4-Nitrobenzohydrazide: Deep Dive into Its Role Across Industry and Research
Historical Development
4-Nitrobenzohydrazide entered scientific literature in the first half of the 20th century when researchers began seeking intermediates to support new dyes and agrochemicals. Hydrazide derivatives rose in popularity during the mid-century push for innovative pharmaceuticals. 4-Nitrobenzohydrazide drew attention thanks to its reactive nitro group and the hydrazide moiety, which together support unique reactivity. Over decades, chemists have adopted this compound for both its versatility and relatively straightforward synthesis. Its position among specialty chemicals grew as heyday advances in synthetic organic chemistry demanded building blocks tolerant of tough reaction conditions.
Product Overview
The chemical features a benzene core with a para-positioned nitro group and a hydrazide substituent. This setup creates a molecule with enhanced nucleophilicity at the hydrazide site and electrophilicity at the nitro group. These characteristics make it attractive for forming hydrazones, exploring nitro reductions, and constructing advanced intermediates. As manufacturers pushed to deliver pure and reproducible product, technical standards formed around crystalline powders, often available in light yellow to orange hues, and packages designed for laboratory or industrial environments.
Physical & Chemical Properties
Most samples present as pale yellow crystals, shaped by strong intermolecular forces owing to the nitro and hydrazide functionalities. The compound’s molecular weight stands at roughly 181.14 g/mol. Melting points typically fall between 220°C-225°C, depending on purity—an impressive temperature buffer that protects the hydrazide group during multi-step syntheses. 4-Nitrobenzohydrazide’s moderate solubility in polar organic solvents like dimethylformamide and dimethyl sulfoxide aids its utility in organic reactions. Its nitro group invites reductions, while the hydrazide often reacts cleanly with a range of electrophiles.
Technical Specifications & Labeling
Producers supply technical sheets reporting assay percentages, usually not lower than 98%, and identify related impurities using high-performance liquid chromatography (HPLC). Moisture content and inorganic residue get flagged, as water traces may catalyze unwanted side reactions. Labels warn about toxicological risks, highlighting both the nitro group’s reputation for reactivity and the potential sensitizing effect of the hydrazide. Standard containers come with tamper-evident seals plus batch traceability codes, supporting both laboratory transparency and compliance with regulatory guidelines.
Preparation Method
Synthesizing 4-nitrobenzohydrazide typically starts with 4-nitrobenzoic acid as a precursor. Treating this with thionyl chloride produces the acyl chloride, which then reacts with hydrazine hydrate. Reflux conditions allow near-complete conversion. After the reaction, filtration and recrystallization from ethanol give a solid with minimal byproducts. Alternative protocols use carboxylic acid derivatives, though direct hydrazinolysis of esters sometimes helps avoid chlorinated reagents. Purification steps typically include charcoal treatment and multiple crystallizations, tailored to meet tight impurity limits.
Chemical Reactions & Modifications
The molecule behaves as both a nucleophile and an electrophile. The nitro group withstands many reaction conditions, though catalytic hydrogenation or chemical reduction—often using tin(II) chloride or iron filings—converts it to 4-aminobenzohydrazide. This opens new paths toward heterocycle formation, specialty dyes, and pharmaceutical frameworks. The hydrazide portion readily condenses with aldehydes and ketones, generating hydrazones that chemists use to investigate carbonyl compounds or milk further into advanced ligands. Derivatization at the hydrazide can yield acyl, sulfonyl, or alkylated analogs, broadening its reach into drug design.
Synonyms & Product Names
Across catalogs and chemical literature, the compound appears under several names. Common synonyms include p-nitrobenzohydrazide, 4-nitrobenzoic acid hydrazide, and N'-amino-4-nitrobenzamide. Each major supplier may use subtle branding, but the IUPAC name leaves little room for ambiguity. These names often appear on safety data sheets, transaction documentation, and regulatory filings.
Safety & Operational Standards
Handling 4-nitrobenzohydrazide without robust safety practices invites risks, as exposure may cause skin or respiratory irritation. The nitro group and hydrazide both raise red flags for mutagenicity. Researchers using this compound rely on personal protective equipment, fume hoods, and proper waste containers, as spills or inhalation hazards can complicate cleanup operations. Safety documentation recommends keeping open flames away from organic hydrazides due to rarely observed but well-documented instability. Organizations must train staff on these points, audit storage conditions, and periodically review regulatory changes.
