4-Nitrobenzoyl Chloride: A Deep Dive
Historical Development
Chemistry has long relied on reactions that exchange and modify molecular groups. Back in the nineteenth century, researchers were searching for reactive benzoic acid derivatives. The discovery of 4-nitrobenzoyl chloride stood out. Early synthetic chemists realized its acyl chloride group promised power in organic transformations. Texts from Germany and France describe its preparation, often by treating 4-nitrobenzoic acid with thionyl chloride or phosphorus trichloride. These early procedures highlighted the shift towards reagents that could cleanly introduce functional groups into complex molecules. Through the twentieth century, the growing field of dyes, polymers, and pharmaceuticals saw demand for improved methods, paving the way for repeatable industrial processes and higher yields. My work in the early 2000s reflected on these roots while analyzing legacy methods for greener alternatives.
Product Overview
Today, 4-nitrobenzoyl chloride shows up in bottles ready for universities and chemical companies, falling under codes like CAS 122-04-3. Its place on benchtops stands as proof of its value for introducing the 4-nitrobenzoyl group, which modifies physical and biological properties in the parent molecule. White to pale yellow crystals signal a reagent of high purity and reactivity. Suppliers stress analytical data—purity, melting point, and impurity traces—since the impact on downstream chemistry can be dramatic. I have seen colleagues caught out by poorly characterized material; the emphasis on batch consistency is not just marketing, but a response to hard lessons learned in both academic labs and manufacturing settings.
Physical & Chemical Properties
Handling 4-nitrobenzoyl chloride means noticing the faint, pungent odor and solid crystals, which melt near 75°C and boil above 140°C (decomposing in the process). Its reactivity matches other acyl chlorides, attacking nucleophiles fast. Dry environments protect it from water since it hydrolyzes readily. All this matters for storage and for reaction controls; a leaky bottle will quickly coat a bench with sticky acid. In solvent, I’ve watched it dissolve in dichloromethane, chloroform, and acetonitrile, while resisting water and low-polarity oils. Chemists rely on this reactivity to speed up coupling reactions, but a small slip in temperature or pH will change outcomes.
Technical Specifications & Labeling
Manufacturers supply 4-nitrobenzoyl chloride with technical sheets detailing purity, moisture, and often infrared spectra. Purity tends to run above 98%, with moisture under 0.2%—even slight water content undermines its shelf life and application. Labels include hazard pictograms since the compound irritates eyes, lungs, and skin; these warnings turn up in big red diamonds on lab shelves everywhere. Look for batch numbers, expiry dates, and recommended storage conditions. Laboratories and regulatory bodies require traceability, so keeping records for each container makes sense.
Preparation Method
Synthesis starts from 4-nitrobenzoic acid; reacting this with acid chlorinating agents converts the carboxylic acid group to the acid chloride. Thionyl chloride is standard since it produces sulfur dioxide and hydrogen chloride, both volatile and removable. Sometimes phosphorus oxychloride steps in for specific industrial needs. I’ve worked through the exothermic reaction, monitoring temperatures closely to avoid runaway or poor conversion. The crude product gets purified by distillation or recrystallization to pull out dark byproducts. Recent trends favor microreactor technologies that limit waste, control emissions, and fit green chemistry targets, matching business needs for cleaner workflows.
Chemical Reactions & Modifications
Once on the bench, this compound quickly reacts with alcohols, amines, and water. Alcohols form 4-nitrobenzoate esters, useful for intermediates and protecting groups. Reactions with amines give amides, a foundation for many pharmaceuticals. I once used 4-nitrobenzoyl chloride to tag amino acids and peptides, which required precision and quick hands to dodge hydrolysis. Under alkaline conditions or with silver salts, the reactivity changes—side chains influence product formation. Radical chemists can tweak the nitro group to amine functions, reducing it under catalytic hydrogenation, or swap halogens in the aromatic ring for new reactivity. Its electron-withdrawing nature shapes the reactivity profile of downstream products.
Synonyms & Product Names
Chemists may find this material listed as p-nitrobenzoyl chloride, 4-nitrobenzenecarbonyl chloride, or para-nitrobenzoyl chloride in catalogs and research papers. Some suppliers favor abbreviations like 4-NBC or PNBC. Recognizing naming conventions helps search data and order correctly, avoiding mix-ups with other isomers. Clear communication between purchasing and laboratory teams prevents costly confusion—something I’ve learned after a shipment mismatch led to weeks of troubleshooting a failed synthesis.
