.73 |
| Magnetic susceptibility (χ) | -64.7 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.5890 |
| Viscosity | 2.399 cP (25°C) |
| Dipole moment | 4.29 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 333.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -2.3 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes severe skin burns and eye damage, may cause respiratory irritation, toxic to aquatic life. |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H332, H335, H410 |
| Precautionary statements | P210, P261, P264, P271, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P332+P313, P337+P313, P362, P403+P233, P501 |
| NFPA 704 (fire diamond) | 3-2-1-X |
| Flash point | 94°C |
| Autoignition temperature | 220 °C |
| Lethal dose or concentration | LD50 oral rat 2400 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 1410 mg/kg |
| NIOSH | CAS7858 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 5 ppm |
| Related compounds | |
| Related compounds |
Benzyl chloride 4-Nitrobenzyl alcohol 4-Nitrobenzaldehyde 4-Nitrobenzoic acid |