Application Area
Chemists have learned to apply 4-nitrobenzohydrazide in several branches of research. Its classic use involves synthesizing hydrazone derivatives for detection or structural labeling, making it a workhorse for analytical chemistry and biochemistry. The molecule’s reactivity lends itself well to pharmaceutical exploration—building blocks for antiviral, antibacterial, and anticancer compounds. In polymers and dyes, the compound’s stability supports applications that require resistance to heat and light. Materials science ventures also take advantage of its redox behavior and ability to introduce nitrogen-rich motifs into aromatic systems, whether for smart sensors or energetic materials.
Research & Development
Ongoing projects worldwide keep pushing the possible with 4-nitrobenzohydrazide. Teams continue publishing new methods for green synthesis that avoid hazardous solvents and minimize chemical waste. Drug discovery programs lean on its framework for hitting new molecular targets, especially using the hydrazide group as a key handle in library synthesis and fragment linking. Researchers working on metal-organic frameworks experiment with functionalizing ligands based on substituted benzohydrazides, pursuing promising advances in catalysis and adsorption. The versatility of this compound keeps it in the sights of innovators focused on value-added materials and sustainable process chemistry.
Toxicity Research
Regulatory bodies pay close attention to nitroarene derivatives, and 4-nitrobenzohydrazide is no exception. Toxicological studies reveal evidence of acute toxicity in animal models, guiding strict occupational exposure limits. Chronic exposure studies remain ongoing, examining any connections to mutagenic or carcinogenic pathways. The hydrazide structure has a documented record for causing allergic reactions. Toxicity management often combines engineering controls with good hygiene practices to keep individual risk low. In laboratory settings, proper waste tracking and minimized use guard both worker safety and environmental impact.
Future Prospects
The journey for 4-nitrobenzohydrazide is far from over. As process chemists turn toward efficiency and sustainability, finding less energy-intensive preparation routes holds promise. Increased data on biological activity may spur new drug discovery programs. Materials scientists eye its suitability in the creation of smart polymers and advanced composites, especially for niche electronics and high-value coatings. As analytical techniques get sharper and regulatory demands tighten, producing ultra-pure product will become even more important. Partnerships between academia and industry will set the pace for how this chemical continues to influence research, manufacturing, and product development on a global scale.
What is the chemical formula of 4-Nitrobenzohydrazide?
Why Chemical Structure Still Matters
Chemical names sometimes sound intimidating. Take 4-Nitrobenzohydrazide, for example. That name might make you think of a complicated compound filled with twists and turns. In practice, knowing the chemical formula brings everything down to earth. For 4-Nitrobenzohydrazide, that formula is C7H7N3O3. Each letter and number in that formula tells a story—a nitro group attached to a benzene ring, with a hydrazide group waiting at the tail. That’s not just textbook chemistry; it’s the stuff underpinning many reactions in today’s research labs.
Why This Compound Draws Attention in Research
I’ve spent time in university labs watching undergrads struggle with chemical nomenclature. The name often leaves people confused or thinking that only experts need to care. But even outside organized chemistry circles, the connectivity in a molecule like 4-Nitrobenzohydrazide helps everyone—from teachers to pharmaceutical engineers—better understand how drugs are designed or why dyes work as they do. The nitro group brings a punch of reactivity. The hydrazide gives it a leg-up for making other molecules, especially when forming bonds with acids or aldehydes.
Safety and Handling Using Real Knowledge
Talking textbook precautions about chemicals doesn’t stick until you see someone get careless. Imagine a finely ground yellow powder that stains everything, stings your nose, and can cause headaches after a day’s exposure. That’s not just 4-Nitrobenzohydrazide, but it does apply. Using the chemical formula C7H7N3O3 helps make sense of what it can do. The nitro group flags potential irritancy and oxidative character, not just for chemists but for anyone handling powders for the first time. Keep goggles on and ventilation running, because molecules like this don’t forgive sloppy glove work.
Environmental and Ethical Impact
Reading the formula, you spot the three nitrogens—more nitrogen than most basic organic molecules. Compounds like this could break down into things you wouldn’t want in groundwater or rivers. Academic studies from Europe and Asia warn about hydrazide-containing chemicals sometimes making their way past ordinary water treatment processes. We can’t ignore what that means for environmental safety. The solution isn’t to ban such substances across the board; rather, responsible disposal steps up. Labs and factories need modern treatment strategies to trap or degrade hydrazide byproducts before they leave the facility.