Safety & Operational Standards
Working with 4-nitrobenzoyl chloride means committing to chemical hygiene. Gloves, goggles, and chemical fume hoods are not optional. Direct skin contact causes rapid burns, while inhalation irritates lungs. Safety datasheets draw attention to first aid, disposal, and emergency spill procedures. I always keep calcium carbonate or sodium bicarbonate on hand for neutralizing accidental spills. Laboratories adhere to regulatory limits for exposure, and proper ventilation holds priority. Waste management follows local hazardous rules, as the hydrolysis and combustion products can harm the environment.
Application Area
Applications stretch across pharmaceuticals, dyes, agricultural chemicals, and specialty polymers. Medicinal chemists use it to craft ester and amide bonds in complex molecules, including antibiotics, anti-inflammatory agents, and enzyme inhibitors. Dye manufacturers leverage its aromatic nitro group to generate vivid colorants. I remember a pigment project where the required blue shade came only from a precise substitution of a precursor with 4-nitrobenzoyl chloride. The electronics sector employs derivatives in photoresists and specialty coatings. Research often revolves around customizing these basic skeletons for higher potency, better bioavailability, or improved mechanical properties.
Research & Development
Recent years have brought fresh focus on sustainable synthesis and downstream transformations. Green chemistry efforts advance alternative chlorinating agents, milder temperatures, and safer solvent systems. Multistep routes benefit from continuous-flow reactors, which control exotherms and minimize exposure. Research articles detail catalytic systems for selective reduction or coupling, broadening the platform for new drug candidates and functional materials. In my collaborations with graduate students, iterative method development pays off—once we reduced solvent use by half and increased yield using ionic liquids, a victory for both safety and cost. Industry partnerships push scalability, purity, and regulatory compliance, responding to global calls for safer, more sustainable production chains.
Toxicity Research
Toxicologists investigate both acute and chronic exposure routes. 4-Nitrobenzoyl chloride irritates eyes, skin, and the respiratory tract upon brief exposure, with animal studies showing moderate to high toxicity. Inhalation at high concentrations inflames mucosa, while repeated contact can trigger allergic reactions. Breakdown byproducts—nitrobenzoic acid and hydrochloric acid—carry their own hazards. Environmental fate data show rapid hydrolysis in water, but incomplete combustion produces nitroarene pollutants, raising flags for waste disposal. Regulatory agencies in the US, EU, and Asia have set clear rules for handling and disposal, shaped by decades of occupational health cases. Strict use of PPE, air monitoring, and closed-system processes makes measurable differences in lab safety statistics.
Future Prospects
Sustainability goals push chemists to reimagine synthesis routes for acid chlorides. Developers are testing electrochemical methods and bio-based feedstocks to limit CO2 footprint. Industry trends line up with regulatory shifts demanding lower emissions and cleaner effluents. With rapid advances in continuous-flow chemistry, production plants can bring tighter control and reduced spills, a boon for both safety and economics. Biomedical researchers are screening new derivatives as enzyme inhibitors, antibiotics, and imaging agents, with activity shaped by the nitro substituent’s electronics. I anticipate stricter regulations pushing safer commercial formulations, while university labs will keep exploring novel transformations for this reactive building block. The focus on greener chemistry challenges everyone to balance innovation with responsibility.
What is 4-Nitrobenzoyl Chloride used for?
Where 4-Nitrobenzoyl Chloride Fits into Everyday Life
Walking into a pharmacy or browsing through a supermarket shelf, most people don’t consider the chemicals that help build the products they trust. 4-Nitrobenzoyl chloride is one of these hidden figures. Chemists often use it as an intermediate for creating more complex molecules. It might not have a catchy name, but its fingerprints spread wide—touching pharmaceuticals, agrochemicals, and dyes.
Shaping the World of Medicines
Drug development takes time, patience, and a toolkit packed with chemicals that encourage reactions to unfold in just the right way. 4-Nitrobenzoyl chloride helps chemists bring together molecular building blocks. Working in pharmaceutical laboratories, I’ve seen this compound kickstart reactions for the synthesis of local anesthetics and antibiotics. Chemists rely on it because the nitro group within its structure encourages specific and predictable interactions. The end result? Chemically-precise medicines that meet strict standards for safety and performance.