Real Potential in Medicine and Industry
Look at drug libraries or chemical catalogs, and 4-Nitrobenzohydrazide appears in a long list of building blocks. Medicinal chemistry finds hydrazide derivatives useful when searching for new antibiotics or anti-tubercular drugs. Some researchers test these molecules for their power to kill certain pathogens or even act as color-forming agents in test kits. That starts from a basic knowledge of the chemical formula, giving insight into what transformations you can expect in the lab and which new properties might emerge in daily use.
Solutions Rooted in Chemistry Literacy
Learning chemical formulas isn’t about passing exams. Anyone—student, teacher, or manufacturer—gains both safety and creative power from this kind of literacy. Organizations need to make ingredient information transparent, down to each atomic component. And ongoing training means fewer lost hours from accident and misadventure. For anyone overseeing labs or industrial processes, investment in modern waste remediation stands out as more than compliance—it's basic stewardship.
What are the typical applications of 4-Nitrobenzohydrazide?
In the Lab: Stepping Stone for New Compounds
Many chemists, especially those working in organic synthesis, look at 4-nitrobenzohydrazide as a versatile tool. This compound gives researchers a path toward more complex molecules. The nitro group and hydrazide group both open doors for all sorts of chemical reactions. If someone wants to make heterocyclic rings—a backbone in many drugs—or explore new dyes, this molecule is likely on their shelf.
I remember chatting with a friend who ran a small synthesis lab. She always kept a few grams on hand for those unexpected project twists. If a reaction called for a functional group transfer or formation of novel azo dyes, this molecule’s dual reactive sites often set her on the right track. Publications in journals like the Journal of Organic Chemistry often mention its use in building blocks for pharmaceuticals. These rings and complex structures rarely come from straightforward, one-step reactions; you need reliable intermediates, and this hydrazide fits the bill.
Medicinal Chemistry: Chasing the Next Drug Candidate
Those nitro and hydrazide groups aren’t just for show—they're key players in medicinal chemistry. Researchers use 4-nitrobenzohydrazide to build intermediates in the search for new antibiotics, antifungals, and anti-inflammatory drugs. For example, linking this molecule with other aromatic aldehydes creates hydrazones that researchers test for a range of biological activities. Not every hydrazone comes out a winner, but these experiments generate the data needed to move closer to a blockbuster drug.
As a graduate student, I saw plenty of failed syntheses and unexpected color changes, but also witnessed how a single compound tweak—like swapping a methyl group for a nitro group—changed everything. The literature backs this up: studies reported in Bioorganic & Medicinal Chemistry highlight nitrobenzohydrazide derivatives as potent inhibitors of certain enzymes. These findings keep driving new experiments and patent applications year after year.
Dyestuffs and Pigments: Coloring Our World
Chemists use this compound to form new azo dyes. The azo coupling reactions involving 4-nitrobenzohydrazide often produce vibrant colors, which end up in textile labs and ink factories. Azo dyes aren’t just about color—they need to be stable, safe, and resistant to sunlight or washing. The predictable chemistry of this molecule helps specialists fine-tune shades that manufacturers want.
People who work in dye development have shared with me how a small change at the molecular level sometimes brings about the vivid yellow or deep red that a client expects. Research papers, particularly those from the textile industry, still reference 4-nitrobenzohydrazide as a foundation for color chemistry.
Looking at Risks and Handling
Anyone using 4-nitrobenzohydrazide should think about safety. The nitro group in many organic chemicals signals caution, as it can present handling risks; this compound isn’t something to toss around the bench without gloves and a fume hood. Regulatory agencies recommend clear labeling and proper disposal protocols. No shortcut—attention paid early prevents headaches later.
Chemical safety databases point out not only toxicological hazards but environmental ones, too. Keeping up with safety data sheets and double-checking protocols prevents accidents, which can be more than just a minor lab mishap.
Room to Improve and Innovate
Industries and academic labs keep looking for ways to use compounds like 4-nitrobenzohydrazide more safely and efficiently. Ongoing research targets greener reaction conditions, better waste management, and safer storage. Efforts from both grassroots lab workers and regulatory bodies shape how this compound gets used responsibly. Open communication in the scientific community helps everyone make smarter, safer choices in the lab and beyond.
What are the storage and handling precautions for 4-Nitrobenzohydrazide?
Why Storage Really Matters with 4-Nitrobenzohydrazide
I’ve seen labs come to a complete standstill because of a chemical spill or an unexpected reaction during storage. That’s enough to put respect into anyone handling something as sensitive as 4-nitrobenzohydrazide. Even though it isn’t the most volatile chemical out there, this compound demands attention. Its nitro-group and hydrazide structure give it both reactivity and sensitivity, a mix that can spell trouble for careless storage.
The material looks unassuming—a pale, powdery solid—but its properties tell a different story. It’s sensitive to heat, friction, and sometimes decomposes in the presence of incompatible substances. Storing it in a cool, dry, and well-ventilated area helps prevent any surprises. Heat turns a stable chemical into an unpredictable one, and moisture plays against its integrity. Sealing it tightly and keeping humidity levels low reduces the chance of dangerous decomposition or clumping, both of which I’ve unfortunately witnessed.
Material Compatibility and Segregation
For me, the golden rule is never jam a shelf with every chemical possible. 4-nitrobenzohydrazide especially deserves segregation from acids, oxidizers, and strong bases. Some labs cut corners here, but that’s asking for trouble. I keep it away from anything reactive, since accidental mixing can start a reaction chain that’s not easy to control. Glass containers work better than plastics because hydrazides sometimes corrode softer materials. Proper labels that include the compound’s full name, the date received, and hazard class help everyone stay on the same page.
Personal Protection During Handling
Anyone who’s scooped out hydrazides without gloves probably remembers the tingling or irritation that follows. Direct contact might leave burns or rashes, so I insist on gloves and safety goggles every time. A dust mask prevents inhalation, something that gets overlooked when working with powders. Chemical-resistant lab coats and closed shoes offer another layer of defense, especially since it’s easier to sweep off spills from proper clothing than bare skin.
Ventilation helps keep fumes at bay. Even when opening a new bottle, I work in a fume hood if possible. That way, any stray dust or vapors move away from my face. I’ve heard too many stories of colleagues taking short breaths after “quickly” working outside a dedicated hood. It’s never worth the risk.
Waste Management and Spill Response
Disposing of any nitro or hydrazide compound requires planning, not improvisation. Collecting waste in dedicated, clearly labeled containers prevents accidental mixing that could lead to fire or toxic fume release. I always check the container material and confirm with local hazardous waste protocols, since this material rarely goes down the drain without running afoul of environmental rules. Quick clean-up of spills—using inert absorbents like sand—prevents the powder from spreading. Never sweep it dry or let it blow around. Wet paper towels don’t absorb, they spread. A little vigilance means a lot fewer headaches later.
Training, Documentation, and Ongoing Vigilance
Written procedures and regular safety refreshers keep everyone alert. In my experience, even experienced chemists grow forgetful over time, especially if they haven’t handled hydrazides in a while. Standard operating procedures posted near the storage area make safe handling an everyday habit, not a forgotten protocol. Incident logs and inspection records help spot small problems before they turn serious. Routine checks on container integrity, label clarity, and storage environment stop issues before they start.
With the right respect, 4-nitrobenzohydrazide doesn’t have to be intimidating. Knowledge, experience, and a few simple habits keep labs safe. This is an area I never cut corners on—because once trouble starts, it doesn’t back down easily.
What is the purity and appearance of 4-Nitrobenzohydrazide?
What Stands Out About Chemical Purity
Purity takes center stage for anyone working with 4-Nitrobenzohydrazide, whether you’re at a lab bench or managing quality control for a producer. In my own work in research, the measurement of purity earns more than just a passing glance—it’s a daily priority. For 4-Nitrobenzohydrazide, top-quality samples usually show a purity of at least 98%, checked through techniques like high-performance liquid chromatography (HPLC) or NMR. Anything less brings risks: unwanted byproducts during reactions, unreliable test results, or safety hazards down the line.
One time, a batch came in at less than 95%. Chasing down the impurity cost our team both time and resources. Not only did it throw off the results, it also triggered safety questions, since low-purity chemicals often carry unpredictable and sometimes dangerous side products. Reliable suppliers put their reputation on the line by publishing clear quality control data, showing transparency and commitment to safety.