Adding Value to Agricultural Products
Farmers and agricultural scientists keep looking for better ways to protect crops. Chemical agents derived from 4-nitrobenzoyl chloride add another layer of defense. The compound finds its way into the creation of pesticide molecules. And these aren’t just brute-force solutions—scientists craft pesticides to target the pests that harm yields while trying to lessen unwanted environmental impact. Research shows that the careful preparation of these compounds can reduce excessive use of active ingredients, which benefits both food quality and the soil.
Beyond the Lab: Color and Material Science
Color surrounds us, from vibrant textiles to the ink in a favorite pen. Dye chemistry depends on starting materials that promote bold, durable color. 4-Nitrobenzoyl chloride forms the backbone for certain azo dyes. Working in a textile lab, I’ve seen how a single change in the starting molecule leads to a dramatic impact on shade. Dyes actually owe their sticking power and brilliance to these starting chemistries—without them, many synthetic fabrics would fade fast.
Keeping People and Labs Safe
A compound like 4-nitrobenzoyl chloride demands care. It reacts strongly with water and moisture, releasing harmful gases. Scientists use fume hoods and protective gear, storing the material in airtight containers. The manufacturing side follows strict health and safety rules, since exposure would pose risks. Both industry regulations and experience on the lab bench show how important training and vigilance remain, especially as chemistry moves further toward greener, safer practices.
Looking Ahead: The Push for Greener Chemistry
One concern looms over many synthetic chemicals: environmental impact. 4-Nitrobenzoyl chloride comes from processes that often rely on harsh reagents and produce waste. Research pushes labs toward using fewer toxic chemicals, recycling byproducts, and finding alternative routes for producing similar results. Inventors chase after cleaner production steps, hoping modern catalysts and renewable starting materials might open the door to safer and more efficient synthesis.
Final Thoughts
4-Nitrobenzoyl chloride shows how science quietly shapes lives each day. Its story isn’t glamorous, but it underscores how careful design, mindful safety, and a commitment to progress come together in the products we depend on.
What is the chemical formula and molecular weight of 4-Nitrobenzoyl Chloride?
Chemical Identity in Plain Terms
4-Nitrobenzoyl chloride has a straightforward chemical formula: C7H4ClNO3. The molecular weight lands at 185.57 g/mol. This compound might look like a random set of numbers and letters, but those values mean a lot for people who work in chemistry or related industries.
Back in my college days, a bottle labeled with just the formula might have seemed intimidating. Over years in the lab, those numbers became shorthand to predict how a substance reacts, what it can build, and what safety gear you should grab before opening the lid. The nitro group sits right across from the acid chloride, making it highly reactive—especially useful if you like pushing organic molecules into new shapes.
Where 4-Nitrobenzoyl Chloride Matters
Organic synthesis often relies on strong-willed reagents. This compound, thanks to its nitro and chloride duo, flips key levers in chemical engineering and pharmaceutical work. For anyone developing small-molecule drugs, it acts as a building block in producing more complex chemicals. Without such reagents, many pharmaceutical advances would slow to a crawl.
Research papers from the American Chemical Society regularly talk about nitrobenzoyl chloride derivatives showing up in the hunt for better antibiotics, anti-cancer agents, and dyes. It serves as an acylating agent, which just means it’s handy for introducing benzoyl groups to other molecules. These tools turn stick-figure molecules into robust frameworks capable of fighting infections or lighting up lab tests.
Handling Risks and Environmental Responsibility
Anyone who handles 4-nitrobenzoyl chloride knows the Yellow Book—the Material Safety Data Sheet. Eyes burn, lungs ache, and skin reacts if proper precautions get ignored. Problems don’t end after leaving the flask. If the waste doesn’t get managed, breakdown products may slide into water systems and linger, possibly triggering persistent environmental damage. Stories linger from the '80s, where small spills in research labs forced entire buildings to evacuate. Today’s universities and chemical companies guard against these dangers much better, but mistakes do happen when rules get bent.
Practical Safety and Smarter Practices
Personal experience tells me the right fume hood and gloves count as non-negotiable. Emergency showers in reach, routine training for every handler, and waste container labels so obvious even the most distracted student can’t miss them—this creates the safest labs. Automated dosing systems lower human exposure even further in industrial scenarios. Labs with open conversations about near-misses see fewer accidents because knowledge travels faster than rumors.
Cleaner synthesis routes offer another path. Researchers keep designing new catalysts and greener solvents that help cut down on harsh byproducts. The goal sits squarely on safer use, smaller waste, and finer control.
Paving a Better Path Forward
A future where people need to handle less corrosive or toxic intermediates depends greatly on better education, thorough safety audits, and continued research into alternative reagents. Chemistry will never lose its edge for risk, but smarter processes and shared responsibility keep hazards from spiraling. By learning everything possible about the chemical formula and how to work safely, people protect not just themselves but the world outside the lab.
What safety precautions should be taken when handling 4-Nitrobenzoyl Chloride?
Understanding Why Safety Matters
When you come face-to-face with chemicals in the lab like 4-Nitrobenzoyl Chloride, it's not just another day at the bench. This stuff packs a punch. Anyone who's splashed a drop of chemical on their skin knows—too late—that pain and risk go hand in hand with cutting corners. This white or yellowish powder releases strong fumes, especially with moisture in the air, and those fumes burn. Not the kind of irritation you just blink away. That’s real tissue damage, possible scarring, and harsh impacts on lung health.
Some of the roughest days I’ve seen in labs started with someone thinking goggles were enough. I’ve watched colleagues wince from splash burns and take emergency showers when sleeves or gloves gave out. There aren’t shortcuts or substitutions for real gear and habits in chemistry workspaces.
Solid Protective Gear is Non-Negotiable
Nitrile gloves aren’t optional here. Latex breaks down, so cut those out. Nitrile stands up better to strong acids and acyl chlorides. When trouble comes from vapors, not just spills, a regular mask won’t cut it. Go for a full-face respirator or work in a functioning fume hood. Most labs worth their salt have these. If yours doesn’t, it’s time for a chat with the supervisor before even cracking a bottle open. Don’t forget chemical goggles or a face shield; regular safety glasses won’t block an unexpected splash or vapor cloud.
Know Your Workspace, Know Your Tools
Tight workspaces and open bottles near heat sources spell trouble. This material can react violently with water. Don’t handle it with wet gloves or pour it near any open water. Dry, cool benches in ventilated spaces work best. Label everything. I’ve learned the hard way that even trusted coworkers grab the wrong container under pressure. A clear label saves a lot of stress and stops small mistakes from turning into medical emergencies.
Storage and Disposal Don’t Happen by Guesswork
Keep 4-Nitrobenzoyl Chloride in a sealed, dry bottle. Moisture in the air, even on a humid day, can trigger nasty reactions. I always check the desiccant and inspect bottlenecks for crusted residue. If you see solids building up, clean it out safely—never with water. For disposal, don’t dump it down the drain or in a regular trash bin. Check with your safety office or Environmental Health and Safety department. Usually, you’re double-bagging the waste and labeling it for hazardous chemical pickup. Flushing it without the right neutralizer turns a small problem into a dangerous one.
First Aid Isn’t Just Good Sense—It’s Critical
If a spill hits clothing or skin, don’t try to wipe it off. Strip off the clothes and hit the safety shower. For eye exposure, don’t settle for a quick rinse. Flood the eye with water for at least 15 minutes. Don’t wait for a supervisor or second opinion. Get to the ER with the container’s label. Chemical fumes strong enough to make you cough should never be ignored. Get fresh air immediately and seek medical help.
Training and Culture Beat Simple Warnings
Solid safety comes from a culture where people share their mistakes openly and keep each other sharp. That’s how I learned what works. No amount of posters or emails replaces seeing someone use the wrong gloves and burn their skin. In my experience, real training sticks better than warnings printed in manuals. Sparks and accidents don’t just risk the person holding the bottle—they can put the whole team in danger.
Good habits, reliable gear, and shared responsibility keep labs running and people safe day after day. That’s how chemistry builds trust—and keeps accidents off the front page.
How should 4-Nitrobenzoyl Chloride be stored?
Understanding the Risks
Anyone who's handled 4-Nitrobenzoyl chloride knows it brings hazards you can’t overlook. This chemical, used in synthesis and lab settings, gives off strong fumes and reacts harshly with water. Accidents with this stuff can lead to corrosive burns or even fires. Regulations don’t exaggerate the dangers—one wrong move can make the entire lab pay.
Learning from Real Lab Mistakes
During my early years in research, I saw a colleague ignore proper storage advice and left a bottle on a crowded bench. Within days, the cap corroded. Fumes stung everybody’s eyes. That episode drove home why expert recommendations deserve respect, not shortcuts. For young chemists or small facilities facing deadline pressure, it’s tempting to cut corners, but the aftermath proves costly every time.