The Visual Markers: What to Look for in Appearance
4-Nitrobenzohydrazide typically shows up as a pale yellow to yellow crystalline powder. In my experience, any departure from this suggested closer inspection. Off-color means you might be looking at contamination or degradation, which can turn the powder orange, brown, or even red if things have really gone off track due to oxidation.
Texture tells another part of the story. A fine, even powder handles well and dissolves without fuss. Clumping or gritty particles, on the other hand, often hint at moisture or leftover reactants, neither of which do any favors in analytical reactions. A clean, dry, free-flowing powder offers confidence that the material stores well and doesn’t carry hidden surprises.
Why Purity and Appearance Matter
Whether it’s for drug development, dyes, or chemical synthesis, users rely on pure, well-characterized chemicals for serious work. Pharmaceutical labs, in particular, demand high purity for any intermediate because impurities carry forward and affect the final medicine’s safety profile. For dye work, minor impurities can skew color or produce unstable results. Even small impurities can act as poisons in catalytic reactions.
If something looks off, the easiest fix is to check with the supplier for their latest batch record or certificate of analysis. I have found that even big suppliers occasionally miss a batch that gets exposed to moisture or heat during shipping, changing both color and purity. Good storage—cool, dry, and away from light—helps keep the powder in top shape over time.
Investing in Quality and Trust
The marketplace doesn’t always make things easy, with price competition pushing some suppliers to cut corners. Too many buyers worry most about cost instead of provenance. Supporting producers with clear, traceable supply chains and strict quality checks builds a safer industry for all. I’ve found that investing upfront in trusted chemicals pays off many times over by cutting troubleshooting and safety worries later.
To keep chemicals like 4-Nitrobenzohydrazide safe for serious lab work, nothing beats careful storage, reliable sourcing, and a sharp eye for unexpected changes in color or texture. This approach minimizes risk and keeps research and production on solid ground.
Is 4-Nitrobenzohydrazide hazardous or toxic?
Understanding the Chemical’s Profile
4-Nitrobenzohydrazide pops up in conversations among chemists and lab professionals. It looks like a fairly average compound at first glance: crystalline, pale color, not particularly volatile. The fact that it contains a nitro group and a hydrazide functional group should trigger some caution right away. In my experience working in university labs, those chemical features usually spell extra care. Nitro compounds, for example, carry a reputation for toxicity and sometimes explosive behavior, depending on the context.
Known Health Hazards
Looking through safety data for 4-Nitrobenzohydrazide, certain risks stand out. Direct contact causes irritation to eyes, skin, and airways. Dust from powders can easily get airborne, leading to inhalation risks—something that becomes a real issue in tight quarters or with poor ventilation. The combination of nitro and hydrazide groups suggests a strong potential for toxicity. Similar compounds in the same family have known harmful effects, such as affecting blood cells or the nervous system after prolonged or heavy exposure. Acute inhalation or accidental ingestion would likely trigger nausea, dizziness, maybe convulsions in serious exposures—symptoms that mean get to the doctor right away.
Regulatory bodies in Europe and North America treat 4-Nitrobenzohydrazide as a hazardous material. The Globally Harmonized System for Classification and Labelling of Chemicals (GHS) flags it for health risks. Many universities require gloves, goggles, and lab coats when handling it. Based on chemical structure and available toxicity scores, there’s a good chance the compound turns out harmful to aquatic life. Lab manuals underline the importance of proper disposal—never down the drain, always inside hazardous waste containers so local water supply stays protected.
Gaps in Data and the Importance of Precaution
Some people point out the gaps in hard data: long-term studies about its effects on people or ecosystems don’t exist in detail. Still, you can draw lessons from similar molecules. Both nitro and hydrazide compounds have records of causing cellular damage, oxidative stress, and sometimes being possible carcinogens. Anyone who’s spent semesters in organic synthesis has watched the safety lectures about handling powders and vapors, particularly with unknown or only partially understood toxicities. That’s not just about ticking a box on a checklist—problems show up when folks get careless. Even if there’s no incident this time, building bad habits with one chemical leads to risks with another.