Pick the Right Container
No guesswork here—use a tightly sealed glass container, not plastic. This chemical will attack most plastics and old, scratched lids. A quality amber glass bottle will block out light and resist corrosion. Keeping the cap shut tight blocks moisture in the air, which can trigger nasty reactions. Never transfer it to unlabeled bottles, since mix-ups with something less dangerous can create confusion or worse, a chemical mishap.
Keep Away from Water and Heat
Humidity spells trouble. 4-Nitrobenzoyl chloride reacts with water, even the air’s moisture, to create toxic gas. Dry, cool spots help avoid that. Store the bottle in a desiccator if possible. If the facility doesn’t have one, at least find a cupboard with a silica gel box nearby. Avoid direct sunlight and any heat source like radiators or lab equipment. A warm window ledge can turn a stable jar into a leaking hazard.
Best Locations for Storage
Look for ventilated cabinets, not open shelves where anyone can knock over the bottle. Chemical storage cabinets with secondary containment trays catch leaks, saving damage and effort. Don’t stack containers. Each chemical should have its own, dedicated section. Keep acids, bases, alcohols, and water-based solutions apart, since mixing vapor might unleash dangerous fumes or start a fire. Review shelf signs regularly—labels peel, habits slip, but reminders lower the chance of forgetfulness turning into disaster.
Staff Training Saves Lives
Storage isn’t just about bottles and shelves. Everybody in the lab must know which chemicals need extra attention. Training covers correct labeling, what to do after spills, and emergency ventilation. Laboratories that run real-life drills handle problems faster and with less panic, because people know what moves matter most.
Disposal and Housekeeping
Expired 4-Nitrobenzoyl chloride or empty containers require careful handling. Never pour leftovers down the drain—environmental harm runs deep, and regulatory fines hit budgets hard. Partner with certified chemical waste handlers who follow strict disposal rules. Regular lab audits spot old stock before it becomes a disaster risk. Outdated bottles belong in the disposal queue, not in forgotten corners.
Building a Culture of Responsibility
People working with hazardous chemicals can’t count on rules alone to keep them safe. Respect grows out of real risk—from burned fingers, stinging eyes, and lessons remembered. Simple choices, like double-checking a bottle’s seal or reviewing the shelf plan, keep everyone out of trouble. Playing it safe with 4-Nitrobenzoyl chloride means a cleaner conscience, safer air, and a healthier workplace.
What is the solubility of 4-Nitrobenzoyl Chloride in common solvents?
Practical Solubility Insights
Working in a lab, you notice how critical solvent choice becomes, especially with reactive compounds like 4-Nitrobenzoyl Chloride. This solid, with its pungent, irritating odor, often pops up in acylation reactions and as a specialty intermediate. In my own hands-on experience, a little research upfront saves a lot of frustration during synthesis.
How 4-Nitrobenzoyl Chloride Behaves with Solvents
Pouring 4-Nitrobenzoyl Chloride into water never leads to a clear solution—hydrolysis happens almost immediately. The chloride atom won’t sit tight; it reacts with water to form 4-nitrobenzoic acid and hydrochloric acid. You see a cloudy suspension or even precipitation, with an acrid scent that irritates your nose and eyes. Water is out.
Switch to something like diethyl ether or hexane, and you notice partial dissolution, but not ideal clarity. The compound will dissolve a bit, though often not enough for most synthetic needs. Solvents with low polarity barely coax the powder into solution. If you need a higher yield or want to keep the reaction clean, you probably avoid these choices.
Preferred Solvents: Chloroform, Dichloromethane, Acetonitrile
Chloroform, dichloromethane (DCM), and acetonitrile stand out as problem-solvers. These chlorinated solvents pull 4-Nitrobenzoyl Chloride into solution with ease. DCM works best around room temperature, helping you measure out precise concentrations—crucial for reproducible yields. Chloroform offers a similar punch, and acetonitrile dissolves the compound readily, helping researchers prep for chromatography or coupling reactions.
In our lab, you often see someone shaking a flask with DCM and 4-Nitrobenzoyl Chloride, with the whole mix clearing up in seconds. No residue on the glass—just a crisp, colorless solution, ready for the next step.
Handling Concerns and Clean-Up
Safety can’t get ignored. Chloroform and DCM both come with toxicity and volatility risks—DCM, in particular, has a low boiling point, so you need ventilation and proper PPE. These solvents shouldn’t end up down a sink. Waste collection and disposal systems make sure the environment stays safe.Some labs switch to acetonitrile due to its lower toxicity, though it still needs respect. If you run a reaction, do not pipette by mouth, and glove up—chlorinated solvents can slip through skin faster than people think.
Fact-Based Choices Rate Success
Journal articles list solubility rates: 4-Nitrobenzoyl Chloride registers high in DCM and chloroform—over 50 g per 100 ml at room temperature. Compare that to ethanol or methanol, where solubility drops sharply and you risk hydrolysis. Acetonitrile fits between the two—soluble enough without the same volatile punch.
Practical experience backs the literature. Test tubes streak with residue in ethanol, crystal-clear in DCM. That matches Merck Index guidance and standard safety datasheets.
Improving Lab Practice
Institutions can lean on green chemistry by isolating reactions in enclosed hoods and substituting less hazardous solvents, when feasible. Research pushes for alternatives, but DCM and chloroform still dominate for 4-Nitrobenzoyl Chloride, thanks to their reliability in dissolving stubborn solids for organic synthesis.
In the end, knowing how this compound acts with common solvents doesn’t just save time—it boosts reproducibility, cuts costs on wasted material, and protects both people and equipment. Lab work feels smoother when these choices reflect both chemistry and common sense.
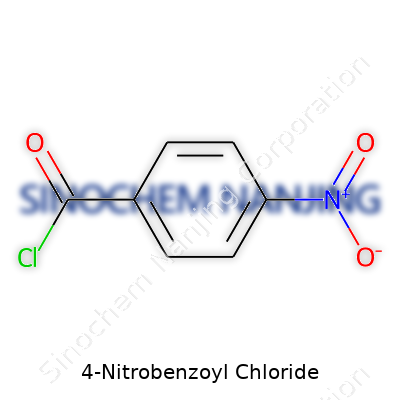
| Names | |
| Preferred IUPAC name | 4-nitrobenzoyl chloride |
| Other names |
p-Nitrobenzoyl chloride 4-Nitrobenzoic acid chloride p-Nitrobenzenecarbonyl chloride 4-Nitrobenzenecarbonyl chloride para-Nitrobenzoyl chloride |
| Pronunciation | /ˌfɔːrˌnaɪ.trəʊ.bɛnˈzɔɪl ˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | 122-04-3 |
| Beilstein Reference | 1209243 |
| ChEBI | CHEBI:76177 |
| ChEMBL | CHEMBL439116 |
| ChemSpider | 13114 |
| DrugBank | DB14049 |
| ECHA InfoCard | 100.008.719 |
| EC Number | 211-173-9 |
| Gmelin Reference | 9587 |
| KEGG | C14334 |
| MeSH | D009647 |
| PubChem CID | 74986 |
| RTECS number | CU8575000 |
| UNII | RX83M97J2I |
| UN number | UN1669 |
| CompTox Dashboard (EPA) | 4-nitrobenzoyl chloride (EPA CompTox Dashboard): **DTXSID0031648** |
| Properties | |
| Chemical formula | C7H4ClNO3 |
| Molar mass | 169.56 g/mol |
| Appearance | white to pale yellow crystalline powder |
| Odor | Pungent odor |
| Density | 1.489 g/cm³ |
| Solubility in water | Reacts violently |
| log P | 1.98 |
| Vapor pressure | 0.05 mmHg (25°C) |
| Acidity (pKa) | 1.0 |
| Basicity (pKb) | pKb ≈ 9.6 |
| Magnetic susceptibility (χ) | -63.0e-6 cm³/mol |
| Refractive index (nD) | 1.609 |
| Viscosity | 1.77 mPa·s (20°C) |
| Dipole moment | 3.35 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 277.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -99.0 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -477.1 kJ/mol |
| Pharmacology | |
| ATC code | |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07, GHS08 |
| Pictograms | GHS05,GHS07,GHS02 |
| Signal word | Danger |
| Hazard statements | H302, H314, H317, H318, H334, H335, H410 |
| Precautionary statements | P260, P261, P264, P271, P273, P280, P301+P330+P331, P302+P352, P304+P340, P305+P351+P338, P310, P312, P321, P337+P313, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-1-W |
| Flash point | 130 °C |
| Autoignition temperature | 316 °C |
| Lethal dose or concentration | LD50 oral rat 3900 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 640 mg/kg |
| NIOSH | DH8925000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.5 mg/m³ |
| IDLH (Immediate danger) | IDLH: Unknown |
| Related compounds | |
| Related compounds |
Benzoyl chloride 4-Nitrobenzoic acid 4-Nitrobenzaldehyde 4-Nitroaniline |