Taking Practical Steps
Protecting yourself starts with the basics: good gloves, a lab coat, and always working in a fume hood. In small educational labs, those seem like overkill until someone finds out the hard way that some chemical dust isn’t visible and sticks to hands and equipment. Never underestimate the effect of cross-contamination—just a fleck on a doorknob travels classrooms or homes. Storage matters, too: keeping the chemical in tightly sealed containers, labeled with hazard warnings, removes a lot of uncertainty for the next person who handles it.
Some practical solutions focus on training: anybody in contact with 4-Nitrobenzohydrazide deserves clear, simple safety information and reminders about precaution. Higher education programs benefit from building strong safety cultures, making it just as routine as putting on sneakers. In industry, periodic reviews and proper engineering controls keep exposures low. Looking forward, researchers can keep pressing for more thorough studies— both for workers and the environment—so decision-making becomes more data-driven rather than guesswork.
Final Thoughts
Trust builds in chemistry by respecting each material's risks, even if data are incomplete. Emphasizing training, protection, and responsible disposal keeps labs safer and the environment cleaner. In my own work, that mindset moves beyond compliance and into genuine care for colleagues and communities.
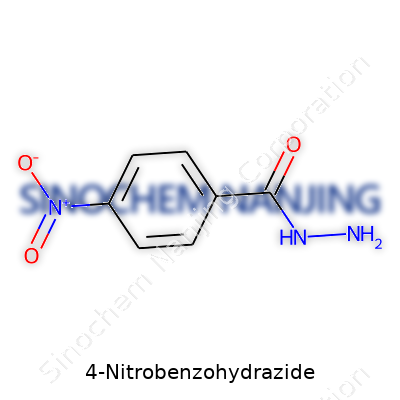
| Names | |
| Preferred IUPAC name | 4-nitrobenzohydrazide |
| Other names |
4-Nitrobenzhydrazide p-Nitrobenzhydrazide 4-Nitrobenzoic acid hydrazide p-Nitrobenzoic acid hydrazide |
| Pronunciation | /ˌfɔːrˌnaɪtroʊˌbɛnzoʊ.haɪˈdræzaɪd/ |
| Identifiers | |
| CAS Number | 619-94-3 |
| 3D model (JSmol) | `/data/structures/all/3D/61947/61947.pdb` |
| Beilstein Reference | 136444 |
| ChEBI | CHEBI:36809 |
| ChEMBL | CHEMBL417246 |
| ChemSpider | 25123077 |
| DrugBank | DB07845 |
| ECHA InfoCard | ECHA InfoCard: 100.038.318 |
| EC Number | 619-94-1 |
| Gmelin Reference | 727844 |
| KEGG | C14311 |
| MeSH | D014782 |
| PubChem CID | 98085 |
| RTECS number | CU8750000 |
| UNII | 5O5L9D2M1P |
| UN number | 3466 |
| CompTox Dashboard (EPA) | 4-Nitrobenzohydrazide: "DTXSID10142160 |
| Properties | |
| Chemical formula | C7H7N3O3 |
| Molar mass | 166.14 g/mol |
| Appearance | White to light yellow crystalline powder |
| Odor | Odorless |
| Density | 1.53 g/cm3 |
| Solubility in water | slightly soluble |
| log P | 0.08 |
| Acidity (pKa) | 11.02 |
| Basicity (pKb) | 13.04 |
| Magnetic susceptibility (χ) | -64.7×10^-6 cm³/mol |
| Dipole moment | 3.53 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | Std molar entropy (S⦵298) of 4-Nitrobenzohydrazide is 251.0 J·mol⁻¹·K⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. May cause respiratory irritation. |
| GHS labelling | GHS labelling of 4-Nitrobenzohydrazide: GHS07, GHS09 |
| Pictograms | GHS07, GHS09 |
| Signal word | Danger |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | Precautionary statements: P261, P280, P302+P352, P305+P351+P338, P332+P313, P337+P313 |
| NFPA 704 (fire diamond) | 2-3-1-Ξ |
| Flash point | 164°C |
| Lethal dose or concentration | LD50 (oral, rat): >2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 >2000 mg/kg |
| PEL (Permissible) | No established PEL |
| REL (Recommended) | 10 mg |
| Related compounds | |
| Related compounds |
Benzohydrazide 4-Nitrobenzoic acid 4-Nitrobenzoyl chloride 4-Nitrophenylhydrazine Hydrazine hydrate |

